Effects of Different Stroking Styles on Behaviour and Cardiac Parameters in Heifers
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals, Housing and Management
2.2. Selection and Habituation
2.3. Experimental Design
2.4. Experimental Procedure
2.4.1. General Procedure
2.4.2. Stroking Styles
2.5. Behavioural Observations
2.6. Heart Rate Measurements
2.7. Statistical Analysis
2.7.1. Behavioural Data
2.7.2. Cardiac Data
3. Results
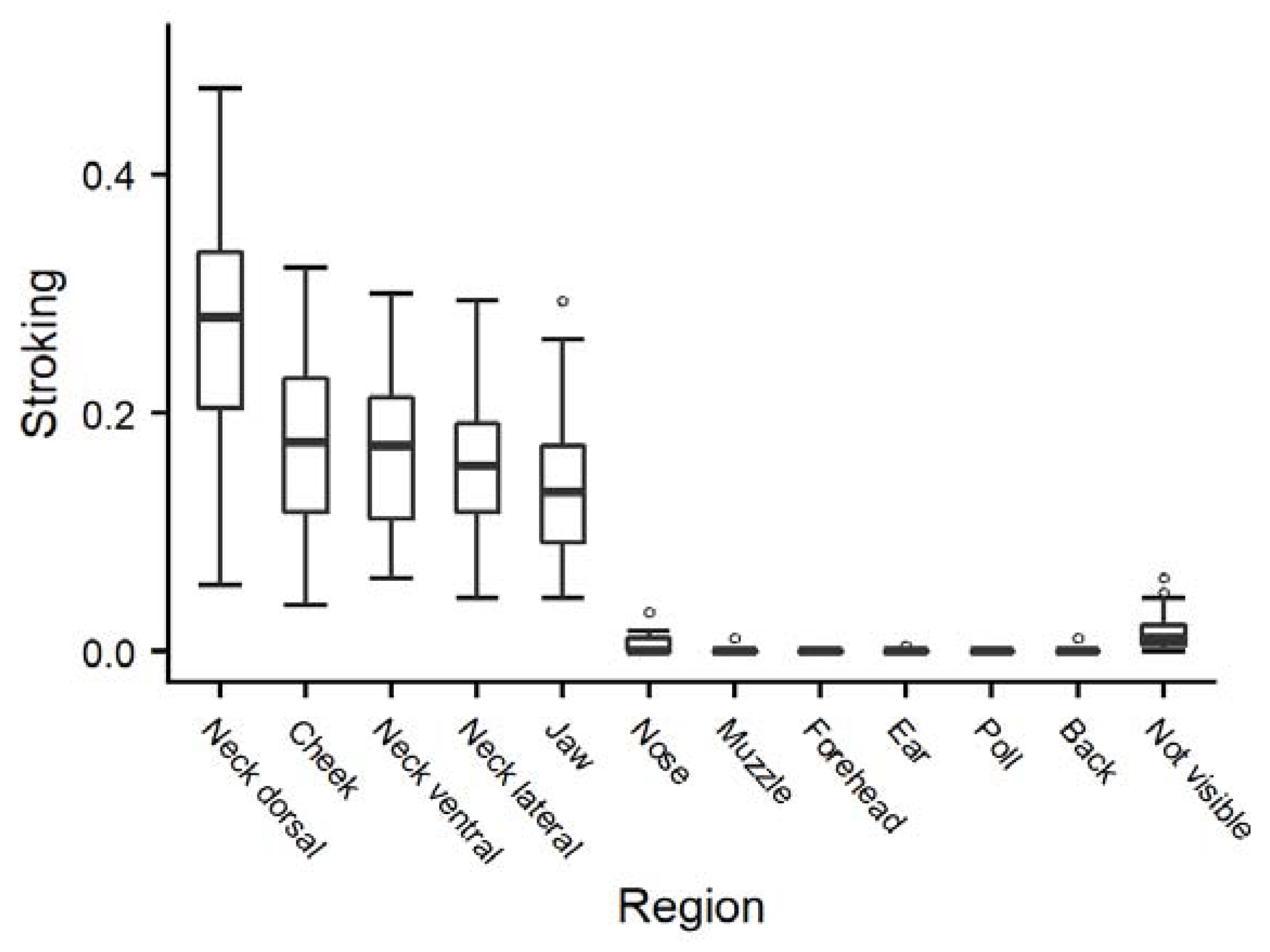
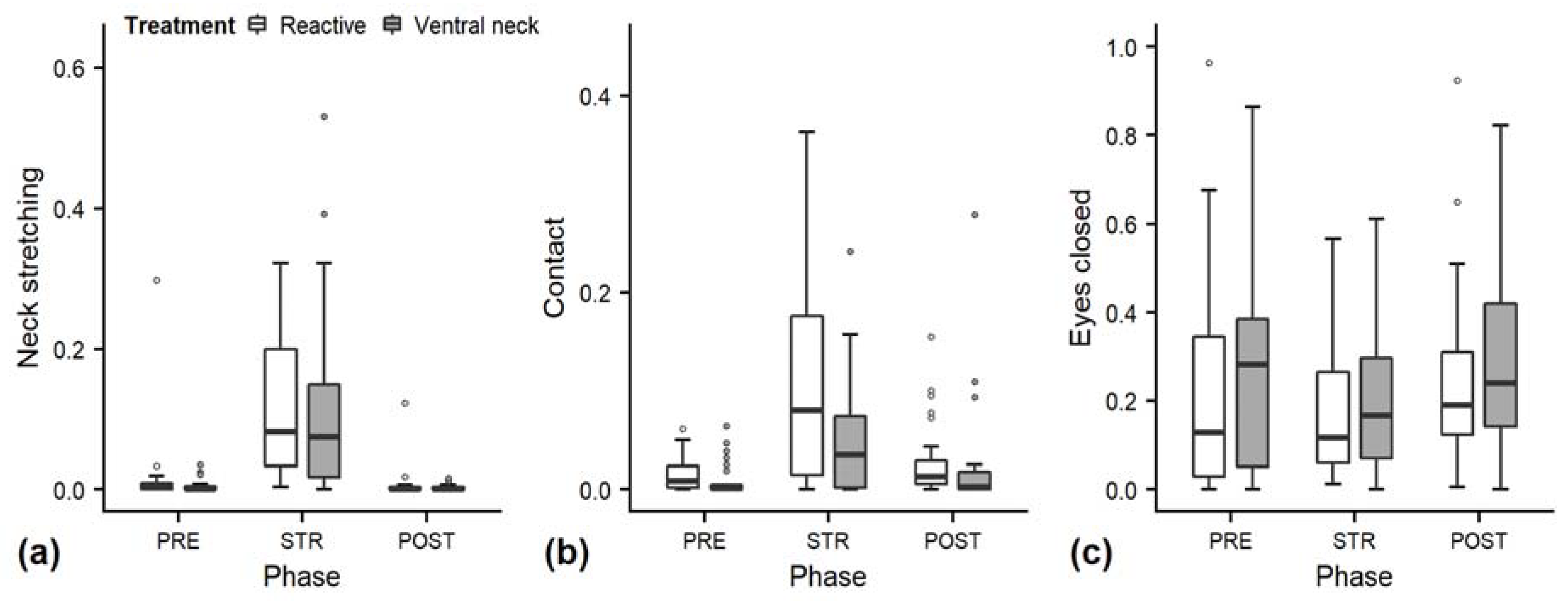
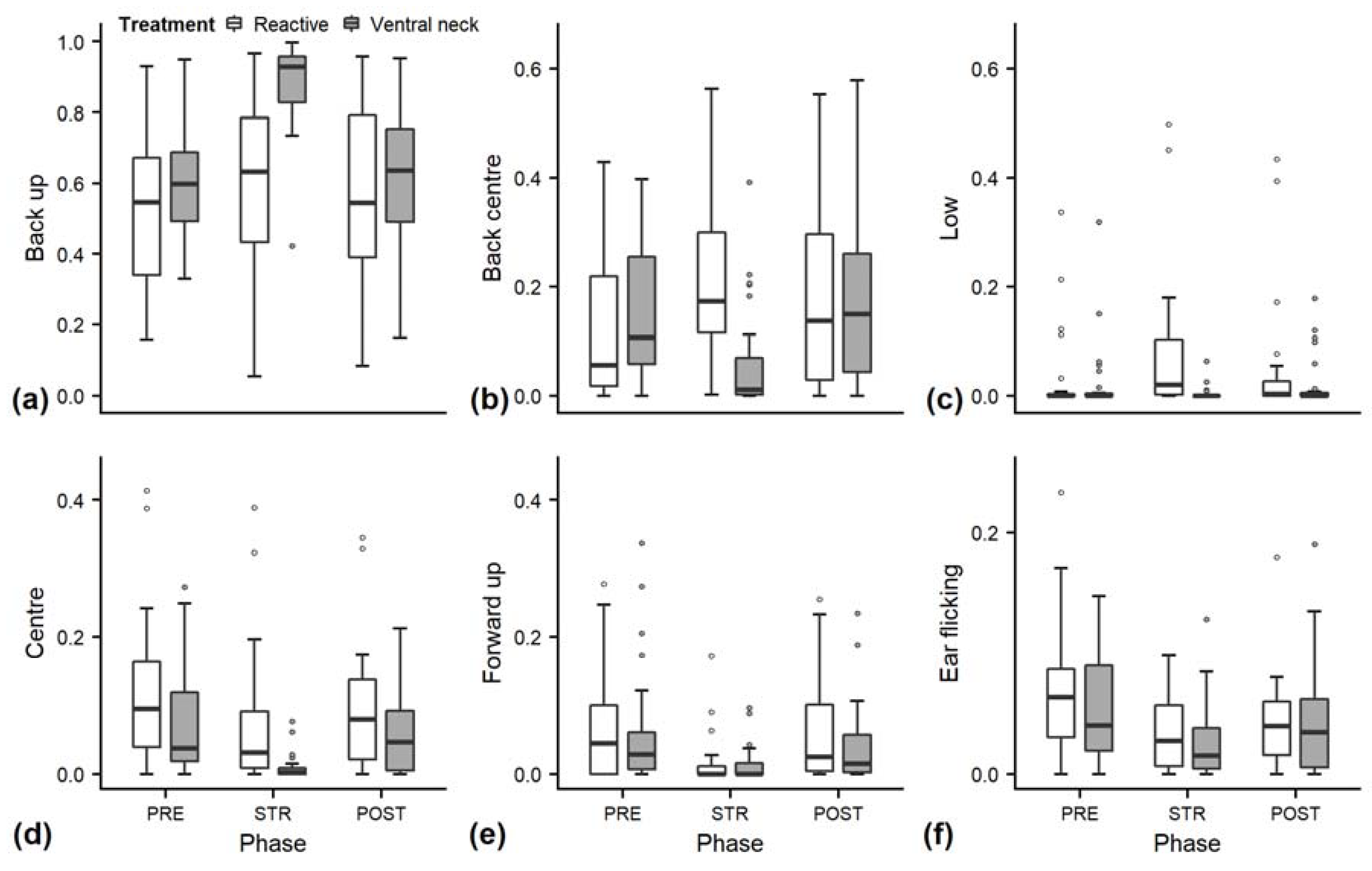
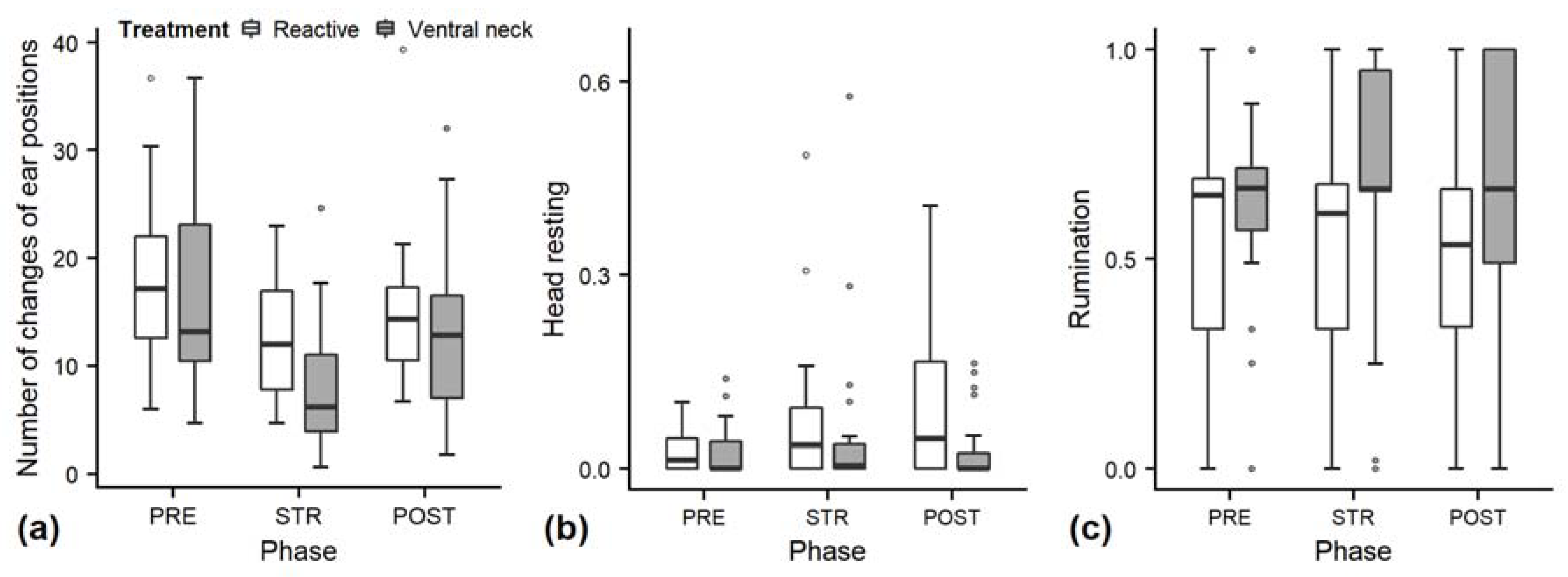
3.1. Behaviour during Gentle Interactions
3.2. Cardiac Data
4. Discussion
4.1. General Effects of Gentle Tactile Interactions on Behaviour and Cardiac Parameters
4.2. Effects of Stroking Style
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yeates, J.W.; Main, D.C.J. Assessment of positive welfare: A review. Vet. J. 2008, 175, 293–300. [Google Scholar] [CrossRef]
- Boissy, A.; Manteuffel, G.; Jensen, M.B.; Moe, R.O.; Spruijt, B.; Keeling, L.J.; Winckler, C.; Forkman, B.; Dimitrov, I.; Langbein, J.; et al. Assessment of positive emotions in animals to improve their welfare. Physiol. Behav. 2007, 92, 375–397. [Google Scholar] [CrossRef]
- Mellor, D. Animal emotions, behaviour and the promotion of positive welfare states. N. Z. Vet. J. 2012, 60, 1–8. [Google Scholar] [CrossRef]
- Boivin, X.; Lensink, J.; Tallet, C.; Veissier, I. Stockmanship and farm animal welfare. Anim. Welf. 2003, 12, 479–492. [Google Scholar]
