New Insights on the Evolution of the Sweet Taste Receptor of Primates Adapted to Harsh Environments
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement and Humane Care Guidelines
2.2. Taxonomic Sampling
2.3. DNA Extraction, Amplification, and Sequencing
2.4. Evolutionary Analyses
2.5. Divergence Time Estimates
3. Results
3.1. Evolutionary Analyses
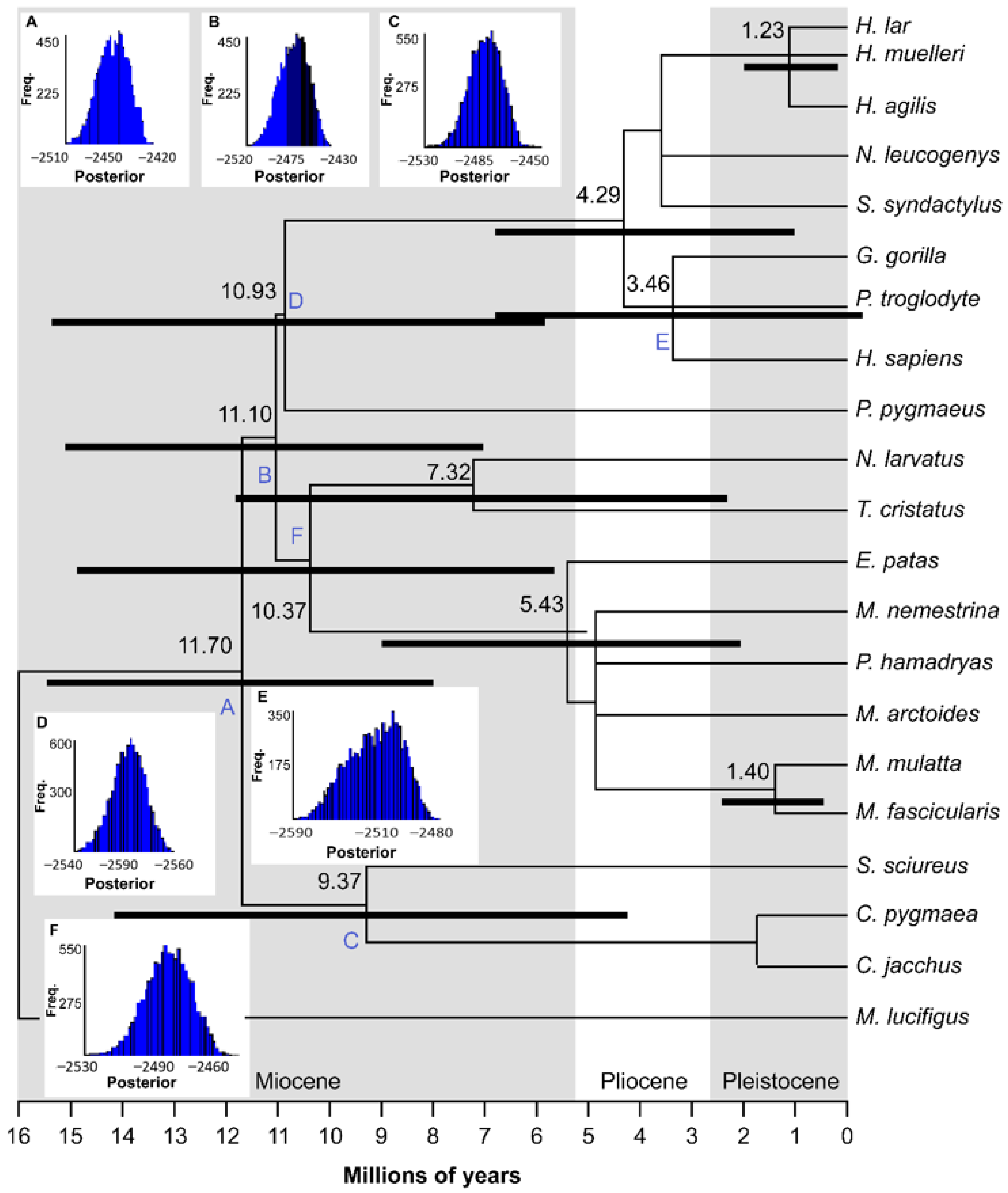
3.2. Molecular Clock Analyses
4. Discussion
4.1. NWM (Family Cebidae)
4.2. OWM (Family Cercopithecidae)
4.3. Apes and Humans
4.4. Tracing the Evolutionary Pathway: A Reassessment of the Time of Divergence for Anthropoid Primates
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kinnamon, S.C.; Margolskee, R.F. Mechanisms of taste transduction. Curr. Opin. Neurobiol. 1996, 6, 506–513. [Google Scholar] [CrossRef]
- Lindemann, B. Taste reception. Physiol. Rev. 1996, 76, 719–766. [Google Scholar] [CrossRef] [PubMed]
- Bachmanov, A.A.; Beauchamp, G.K. Taste receptor genes. Annu. Rev. Nutr. 2007, 27, 389–414. [Google Scholar] [CrossRef]
- Zhao, G.Q.; Zhang, Y.; Hoon, M.A.; Chandrashekar, J.; Erlenbach, I.; Ryba, N.J.P.; Zuker, C.S. The receptors for mammalian sweet and umami taste. Cell. 2003, 115, 255–266. [Google Scholar] [CrossRef]
- Zhao, H.; Zhou, Y.; Pinto, C.M.; Charles-Dominique, P.; Galindo-Gonzalez, J.; Zhang, S.; Zhang, J. Evolution of the sweet taste receptor gene Tas1r2 in bats. Mol. Biol. Evol. 2010, 27, 2642–2650. [Google Scholar] [CrossRef]
- Li, X.; Staszewski, L.; Xu, H.; Durick, K.; Zoller, M.; Adler, E. Human receptors for sweet and umami taste. Proc. Natl. Acad. Sci. USA 2002, 99, 4692–4696. [Google Scholar] [CrossRef] [PubMed]
- Shi, P.; Zhang, J. Contrasting modes of evolution between vertebrate sweet/umami receptor genes and bitter receptor genes. Mol. Biol. Evol. 2006, 23, 292–300. [Google Scholar] [CrossRef]
- Li, R.; Fan, W.; Tian, G.; Zhu, H.; He, L.; Cai, J.; Huang, Q.; Cai, Q.; Li, B.; Bai, Y.; et al. The sequence and de novo assembly of the giant panda genome. Nature 2010, 463, 311–317. [Google Scholar] [CrossRef]
- Li, X.; Li, W.; Wang, H.; Cao, J.; Maehashi, K.; Huang, L.; Bachmanov, A.A.; Reed, D.R.; Legrand-Defretin, V.; Beauchamp, G.K.; et al. Pseudogenization of a sweet receptor gene accounts for cats’ indifference toward sugar. PLoS Genet. 2005, 1, 27–35. [Google Scholar] [CrossRef]
- Bennett, E.L.; Gombek, F. Proboscis Monkeys of Borneo, 1st ed.; Natural History Publications (Borneo) Sdn. Bhd. & Koktas Sabah: Ranau, Malaysia, 1993; ISBN 978-9838120012. [Google Scholar]
- Boonratana, R. The Ecology and Behaviour of the Proboscis Monkey (Nasalis larvatus) in the Lower Kinabatangan, Sabah. Ph.D. Thesis, Mahidol University, Nakhon Pathom, Thailand, 1993. [Google Scholar]
