Feather Corticosterone Measurements of Greater Flamingos Living under Different Forms of Flight Restraint
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Zoological Institutions and Behavioral Observations
2.3. Feather Collection
2.4. Corticosterone Extraction and Measurement
2.5. Statistical Analysis
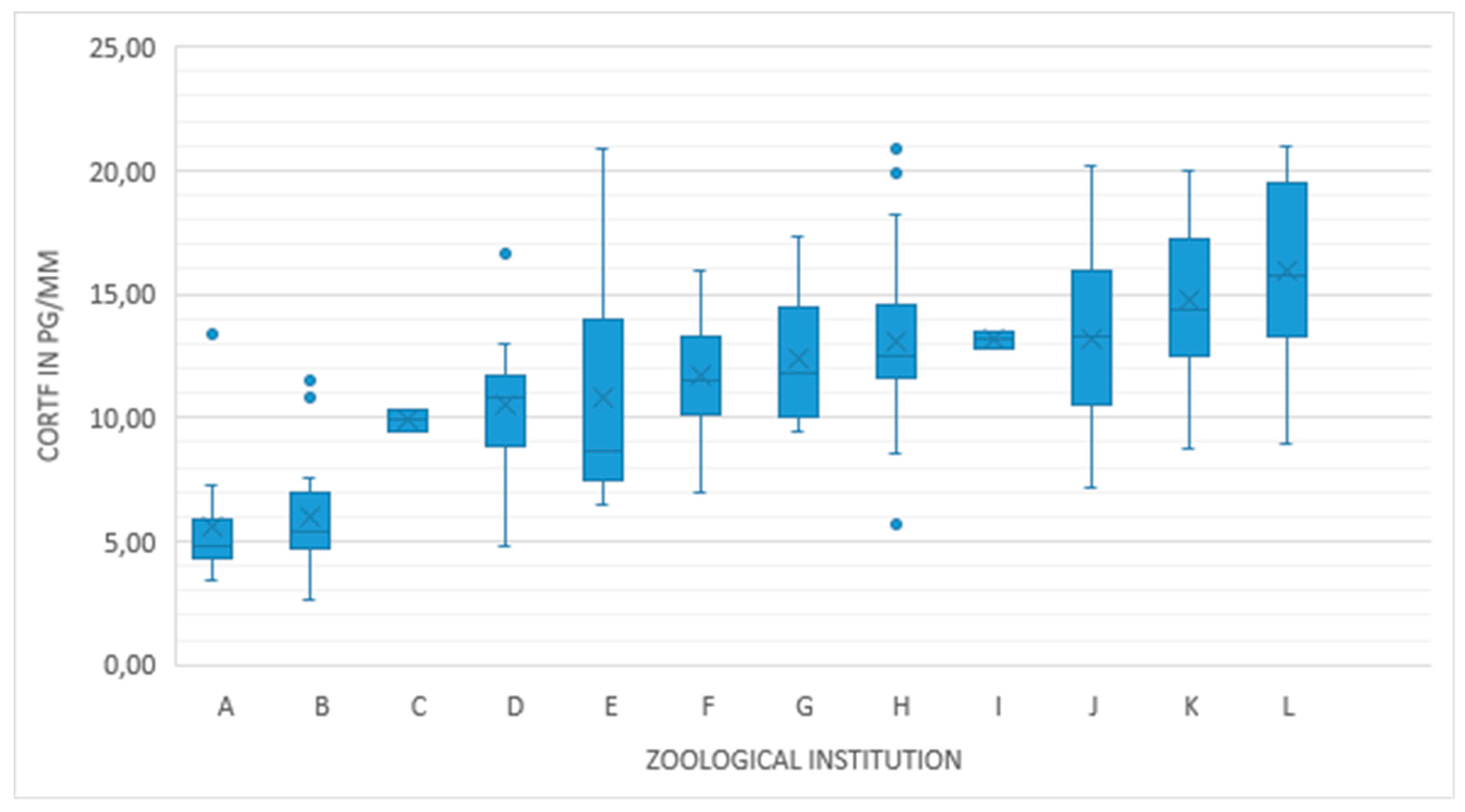
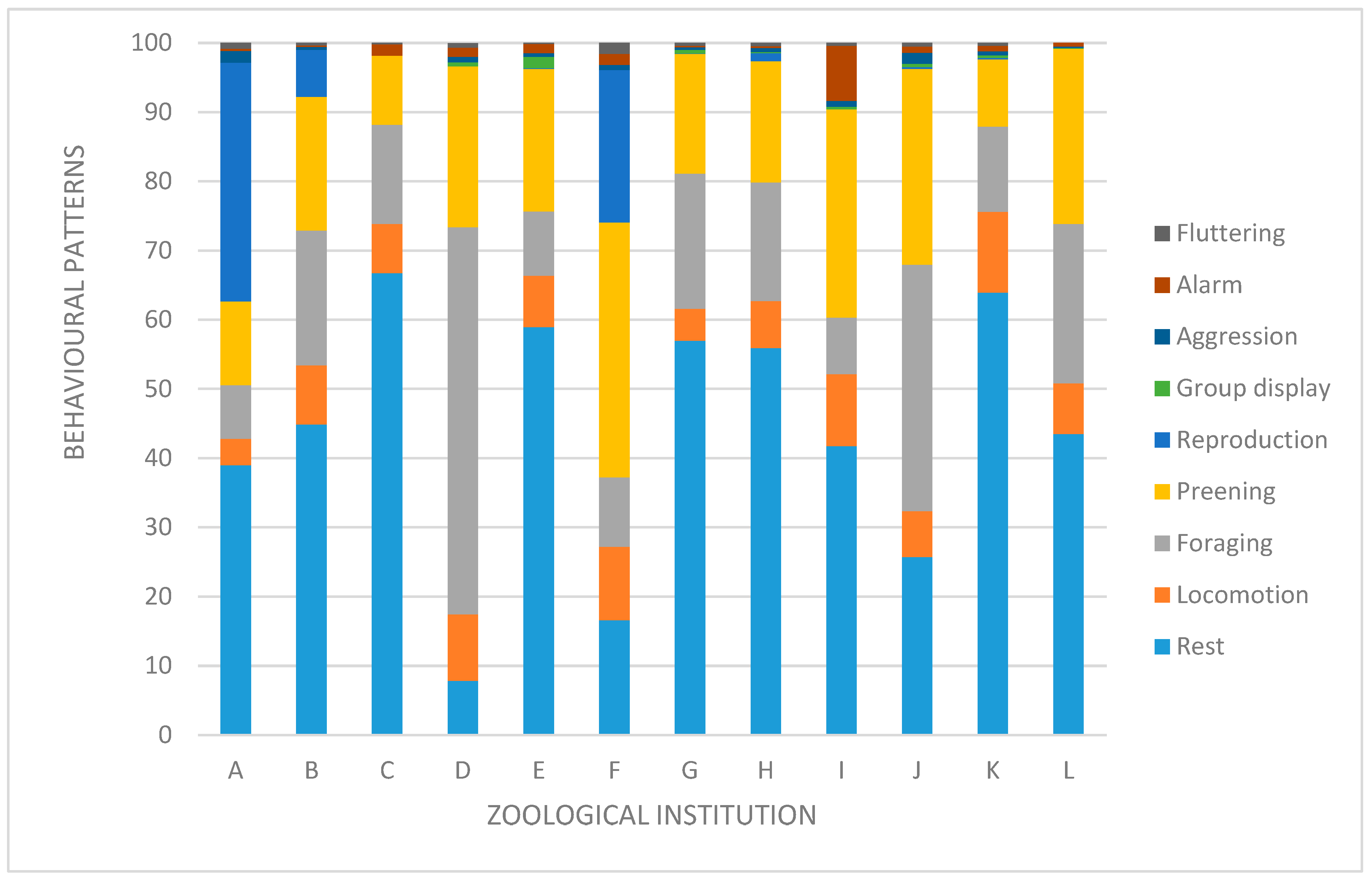
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Paul-Murphy, J. Foundations in Avian Welfare. In Current Therapy in Avian Medicine and Surgery; Speer, B.L., Ed.; Elsevier: St. Louis, MO, USA, 2016. [Google Scholar]
- Reese, L.; Ladwig-Wiegard, M.; Fersen, L.; von Haase, G.; Will, H.; Merle, R.; Encke, D.; Maegdefrau, H.; Baumgartner, K.; Thöne-Reineke, C. Deflighting zoo birds and its welfare considerations. Anim. Welfare 2020, 29, 69–80. [Google Scholar] [CrossRef]
- Beckmann, M.; Thal, D. Flugunfähigkeitsbewirkende Behandlungen von Zoovögeln—Rechtliche Rahmenbedingungen des Tier- und Naturschutzrechts. Natur und Recht 2017, 39, 154–163. [Google Scholar] [CrossRef]
- Maisack, C.; Schmidt, T. Zum Flugunfähigmachen von Vögeln in Zoos und privaten Geflügelhaltungen. Natur und Recht 2017, 39, 734–741. [Google Scholar] [CrossRef]
- Bračko, A.; King, C.E. Advantages of aviaries and the Aviary Database Project: A new approach to an old housing option for birds. Int. Zoo Yearb. 2014, 48, 166–183. [Google Scholar] [CrossRef]
- Klausen, B. A mixed-species exhibit for African water birds (including pelicans, flamingos, spoonbills and storks) at Odense Zoo, Denmark: breeding success, animal welfare and education. Int. Zoo Yearb. 2014, 48, 61–68. [Google Scholar] [CrossRef]
- Schmidt, T.; Jäger, C. Das Flugunfähigmachen von Vögeln in zoologischen Einrichtungen unter Tierschutzgesichtspunkten. ATD 2015, 163–167. [Google Scholar]
- Tyson, E. For an End to Pinioning: The Case Against the Legal Mutilation of Birds in Captivity. J. Anim. Ethics 2014, 4, 1–4. [Google Scholar] [CrossRef]
- Captive Animals’ Protection Society (CAPS) Mutilated for your viewing pleasure-Pinioning birds in English zoos 2013. Available online: https://www.yumpu.com/en/document/view/11988590/mutilated-captive-animals-protection-society (accessed on 14 February 2020).