- Waiblinger, S. Anthrozoology: Human-animal interactions in domesticated and wild animals. In Anthrozoology; Oxford University Press: Oxford, UK, 2018; pp. 32–58. [Google Scholar]
- Waiblinger, S.; Boivin, X.; Pedersen, V.; Tosi, M.V.; Janczak, A.M.; Visser, E.K.; Jones, R.B. Assessing the human-animal relationship in farmed species: A critical review. Appl. Anim. Behav. Sci. 2006, 101, 185–242. [Google Scholar] [CrossRef]
- Coulon, M.; Nowak, R.; Peyrat, J.; Chandèze, H.; Boissy, A.; Boivin, X. Do Lambs Perceive Regular Human Stroking as Pleasant? Behavior and Heart Rate Variability Analyses. PLoS ONE 2015, 10, 1–14. [Google Scholar] [CrossRef]
- Reefmann, N.; Wechsler, B.; Gygax, L. Behavioural and physiological assessment of positive and negative emotion in sheep. Anim. Behav. 2009, 78, 651–659. [Google Scholar] [CrossRef]
- Schmied, C.; Waiblinger, S.; Scharl, T.; Leisch, F.; Boivin, X. Stroking of different body regions by a human: Effects on behaviour and heart rate of dairy cows. Appl. Anim. Behav. Sci. 2008, 109, 25–38. [Google Scholar] [CrossRef]
- Boivin, X.; Garel, J.P.; Durier, C.; Le Neindre, P. Is gentling by people rewarding for beef calves? Appl. Anim. Behav. Sci. 1998, 61, 1–12. [Google Scholar] [CrossRef]
- Hennessy, M.B.; Williams, M.T.; Miller, D.D.; Douglas, C.W.; Voith, V.L. Influence of male and female petters on plasma cortisol and behaviour: Can human interaction reduce the stress of dogs in a public animal shelter? Appl. Anim. Behav. Sci. 1998, 61, 63–77. [Google Scholar] [CrossRef]
