Thermoregulatory Responses of Heat Acclimatized Buffaloes to Simulated Heat Waves
Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Animals and Experimental Conditions
2.2. Experimental Design
2.3. Physiological Variables
2.4. Blood Parameters
2.5. Statistical Analysis
3. Results
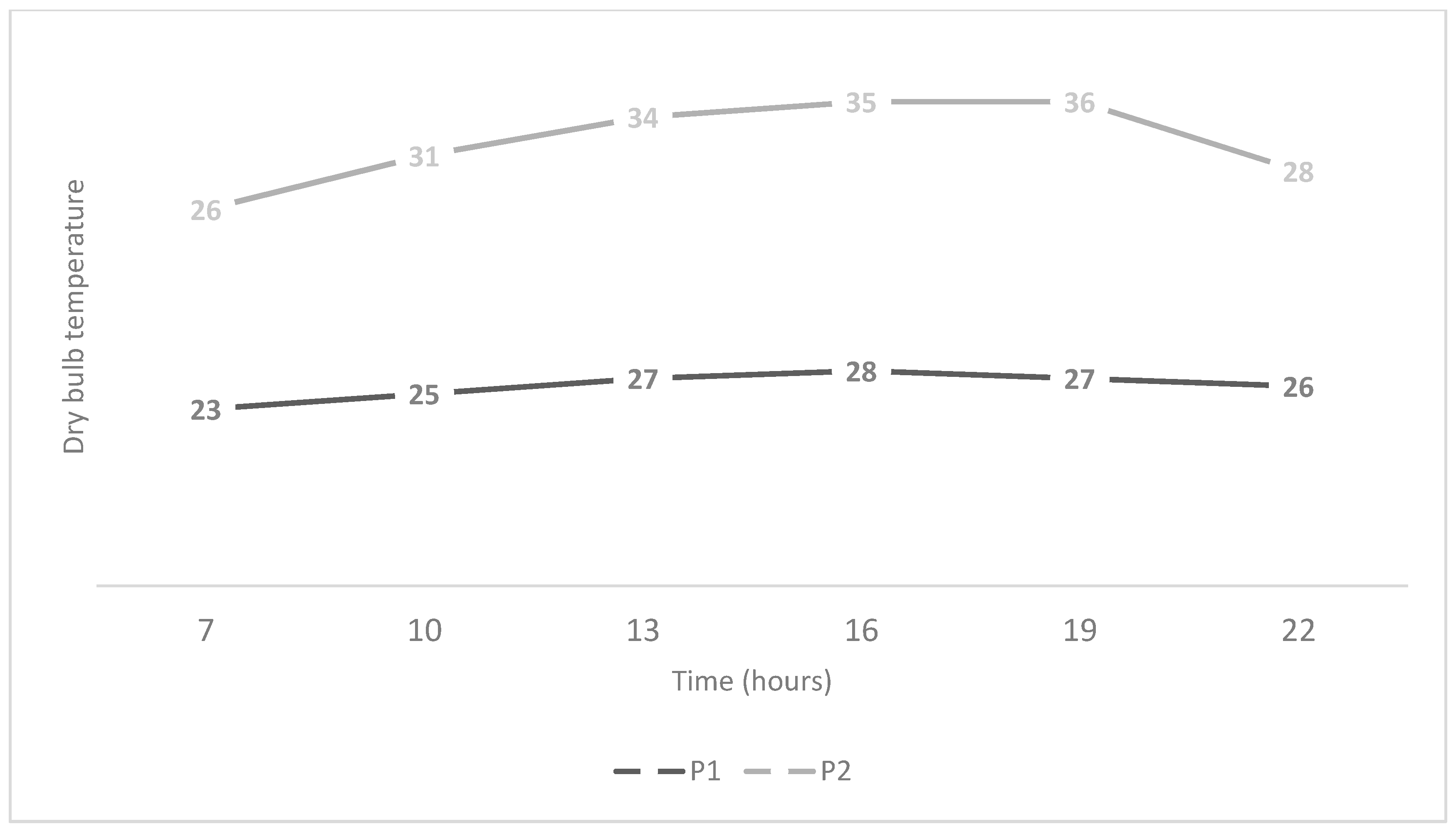
3.1. Coat and Epidermis Temperatures
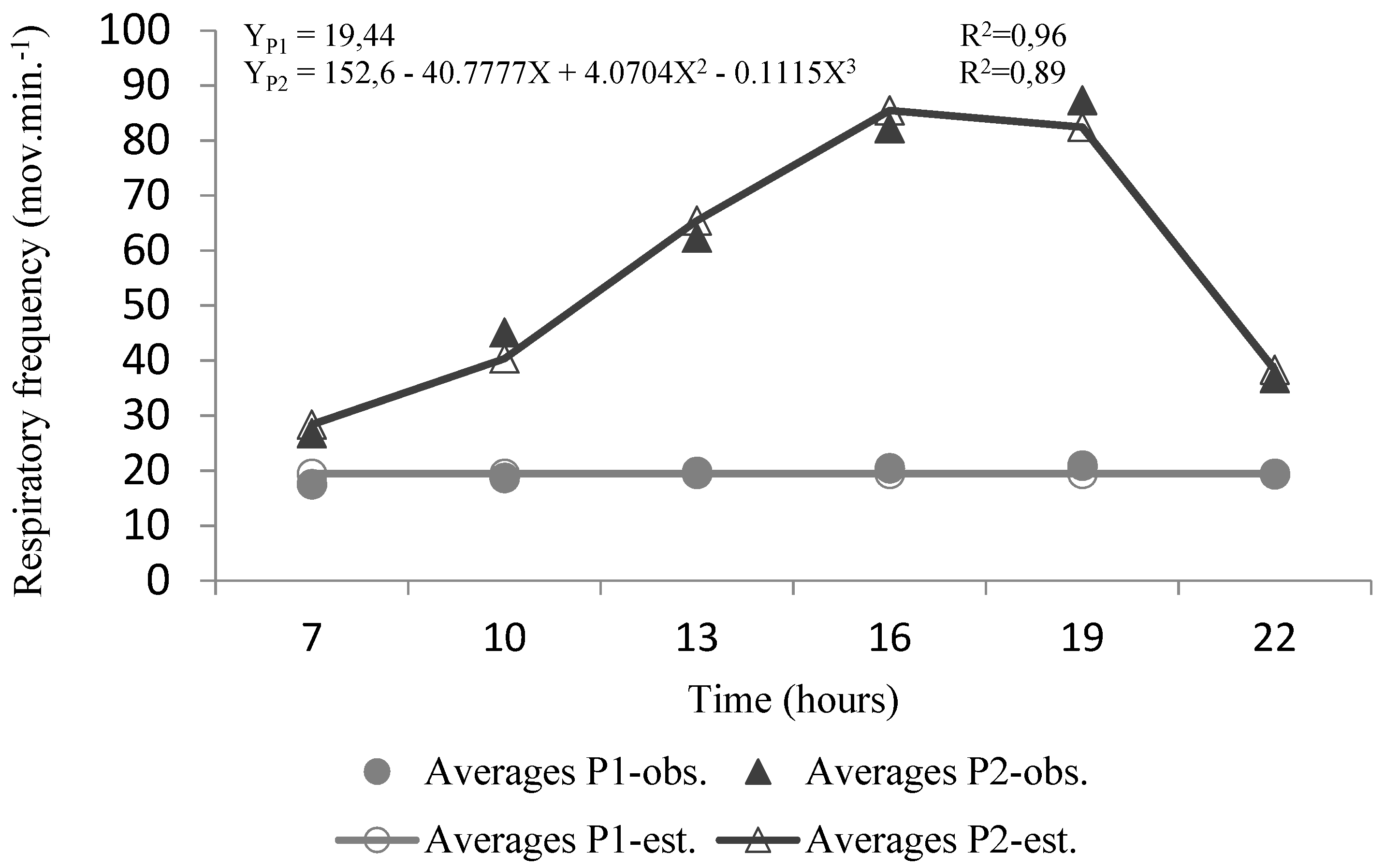
3.2. Respiratory Frequency
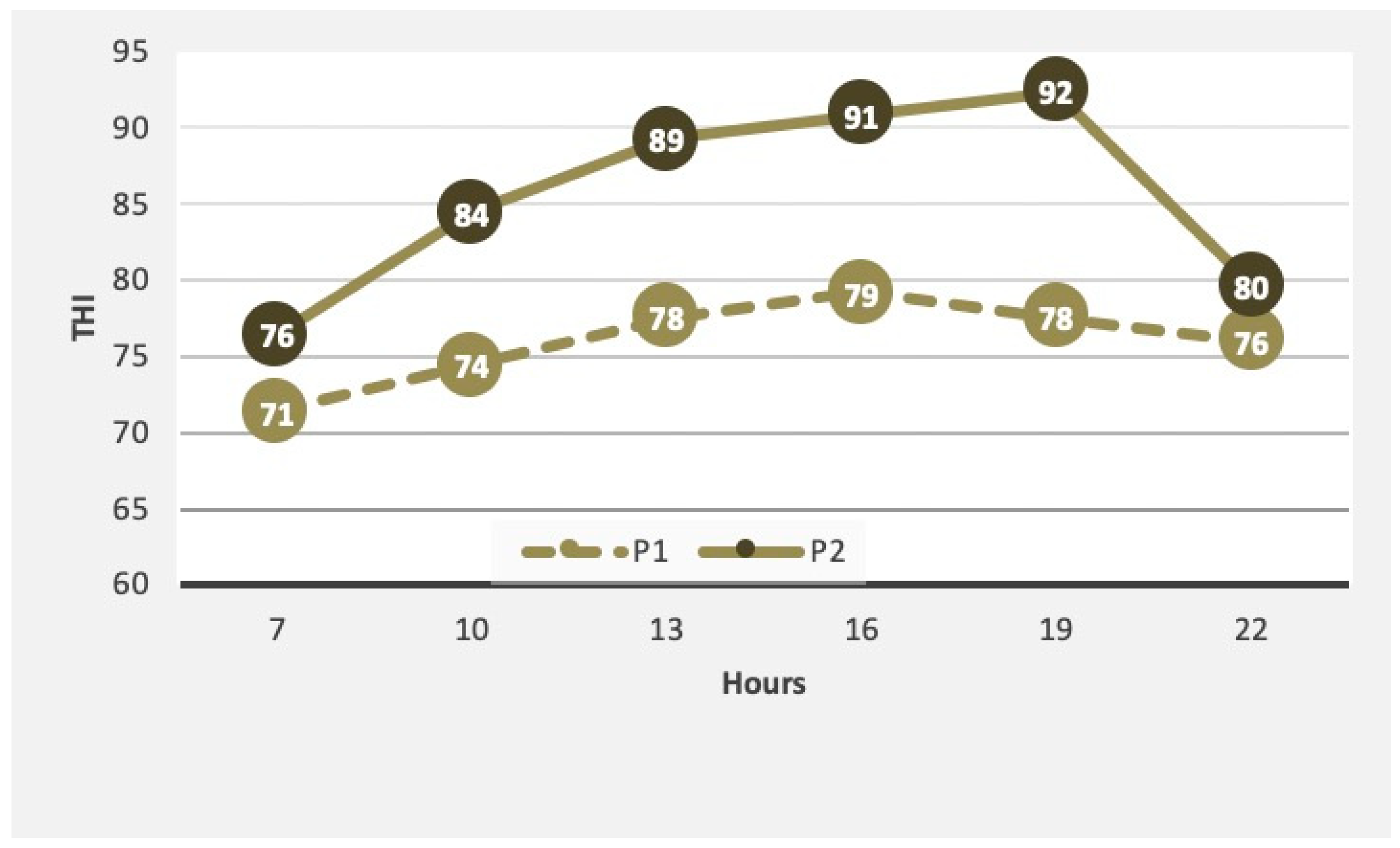
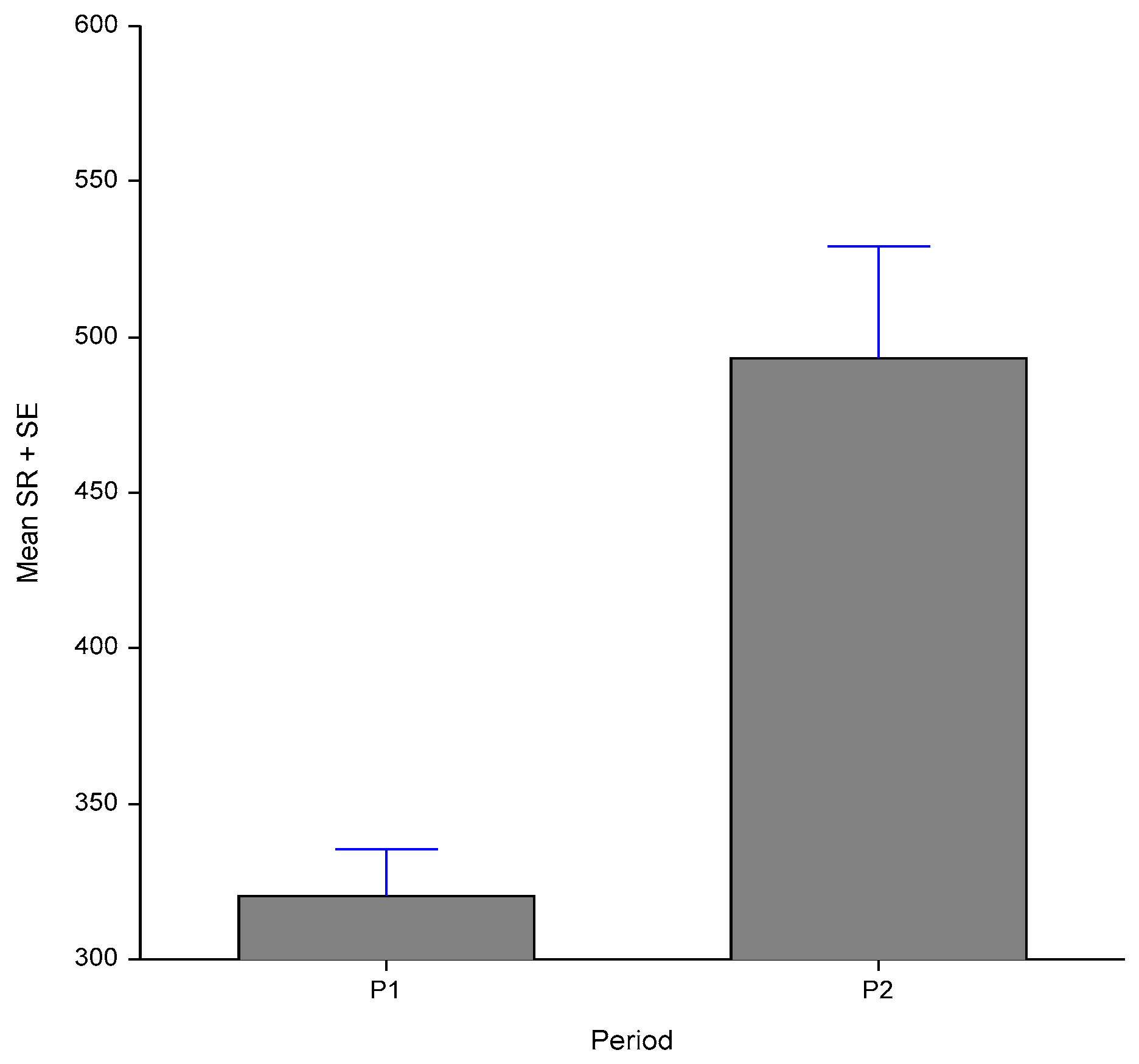
3.3. Sweating Rate
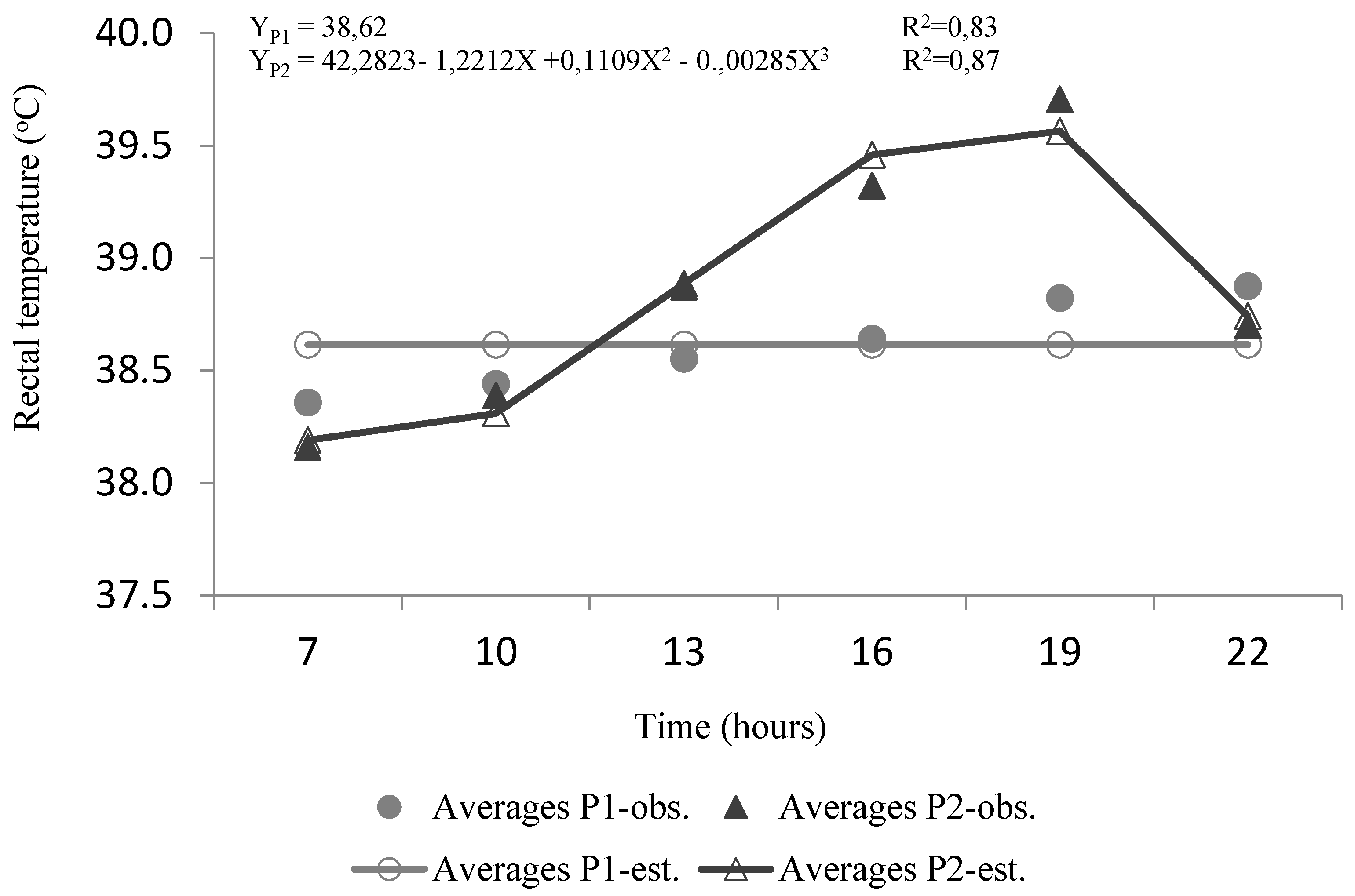
3.4. Rectal Temperature, Heat Storage, and Cumulative Heat Storage
3.5. Hematological White Blood Cells and Electrolytic Variables
4. Discussion
4.1. Coat, Epidermis Temperatures and Sensible Heat Loss
4.2. Respiratory Frequency, Sweating Rate and Evaporative Heat Loss
4.3. Rectal Temperature and Heat Storage
4.4. Hemogram, Leukogram and Electrolytes
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Mearns, L.O.; Katz, R.W.; Schneider, S.H. Extreme high–temperature events; changes in their probabilities with changes in mean temperature. J. Clim. Appl. Meteorol. 1984, 23, 1601–1613. [Google Scholar] [CrossRef]
- Hahn, G.L.; Mader, T.L. Heat waves in relation to thermoregulation, feeding behavior and mortality of feedlot cattle. In Livestock Environment V, Proceedings of the 5th International Symposium ASAE, St. Joseph, Bloomington, MA, USA, 29–30 May 1997; Bottcher, R.W., Hoff, S.J., Eds.; USDA: Washington, DC, USA, 1997; pp. 563–571. [Google Scholar]
- Nienaber, J.A.; Hahn, G.L. Livestock production system management responses to thermal challenges. Int. J. Biometeorol. 2007, 52, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Mader, T.L.; Hungerford, L.L.; Nienaber, J.A.; Buhman, M.J.; Davis, M.S.; Hahn, G.L.; Cerkoney, W.M.; Holt, S.M. Heat stress mortality in Midwest feedlots. J. Anim. Sci. 2001, 79, 2. [Google Scholar]
- Vitali, A.; Felici, A.; Esposito, S.; Bernabucci, U.; Bertocchi, L.; Maresca, C.; Nardone, A.; Lacetera, N. The effect of heat waves on dairy cow mortality. J. Dairy Sci. 2015, 98, 4572–4579. [Google Scholar] [CrossRef]
- Robinson, P.J. On the definition of a heat wave. J. Appl. Meteorol. 2001, 40, 762–775. [Google Scholar] [CrossRef]