- Dekker, J. (Van Hall Larenstein, University of Applied Sciences, Leeuwarden and Velp, Netherlands). Future Prospects of Commonly Kept Pinioned Bird Species at EAZA Zoos. Personal communication, 2016.
- Dollinger, P.; Pagel, T.; Baumgartner, K.; Encke, D.; Engel, H.; Filz, A. Flugunfähigmachen von Vögeln—Für und Wider. Zool. Gart. 2014, 82, 293–339. [Google Scholar] [CrossRef]
- Hesterman, H.; Gregory, N.G.; Boardman, W.S.J. Deflighting Procedures and their Welfare Implications in Captive Birds. Anim Welfare 2001, 10, 405–419. [Google Scholar]
- Smith, K. Housing and Enclosure Requirements. In Flamingo Husbrandry Guidelines—A Joint Effort of the AZA and EAZA in Cooperation with WWT; Brown, C., King, C., Eds.; Dallas Zoo: Dallas, TX, USA, 2005. [Google Scholar]
- Carr, N. An analysis of zoo visitors’ favourite and least favourite animals. Tour. Manag. Perspect. 2016, 20, 70–76. [Google Scholar] [CrossRef]
- Brown, C.; King, C. Flamingo Husbandry Guidelines—A Joint Effort of the AZA and EAZA in Cooperation with WWT; Dallas Zoo: Dallas, TX, USA, 2005. [Google Scholar]
- Johann, A. Die neue Flamingo-Anlage im NaturZoo Rheine. Zool. Gart. 2007, 77, 1–14. [Google Scholar] [CrossRef]
- Studer-Thiersch, A. Behavioral Demands on a New Exhibit for Greater Flamingos at the Basle Zoo, Switzerland. Waterbirds 2000, 23, 185–192. [Google Scholar] [CrossRef]
- Studer-Thiersch, A. What 19 Years of Observation on Captive Greater Flamingos Suggests about Adaptations to Breeding under Irregular Conditions. Waterbirds 2000, 23, 150–159. [Google Scholar] [CrossRef]
- Rose, P.E.; Croft, D.P.; Lee, R. A review of captive flamingo (Phoenicopteridae) welfare: a synthesis of current knowledge and future directions. Int. Zoo Yearb. 2014, 48, 139–155. [Google Scholar] [CrossRef]
- Rose, P.E.; Brereton, J.E.; Croft, D.P. Measuring welfare in captive flamingos: Activity patterns and exhibit usage in zoo-housed birds. Appl. Anim. Behav. Sci. 2018, 205, 115–125. [Google Scholar] [CrossRef]
- Rose, P.E.; Croft, D.P. Evidence of directed interactions between individuals in captive flamingo flocks. Wildfowl 2015, 65, 121–132. [Google Scholar]
- Richardson, T.; Pickering, S.W.; Shannon, P. Natural History. In Flamingo Husbrandry Guidelines—A Joint Effort of the AZA and EAZA in Cooperation with WWT; Brown, C., King, C., Eds.; Dallas Zoo: Dallas, TX, USA, 2005. [Google Scholar]
- Studer-Thiersch, A. Personal communication, Basle, Switzerland. 2016.