- Lürzel, S.; Windschnurer, I.; Futschik, A.; Waiblinger, S. Gentle interactions decrease the fear of humans in dairy heifers independently of early experience of stroking. Appl. Anim. Behav. Sci. 2016, 178, 16–22. [Google Scholar] [CrossRef]
- Reinhardt, C.; Reinhardt, A.; Reinhardt, V. Social behaviour and reproductive performance in semi-wild Scottish Highland cattle. Appl. Anim. Behav. Sci. 1986. [Google Scholar] [CrossRef]
- Sato, S.; Sako, S.; Maeda, A. Social licking patterns in cattle (Bos taurus): Influence of environmental and social factors. Appl. Anim. Behav. Sci. 1991, 32, 3–12. [Google Scholar] [CrossRef]
- Laister, S.; Stockinger, B.; Regner, A.-M.M.; Zenger, K.; Knierim, U.; Winckler, C. Social licking in dairy cattle—Effects on heart rate in performers and receivers. Appl. Anim. Behav. Sci. 2011, 130, 81–90. [Google Scholar] [CrossRef]
- Bertenshaw, C.E.; Rowlinson, P. Exploring heifers’ perception of “positive” treatment through their motivation to pursue a retreated human. Anim. Welf. 2008, 17, 313–319. [Google Scholar]
- Schulze Westerath, H.; Gygax, L.; Hillmann, E. Are special feed and being brushed judged as positive by calves? Appl. Anim. Behav. Sci. 2014, 156, 12–21. [Google Scholar] [CrossRef]
- Schmied, C.; Boivin, X.; Waiblinger, S. Stroking Different Body Regions of Dairy Cows: Effects on Avoidance and Approach Behavior Toward Humans. J. Dairy Sci. 2008, 91, 596–605. [Google Scholar] [CrossRef]