- Lehman, S.M.; Fleagle, J.G. Biogeography and primates: A review. In Primate Biogeography; Springer: Boston, MA, USA, 2006; pp. 1–58. ISBN 978-0-387-29871-9. [Google Scholar]
- McNeilage, A. Diet and habitat use of two mountain gorilla groups in contrasting habitats in the Virungas. In Mountain Gorillas: Three Decades of Research at Karisoke, 1st ed.; Robbins, M.M., Sicotte, P., Stewart, K.J., Eds.; Cambridge University Press: Cambridge, UK, 2001; pp. 265–292. ISBN 978-0521780049. [Google Scholar]
- Nurcahyo, A. Daily ranging, home-range, foods, feeding and calling in Siamang (Hylobates syndactylus). In Bukit Barisan Selatan National Park in Space and Time. 2000–2001 Research Report; WCS-IP/PHKA: Bogor, Indonesia, 2001; pp. 35–52. [Google Scholar]
- Swedell, L. Ranging behavior, group size and behavioral flexibility in Ethiopian Hamadryas baboons (Papio hamadryas hamadryas). Folia Primatol. 2002, 73, 95–103. [Google Scholar] [CrossRef]
- Zaw, K.; Meffre, S.; Takai, M.; Suzuki, H.; Burrett, C.; Htike, T.; Thein, Z.M.M.; Tsubamoto, T.; Egi, N.; Maung, M. The oldest anthropoid primates in SE Asia: Evidence from LA-ICP-MS U-Pb zircon age in the Late Middle Eocene Pondaung Formation. Myanmar. Gondwana Res. 2014, 26, 122–131. [Google Scholar] [CrossRef]
- Beard, K.C.; Marivaux, L.; Tun, S.T.; Soe, A.N.; Chaimanee, Y.; Htoon, W.; Marandat, B.; Aung, H.H.; Jaeger, J.J. New sivaladapid primates from the Eocene Pondaung Formation of Myanmar and the anthropoid status of Amphipithecidae. Bull. Carnegie Mus. Nat. Hist. 2007, 39, 67–76. [Google Scholar] [CrossRef]
- Takai, M.; Sein, C.; Tsubamoto, T.; Egi, N.; Maung, M.; Shigehara, N. A new eosimiid from the latest middle Eocene in Pondaung, central Myanmar. Anthropol. Sci. 2005, 113, 17–25. [Google Scholar] [CrossRef]
- Finstermeier, K.; Zinner, D.; Brameier, M.; Meyer, M.; Kreuz, E.; Hofreiter, M.; Roos, C. A mitogenomic phylogeny of living primates. PLoS ONE 2013, 8, e69504. [Google Scholar] [CrossRef] [PubMed]
- Perelman, P.; Johnson, W.E.; Roos, C.; Seuanez, H.N.; Horvath, J.E.; Moreira, M.A.M.; Kessing, B.; Pontius, J.; Roelke, M.; Rumpler, Y.; et al. A molecular phylogeny of living primates. PLoS Genet. 2011, 7, e1001342. [Google Scholar] [CrossRef]
- Seiffert, E.R.; Simons, E.L.; Clyde, W.C.; Rossie, J.B.; Attia, Y.; Bown, T.M.; Chatrath, P.; Mathison, M.E. Basal anthropoids from Egypt and the antiquity of Africa’s higher primate radiation. Science 2005, 310, 300–304. [Google Scholar] [CrossRef]
- Steiper, M.E.; Young, N.M. Primate molecular divergence dates. Mol. Phylogenet. Evol. 2006, 41, 384–394. [Google Scholar] [CrossRef]
- Schrago, C.G.; Russo, C.A. Timing the origin of New World monkeys. Mol. Biol. Evol. 2003, 20, 1620–1625. [Google Scholar] [CrossRef]
- Hasegawa, M.; Thorne, J.L.; Kishino, H. Time scale of eutherian evolution estimated without assuming a constant rate of molecular evolution. Genes Genet. Syst. 2003, 78, 267–283. [Google Scholar] [CrossRef]