- Sapolsky, R.M.; Romero, L.M.; Munck, A.U. How Do Glucocorticoids Influence Stress Responses? Integrating Permissive, Suppressive, Stimulatory, and Preparative Actions. Endocr. Rev. 2000, 21, 55–89. [Google Scholar]
- Vera, F.; Zenuto, R.; Antenucci, C.D. Expanding the actions of cortisol and corticosterone in wild vertebrates: A necessary step to overcome the emerging challenges. Gen. Comp. Endocrinol. 2017, 246, 337–353. [Google Scholar] [CrossRef]
- Cockrem, J.F. Individual variation in glucocorticoid stress responses in animals. Gen. Comp. Endocrinol. 2013, 181, 45–58. [Google Scholar] [CrossRef]
- Dickens, M.J.; Romero, L.M. A consensus endocrine profile for chronically stressed wild animals does not exist. Gen. Comp. Endocrinol. 2013, 191, 177–189. [Google Scholar] [CrossRef]
- McEwen, B.S.; Wingfield, J.C. The concept of allostasis in biology and biomedicine. Horm. Behav. 2003, 43, 2–15. [Google Scholar] [CrossRef]
- Sterling, P.; Eyer, J. Allostasis: A new paradigm to explain arousal pathology. In Handbook of Life Stress, Cognition and Health; John Wiley & Sons: Oxford, UK, 1988; pp. 629–649. [Google Scholar]
- Romero, L.M.; Dickens, M.J.; Cyr, N.E. The reactive scope model—A new model integrating homeostasis, allostasis, and stress. Horm. Behav. 2009, 55, 375–389. [Google Scholar] [CrossRef] [PubMed]
- Romero, L.M.; Fairhurst, G.D. Measuring corticosterone in feathers: Strengths, limitations, and suggestions for the future. Comp. Biochem. Phys. A 2016, 202, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Bortolotti, G.R.; Marchant, T.A.; Blas, J.; German, T. Corticosterone in feathers is a long-term, integrated measure of avian stress physiology. Funct. Ecol. 2008, 22, 494–500. [Google Scholar] [CrossRef]
- Fairhurst, G.D.; Frey, M.D.; Reichert, J.F.; Szelest, I.; Kelly, D.M.; Bortolotti, G.R. Does Environmental Enrichment Reduce Stress? An Integrated Measure of Corticosterone from Feathers Provides a Novel Perspective. PLoS ONE 2011, 6, e17663. [Google Scholar] [CrossRef]
- Lattin, C.R.; Reed, J.M.; DesRochers, D.W.; Romero, L.M. Elevated corticosterone in feathers correlates with corticosterone-induced decreased feather quality: A validation study. J. Avian Biol. 2011, 42, 247–252. [Google Scholar] [CrossRef]