- Lürzel, S.; Münsch, C.; Windschnurer, I.; Futschik, A.; Palme, R.; Waiblinger, S. The influence of gentle interactions on avoidance distance towards humans, weight gain and physiological parameters in group-housed dairy calves. Appl. Anim. Behav. Sci. 2015, 172, 9–16. [Google Scholar] [CrossRef]
- Windschnurer, I.; Barth, K.; Waiblinger, S. Can stroking during milking decrease avoidance distances of cows towards humans? Anim. Welf. 2009, 18, 507–513. [Google Scholar]
- Schmied, C.; Boivin, X.; Waiblinger, S. Ethogramm des sozialen Leckens beim Rind: Untersuchungen in einer Mutterkuhherde. KTBL-Schrift 441 2005, 86–92. [Google Scholar]
- Sambraus, H.H. Das soziale Lecken des Rindes. Z. Tierpsychol. 1969, 26, 805–810. [Google Scholar]
- Lansade, L.; Nowak, R.; Lainé, A.L.; Leterrier, C.; Bonneau, C.; Parias, C.; Bertin, A. Facial expression and oxytocin as possible markers of positive emotions in horses. Sci. Rep. 2018, 8, 14680. [Google Scholar] [CrossRef]
- Mendl, M.; Burman, O.H.P.; Paul, E.S. An integrative and functional framework for the study of animal emotion and mood. Proc. R. Soc. B Biol. Sci. 2010, 277, 2895–2904. [Google Scholar] [CrossRef]
- Descovich, K.A.; Wathan, J.; Leach, M.C.; Buchanan-Smith, H.M.; Flecknell, P.; Farningham, D.; Vick, S.J. Facial expression: An under-utilized tool for the assessment of welfare in mammals. ALTEX 2017. [Google Scholar] [CrossRef]
- De Oliveira, D.; Keeling, L.J. Routine activities and emotion in the life of dairy cows: Integrating body language into an affective state framework. PLoS ONE 2018, 13, e0195674. [Google Scholar] [CrossRef]
