The Influence of Anandamide on the Anterior Pituitary Hormone Secretion in Ewes—Ex Vivo Study
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Experimental Procedure
Incubation of Pituitary Explants
2.3. Assays
2.3.1. Radioimmunoassay for LH
2.3.2. Radioimmunoassay for FSH
2.3.3. Relative Gene Expression Assay
2.3.4. Western Blot Assays for GnRHR Expression in the AP
2.4. Statistical Analysis
3. Results
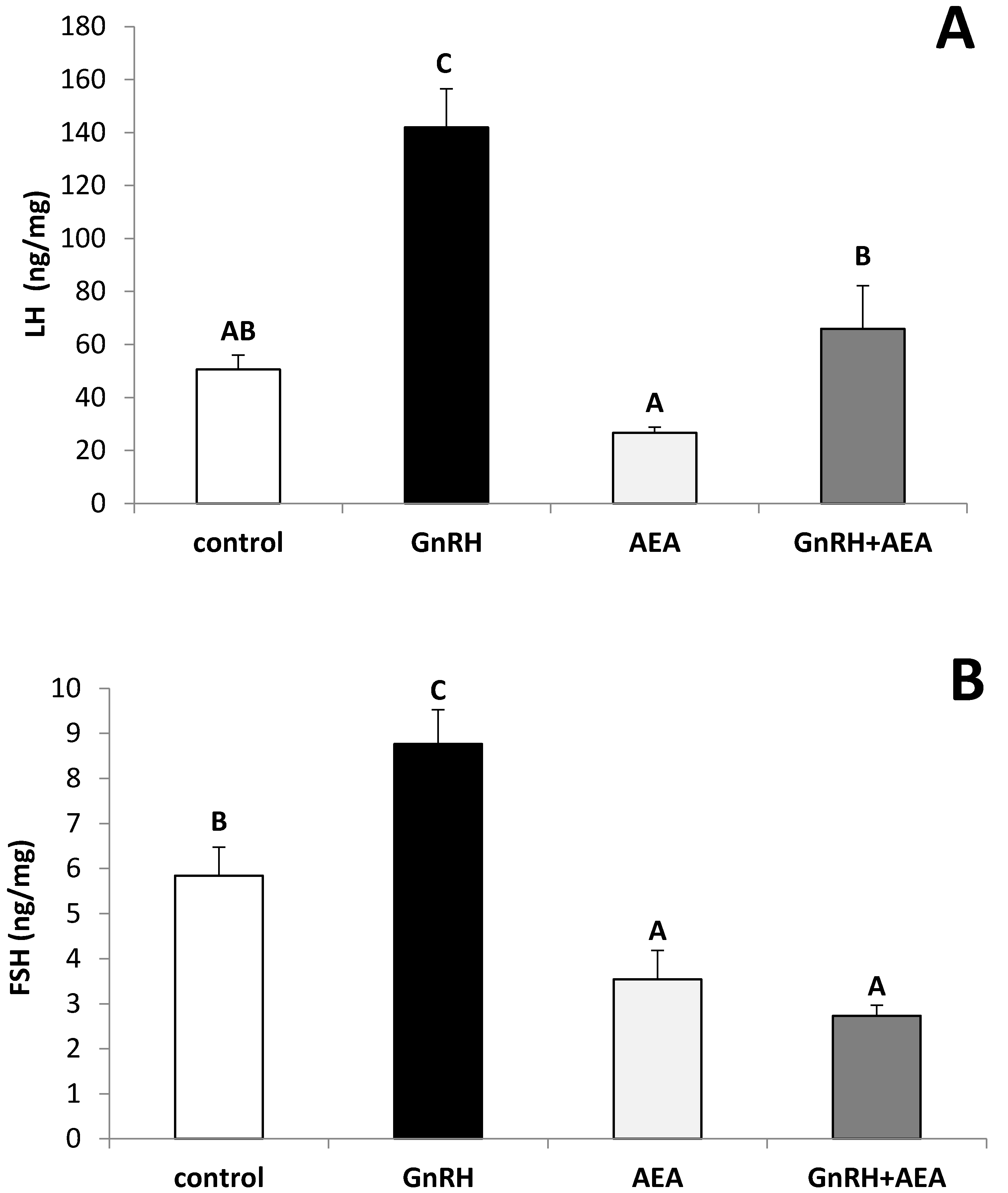
3.1. The Ex Vivo Influence of GnRH and Anandamide on the LH and FSH Release from the AP Explants
3.2. The Ex Vivo Influence of GnRH and Anandamide on the LHβ and FSHβ Gene Expression in the AP Explants
3.3. The Ex Vivo Effect of GnRH and Anandamide on the GnRHR Gene Expression and GnRHR Protein Expression in the AP Explants
3.4. The Ex Vivo Effect of GnRH and Anandamide on the Gene Expression of CNR1 in the AP Explants
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tomaszewska-Zaremba, D.; Herman, A.P.; Misztal, T. Does central IL-1β affect GnRH secretion in the hypothalamus of anoestrous ewes via different regulatory pathways? J. Anim. Feed Sci. 2013, 22, 5–12. [Google Scholar] [CrossRef]
- Bloch, E.; Thysen, B.; Morrill, G.A.; Gardner, E.; Fujimoto, G. Effects of cannabinoids on reproduction and development. Vitam Horm. 1978, 36, 203–258. [Google Scholar] [PubMed]
- Murphy, L.L.; Muñoz, R.M.; Adrian, B.A.; Villanúa, M.A. Function of cannabinoid receptors in the neuroendocrine regulation of hormone secretion. Neurobiol. Dis. 1998, 5, 32–46. [Google Scholar] [CrossRef] [PubMed]
- Pagotto, U.; Pasquali, R.J. Endocannabinoids and energy metabolism. Endocrinol. Investig. 2006, 29 (Suppl. 3), 66–76. [Google Scholar]
- Wang, H.; Dey, S.K.; Maccarrone, M. Jekyll and Hyde: Two faces of cannabinoid signaling in male and female fertility. Endocr. Rev. 2006, 27, 427–448. [Google Scholar] [CrossRef] [PubMed]
- Wenger, T.; Tóth, B.E.; Martin, B.R. Effects of anandamide (endogen cannabinoid) on anterior pituitary hormone secretion in adult ovariectomized rats. Life Sci. 1995, 56, 2057–2063. [Google Scholar] [CrossRef]
- Rettori, V.; Wenger, T.; Snyder, G.; Dalterio, S.; McCann, S.M. Hypothalamic action of delta-9-tetrahydrocannabinol to inhibit the release of prolactin and growth hormone in the rat. Neuroendocrinology 1988, 47, 498–503. [Google Scholar] [CrossRef]
- Fasano, S.; Meccariello, R.; Cobellis, G.; Chianese, R.; Cacciola, G.; Chioccarelli, T.; Pierantoni, R. The endocannabinoid system: An ancient signaling involved in the control of male fertility. Ann. N. Y. Acad. Sci. 2009, 1163, 112–124. [Google Scholar] [CrossRef]
- McPartland, J.M.; Matias, I.; Di Marzo, V.; Glass, M. Evolutionary origins of the endocannabinoid system. Gene 2006, 370, 64–74. [Google Scholar] [CrossRef]
- Zou, S.; Kumar, U. Cannabinoid Receptors and the Endocannabinoid System: Signaling and Function in the Central Nervous System. Int. J. Mol. Sci. 2018, 19, E833. [Google Scholar]