- Steiper, M.E.; Young, N.M.; Sukarna, T.Y. Genomic data support the hominoid slowdown and an Early Oligocene estimate for the hominoid-cercopithecoid divergence. Proc. Natl Acad. Sci. USA 2004, 101, 17021–17026. [Google Scholar] [CrossRef]
- McCarthy, C. CHROMAS Program; Schools of Health Science, Griffith University, Gold Coast Campus: Queensland, Australia, 1997. [Google Scholar]
- Higgins, D.; Thompson, J.; Gibson, T. CLUSTAL W: Improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice. Nucleic Acids Res. 1994, 22, 4673–4680. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across computing platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef] [PubMed]
- Huelsenbeck, J.P.; Ronquist, F. MrBayes: Bayesian inference of phylogeny. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef] [PubMed]
- Posada, D.; Crandall, K.A. Modeltest: Testing the model of DNA substitution. Bioinformatics 1998, 14, 817–818. [Google Scholar] [CrossRef] [PubMed]
- Kimura, M. A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J. Mol. Evol. 1980, 16, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Drummond, A.J.; Rambaut, A. BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol. Biol. 2007, 7, 214. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, H.J.; Ho, S.Y.W.; Barnes, I.; Groves, C. Estimating the phylogeny and divergence times of primates using a supermatrix approach. BMC Evol. Biol. 2009, 9, 259. [Google Scholar] [CrossRef]
- Fabre, P.H.; Rodrigues, A.; Douzery, E.J.P. Patterns of macroevolution among primates inferred from a supermatrix of mitochondrial and nuclear DNA. Mol. Phylogenet. Evol. 2009, 53, 808–825. [Google Scholar] [CrossRef]
- Matsui, A.; Rakotondraparany, F.; Munechika, I.; Hasegawa, M.; Horai, S. Molecular phylogeny and evolution of prosimians based on complete sequences of mitochondrial DNAs. Gene 2009, 441, 53–66. [Google Scholar] [CrossRef]
- Wilkinson, R.D.; Steiper, M.E.; Soligo, C.; Martin, R.D.; Yang, Z.; Tavare, S. Dating primate divergences through an integrated analysis of palaeontological and molecular data. Syst. Biol. 2011, 60, 16–31. [Google Scholar] [CrossRef]
- Springer, M.S.; Meredith, R.W.; Gatesy, J.; Emerling, C.; Park, J.; Rabosky, D.L.; Stadler, T.; Steiner, C.; Ryder, O.; Janečka, J.E.; et al. Macroevolutionary dynamics and historical biogeography of primate diversification inferred from a species supermatrix. PLoS ONE 2012, 7, e49521. [Google Scholar] [CrossRef] [PubMed]
- Pozzi, L.; Hodgson, J.A.; Burrell, A.S.; Sterner, K.N.; Raaum, R.L.; Disotell, T.R. Primate phylogenetic relationships and divergence dates inferred from complete mitochondrial genomes. Mol. Phylogenet. Evol. 2014, 75, 165–183. [Google Scholar] [CrossRef] [PubMed]