- Lambert, H.; Carder, G. Positive and negative emotions in dairy cows: Can ear postures be used as a measure? Behav. Processes 2019, 158, 172–180. [Google Scholar] [CrossRef]
- Battini, M.; Agostini, A.; Mattiello, S. Understanding Cows’ Emotions on Farm: Are Eye White and Ear Posture Reliable Indicators? Animals 2019, 9, 477. [Google Scholar] [CrossRef]
- Mandel, R.; Wenker, M.L.; van Reenen, K.; Keil, N.M.; Hillmann, E. Can access to an automated grooming brush and/or a mirror reduce stress of dairy cows kept in social isolation? Appl. Anim. Behav. Sci. 2019, 211, 1–8. [Google Scholar] [CrossRef]
- Von Borell, E.; Langbein, J.; Després, G.; Hansen, S.; Leterrier, C.; Marchant-Forde, J.; Marchant-Forde, R.; Minero, M.; Mohr, E.; Prunier, A.; et al. Heart rate variability as a measure of autonomic regulation of cardiac activity for assessing stress and welfare in farm animals—A review. Physiol. Behav. 2007, 92, 293–316. [Google Scholar] [CrossRef]
- Martin, P.; Bateson, P. Measuring Behaviour. An Introductory Guide; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Proctor, H.S.; Carder, G. Can ear postures reliably measure the positive emotional state of cows? Appl. Anim. Behav. Sci. 2014, 161, 20–27. [Google Scholar] [CrossRef]
- Hagen, K.; Langbein, J.; Schmied, C.; Lexer, D.; Waiblinger, S. Heart rate variability in dairy cows—Influences of breed and milking system. Physiol. Behav. 2005, 85, 195–204. [Google Scholar] [CrossRef] [PubMed]
- R Core Team R: A Language and Environment for Statistical Computing. Available online: https://www.R-project.org. (accessed on 4 March 2020).