- Meccariello, R.; Chianese, R.; Fasano, S.; Pierantoni, R. Endocannabinoids and Kisspeptins: Two Modulators in Fight for the Regulation of GnRH Activity. In Book: Gonadotropin; Vizcarra, J., Ed.; Intech: London, UK, 2013; pp. 57–88. [Google Scholar]
- Devane, W.A.; Hanus, L.; Breuer, A.; Pertwee, R.G.; Stevenson, L.A.; Griffin, G.; Gibson, D.; Mandelbaum, A.; Etinger, A.; Mechoulam, R. Isolation and structure of a brain constituent that binds to the cannabinoid receptor. Science 1992, 258, 1946–1949. [Google Scholar] [CrossRef] [PubMed]
- Brown, S.M.; Wager-Miller, J.; Mackie, K. Cloning and molecular characterization of the rat CB2 cannabinoid receptor. Biochim. Biophys. Acta 2002, 1576, 255–264. [Google Scholar] [CrossRef]
- Galiègue, S.; Mary, S.; Marchand, J.; Dussossoy, D.; Carrière, D.; Carayon, P.; Bouaboula, M.; Shire, D.; Le Fur, G.; Casellas, P. Expression of central and peripheral cannabinoid receptors in human immune tissues and leukocyte subpopulations. Eur. J. Biochem. 1995, 232, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, L.A.; Lolait, S.J.; Brownstein, M.J.; Young, A.C.; Bonner, T.I. Structure of a cannabinoid receptor and functional expression of the cloned cDNA. Nature 1990, 346, 561–564. [Google Scholar] [CrossRef] [PubMed]
- Shire, D.; Calandra, B.; Rinaldi-Carmona, M.; Oustric, D.; Pessègue, B.; Bonnin-Cabanne, O.; Le Fur, G.; Caput, D.; Ferrara, P. Molecular cloning, expression and function of the murine CB2 peripheral cannabinoid receptor. Biochim. Biophys. Acta 1996, 1307, 132–136. [Google Scholar] [CrossRef]
- Lauckner, J.E.; Jensen, J.B.; Chen, H.Y.; Lu, H.C.; Hille, B.; Mackie, K. GPR55 is a cannabinoid receptor that increases intracellular calcium and inhibits M current. Proc. Natl. Acad. Sci. USA 2008, 105, 2699–2704. [Google Scholar] [CrossRef] [PubMed]
- Herkenham, M.; Lynn, A.B.; Johnson, M.R.; Melvin, L.S.; de Costa, B.R.; Rice, K.C. Characterization and localization of cannabinoid receptors in rat brain: A quantitative in vitro autoradiographic study. J. Neurosci. 1991, 11, 563–583. [Google Scholar] [CrossRef]
- González, S.; Manzanares, J.; Berrendero, F.; Wenger, T.; Corchero, J.; Bisogno, T.; Romero, J.; Fuentes, J.A.; Di Marzo, V.; Ramos, J.A.; et al. Identification of endocannabinoids and cannabinoid CB(1) receptor mRNA in the pituitary gland. Neuroendocrinology 1999, 70, 137–145. [Google Scholar] [CrossRef]
- González, S.; Bisogno, T.; Wenger, T.; Manzanares, J.; Milone, A.; Berrendero, F.; Di Marzo, V.; Ramos, J.A.; Fernández-Ruiz, J.J. Sex steroid influence on cannabinoid CB(1) receptor mRNA and endocannabinoid levels in the anterior pituitary gland. Biochim. Biophys. Res. Commun. 2000, 270, 260–266. [Google Scholar] [CrossRef]
- Lynn, A.B.; Herkenham, M. Localization of cannabinoid receptors and nonsaturable high-density cannabinoid binding sites in peripheral tissues of the rat: Implications for receptor-mediated immune modulation by cannabinoids. J. Pharmacol. Exp. Ther. 1994, 268, 1612–1623. [Google Scholar]
