Proteomic Analysis of Fresh and Liquid-Stored Boar Spermatozoa
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Semen Samples and Analysis
2.2. The Extraction of Spermatozoa Proteins
Protein Quantification
2.3. Gel Electrophoresis
Image Scan and Data Analysis
2.4. In-Gel Enzymatic Digestion and Mass Fingerprinting
Matrix-Assisted Laser Desorption/Ionization Time-of-Flight Mass Spectrometry (MALDI-TOF/MS) Analysis
3. Results and Discussion
3.1. Sperm Parameters in Fresh and Liquid Stored Semen
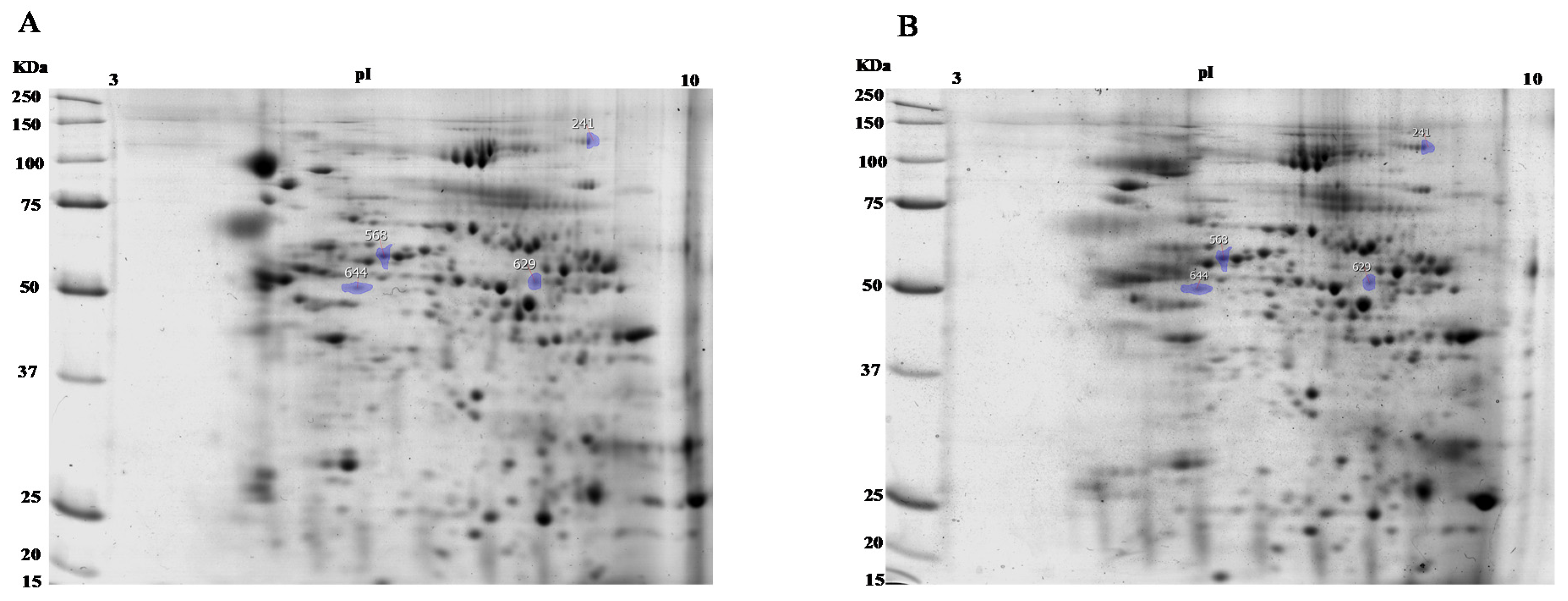
3.2. Two-Dimensional (2-D) Electrophoresis and Mass Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Estrada, E.; Rodriguez-Gil, J.E.; Roncha, L.G.; Balasch, S.; Bonet, S.; Yeste, M. Supplementing cryopreservation media with reduced glutathione increases fertility and prolifacy of sows inseminated with frozen-thawed boar semen. Andrology 2014, 2, 88–99. [Google Scholar] [CrossRef]
- Pinart, E.; Yeste, M.; Prieto-Martínez, N.; Reixach, J.; Bonet, S. Sperm quality and fertility of boar seminal doses after 2 days of storage: Does the type of extender really matter? Theriogenology 2015, 83, 1428–1437. [Google Scholar] [CrossRef] [PubMed]
- Waberski, D.; Henning, H.; Petrunkina, A.M. Assessment of storage effects in liquid preserved boar semen. Reprod. Domest. Anim. 2011, 46, 45–48. [Google Scholar] [CrossRef] [PubMed]
- Llavanera, M.; Delgado-Bermúdez, A.; Olives, S.; Mateo-Otero, Y.; Recuero, S.; Bonet, S.; Fernández-Fuertes, B.; Yeste, M.; Barranco, I. Glutathione S-Transferases play a crucial role in mitochondrial function, plasma membrane stability and oxidative regulation of mammalian sperm. Antioxidants 2020, 9, 100. [Google Scholar] [CrossRef] [PubMed]
- Bonet, S.; Briz, M.D.; Yeste, M. A proper assessment of boar sperm function may not only require conventional analyses but also others focused on molecular markers of epididymal maturation. Reprod. Domest. Anim. 2012, 47, 52–64. [Google Scholar] [CrossRef]
- Kummer, A.B.; Gaggini, T.S.; Bernardi, M.L.; McManus, C.; Gonçales, E.M.; Wentz, I.; Bortolozzo, F.P. Multivariate analyses for determining the association of field porcine fertility with sperm motion traits analysed by computer-assisted semen analysis and with sperm morphology. Reprod. Domest. Anim. 2013, 48, 747–754. [Google Scholar] [CrossRef]
- Pinart, E.; Yeste, M.; Puigmulé, M.; Barrera, X.; Bonet, S. Acrosin activity is a suitable indicator of boar semen preservation at 17 °C when increasing environmental temperature and radiation. Theriogenology 2013, 80, 234–247. [Google Scholar] [CrossRef]
- Silva, C.G.; Cunha, E.R.; Blume, G.R.; Malaquias, J.V.; Báo, S.N.; Martins, C.F. Cryopreservation of boar sperm comparing different cryoprotectants associated in media based on powdered cococnut water, lactose and trehalose. Cryobiology 2015, 70, 90–94. [Google Scholar] [CrossRef]