- Russo, S.; Dosio, A.; Graversen, R.G.; Sillmann, J.; Carrao, H.; Dunbar, M.B.; Vogt, J.V. Magnitude of extreme heat waves in present climate and their projection in a warming world. J. Geophys. Res. Atmosph. 2014, 119, 12–500. [Google Scholar] [CrossRef]
- Beniston, M.; Stephenson, D.B.; Christensen, O.B.; Ferro, C.A.; Frei, C.; Goyette, S.; Halsnaes, K.; Holt, T.; Jylhä, K.; Koffi, B.; et al. Future extreme events in European climate: An exploration of regional climate model projections. Clim. Change 2007, 81, 71–95. [Google Scholar] [CrossRef]
- Marai, I.F.M.; Haeeb, A.A.M. Buffalo’s biological functions as affected by heat stress: A review. Livest. Sci. 2010, 127, 89–109. [Google Scholar] [CrossRef]
- Shafie, M.M. Biological adaptation of Buffaloes to climatic conditions. In Prospects of buffalo production in the Mediterranean and the Middle East; International Center for Agriculture: Doki (Cairo), Egypt, 1992; pp. 176–185. [Google Scholar]
- Aggarwal, A.; Singh, M. Changes in skin and rectal temperature in lactating buffaloes provided with showers and wallowing during hot–dry season. Trop. Anim. Health Prod. 2008, 40, 223–228. [Google Scholar] [CrossRef]
- Joshi, B.C.; McDowell, R.E.; Sadhu, D.P. Body surface evaporation rates at low and high temperatures in Murrah buffalo. J. Dairy Sci. 1968, 51, 1689–1692. [Google Scholar] [CrossRef]
- Moran, J.B. Heat tolerance of Brahman cross, buffalo, bauteng and shorthorn steers during exposure to sun and as a result of exercise. Aust. J. Exp. Agric. 1973, 24, 775–782. [Google Scholar] [CrossRef]
- Badreldin, A.L.; Ghany, M.A. Adaptive mechanisms of buffaloes to ambient temperature. Nature 1952, 170, 457–458. [Google Scholar] [CrossRef] [PubMed]
- Chikamune, T.; Kanai, Y.; Ichikawa, T.; Homma, H.; Shimizu, H. Influence of solar radiation and effects of water spray on thermoregulatory responses and heat production in swamp buffaloes. Jpn. J. Trop. Agric. 1987, 31, 1–5. [Google Scholar]
- Wankar, A.K.; Singh, G.; Yadav, B. Thermoregulatory and adaptive responses of adult buffaloes (Bubalus bubalis) during hyperthermia: Physiological, behavioral and metabolic approach. Vet. World 2014, 7, 825–830. [Google Scholar] [CrossRef]