- Wenger, T.; Fernández-Ruiz, J.J.; Ramos, J.A. Immunocytochemical demonstration of CB1 cannabinoid receptors in the anterior lobe of the pituitary gland. J. Neuroendocrinol. 1999, 11, 873–878. [Google Scholar] [CrossRef] [PubMed]
- Cesa, R.; Guastalla, A.; Cottone, E.; Mackie, K.; Beltramo, M.; Franzoni, M.F. Relationships between CB1 cannabinoid receptors and pituitary endocrine cells in Xenopus laevis: An immunohistochemical study. Gen. Comp. Endocrinol. 2002, 125, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Yasuo, S.; Unfried, C.; Kettner, M.; Geisslinger, G.; Korf, H.W. Localization of an endocannabinoid system in the hypophysial pars tuberalis and pars distalis of man. Cell Tissue Res. 2010, 342, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Wojtulewicz, K.; Tomaszewska-Zaremba, D.; Herman, A.P. Endotoxin-Induced Inflammation Suppresses the Effect of Melatonin on the Release of LH from the Ovine Pars Tuberalis Explants-Ex Vivo Study. Molecules 2017, 22, 1933. [Google Scholar] [CrossRef]
- Stupnicki, R.; Madej, A. Radioimmunoassay of LH in blood plasma of farm animals. Endocrinologie 1976, 68, 6–13. [Google Scholar]
- L’Hermite, M.; Niswender, G.D.; Reichert, L.E.; Midgley, A.R. Serum follicle stimulating hormone in sheep as measured by radioimmunoassay. Biol. Reprod. 1972, 6, 325–332. [Google Scholar] [CrossRef]
- Rasmussen, R. Quantification on the LightCycler. In Rapid Cycle Real-Time PCR Methods and Applications; Meuer, S., Wittwer, C., Nakagawara, K., Eds.; Springer: Berlin, Germany, 2001; pp. 21–34. [Google Scholar]
- Herman, A.P.; Krawczyńska, A.; Bochenek, J.; Antushevich, H.; Herman, A.; Tomaszewska-Zaremba, D. Peripheral injection of SB203580 inhibits the inflammatory-dependent synthesis of proinflammatory cytokines in the hypothalamus. BioMed. Res. Internat. 2014, 2014, 475152. [Google Scholar] [CrossRef]
- Herman, A.P.; Krawczyńska, A.; Bochenek, J.; Haziak, K.; Romanowicz, K.; Misztal, T.; Antushevich, H.; Herman, A.; Tomaszewska Zaremba, D. The effect of rivastigmine on the LPS-induced suppression of GnRH/LH secretion during the follicular phase of the estrous cycle in ewes. Anim. Reprod. Sci. 2013, 38, 203–212. [Google Scholar] [CrossRef]
- Wenger, T.; Jamali, K.A.; Juanéda, C.; Bácsy, E.; Tramu, G. The endogenous cannabinoid, anandamide regulates anterior pituitary secretion in vitro. Addict. Biol. 2000, 5, 59–64. [Google Scholar] [CrossRef]
- Giannikou, P.; Yiannakakis, N.; Frangkakis, G.; Probonas, K.; Wenger, T. Anandamide (endogenous cannabinoid) decreases serum prolactin in pregnant rat. Neuroendocrinol. Lett. 1995, 17, 281–287. [Google Scholar]
- Wenger, T.; Jamali, K.A.; Juanéda, C.; Leonardelli, J.; Tramu, G. Arachidonyl ethanolamide (anandamide) activates the parvocellular part of hypothalamic paraventricular nucleus. Biochem. Bioph. Res. Commun. 1997, 238, 724–728. [Google Scholar] [CrossRef] [PubMed]