- De Lazari, F.L.; Sontag, E.R.; Schneider, A.; Araripe Moura, A.A.; Vasconcelos, F.R.; Nagano, C.S.; Dalberto, P.F.; Bizarro, C.V.; Mattos, R.C.; Mascarenhas Jobim, M.A.; et al. Proteomic identification of boar seminal plasma proteins related to sperm resistence to cooling at 17 °C. Theriogenology 2019. [Google Scholar] [CrossRef]
- Parrilla, I.; Perez-Patiño, C.; Li, J.; Barranco, I.; Padilla, L.; Rodriguez-Martinez, H.; Martinez, E.A.; Roca, J. Boar semen proteomics and sperm preservation. Theriogenology 2019, 137, 23–29. [Google Scholar] [CrossRef]
- Weber, A.; Argenti, L.E.; Binato de Souza, A.P.; Santi, L.; Beys-da-Silva, W.O.; Yates, J.R.; Bustamante-Filho, I.C. Ready for the journey: A comparative proteome profiling of porcine cauda epididymal fluid and spermatozoa. Cell Tissue Res. 2020, 379, 389–405. [Google Scholar] [CrossRef] [PubMed]
- Pintado, B.; de la Fuente, J.; Roldan, E.R. Permeability of boar and bull spermatozoa to the nucleic acid stains propidium iodide or Hoechst 33258, or to eosin: Accuracy in the assessment of cell viability. J. Reprod. Fertil. 2000, 118, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Kovács, A.; Foote, R.H. Viability and acrosome staining of bull, boar and rabbit spermatozoa. Biotech. Histochem. 1992, 67, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Mattioli, M.; Barboni, B.; Lucidi, P.; Seren, E. Identification of capacitation in boar spermatozoa by chlortetracycline staining. Theriogenology 1996, 45, 373–381. [Google Scholar] [CrossRef]
- Piras, C.; Soggiou, A.; Bonizzi, L.; Greco, V.; Ricchi, M.; Arrigoni, N.; Bassols, A.; Urbani, A.; Roncada, P. Identification of immunoreactive proteins of Mycobacterium avium subsp. paratuberculosis. Proteomics 2015, 15, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Kwon, W.S.; Rahman, M.S.; Lee, J.S.; Yoon, S.J.; Park, Y.J.; Pang, M.G. Discovery of predictive biomarkers for litter size in boar spermatozoa. Mol. Cell. Proteom. 2015, 14, 1230–1240. [Google Scholar] [CrossRef]
- Feitsma, H.; Broekhuijse, M.L.; Gadella, B.M. Do CASA systems satisfy consumers demands? A critical analysis. Reprod. Domest. Anim. 2011, 46, 49–51. [Google Scholar] [CrossRef]
- Broekhuijse, M.L.; Soštarić, E.; Feitsma, H.; Gadella, B.M. The value of microscopic semen motility assessment at collection for a commercial artificial insemination center, a retrospective study on factors explaining variation in pig fertility. Theriogenology 2012, 77, 1466–1479. [Google Scholar] [CrossRef]
- Henning, H.; Petrunkina, A.M.; Harrison, R.A.P.; Waberski, D. Bivalent response to long-term storage in liquid-preserved boar semen: A flow cytometric analysis. Cytom. Part A 2012, 81, 576–587. [Google Scholar] [CrossRef]