- Koga, A.; Kurata, K.; Furukawa, R.; Nakajima, M.; Homma, H.; Kanai, Y.; Chikamune, T. Rectal and skin temperature difference regulated by blood volume in swamp buffaloes in hot conditions: Comparative study of thermo–regulation in buffaloes and cattle. Anim. Sci. Tech. (Jpn.) 1998, 69, 81–89. [Google Scholar]
- Desta, T.T. Introduction of domestic buffalo (Bubalus bubalis) into Ethiopia would be feasible. Renew. Agric. Food Syst. 2012, 27, 305–313. [Google Scholar] [CrossRef]
- Collier, R.J.; Dahl, G.E.; Van Baale, M.J. Major Advances Associated with Environmental Effects on Dairy Cattle. J. Dairy Sci. 2006, 89, 1244–1253. [Google Scholar] [CrossRef]
- Korde, J.P.; Singh, G.; Varshney, V.P.; Shukla, D.C. Effects of long–term heat exposure on adaptive mechanism of blood acid–base in Buffalo calves. Asian. Aust. J. Anim. Sci. 2007, 13, 329–332. [Google Scholar] [CrossRef]
- Banerjee, D.; Ashutosh. Effect of thermal exposure on diurnal rhythms of physiological parameters and feed, water intake in Tharparkar and Karan Fries heifers. Biol. Rhythm Res. 2011, 42, 39–51. [Google Scholar] [CrossRef]
- Pereira, A.M.; Baccari, F., Jr.; Titto, E.A.; Almeida, J.A. Effect of thermal stress on physiological parameters, feed intake and plasma thyroid hormones concentration in Alentejana, Mertolenga, Frisian and Limousine cattle breeds. Int. J. Biometeorol. 2008, 52, 199–208. [Google Scholar] [CrossRef]
- Mirkena, T.; Duguma, G.; Haile, A.; Tibbo, M.; Okeyo, A.M.; Wurzinger, M.; Sölkner, J. Genetics of adaptation in domestic farm animals: A review. Livest. Sci. 2010, 132, 1–12. [Google Scholar] [CrossRef]
- Brown–Brandl, T.M.; Eigenberg, R.A.; Hahn, G.L.; Nienaber, J.A.; Mader, T.L.; Spiers, D.E.; Parkhurst, A.M. Analyses of thermoregulatory responses of feeder cattle exposed to simulated heat waves. Int. J. Biometeorol. 2005, 49, 285–296. [Google Scholar] [CrossRef]
- Collier, R.J.; Collier, J.L.; Rhoads, R.P.; Baumgard, L.H. Invited review: Genes involved in the bovine heat stress response. J. Dairy Sci. 2008, 91, 445–454. [Google Scholar] [CrossRef]
- Silanikove, N. Effects of heat stress on the welfare of extensively managed domestic ruminants. Livest. Prod. Sci. 2000, 67, 1–18. [Google Scholar] [CrossRef]
- Tattersall, G.J.; Sinclair, B.J.; Withers, P.C.; Fields, P.A.; Seebacher, F.; Cooper, C.E.; Maloney, S.K. Coping with thermal challenges: physiological adaptations to environmental temperatures. Compr. Physiol. 2012, 2, 2151–2202. [Google Scholar]