- Kennedy, E.A.; Lattin, C.R.; Romero, L.M.; Dearborn, D.C. Feather coloration in museum specimens is related to feather corticosterone. Behav. Ecol. Sociobiol. 2013, 67, 341–348. [Google Scholar] [CrossRef]
- Monclús, L.; Carbajal, A.; Tallo-Parra, O.; Sabés-Alsina, M.; Darwich, L.; Molina-López, R.A.; Lopez-Bejar, M. Relationship between feather corticosterone and subsequent health status and survival in wild Eurasian Sparrowhawk. J. Ornithol. 2017, 158, 773–783. [Google Scholar] [CrossRef]
- Bortolotti, G.R.; Marchant, T.; Blas, J.; Cabezas, S. Tracking stress: Localisation, deposition and stability of corticosterone in feathers. J. Exp. Biol. 2009, 212, 1477–1482. [Google Scholar] [CrossRef]
- Bortolotti, G.R. Flaws and pitfalls in the chemical analysis of feathers: Bad news–good news for avian chemoecology and toxicology. Ecol. Appl. 2010, 20, 1766–1774. [Google Scholar] [CrossRef]
- Monclús, L.; Ballesteros-Cano, R.; De La Puente, J.; Lacorte, S.; Lopez-Bejar, M. Influence of persistent organic pollutants on the endocrine stress response in free-living and captive red kites (Milvus milvus). Environ. Pollut. 2018, 242, 329–337. [Google Scholar] [CrossRef] [PubMed]
- Fairhurst, G.D.; Marchant, T.A.; Soos, C.; Machin, K.L.; Clark, R.G. Experimental relationships between levels of corticosterone in plasma and feathers in a free-living bird. J. Exp. Biol. 2013, 216, 4071–4081. [Google Scholar] [CrossRef]
- Jenni-Eiermann, S.; Helfenstein, F.; Vallat, A.; Glauser, G.; Jenni, L. Corticosterone: effects on feather quality and deposition into feathers. Methods Ecol. Evol. 2014, 6, 237–246. [Google Scholar] [CrossRef]
- Kouwenberg, A.-L.; Hipfner, J.M.; McKay, D.W.; Storey, A.E. Corticosterone and stable isotopes in feathers predict egg size in Atlantic Puffins Fratercula arctica. Ibis 2013, 155, 413–418. [Google Scholar] [CrossRef]
- Monclús, L.; Lopez-Bejar, M.; De la Puente, J.; Covaci, A.; Jaspers, V.L.B. First evaluation of the use of down feathers for monitoring persistent organic pollutants and organophosphate ester flame retardants: A pilot study using nestlings of the endangered cinereous vulture (Aegypius monachus). Environ. Pollut. 2018, 238, 413–420. [Google Scholar] [CrossRef]
- Koren, L.; Nakagawa, S.; Burke, T.; Soma, K.K.; Wynne-Edwards, K.E.; Geffen, E. Non-breeding feather concentrations of testosterone, corticosterone and cortisol are associated with subsequent survival in wild house sparrows. Proc. R. Soc. B 2012, 279, 1560–1566. [Google Scholar] [CrossRef]