- Wenger, T.; Croix, D.; Tramu, G. The effect of chronic prepubertal administration of marihuana (delta-9-tetrahydrocannabinol) on the onset of puberty and the postpubertal reproductive functions in female rats. Biol. Reprod. 1988, 39, 540–545. [Google Scholar] [CrossRef] [PubMed]
- Steger, R.W.; Silverman, A.Y.; Siler-Khodr, T.M.; Asch, R.H. The effects of delta 9-tetrahydrocannabinol on the positive and negative feedback control of luteinizing hormone release. Life Sci. 1980, 27, 1911–1916. [Google Scholar] [CrossRef]
- Scorticati, C.; Fernández-Solari, J.; De Laurentiis, A.; Mohn, C.; Prestifilippo, J.; Lasaga, M.; Seilicovich, A.; Billi, S.; Franchi, A.; McCann, S.M.; et al. The inhibitory effect of anandamide on luteinizing hormone-releasing hormone secretion is reversed by estrogen. Proc. Natl. Acad. Sci. USA 2004, 101, 11891–11896. [Google Scholar] [CrossRef] [PubMed]
- Gorzalka, B.B.; Dang, S.S. Minireview: Endocannabinoids and gonadal hormones: Bidirectional interactions in physiology and behavior. Endocrinology 2012, 153, 1016–1024. [Google Scholar] [CrossRef]
- Chianase, R.; Ciaramella, V.; Fasano, S.; Pieranton, R.; Meccariello, R. Anandamide modulates the expression of GnRH-II and GnRHRs in frog, Rana esculenta, diencephalon. Gen. Com. Endocrinol. 2011, 173, 389–395. [Google Scholar] [CrossRef]
- Fox, S.R.; Hoefer, M.T.; Bartke, A.; Smith, M.S. Suppression ofpulsatile LH secretion, pituitary GnRH receptor content and pituitary responsiveness to GnRH by hyperprolactinemia in the male rat. Neuroendocrinology 1987, 46, 350–359. [Google Scholar] [CrossRef]
- Nett, T.M.; Turzillo, A.M.; Baratta, M.; Rispoli, L.A. Pituitary effects of steroid hormones on secretion of follicle-stimulating hormone and luteinizing hormone. Domest. Anim. Endocrinol. 2002, 23, 33–42. [Google Scholar] [CrossRef]
- Rispoli, L.A.; Nett, T.M. Pituitary gonadotropin-releasing hormone (GnRH) receptor: Structure, distribution and regulation of expression. Anim. Reprod. Sci. 2005, 88, 57–74. [Google Scholar] [CrossRef]
- Haziak, K.; Herman, A.P.; Tomaszewska-Zaremba, D. The effect of LPS on LH release and gene expression of LH-β, GnRH-R and TLR4 in the anterior pituitary of follicular phase ewes—An in vitro study. J. Anim. Feed Sci. 2013, 22, 97–105. [Google Scholar] [CrossRef]
- Dall’Aglio, C.; Millán, P.; Maranesi, M.; Rebollar, P.G.; Brecchia, G.; Zerani, M.; Gobbetti, A.; Gonzalez-Mariscal, G.; Boiti, C. Expression of the cannabinoid receptor type 1 in the pituitary of rabbits and its role in the control of LH secretion. Domest. Anim. Endocrinol. 2013, 45, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, A.; Ekuta, J.E.; Onaivi, E.S. Neurobehavioral effects of anandamide and cannabinoid receptor gene expression in mice. Brain Res. Bull. 1998, 45, 67–74. [Google Scholar] [CrossRef]