- Nylander, J.A.A. MrModeltest 2.3; Program Distributed by the Author; Evolutionary Biology Centre, Uppsala University: Uppsala, Sweden, 2004. [Google Scholar]
- Swofford, D.L.; Olsen, G.J.; Waddell, P.J.; Hills, D.M. Phylogenetic Inference. In Molecular Systematic, 2nd ed.; Hillis, D.M., Moritz, C., Mable, B.K., Eds.; Sinauer Associates: Sunderland, MA, USA, 1996; pp. 407–509. ISBN 978-0878932825. [Google Scholar]
- Goldberg, A.; Wildman, D.E.; Schmidt, T.R.; Huttemann, M.; Goodman, M.; Weiss, M.L.; Grossman, L.I. Adaptive evolution of cytochrome c oxidase subunit VIII in anthropoid primates. Proc. Natl. Acad. Sci. USA 2003, 100, 5873–5878. [Google Scholar] [CrossRef] [PubMed]
- Opazo, J.C.; Wildman, D.E.; Prychitko, T.; Johnson, R.M.; Goodman, M. Phylogenetic relationships and divergence times among New World monkeys (Platyrrhini, Primates). Mol. Phylogenet. Evol. 2006, 40, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Takai, M.; Anaya, F.; Shigehara, N.; Setoguchi, T. New fossil materials of the earliest new world monkey, Branisella boliviana, and the problem of platyrrhine origins. Am. J. Phys. Anthropol. 2000, 111, 263–281. [Google Scholar] [CrossRef]
- Takai, M.; Anaya, F. New specimens of the oldest fossil platyrrhine, Branisella boliviana, from Salla, Bolivia. Am. J. Phys. Anthropol. 1996, 99, 301–317. [Google Scholar] [CrossRef]
- Hodgson, J.A.; Sterner, K.N.; Matthews, L.J.; Burrell, A.S.; Jani, R.A.; Raaum, R.L.; Stewart, C.; Disotell, T.R. Successive radiations, not stasis, in the South American primate fauna. Proc. Natl. Acad. Sci. USA 2009, 106, 5534–5539. [Google Scholar] [CrossRef]
- Poux, C.; Chevret, P.; Huchon, D.; Jong, W.W.; Douzery, E.J.P. Arrival and diversification of caviomorph rodents and platyrrhine primates in South America. Syst. Biol. 2006, 55, 228–244. [Google Scholar] [CrossRef]
- Genoud, M.; Martin, R.D.; Glasser, D. Rate of metabolism in the smallest simian primate, the pygmy marmoset (Cebuella pygmaea). Am. J. Primatol. 1997, 41, 229–245. [Google Scholar] [CrossRef]
- Kinzey, W.G. Synopsis of New World primates (16 genera). In New World Primates: Ecology, Evolution and Behaviour; Kinzey, W.G., Ed.; Aldine de Gruyter: New York, NY, USA, 1997; pp. 169–324. ISBN 978-0202367507. [Google Scholar]
- Rylands, A.B.; Faria, D.S. Habitats, feeding ecology and home range size in the genus Callithrix. In Marmosets and Tamarins: Systematic, Behaviour and Ecology; Rylands, A.B., Ed.; Oxford University Press: Oxford, UK, 1993; pp. 262–272. ISBN 978-0198540229. [Google Scholar]
- Ford, S.M. Callitrichids as phyletic dwarfs, and the place of Callitrichidae in Platyrrhini. Primates 1980, 21, 31–43. [Google Scholar] [CrossRef]
- Ford, S.M.; Davis, L.C. Systematics and body size: Implications for feeding adaptations in New World monkeys. Am. J. Phys. Anthropol. 1992, 88, 415–468. [Google Scholar] [CrossRef] [PubMed]