- Singh, S.P.; Newton, W.M. Acclimation of young calves to high temperatures: Physiologic responses. Am. J. Vet. Res. 1978, 39, 795–797. [Google Scholar]
- Beatty, D.T.; Barnes, A.; Taylor, E.; Pethick, D.; McCarthy, M.; Maloney, S.K. Physiological responses of Bos taurus and Bos indicus cattle to prolonged continuous heat and humidity. J. Anim. Sci. 2006, 84, 972–985. [Google Scholar] [CrossRef]
- Hooda, O.K.; Singh, G. Changes in serum electrolytes, mineral sand physiological responses in buffalo heifers exposed to high temperature in a psychrometric chamber. Indian J. Anim. Nutr. 2009, 26, 327–332. [Google Scholar]
- Hahn, G.L. Dynamic responses of cattle to thermal heat loads. J. Anim. Sci. 1999, 77 (Suppl. 2), 10–20. [Google Scholar] [CrossRef]
- McGovern, R.R.; Bruce, J.M. A model of the thermal balance for cattle in hot conditions. J. Agric. Eng. Res. 2000, 77, 81–92. [Google Scholar] [CrossRef]
- Birgel, E.H. Hematologia Clínica Veterinária. In Patologia Clínica Veterinária; USP: Rockville, MD, USA, 1982; Volume I. [Google Scholar]
- Lagerspetz, K.Y.H. What is thermal acclimation? J. Thermal Biol. 2006, 31, 332–336. [Google Scholar] [CrossRef]
- Alnaimy, A.; Habbeb, M.; Fayaz, M.; Marai, M.; Kamal, T.H. Heat Stress. In Farm Animals and the Environment, 1st ed.; Phillpis, C., Piggins, D., Eds.; CAB International: Oxon, UK, 1992; pp. 27–47. [Google Scholar]
- Haque, N.; Ludri, A.; Hassain, S.A.; Ashutosh, M. Alteration of metabolic profiles in young and adult Murrah buffaloes exposed to acute heat stress. Iranian J. Appl. Anim. Sci. 2012, 1, 23–29. [Google Scholar]
- Amakiri, S.F.; Funsho, O.N. Studies of rectal temperature, respiratory rates and heat tolerance in cattle in the humid tropics. Anim. Prod. 1979, 28, 329–335. [Google Scholar] [CrossRef]
- Koga, A.; Sugiyama, M.; Del Barrio, A.N.; Lapitan, R.M.; Arenda, B.R.; Robles, A.Y.; Kanai, Y. Comparison of the thermoregulatory response of buffaloes and tropical cattle, using fluctuations in rectal temperature, skin temperature and haematocrit as an index. J. Agric. Sci. 2004, 142, 351–355. [Google Scholar] [CrossRef]
- Titto, E.A.; Russo, H.G.; Lima, C.G. Efeito do banho de água sobre o conforto térmico de bubalinos. In Actas do XX Congresso de Zootecnia; APEZ: Santarém, Portugal, 1997; pp. 23–24. [Google Scholar]