- Baayen, R.H. Analyzing linguistic data: A practical introduction to statistics using R; Cambridge University Press: Cambridge, UK, 2008; ISBN 9780511801686. [Google Scholar]
- Bolker, B.M. Ecological models and data in R; Princeton University Press: Princeton, NJ, USA, 2008; ISBN 9781400840908. [Google Scholar]
- McCullagh, P.; Nelder, J.A. Generalized Linear Models, 2nd ed.; Chapman & Hall/CRC: Boca Raton, FL, USA, 1989; ISBN 0412317605. [Google Scholar]
- Brooks, M.E.; Kristensen, K.; van Benthem, K.J.; Magnusson, A.; Berg, C.W.; Nielsen, A.; Skaug, H.J.; Machler, M.; Bolker, B.M. glmmTMB Balances Speed and Flexibility Among Packages for Zero-inflated Generalized Linear Mixed Modeling. R J. 2017, 9, 378–400. [Google Scholar] [CrossRef]
- Smithson, M.; Verkuilen, J. A better lemon squeezer? Maximum-likelihood regression with beta-distributed dependent variables. Psychol. Methods 2006. [Google Scholar] [CrossRef]
- Barr, D.J.; Levy, R.; Scheepers, C.; Tily, H.J. Random effects structure for confirmatory hypothesis testing: Keep it maximal. J. Mem. Lang. 2013, 68, 255–278. [Google Scholar] [CrossRef]
- Forstmeier, W.; Schielzeth, H. Cryptic multiple hypotheses testing in linear models: Overestimated effect sizes and the winner’s curse. Behav. Ecol. Sociobiol. 2011, 65, 47–55. [Google Scholar] [CrossRef]
- Stoehr, A.M. Are significance threshholds appropriate for the study of animal behaviour? Anim. Behav. 1999, 57, F22–F25. [Google Scholar] [CrossRef]
- Nieuwenhuis, R.; te Grotenhuis, M.; Pelzer, B. Influence.ME: Tools for detecting influential data in mixed effects models. R J. 2012, 4, 38–47. [Google Scholar] [CrossRef]
- Gelman, A.; Hill, J. Data Analysis Using Regression and Multilevel/Hierarchical Models; Cambridge University Press: New York, NY, USA, 2006. [Google Scholar]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Wilke, C.O. cowplot: Streamlined Plot Theme and Plot Annotations for “ggplot2”. R package version 1.0.0. Available online: https://cran.r-project.org/package=cowplot. (accessed on 4 March 2020).
- Sacha, J. Interaction between Heart Rate and Heart Rate Variability. Ann. Noninvasive Electrocardiol. 2014, 19, 207–216. [Google Scholar] [CrossRef]
- Mccraty, R.; Shaffer, F. Heart Rate Variability: New Perspectives on Physiological Mechanisms, Assessment of Self-regulatory Capacity, and Health Risk. Glob. Adv. Heal. Med. 2015, 4, 46–61. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. 1995, 57. [Google Scholar] [CrossRef]
- Lenth, R.V. Emmeans: Estimated Marginal Means, aka Least-Squares Means. R Package Version 1.4.4. Available online: https://cran.r-project.org/package=emmeans. (accessed on 4 March 2020).
- Waiblinger, S.; Menke, C.; Korff, J.; Bucher, A. Previous handling and gentle interactions affect behaviour and heart rate of dairy cows during a veterinary procedure. Appl. Anim. Behav. Sci. 2004, 85, 31–42. [Google Scholar] [CrossRef]
- Désiré, L.; Boissy, A.; Veissier, I. Emotions in farm animals: A new approach to animal welfare in applied ethology. Behav. Processes 2002, 60, 165–180. [Google Scholar] [CrossRef]
- Fraser, D.; Duncan, I. ‘Pleasures’, ‘Pains’ and Animal Welfare: Toward a Natural History of Affect. Anim. Welf. Collect. 1998, 7, 383–396. [Google Scholar]
- Sato, S.; Tarumizu, K. Heart rates before, during and after allo-grooming in cattle (Bos taurus). J. Ethol. 1993, 11, 149–150. [Google Scholar] [CrossRef]
- Reefmann, N.; Bütikofer Kaszàs, F.; Wechsler, B.; Gygax, L. Ear and tail postures as indicators of emotional valence in sheep. Appl. Anim. Behav. Sci. 2009, 118, 199–207. [Google Scholar] [CrossRef]
- Heinrich, A.; Duffield, T.F.; Lissemore, K.D.; Millman, S.T. The effect of meloxicam on behavior and pain sensitivity of dairy calves following cautery dehorning with a local anesthetic. J. Dairy Sci. 2010, 93, 2450–2457. [Google Scholar] [CrossRef]
- Neave, H.W.; Daros, R.R.; Costa, J.H.C.; Von Keyserlingk, M.A.G.; Weary, D.M. Pain and pessimism: Dairy calves exhibit negative judgement bias following hot-iron disbudding. PLoS ONE 2013, 8, 8–13. [Google Scholar] [CrossRef]