- Sarah, T.P. Observations of the foraging behaviour of squirrel monkeys (Saimiri sciureus boliviensis) at the Tulsa Zoological Park. Bios 1993, 64, 64–71. [Google Scholar]
- Perez, S.I.; Tejedor, M.F.; Novo, N.M.; Aristide, L. Divergence times and the evolutionary radiation of New World monkeys (Platyrrhini, Primates): An analysis of fossil and molecular data. PLoS ONE 2013, 8, e68029. [Google Scholar] [CrossRef] [PubMed]
- Caton, J.M. Digestive strategy of the Asian colobine genus Trachypithecus. Primates 1999, 40, 311–325. [Google Scholar] [CrossRef]
- Harding, L.E. Trachypithecus cristatus (Primates: Cercopithecidae). Mamm. Species 2010, 42, 149–165. [Google Scholar] [CrossRef]
- Kombi, M.B.; Abdullah, M.T. A review of the proboscis monkey (Nasalis larvatus) in Borneo, with reference to the population in Bako National Park, Sarawak, Malaysian Borneo. Trop. Nat. Hist. 2016, 16, 42–56. [Google Scholar]
- Isbell, L.A. Diet for a small primate: Insectivory and gummivory in the (large) patas monkey (Erythrocebus patas). Am. J. Primatol. 1998, 45, 381–398. [Google Scholar] [CrossRef]
- Kummer, H. Social Organisation of Hamdryas Baboons: A Field Study; Karger and University Press: Basel, Switzerland, 1968. [Google Scholar]
- Lindburg, D.G. The rhesus monkeys in north India: An ecological and behavioural study. In Primate Behaviour: Developments in the Field and Laboratory Research; Rosenblum, L.A., Ed.; Academic Press: New York, NY, USA, 1971; pp. 83–104. ISBN 978-0125340021. [Google Scholar]
- Lee, M.S. Molecular phylogenies become functional. Trends Ecol. Evol. 1999, 14, 177–178. [Google Scholar] [CrossRef]
- Kendall, M.; Colijn, C. Mapping phylogenetic trees to reveal distinct patterns of evolution. Mol. Biol. Evol. 2016, 33, 2735–2743. [Google Scholar] [CrossRef]
- Phillipps, Q.; Phillipps, K. Phillipps’ Field Guide to the Mammals of Borneo and Their Ecology, 2nd ed.; John Beaufoy Publishing Ltd.: Oxford, UK, 2018; ISBN 978-1-912081-95-0. [Google Scholar]
- Rowe, N. Pictorial Guide to the Living Primates; Pogonias Press: East Hampton, NY, USA, 1996. [Google Scholar]
- Arnason, U.; Adegoke, J.A.; Gullberg, A.; Harley, E.H.; Janke, A.; Kullberg, M. Mitogenomic relationships of placental mammals and molecular estimates of their divergences. Gene 2008, 421, 37–51. [Google Scholar] [CrossRef]
- Jameson, N.M.; Hou, Z.C.; Sterner, K.N.; Weckle, A.; Goodman, M.; Steiper, M.E.; Wildman, D.E. Genomic data reject the hypothesis of a prosimian primate clade. J. Hum. Evol. 2011, 61, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Harrison, M.E.; Chivers, D.J. The orang-utan mating system and the unflanged male: A product of increased food stress during the late Miocene and Pliocene? J. Hum. Evol. 2007, 52, 275–293. [Google Scholar] [CrossRef] [PubMed]