- Sild, E.; Meitern, R.; Männiste, M.; Karu, U.; Hõrak, P. High feather corticosterone indicates better coccidian infection resistance in greenfinches. Gen. Comp. Endocrin. 2014, 204, 203–210. [Google Scholar] [CrossRef]
- Will, A.P.; Suzuki, Y.; Elliott, K.H.; Hatch, S.A.; Watanuki, Y.; Kitaysky, A.S. Feather corticosterone reveals developmental stress in seabirds. J. Exp. Biol. 2014, 217, 2371–2376. [Google Scholar] [CrossRef]
- Patterson, A.G.L.; Kitaysky, A.S.; Lyons, D.E.; Roby, D.D. Nutritional stress affects corticosterone deposition in feathers of Caspian tern chicks. J. Avian Biol. 2015, 46, 18–24. [Google Scholar] [CrossRef]
- Sileo, L.; Tuite, C.H.; Hopcraft, J.B.D. Plumages and primary moult in Lesser Flamingos. Wildfowl 1977, 28, 4. [Google Scholar]
- Johnson, A.; Cézilly, F.; Boy, V. Plumage development and maturation in the Greater Flamingo, Phoenicopterus ruber roseus. Ardea 1993, 81, 25–33. [Google Scholar]
- Shannon, P. Plumages and Molt Patterns in Captive Caribbean Flamingos. Waterbirds 2000, 23, 160. [Google Scholar] [CrossRef]
- Altmann, J. Observational Study of Behavior: Sampling Methods. Behaviour 1974, 49, 227–266. [Google Scholar] [CrossRef] [PubMed]
- Arengo, F.; Baldassarre, G.A. Effects of Food Density on the Behavior and Distribution of Nonbreeding American Flamingos in Yucatan, Mexico. Condor 1995, 97, 325–334. [Google Scholar] [CrossRef]
- Bouaguel, L.; Saheb, M.; Bensaci, E.; Bougoudjil, S.; Bouslama, Z.; Houhamdi, M. Status and Diurnal Behavior of the Greater Flamingo Phoenicopterus Roseus in Algerian Eastern High Plains. Ann. Biol. Res. 2013, 4, 232–237. [Google Scholar]
- Espino-Barros, R.; Baldassarre, G.A. Numbers, Migration Chronology, and Activity Patterns of Nonbreeding Caribbean Flamingos in Yucatan, Mexico. Condor 1989, 91, 592–597. [Google Scholar] [CrossRef]
- Khaleghizadeh, A. Diurnal Behaviour of the Greater Flamingo Phoenicopterus roseus during a Tidal Cycle on the Bandar Abbas Coast, Persian Gulf. Ann. Biol. Res. 2010, 6, 232–237. [Google Scholar]
- Studer-Thiersch, A. Die Balz der Flamingogattung Phoenicopterus, unter besonderer Berücksichtigung von Ph. ruber roseus. Z. Tierpsychol. 1974, 36, 212–266. [Google Scholar] [CrossRef]