- Gaughan, J.B.; Mader, T.L.; Holt, S.M.; Sullivan, M.L.; Hahn, G.L. Assessing the heat tolerance of 17 beef cattle genotypes. Int. J. Biometeorol. 2010, 54, 617–627. [Google Scholar] [CrossRef]
- Thompson, V.A.; Fadel, J.G.; Sainz, R.D. Meta–analysis to predict sweating and respiration rates for Bos indicus, Bos taurus, and their crossbreds. J. Anim. Sci. 2011, 89, 3973–3982. [Google Scholar] [CrossRef]
- Gaughan, J.B.; Holt, S.M.; Hahn, G.L.; Mader, T.L.; Eigenberg, R. Respiration Rate – Is it a good measure of stress in catle? J. Anim. Sci. 2000, 13 (Suppl. C), 329–332. [Google Scholar]
- Pereira, A.M.F.; Titto, E.L.; Infante, P.; Titto, C.G.; Geraldo, A.M.; Alves, A.; Leme, T.M.; Baccari, F., Jr.; Almeida, J.A. Evaporative heat loss in Bos taurus: Do different cattle breeds cope with heat stress in the same way? J. Therm. Biol. 2014, 45, 87–95. [Google Scholar] [CrossRef]
- Chaiyabutr, N.; Burassakurl, C.; Moanbchurogen, V.; Loypetjra, P.; Pichaichrnatong, A. Effect of acute heat stress on changes in the rate of liquid flow from the rumen and turnover of swamp buffalo. J. Agric. (Camb.) 1987, 108, 549–553. [Google Scholar] [CrossRef]
- Das, S.K.; Upadhyay, R.C.; Madan, M.L. Heat stress in buffalo calves. Livest. Prod. Sci. 1999, 61, 71–78. [Google Scholar] [CrossRef]
- Aggarwal, A.; Upadhyay, R.C. Studies on evaporative heat loss from skin and pulmonary surfaces in male buffaloes exposed to solar radiations. Buffalo J. 1998, 2, 179–187. [Google Scholar]
- Verma, D.L.; Lal, S.N.; Singh, G.; Parkash, O.M.; Parkash, O. Effect of season on biological responses and productivity of buffaloes. Int. J. Anim. Sci. 2000, 15, 234–244. [Google Scholar]
- Pereira, A.F.; Baccari, F., Jr.; Titto, E.L.; Almeida, J.A. Effect of shade after exposure to direct solar radiation on body temperature dynamic responses in Alentejana and Limousine beef cattle. In Proceeding of the VI International Livestock Environment Symposium, Louisville, KY, USA, 21–23 May 2001; pp. 235–240. [Google Scholar]
- Pereira, A.M.F. Adaptation to Mediterranean Geophysical Environment of Native and Exotic Cattle—The heat Tolerance. Ph.D. Thesis, University of Évora, Évora, Portugal, 2004. [Google Scholar]