- Knorr, G.; Lohmann, G. Climate warming during Antarctic ice sheet expansion at the Middle Miocene transition. Nat. Geosci. 2014, 7, 376–381. [Google Scholar] [CrossRef]
- Lewis, A.R.; Marchant, D.R.; Ashworth, A.C.; Hemming, S.R.; Machlus, M.L. Major middle Miocene global climate change: Evidence from East Antarctica and the Transantarctic Mountains. Geol. Soc. Am. Bull. 2007, 119, 1449–1461. [Google Scholar] [CrossRef]
- Huang, R.; Chen, J.; Huang, G. Characteristics and variations of the East Asian monsoon system and its impacts on climate disasters in China. Adv. Atmos. Sci. 2007, 24, 993–1023. [Google Scholar] [CrossRef]
- Yelo, B.A.G.; Cano, A.R.G.; Cantalapiedra, J.L.; Alcalde, G.M.; Sanisidro, O.; Oliver, A.; Hernández-Ballarín, V.; López-Guerrero, P.; Fraile, S.; Fernández, M.H. Palaeoenvironmental analysis of the Aragonian (middle Miocene) mammalian faunas from the Madrid Basin based on body-size structure. J. Iber. Geol. 2014, 40, 129–140. [Google Scholar]
- Yen, Y.L.; Billa, L.; Singh, A. Effect of climate change on seasonal monsoon in Asia and its impact on the variability of monsoon rainfall in Southeast Asia. Geosci. Front. 2014, 6, 817–823. [Google Scholar] [CrossRef]
- Cerling, T.E.; Wang, Y.; Quade, J. Expansion of C4 ecosystems as an indicator of global ecological change in the late Miocene. Nature 1993, 361, 344–345. [Google Scholar] [CrossRef]
- Ehleringer, J.R.; Cerling, T.E.; Helliker, B.R. C4 photosynthesis, atmospheric CO2 and climate. Oecologia 1997, 112, 285–299. [Google Scholar] [CrossRef] [PubMed]
- Ehleringer, J.R.; Cerling, T.E. C3 and C4 photosynthesis. In Encyclopedia of Global Environmental Change; Munn, T., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2002; pp. 186–190. ISBN 978-0471977964. [Google Scholar]
- Lewis, A.R.; Marchant, D.R.; Ashworth, A.C.; Hedenas, L.; Hemming, S.R.; Johnson, J.V.; Leng, M.J.; Machlus, M.L.; Newton, A.E.; Raine, J.I.; et al. Mid-Miocene cooling and the extinction of tundra in continental Antarctica. Proc. Natl. Acad. Sci. USA 2008, 105, 10676–10680. [Google Scholar] [CrossRef]
- Wich, S.A.; Schaik, C.P.V. The impact of El Nino on mast fruiting in Sumatra and elsewhere in Malesia. J. Trop. Ecol. 2000, 16, 563–577. [Google Scholar] [CrossRef]
- Wright, S.J.; Carrasco, C.; Calderon, O.; Paton, S. The El Nino Southern Oscillation, variable fruit production, and famine in a tropical forest. Ecology 1999, 80, 1632–1647. [Google Scholar] [CrossRef]
- Jablonski, N.G.; Whitfort, M.J.; Roberts-Smith, N.; Qinqi, X. The influence of life history and diet on the distribution of catarrhine primates during the Pleistocene in eastern Asia. J. Hum. Evol. 2000, 39, 131–157. [Google Scholar] [CrossRef] [PubMed]
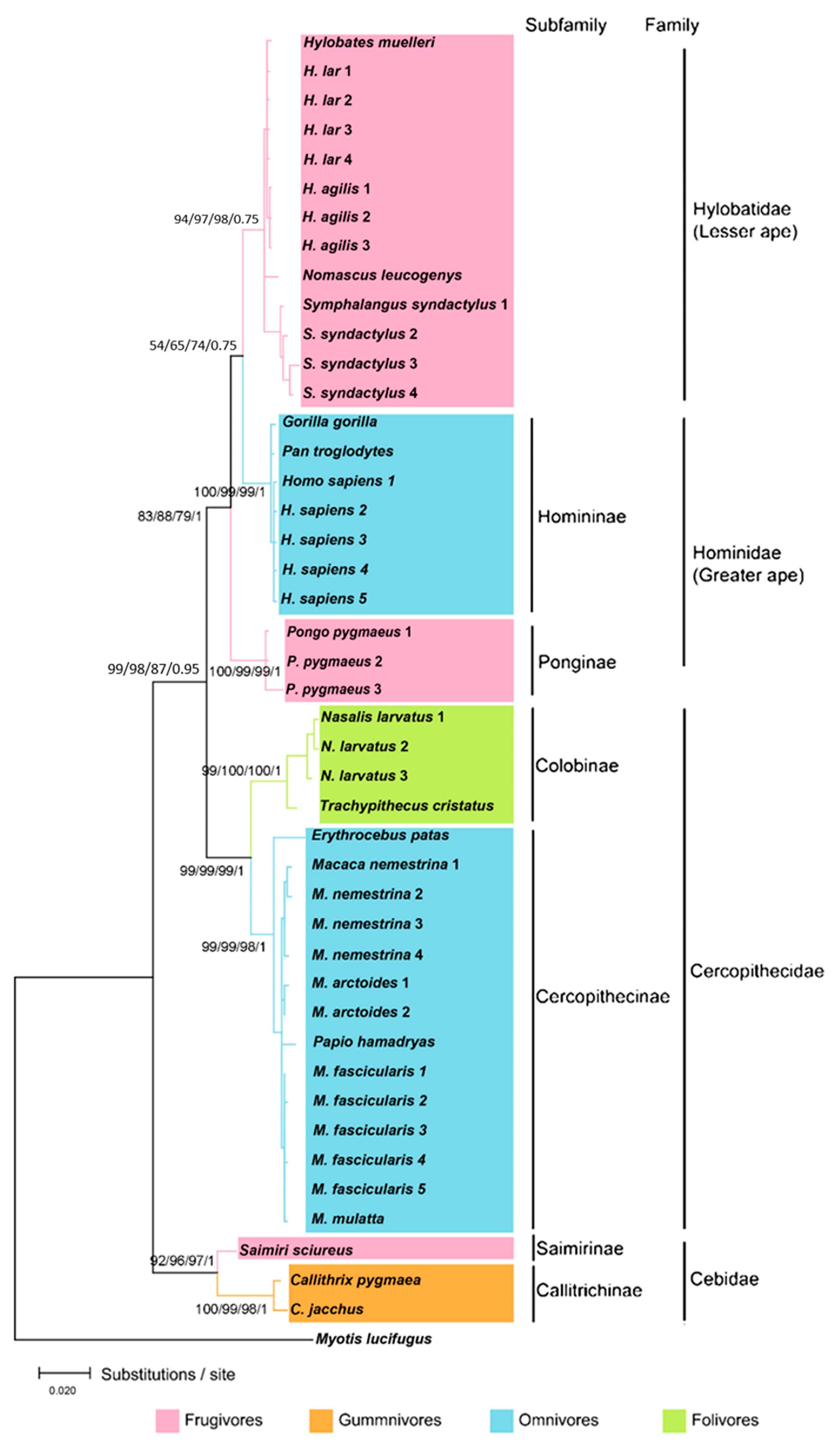
| Dietary Groups | Primate | Preferred Diet |