- Leahy, T.; Gadella, B.M. Capacitation and capacitation-like sperm surface changes induced by handling boar semen. Reprod. Domest. Anim. 2011, 46, 7–13. [Google Scholar] [CrossRef]
- Ren, Z.Q.; Wang, Y.; Xu, Y.J.; Wang, L.J.; Lei, M.G.; Zuo, B.; Li, F.E.; Xu, D.Q.; Zheng, R.; Deng, C.Y.; et al. Identification of a differentially expressed gene, ACL, between Meishan x Large White and Large White x Meishan F1 hybrids and their parents. Genet. Sel. Evol. 2008, 40, 625–637. [Google Scholar] [CrossRef] [PubMed]
- Cappello, A.R.; Guido, C.; Santoro, A.; Santoro, M.; Capobianco, L.; Montanaro, D.; Madeo, M.; Andò, S.; Dolce, V.; Aquila, S. The mitochondrial citrate carrier (CIC) is present and regulates insulin secretion by human male gamete. Endocrinology 2012, 153, 1743–1754. [Google Scholar] [CrossRef] [PubMed]
- Novak, S.; Smith, T.A.; Paradis, F.; Burwash, L.; Dyck, M.K.; Foxcroft, G.R.; Dixon, W.T. Biomarkers of in vivo fertility in sperm and seminal plasma of fertile stallions. Theriogenology 2010, 74, 956–967. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, A.K.; Rama Sastry, B.V. Adenosine 5’-triphosphate-citrate lyase activity in mammalian spermatozoa: A new radiometric method and characterization of the enzyme in spermatozoa. Biochem. Pharmacol. 1980, 29, 2589–2594. [Google Scholar] [CrossRef]
- Chen, X.; Zhu, H.; Hu, C.; Hao, H.S.; Zhang, J.; Li, K.; Zhao, X.; Qin, T.; Zhao, K.; Zhu, H.; et al. Identification of differentially expressed proteins between fresh and frozen-thawed boar spermatozoa by iTRAQ-coupled 2D LC-MS/MS. Reproduction 2014, 147, 321–330. [Google Scholar] [CrossRef]
- Dun, M.D.; Aitken, R.J.; Nixon, B. The role of molecular chaperones in spermatogenesis and the post-testicular maturation of mammalian spermatozoa. Hum. Reprod. Update 2012, 18, 420–435. [Google Scholar] [CrossRef]
- Byrne, K.; Leahy, T.; McCulloch, R.; Colgrave, M.L.; Holland, M.K. Comprehensive mapping of the bull sperm surface proteome. Proteomics 2012, 12, 3559–3579. [Google Scholar] [CrossRef]
- D’Amours, O.; Frenette, G.; Fortier, M.; Leclerc, P.; Sullivan, R. Proteomic comparison of detergent extracted sperm proteins from bulls with different fertility indexes. Reproduction 2010, 139, 545–556. [Google Scholar] [CrossRef]
- Wang, L.; Beserra, C.; Garbers, D.L. A novel aminophospholipid transporter exclusively expressed in spermatozoa is required for membrane lipid asymmetry and normal fertilization. Dev. Biol. 2004, 267, 203–215. [Google Scholar] [CrossRef]
- Davalieva, K.; Kiprijanovska, S.; Noveski, P.; Plaseski, T.; Kocevska, B.; Broussard, C.; Plaseska-Karanfilska, D. Proteomic analysis of seminal plasma in men with different spermatogenic impairment. Andrologia 2012, 44, 256–264. [Google Scholar] [CrossRef]