- Koga, A.; Chikamune, T.; Kanai, Y.; Homma, H.; Tajima, A.; Ishikawa, N.; Furukawa, R.; Ueno, T.; Nakajima, M.; Watanabe, T. Effects of high environmental temperatures on some physicochemical parameters of blood and heat production in swamp buffaloes and Holstein cattle. Anim. Sci. Tech. (Jpn.) 1991, 62, 1022–1028. [Google Scholar]
- Krishnan, G.; Singh, G.; Shukla, D.C. Effect of electrolyte supplementation on physiological responses in heat stressed male buffalo calves. Indian J. Anim. Sci. 2011, 79, 34–37. [Google Scholar]
- Louw, G.N. Physiological Animal Ecology, 3rd ed.; Longman: Harlow, UK, 1993; p. 281. [Google Scholar]
- Akhtar, M.Z.; Khan, A.; Khan, M.Z.; Muhammad, G. Haemato–Biochemical Aspects of Parturient Haemoglubinuria in Buffalo. Turk. J. Vet. Anim. Sci. 2007, 31, 119–123. [Google Scholar]
- Ciaramella, P.; Corona, M.; Ambrosio, R.; Consalvo, F.; Persechino, A. Haematological profile on non–lactating Mediterranean buffaloes (Bubalus bubalis) ranging in age from 24 months to 14 years. Res. Vet. Sci. 2005, 79, 77–80. [Google Scholar] [CrossRef]
- Gomes, V.; Moura, J.; Madureira, K.M.; Baptistella, F.; Kitamura, S.; Blagitz, M.; Benesi, F. Valores de referência e influência da idade no eritrograma de bubalinos da raça Murrah. Pesq. Vet. Bras. 2010, 30, 301–304. [Google Scholar] [CrossRef]
- Shafie, M.M. Environmental effects on water buffalo production. World Anim. Rev. 1993, 77, 21–25. [Google Scholar]
- Silva, J.; Araújo, A.; Lourenço Júnior, J.; Santos, N.; Garcia, A.; Nahúm, B. Conforto térmico de búfalas em sistema silvipastoril na Amazônia Oriental. Pesq. Agrop. Bras. 2011, 46, 1364–1371. [Google Scholar] [CrossRef]
- Habibu, B.; Dzenda, T.; Ayo, J.; Yaqub, L.; Kawu, M. Haematological changes and plasma fluid dynamics in livestock during thermal stress, and response to mitigative measures. Livest. Sci. 2018, 214, 189–201. [Google Scholar] [CrossRef]
- Pandey, M.D.; Roy, A. Variation in cardiorespiratory rates, rectal temperature, blood hematocrit and hemoglobin as measures of adaptability in buffaloes to a hot environment. Br. Vet. J. 1969, 125, 463–471. [Google Scholar] [CrossRef]
- Chaiyabutr, N.; Buranakarl, C.; Loypetira, P. Red blood cell potassium types of murrah, swamp and crossbred buffaloes. Int. J. Anim. Sci. 1993, 8, 19. [Google Scholar]