|---|---|---|
| Gummivore | Marmosets | Gum, sap, latex, and resin |
| Folivore | Langurs, snub-nosed monkeys, proboscis monkeys | Shoots, young leaves, and unripe fruits |
| Omnivore | Macaques, tarsiers, slow lorises, humans, baboons, Patas monkeys, gorillas, chimpanzees | Flexible diets including fruits, leaves, insects, bird eggs, small mammals, and amphibians |
| Frugivore | Squirrel monkeys, siamangs, gibbons, orangutans | Ripe fleshy fruits and mature leaves |
| Divergence (Node) | [33] | [34] | [35] | [20] | [36] | [37] | [19] | [22] | [38] |
|---|---|---|---|---|---|---|---|---|---|
| Crown Anthropoidea (A) | 42.8 (40.1–45) | 37.7 (33.3–42.7) | 45.3 (39.4–51.3) | 43.5 (38.6–48.4) | 47.2 (38.9–56.5) | 40.6 (33.6–49.5) | 45.3 (40.7–50.1) | 42.9 (37.3–52.4) | 46.7 (42.4–50.8) |
| Crown Catarrhini (B) | 29.3 (28–30) | 23.9 (23.1–25.9) | 30.5 (28.8–35.3) | 31.6 (25.7–37.9) | 31.0 (25.1–37.7) | 25.1 (19.7–32.8) | 31.9 (28.3–35.7) | 30.5 (26.9–36.4) | 32.1 (29.4–33.8) |
| Crown Platyrrhini (C) | 26.6 (23.5–30) | 14.5 (9.7–19.9) | N/A | 24.8 (20.6–29.3) | 25.1 (20.1–31.0) | 23.3 (19.2–27.5) | 22.0 (19.2–24.4) | 20.8 (18.2–24.9) | 20.9 (17.9–24.4) |
| Crown Hominoidea (D) | 21.5 (19–24.3) | 18.6 (17.1–20.5) | 19.9 (16.7–23) | 20.3 (16.6–24.2) | 19.2 (15.1–24.1) | 17.4 (12.4–23.9) | 20.3 (17.4–23.5) | 18.3 (16.3–20.8) | 22.3 (20.5–23.9) |
| Homo-Pan (E) | N/A | N/A | N/A | 6.5 (6.0–7.0) | 7.5 (5.7–9.6) | N/A | 5.9 | 6.6 (6.0–7.0) | 5.0 |
| Crown Cercopithecinae (F) | 23.4 (22–24.9) | 13.3 (11.6–14.7) | N/A | 17.6 (13.9–21.5) | 14.1 (11.0–17.7) | 13.2 (8.9–18.3) | 22.8 (20.0–25.6) | 9.9 (8.9–11.9) | 20.8 (18.6–22.9) |
| No | Taxon | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | 21 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Hylobates lar | 0.00 | ||||||||||||||||||||
| 2 | H. agilis | 0.13 | 0.00 | |||||||||||||||||||
| 3 | H. muelleri | 0.13 | 0.26 | NA | ||||||||||||||||||
| 4 | S. syndactylus | 0.93 | 1.06 | 1.06 | 0.00 | |||||||||||||||||
| 5 | Nomascus leucogenys | 0.66 | 0.80 | 0.79 | 1.34 | NA | ||||||||||||||||
| 6 | H. sapiens | 2.20 | 2.19 | 2.35 | 2.92 | 2.35 | 0.00 | |||||||||||||||
| 7 | P. troglodytes | 2.20 | 2.19 | 2.35 | 2.92 | 2.65 | 0.26 | NA | ||||||||||||||
| 8 | G. gorilla | 2.20 | 2.19 | 2.35 | 2.92 | 2.65 | 0.26 | 0.26 | NA | |||||||||||||
| 9 | P. pygmaeus | 2.96 | 3.10 | 3.11 | 3.41 | 3.43 | 3.42 | 3.11 | 3.43 | 0.01 | ||||||||||||
| 10 | N. larvatus | 6.60 | 6.76 | 6.78 | 7.53 | 6.79 | 6.97 | 7.34 | 7.34 | 7.61 | 0.00 | |||||||||||
| 11 | T. cristatus | 5.48 | 5.64 | 5.65 | 6.37 | 6.00 | 5.83 | 6.18 | 6.18 | 6.44 | 1.39 | NA | ||||||||||
| 12 | M. nemestrina | 5.60 | 5.75 | 5.77 | 6.14 | 5.78 | 5.60 | 5.95 | 5.95 | 6.09 | 4.12 | 3.43 | 0.00 | |||||||||