- Bogle, O.A.; Kumar, K.; Attardo-Parrinello, C.; Lewis, S.E.M.; Estanyol, J.M.; Ballescà, J.L.; Oliva, R. Identification of protein changes in human spermatozoa throughout the cryopreservation process. Andrology 2017, 5, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Feugang, J.M.; Liao, S.F.; Willard, S.T.; Ryan, P.L. In-depth proteomic analysis of boar spermatozoa through shotgun and gel-based methods. BMC Genom. 2018, 19, 62. [Google Scholar] [CrossRef] [PubMed]
| Semen Trait | Fresh Semen | Stored Semen | P-Value |
|---|---|---|---|
| motility (%) | 85.7 ± 4.3 | 70.7 ± 6.7 | P < 0.05 |
| progressive motility (%) | 49.6 ± 8.7 | 35.4 ± 3.7 | P < 0.05 |
| normal morphology (%) | 76.4 ± 8.7 | 64.6 ± 10.3 | P < 0.05 |
| viability (%) | 92.4 ± 4.6 | 70.2 ± 10.2 | P < 0.05 |
| non-capacitated spermatozoa (%) | 83.9 ± 6.0 | 60.2 ± 8.6 | P < 0.05 |
| acrosomal reacted spermatozoa (%) | 3.6 ± 2.0 | 14.3 ± 6.0 | P < 0.05 |
| Spot No. | Paired t-Test (P) | Fold | Average Normalised Volumes | |
|---|---|---|---|---|
| Fresh Semen | Stored Semen | |||
| 568 | 0.019 | 1.3 | 1.150 × 107 | 8.745 × 106 |
| 644 | 0.032 | 1.3 | 7.770 × 106 | 5.920 × 106 |
| 629 | 0.033 | 1.3 | 3.768 × 106 | 4.937 × 106 |
| 241 | 0.046 | 1.2 | 3.562 × 106 | 4.429 × 106 |
| Spot No. | Entry Number | Description | MS | MS/MS | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Theoretical Mr(kDa)/pI | Mascot Score (†) | % Seq. Coverage | Matched Peptides | Peptide Sequence | Mascot Score (‡) | m/z | z | |||
| 241 | gi/341942463 | ATP citrate lyase | 120.7/7.4 | 131 | 23.8 | 20 | WGDIEFPPPFGR | 52 | 1417.690 | 1 |
| 568 | gi/523580068 | chaperonin containing TCP1 Subunit 5 (epsilon) | 60.1/5.7 | 49 | 18 | 7 | WVGGPEIELIAIATGGR | 96 | 1738.949 | 1 |
| 629 | Not identified | |||||||||
| 644 | gi/343962597 | cytosolic non specific dipeptidase | 53.1/5.2 | 83.8 | 9.3 | 6 | ALQTVFGVEPDLTR | 75 | 1545.827 | 1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Premrov Bajuk, B.; Zrimšek, P.; Zakošek Pipan, M.; Tilocca, B.; Soggiu, A.; Bonizzi, L.; Roncada, P. Proteomic Analysis of Fresh and Liquid-Stored Boar Spermatozoa. Animals 2020, 10, 553. https://doi.org/10.3390/ani10040553
Premrov Bajuk B, Zrimšek P, Zakošek Pipan M, Tilocca B, Soggiu A, Bonizzi L, Roncada P. Proteomic Analysis of Fresh and Liquid-Stored Boar Spermatozoa. Animals. 2020; 10(4):553. https://doi.org/10.3390/ani10040553
Chicago/Turabian StylePremrov Bajuk, Blanka, Petra Zrimšek, Maja Zakošek Pipan, Bruno Tilocca, Alessio Soggiu, Luigi Bonizzi, and Paola Roncada. 2020. "Proteomic Analysis of Fresh and Liquid-Stored Boar Spermatozoa" Animals 10, no. 4: 553. https://doi.org/10.3390/ani10040553
APA StylePremrov Bajuk, B., Zrimšek, P., Zakošek Pipan, M., Tilocca, B., Soggiu, A., Bonizzi, L., & Roncada, P. (2020). Proteomic Analysis of Fresh and Liquid-Stored Boar Spermatozoa. Animals, 10(4), 553. https://doi.org/10.3390/ani10040553