- Kamwanja, L.A.; Chase, C.C.; Gutierrez, J.A.; Guerriero, V.; Olson, T.A.; Hammond, A.C.; Hansen, P.J. Responses of bovine lymphocytes to heat shock as modified by breed and antioxidant status. J. Anim. Sci. 1994, 72, 438–444. [Google Scholar] [CrossRef] [PubMed]
- Lacetera, N.; Bernabucci, U.; Ronchi, B.; Nardone, A. Moderate summer heat stress does not modify immunological parameters of Holstein dairy cows. Int. J. Biometeorol. 2002, 46, 33–37. [Google Scholar] [CrossRef]
- França, T. Valores hematológicos de búfalos em diferentes faixas etárias criados na região do Rio Grande do Sul. Rev. Bras. Cie. Vet. 2011, 18, 51–54. [Google Scholar] [CrossRef]
- Singh, K.M.; Singh, S.; Ganguly, I.; Ganguly, A.; Nachiappan, R.K.; Chopra, A.; Narula, H.K. Evaluation of Indian sheep breeds of arid zone under heat stress condition. Small Rum. Res. 2016, 141, 113–117. [Google Scholar] [CrossRef]
- Weiser, G. Interpretação da resposta leucocitária nas doenças. In Hematologia e bioquímica clínica veterinária; Tharall, M.A., Ed.; Roca: São Paulo, Brasil, 2007; pp. 118–122. [Google Scholar]
- Lacetera, N.; Bernabucci, U.; Scalia, D.; Basirico, L.; Morera, P.; Nardone, A. Heat stress elicits different responses in peripheral blood mononuclear cells from Brown Swiss and Holstein cows. J. Dairy Sci. 2006, 89, 4606–4612. [Google Scholar] [CrossRef]
- Fetman, M.J. Metabolismo de fluídos e eletrólitos. In Hematologia e Bioquímica Veterinária, 1st ed.; Thrall, M.A., Ed.; Editora Roca: São Paulo, Brasil, 2007; pp. 531–542. [Google Scholar]
- Beede, D.K.; Mallonee, P.G.; Schneider, P.L.; Wilcox, C.J.; Collier, R.J. Potassium nutrition of heat–stressed lactating dairy cows. S. Afr. J. Anim. Sci. 1983, 13, 198. [Google Scholar]


| Variables | Treatments | Hours | |||||
|---|---|---|---|---|---|---|---|
| 7 | 10 | 13 | 16 | 19 | 22 | ||
| CT (°C) | P1 | 30.22 dB | 31.49 cB | 33.22 bB | 34.28 aB | 33.53 abB | 32.92 bB |
| P2 | 33.20 cA | 37.32 bA | 38.66 aA | 38.78 bA | 38.94 aA | 33.81cA | |
| TT (°C) | P1 | 32.83 dB | 33.39 cdB | 34.19 bcB | 35.13 aB | 34.52 abB | 33.98 bcB |
| P2 | 34.14 cA | 35.99 bA | 37.31aA | 37.77 aA | 37.96 aA | 34.15 cA | |
| RF (mov/min) | P1 | 17.56 aB | 18.67 aB | 19.78 aB | 20.44 aB | 20.92 aB | 19.33 aB |
| P2 | 26.78 eA | 45.11 cA | 62.33 bA | 82.22 aA | 87.33 aA | 36.78 dA | |