- Schmitz, R.A.; Baldassarre, G.A. Contest Asymmetry and Multiple Bird Conflicts during Foraging among Nonbreeding American Flamingos in Yucatan, Mexico. Condor 1992, 94, 254–259. [Google Scholar] [CrossRef]
- Carbajal, A.; Tallo-Parra, O.; Sabes-Alsina, M.; Mular, I.; Lopez-Bejar, M. Feather corticosterone evaluated by ELISA in broilers: A potential tool to evaluate broiler welfare. Poult. Sci. 2014, 93, 2884–2886. [Google Scholar] [CrossRef]
- Béziers, P.; San-Jose, L.M.; Almasi, B.; Jenni, L.; Roulin, A. Baseline and stress-induced corticosterone levels are heritable and genetically correlated in a barn owl population. Heredity 2019, 123, 337–348. [Google Scholar] [CrossRef]
- Lattin, C.R.; Breuner, C.W.; Michael Romero, L. Does corticosterone regulate the onset of breeding in free-living birds? The CORT-Flexibility Hypothesis and six potential mechanisms for priming corticosteroid function. Horm. Behav. 2016, 78, 107–120. [Google Scholar] [CrossRef]
- Love, O.P.; Breuner, C.W.; Vézina, F.; Williams, T.D. Mediation of a corticosterone-induced reproductive conflict. Horm. Behav. 2004, 46, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Walker, B.G.; Boersma, P.D.; Wingfield, J.C. Habituation of Adult Magellanic Penguins to Human Visitation as Expressed through Behavior and Corticosterone Secretion. Conserv. Biol. 2006, 20, 146–154. [Google Scholar] [CrossRef]
- Rich, E.L.; Romero, L.M. Exposure to chronic stress downregulates corticosterone responses to acute stressors. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2005, 288, R1628–R1636. [Google Scholar] [CrossRef] [PubMed]
- Rees, A.; Harvey, S.; Phillips, J.G. Habituation of the corticosterone response of ducks (Anas platyrhynchos) to daily treadmill exercise. Gen. Comp. Endocrin. 1983, 49, 485–489. [Google Scholar] [CrossRef]
| Zoo | Total Group Size and Classification | Reproduction | Socialization | Sampled Animals + Flight Status |
|---|---|---|---|---|
| A | 64 (II) | B | 1 | 13 (2p + 11r) |
| B | 67 (II) | B | 2 | 19 (10p + 9r) |
| C | 27 (I) | NB | 2 | 9 (9p) |
| D | 22 (I) | NB | 1 | 15 (5a + 10p) |
| E | 15 (I) | NB | 3 | 15 (15p) |
| F | 43 (II) | B | 1 | 10 (10p) |
| G | 2 (I) | NB | 2 | 2 (2p) |
| H | 46 (II) | B | 1 | 37 (27a + 10p) |
| I | 2 (I) | NB | 2 | 2 (2p) |
| J | 7 (I) | B | 2 | 7 (4p + 3r) |
| K | 36 (I) | NB | 3 | 13 (3p + 10r) |
| L | 12 (I) | NB | 3 | 9 (9p) |