- Mooring, M.S.; Blumstein, D.T.; Reisig, D.D.; Osborne, E.R.; Niemeyer, J.M. Insect-repelling behaviour in bovids: Role of mass, tail length, and group size. Biol. J. Linn. Soc. 2007, 91, 383–392. [Google Scholar] [CrossRef]
- Shahin, M. The effects of positive human contact by tactile stimulation on dairy cows with different personalities. Appl. Anim. Behav. Sci. 2018, 204, 23–28. [Google Scholar] [CrossRef]
- Gleerup, K.B.; Andersen, P.H.; Munksgaard, L.; Forkman, B. Pain evaluation in dairy cattle. Appl. Anim. Behav. Sci. 2015, 171, 25–32. [Google Scholar] [CrossRef]
| Behaviour (1) | Definition | |
|---|---|---|
| Inactive ear posture (2) | Ear hanging | The ear loosely hangs downwards (referring to the ground). There is no visible muscle tension, leading often to a slightly bouncing movement when the position is assumed. |
| Active ear postures (2) (3) | Back up | The ear is held behind and above the latero-lateral axis. |
| Back centre | The ear is held behind at the same height as the latero-lateral axis. | |
| Back down | The ear is held behind and below the latero-lateral axis. | |
| Centre up | The ear is held perpendicular to the head and above the latero-lateral axis. | |
| Centre | The ear is held perpendicular to the head along the latero-lateral axis. | |
| Centre down | The ear is held perpendicular to the head and below the latero-lateral axis. | |
| Forward up | The ear is held in front of and above the latero-lateral axis. | |
| Forward centre | The ear is held in front of and at the same height as the latero-lateral axis. | |
| Forward down | The ear is held in front of and below the latero-lateral axis. | |
| Ear flicking | The ear is quickly (within max. 0.5 s) moved back and forth at least once. The behaviour is coded until one of the other ear postures is clearly visible again. The residual movement after the active movement is still part of ear flicking. | |
| Head/neck postures | Held without touching | The head is actively held up and does not touch the stroker. |
| Held with touching | The head is actively held up and touches the stroker. | |
| Rest head without touching | The heifer does not actively carry the head’s weight. The heifer’s head is in contact with the ground, barn equipment, another animal or with the heifer’s leg(s). The heifer’s head is not in contact with the stroker. | |
| Rest head with touching | The heifer does not actively carry the head’s weight. The heifer’s head is lying on the ground, barn equipment, another animal or the heifer’s leg(s) while being in contact with the stroker, or it is lying on the stroker’s leg. | |
| Head shaking/tossing | Successive quick movements of the head. The movements can be rotational or up and down. | |
| Neck stretching | Positioning neck and head actively in an outstretched line, either up, down, or forward. | |
| Eyes (4) | Open | The iris is at least partly visible. |
| Closed | The iris is not visible at all for longer than 0.5 s. | |
| Not visible | Neither eye is visible. | |
| Miscellaneous | Rubbing the stroker | The heifer touches the stroker and moves the touching body part while in contact with the experimenter. The behaviour ends when the contact between the heifer and the person is interrupted for at least 3 s. |
| Rubbing | The heifer moves the head/neck region while in contact with the ground or barn equipment. The behaviour ends when the contact between the heifer’s head/neck region and the ground/equipment has ended. | |
| Nose close | The heifer moves her muzzle towards the stroker within a range of 5 cm. The behaviour ends when the heifer’s nose does not point towards the stroker anymore, leaves the range of 5 cm or if another behaviour of the “miscellaneous” category starts. | |
| Licking the stroker | The heifer’s tongue touches the stroker at least once. The behaviour ends when the heifer’s tongue does not touch the stroker again within 3 s. | |