| RT (°C) | P1 | 38.36 dA | 38.44 cdA | 38.55 bcB | 38.64 bB | 38.82 aB | 38.88 aA |
| P2 | 38.16 fB | 38.39 eA | 38.87 cA | 39.32 bA | 39.71 aA | 38.70 dAt | |
| HS (W/m2/h) | P1 | 0.00 cA | 0.0021 bcB | 0.0028 abB | 0.0023 abcB | 0.0045 aB | 0.0014 bcA |
| P2 | 0.00 dA | 0.0058 cA | 0.012 aA | 0.0112 abA | 0.0096 bA | −0.0251 eB | |
| Treatments | Variables | ||||||
|---|---|---|---|---|---|---|---|
| Er | Hg (g/dL) | Ht (%) | MCV (fl) | MCHC (%) | Na (mEq/L) | K (mEq/L) | |
| P1 | 6.64 A | 12.03 A | 29.83 A | 51.68 A | 19.97 A | 138.83 A | 4.44 A |
| P2 | 6.57 A | 12.15 A | 31.08 A | 48.41 A | 18.83 A | 138.67 A | 3.96 B |
| Treatments | Variables | ||||||
|---|---|---|---|---|---|---|---|
| Leu (Cel/mm3) | Nsg (Cel/mm3) | Lym (Cel/mm3) | Eos (Cel/mm3) | Bas (Cel/mm3) | Mon (Cel/mm3) | Nrd (Cel/mm3) | |
| P1 | 11667 A | 4272.50 A | 6780.33 A | 438.92 A | 46.33 A | 128.58 A | 25.00 A |
| P2 | 11825 A | 5238.13 A | 6154.96 A | 295.08 A | 10.66 A | 88.00 A | 38.17 A |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira, A.M.F.; Vilela, R.A.; Titto, C.G.; Leme-dos-Santos, T.M.C.; Geraldo, A.C.M.; Balieiro, J.C.C.; Calviello, R.F.; Birgel Junior, E.H.; Titto, E.A.L. Thermoregulatory Responses of Heat Acclimatized Buffaloes to Simulated Heat Waves. Animals 2020, 10, 756. https://doi.org/10.3390/ani10050756
Pereira AMF, Vilela RA, Titto CG, Leme-dos-Santos TMC, Geraldo ACM, Balieiro JCC, Calviello RF, Birgel Junior EH, Titto EAL. Thermoregulatory Responses of Heat Acclimatized Buffaloes to Simulated Heat Waves. Animals. 2020; 10(5):756. https://doi.org/10.3390/ani10050756
Chicago/Turabian StylePereira, Alfredo M. F., Reíssa A. Vilela, Cristiane G. Titto, Thays M. C. Leme-dos-Santos, Ana C. M. Geraldo, Júlio C. C. Balieiro, Raquel F. Calviello, Eduardo H. Birgel Junior, and Evaldo A. L. Titto. 2020. "Thermoregulatory Responses of Heat Acclimatized Buffaloes to Simulated Heat Waves" Animals 10, no. 5: 756. https://doi.org/10.3390/ani10050756
APA StylePereira, A. M. F., Vilela, R. A., Titto, C. G., Leme-dos-Santos, T. M. C., Geraldo, A. C. M., Balieiro, J. C. C., Calviello, R. F., Birgel Junior, E. H., & Titto, E. A. L. (2020). Thermoregulatory Responses of Heat Acclimatized Buffaloes to Simulated Heat Waves. Animals, 10(5), 756. https://doi.org/10.3390/ani10050756