| Total | 151 (32a + 86p + 33r) |
| Influencing factor | Estimate logCORTf in pg/mm | p-Value | 95% Confidence Interval | |
|---|---|---|---|---|
| Lower Limit | Upper Limit | |||
| Intercept | 1.067 | <0.001 | 0.929 | 1.205 |
| Female | 0.090 | 0.200 | 0.015 | 0.166 |
| Male | 0 | 0.257 | - | - |
| Airworthy | 0.040 | 0.441 | −0.063 | 0.113 |
| Pinioned | −0.004 | 0.904 | −0.077 | 0.068 |
| Wing clipped | 0 | 0.502 | - | - |
| Socialization = 1 | −0.133 | 0.176 | −0.333 | 0.067 |
| Socialization = 2 | −0.086 | 0.350 | −0.275 | 0.103 |
| Socialization = 3 | 0 | 0.136 | - | - |
| Sex = female * Socialization = 1 | −0.090 | 0.075 | −0.189 | 0.009 |
| Sex = female * Socialization = 2 | 0.107 | 0.059 | −0.217 | 0.004 |
| ex = female * Socialization = 3 | 0 | - | - | - |
| Sex = male * Socialization = 1 | 0 | - | - | - |
| Sex = male * Socialization = 2 | 0 | - | - | - |
| Sex = male * Socialization = 3 | 0 | 0.109 | - | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reese, L.; Baumgartner, K.; von Fersen, L.; Merle, R.; Ladwig-Wiegard, M.; Will, H.; Haase, G.; Tallo-Parra, O.; Carbajal, A.; Lopez-Bejar, M.; et al. Feather Corticosterone Measurements of Greater Flamingos Living under Different Forms of Flight Restraint. Animals 2020, 10, 605. https://doi.org/10.3390/ani10040605
Reese L, Baumgartner K, von Fersen L, Merle R, Ladwig-Wiegard M, Will H, Haase G, Tallo-Parra O, Carbajal A, Lopez-Bejar M, et al. Feather Corticosterone Measurements of Greater Flamingos Living under Different Forms of Flight Restraint. Animals. 2020; 10(4):605. https://doi.org/10.3390/ani10040605
Chicago/Turabian StyleReese, Lukas, Katrin Baumgartner, Lorenzo von Fersen, Roswitha Merle, Mechthild Ladwig-Wiegard, Hermann Will, Gudrun Haase, Oriol Tallo-Parra, Annaïs Carbajal, Manel Lopez-Bejar, and et al. 2020. "Feather Corticosterone Measurements of Greater Flamingos Living under Different Forms of Flight Restraint" Animals 10, no. 4: 605. https://doi.org/10.3390/ani10040605
APA StyleReese, L., Baumgartner, K., von Fersen, L., Merle, R., Ladwig-Wiegard, M., Will, H., Haase, G., Tallo-Parra, O., Carbajal, A., Lopez-Bejar, M., & Thöne-Reineke, C. (2020). Feather Corticosterone Measurements of Greater Flamingos Living under Different Forms of Flight Restraint. Animals, 10(4), 605. https://doi.org/10.3390/ani10040605