- de Miguel, R.; Romero, J.; Muñoz, R.M.; García-Gil, L.; González, S.; Villanua, M.A.; Makriyannis, A.; Ramos, J.A.; Fernández-Ruiz, J.J. Effects of cannabinoids on prolactin and gonadotrophin secretion: Involvement of changes in hypothalamic gamma-aminobutyric acid (GABA) inputs. Biochem. Pharmacol. 1998, 56, 1331–1338. [Google Scholar] [CrossRef]
- Pertwee, R.G. Ligands that target cannabinoid receptors in the brain: From THC to anandamide and beyond. Addict. Biol. 2008, 13, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Mc Millen, C. The sheep—An ideal model for biomedical research? ANZCCART News 2001, 14, 1–4. [Google Scholar]



| GenBank Acc. No. | Gene | Amplicon Size [bp] | Forward/ Reverse | Sequence 5′→3′ | Reference |
|---|---|---|---|---|---|
| NM_001034034 | GAPDH glyceraldehyde-3-phosphate dehydrogenase | 134 | forward | AGAAGGCTGGGGCTCACT | [29] |
| reverse | GGCATTGCTGACAATCTTGA | ||||
| U39357 | ACTB beta actin | 168 | forward | CTTCCTTCCTGGGCATGG | [29] |
| reverse | GGGCAGTGATCTCTTTCTGC | ||||
| BC108088.1 | HDAC1 histone deacetylase1 | 115 | forward | CTGGGGACCTACGGGATATT | [29] |
| reverse | GACATGACCGGCTTGAAAAT | ||||
| NM_001009397 | GnRHR gonadotropin-releasing hormone receptor | 150 | forward | TCTTTGCTGGACCACAGTTAT | [30] |
| reverse | GGCAGCTGAAGGTGAAAAAG | ||||
| U02517 | GnRH gonadotropin-releasing hormone | 123 | forward | GCCCTGGAGGAAAGAGAAAT | [30] |
| reverse | GAGGAGAATGGGACTGGTGA | ||||
| X52488 | LHB luteinizing hormone beta-subunit | 184 | forward | AGATGCTCCAGGGACTGCT | [30] |
| reverse | TGCTTCATGCTGAGGCAGTA | ||||
| X15493 | FSHB follicle stimulating hormone beta-subunit | 131 | forward | TATTGCTACACCCGGGACTT | [30] |
| reverse | TACAGGGAGTCTGCATGGTG | ||||
| NM_001242341 | CNR1 cannabinoid receptor 1 | 104 | forward | GAGGACCGGGGGATGC | Originally designed |
| reverse | CCGTCGAGGATGGACTTC |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomaszewska-Zaremba, D.; Wojtulewicz, K.; Paczesna, K.; Tomczyk, M.; Biernacka, K.; Bochenek, J.; Herman, A.P. The Influence of Anandamide on the Anterior Pituitary Hormone Secretion in Ewes—Ex Vivo Study. Animals 2020, 10, 706. https://doi.org/10.3390/ani10040706
Tomaszewska-Zaremba D, Wojtulewicz K, Paczesna K, Tomczyk M, Biernacka K, Bochenek J, Herman AP. The Influence of Anandamide on the Anterior Pituitary Hormone Secretion in Ewes—Ex Vivo Study. Animals. 2020; 10(4):706. https://doi.org/10.3390/ani10040706
Chicago/Turabian StyleTomaszewska-Zaremba, Dorota, Karolina Wojtulewicz, Kamila Paczesna, Monika Tomczyk, Katarzyna Biernacka, Joanna Bochenek, and Andrzej Przemysław Herman. 2020. "The Influence of Anandamide on the Anterior Pituitary Hormone Secretion in Ewes—Ex Vivo Study" Animals 10, no. 4: 706. https://doi.org/10.3390/ani10040706
APA StyleTomaszewska-Zaremba, D., Wojtulewicz, K., Paczesna, K., Tomczyk, M., Biernacka, K., Bochenek, J., & Herman, A. P. (2020). The Influence of Anandamide on the Anterior Pituitary Hormone Secretion in Ewes—Ex Vivo Study. Animals, 10(4), 706. https://doi.org/10.3390/ani10040706