| 13 | M. fascicularis | 5.60 | 5.75 | 5.77 | 6.14 | 5.77 | 5.59 | 5.95 | 5.95 | 6.09 | 3.79 | 3.11 | 0.31 | 0.00 | ||||||||
| 14 | M. arctoides | 5.67 | 5.82 | 5.85 | 6.09 | 5.85 | 5.67 | 6.03 | 6.03 | 6.08 | 4.18 | 3.49 | 0.28 | 0.36 | 0.00 | |||||||
| 15 | M. mulatta | 5.56 | 5.72 | 5.74 | 6.10 | 5.74 | 5.56 | 5.92 | 5.92 | 6.05 | 3.76 | 3.08 | 0.31 | 0.03 | 0.35 | NA | ||||||
| 16 | P. hamadryas | 6.07 | 6.23 | 6.25 | 6.62 | 6.25 | 5.89 | 6.25 | 6.25 | 6.57 | 4.55 | 3.85 | 0.70 | 0.69 | 0.75 | 0.66 | NA | |||||
| 17 | E. patas | 6.74 | 6.90 | 6.93 | 7.31 | 6.93 | 6.74 | 7.12 | 7.12 | 7.26 | 4.85 | 4.15 | 1.81 | 1.79 | 1.86 | 1.76 | 2.19 | NA | ||||
| 18 | C. pygmaea | 9.05 | 9.22 | 9.25 | 10.37 | 9.66 | 10.07 | 9.65 | 10.07 | 10.16 | 11.50 | 10.99 | 10.95 | 10.96 | 11.05 | 10.92 | 10.96 | 11.90 | NA | |||
| 19 | C. jacchus | 9.11 | 9.28 | 9.31 | 10.43 | 9.31 | 10.35 | 9.93 | 10.35 | 10.23 | 11.14 | 11.06 | 11.03 | 11.04 | 11.13 | 10.99 | 11.17 | 11.98 | 0.79 | NA | ||
| 20 | S. sciureus | 7.79 | 7.96 | 7.98 | 8.37 | 7.98 | 8.97 | 8.57 | 8.97 | 8.07 | 10.35 | 9.45 | 8.80 | 8.81 | 8.89 | 8.77 | 8.94 | 9.69 | 3.08 | 3.37 | NA | |
| 21 | M. lucifugus | 20.56 | 20.79 | 20.86 | 21.08 | 20.86 | 22.36 | 22.06 | 22.34 | 23.37 | 24.24 | 23.48 | 23.88 | 23.31 | 24.04 | 23.31 | 23.88 | 25.38 | 19.97 | 19.78 | 20.35 | NA |
| Node | Relaxed Clock | Strict Clock | ||
|---|---|---|---|---|
| Date (mya) | Date (95% HPD) | Date (mya) | Date (95% HPD) | |
| A | 11.70 | 8.27–15.78 | 10.93 | 6.89–15.93 |
| B | 11.10 | 7.34–15.40 | 10.47 | 6.47–15.66 |
| C | 9.37 | 4.69–14.64 | 8.84 | 4.91–13.16 |
| D | 10.93 | 6.59–16.03 | 11.17 | 7.21–15.39 |
| E | 3.46 | 0.57–7.82 | 2.31 | 0.31–5.16 |
| F | 10.37 | 6.00–15.26 | 9.75 | 6.09–13.60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tamrin, N.A.M.; Zainudin, R.; Esa, Y.; Alias, H.; Isa, M.N.M.; Croft, L.; Abdullah, M.T. New Insights on the Evolution of the Sweet Taste Receptor of Primates Adapted to Harsh Environments. Animals 2020, 10, 2359. https://doi.org/10.3390/ani10122359
Tamrin NAM, Zainudin R, Esa Y, Alias H, Isa MNM, Croft L, Abdullah MT. New Insights on the Evolution of the Sweet Taste Receptor of Primates Adapted to Harsh Environments. Animals. 2020; 10(12):2359. https://doi.org/10.3390/ani10122359
Chicago/Turabian StyleTamrin, Nur Aida Md, Ramlah Zainudin, Yuzine Esa, Halimah Alias, Mohd Noor Mat Isa, Laurence Croft, and Mohd Tajuddin Abdullah. 2020. "New Insights on the Evolution of the Sweet Taste Receptor of Primates Adapted to Harsh Environments" Animals 10, no. 12: 2359. https://doi.org/10.3390/ani10122359
APA StyleTamrin, N. A. M., Zainudin, R., Esa, Y., Alias, H., Isa, M. N. M., Croft, L., & Abdullah, M. T. (2020). New Insights on the Evolution of the Sweet Taste Receptor of Primates Adapted to Harsh Environments. Animals, 10(12), 2359. https://doi.org/10.3390/ani10122359