| Ruminating | The heifer’s jaw moves regularly sideways with a frequency of about one movement per second. This movement is recorded as rumination if it occurs in a series of at least five movements (which may start before and end after the observation). Rumination ends when the jaw movement is paused for more than 10 s. | |
| Calculated measures | Contact | The time in which the heifer’s head and neck area was in contact with the stroker. Sum of durations of “rest head with touching”, “held with touching”, “nose close”, “rubbing experimenter” and “licking experimenter”, not including contact established by stroking. |
| Resting head | Sum of durations of “rest head with touching” and “rest head without touching”. | |
| Ear low | The sum of the durations of the ear hanging or held below the latero-lateral axis (“hanging” + “back down” + “centre down” + “forward down”). | |
| Changes of ear positions | Sum of the frequencies of different ear positions per trial minus 1. | |
| Treatment | Phase | Treatment × Phase | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Parameter | χ2 | df | p | χ2 | df | p | χ2 | df | p |
| HR (bpm) | 1.14 | 1 | 0.29 | 46.99 | 2 | 0.00 | 1.67 | 2 | 0.43 |
| SDNN (ms) | 0.15 | 1 | 0.70 | 1.57 | 2 | 0.46 | 1.88 | 2 | 0.39 |
| RMSSD (ms) | 0.16 | 1 | 0.69 | 0.78 | 2 | 0.68 | 0.88 | 2 | 0.64 |
| LF (ms2) | 0.18 | 1 | 0.68 | 4.64 | 2 | 0.10 | 0.67 | 2 | 0.72 |
| HF (ms2) | 0.08 | 1 | 0.79 | 5.58 | 2 | 0.06 | 0.54 | 2 | 0.76 |
| LF/HF | 0.13 | 1 | 0.71 | 6.99 | 2 | 0.03 | 0.95 | 2 | 0.62 |
| PRE | STR | POST | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Parameter | Treatment | Mean | SE | CLL | CLU | Mean | SE | CLL | CLU | Mean | SE | CLL | CLU |
| HR (bpm) (1) | Reactive | 81.1 | 2.3 | 56.8 | 115.8 | 83.3 | 2.3 | 58.3 | 119.0 | 81.6 | 2.3 | 57.1 | 116.5 |
| Ventral neck | 83.0 | 2.3 | 58.3 | 118.1 | 84.7 | 2.3 | 59.5 | 120.5 | 83.0 | 2.3 | 58.3 | 118.0 | |
| SDNN (ms) (1) | Reactive | 27.3 | 2.0 | 10.8 | 68.6 | 25.6 | 1.9 | 10.2 | 64.3 | 26.6 | 1.9 | 10.6 | 66.9 |
| Ventral neck | 26.6 | 1.9 | 10.9 | 64.9 | 25.5 | 1.8 | 10.4 | 62.3 | 24.2 | 1.7 | 9.9 | 59.0 | |
| RMSSD (ms) (1) | Reactive | 14.7 | 1.3 | 4.6 | 46.3 | 14.1 | 1.3 | 4.5 | 44.3 | 14.2 | 1.3 | 4.5 | 44.9 |
| Ventral neck | 14.2 | 1.3 | 4.7 | 43.6 | 13.1 | 1.2 | 4.3 | 40.1 | 14.0 | 1.2 | 4.6 | 42.8 | |
| HF (ms2) (1) | Reactive | 12.2 | 1.4 | 2.7 | 54.6 | 15.2 | 1.8 | 3.4 | 67.5 | 12.2 | 1.4 | 2.7 | 54.5 |
| Ventral neck | 11.7 | 1.3 | 2.8 | 48.6 | 14.0 | 1.6 | 3.4 | 58.3 | 12.6 | 1.4 | 3.0 | 52.1 | |
| LF (ms2) | Reactive | 71.8 | 2.5 | 40.3 | 103.4 | 68.9 | 2.5 | 37.5 | 100.3 | 74.5 | 2.5 | 43.1 | 106.0 |
| Ventral neck | 73.0 | 2.3 | 43.2 | 102.9 | 68.9 | 2.4 | 39.0 | 98.8 | 72.8 | 2.3 | 43.0 | 102.7 | |
| LF/HF (1) | Reactive | 5.6 | 0.8 | 0.9 | 36.2 | 4.3 | 0.6 | 0.7 | 27.3 | 6.0 | 0.9 | 0.9 | 37.9 |
| Ventral neck | 6.0 | 0.8 | 1.0 | 35.2 | 4.8 | 0.7 | 0.8 | 27.9 | 5.6 | 0.8 | 1.0 | 32.6 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lange, A.; Franzmayr, S.; Wisenöcker, V.; Futschik, A.; Waiblinger, S.; Lürzel, S. Effects of Different Stroking Styles on Behaviour and Cardiac Parameters in Heifers. Animals 2020, 10, 426. https://doi.org/10.3390/ani10030426
Lange A, Franzmayr S, Wisenöcker V, Futschik A, Waiblinger S, Lürzel S. Effects of Different Stroking Styles on Behaviour and Cardiac Parameters in Heifers. Animals. 2020; 10(3):426. https://doi.org/10.3390/ani10030426
Chicago/Turabian StyleLange, Annika, Sandra Franzmayr, Vera Wisenöcker, Andreas Futschik, Susanne Waiblinger, and Stephanie Lürzel. 2020. "Effects of Different Stroking Styles on Behaviour and Cardiac Parameters in Heifers" Animals 10, no. 3: 426. https://doi.org/10.3390/ani10030426
APA StyleLange, A., Franzmayr, S., Wisenöcker, V., Futschik, A., Waiblinger, S., & Lürzel, S. (2020). Effects of Different Stroking Styles on Behaviour and Cardiac Parameters in Heifers. Animals, 10(3), 426. https://doi.org/10.3390/ani10030426





