The Serum and Saliva Proteome of Dogs with Diabetes Mellitus
Abstract
Simple Summary
Abstract
1. Background
2. Results
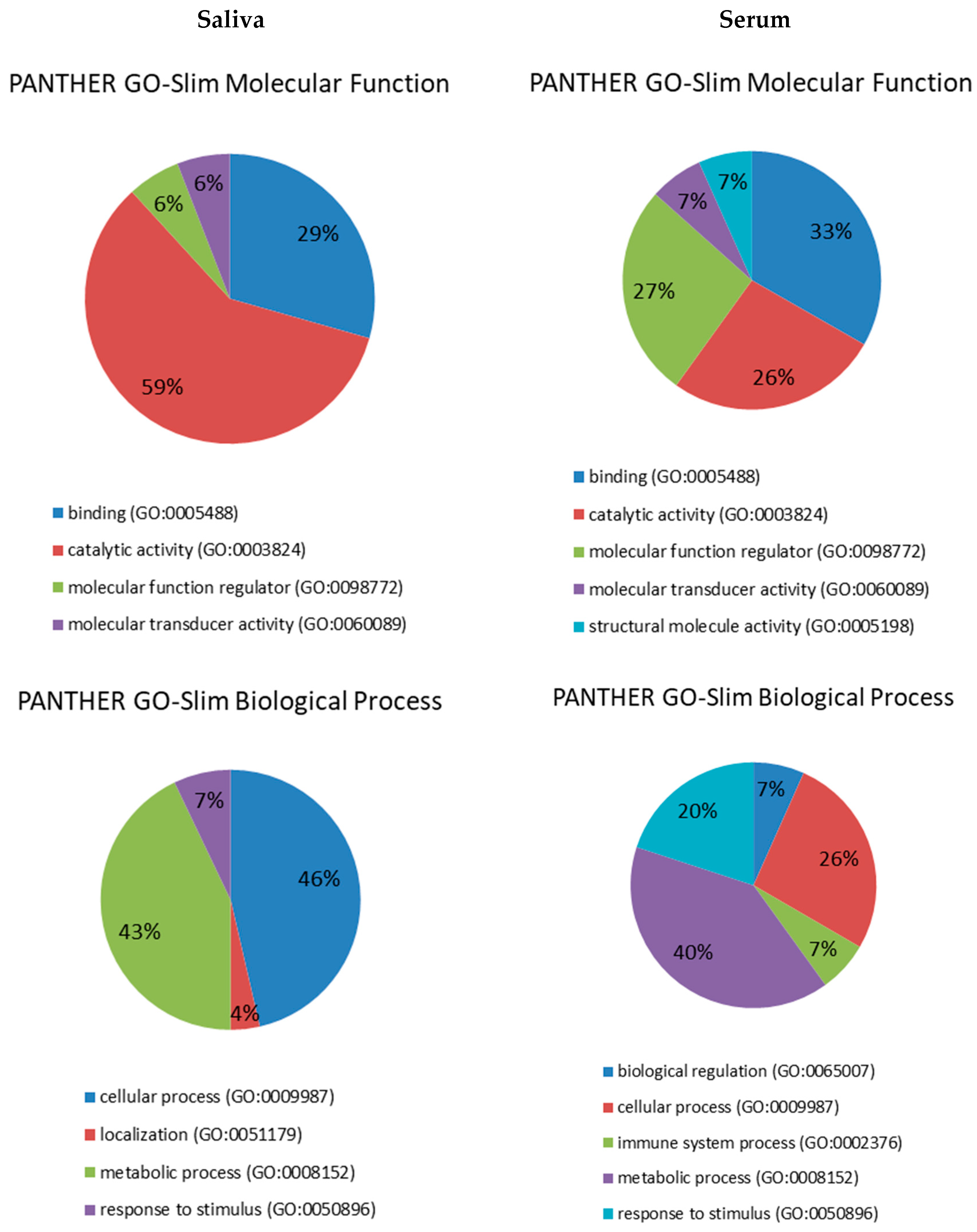
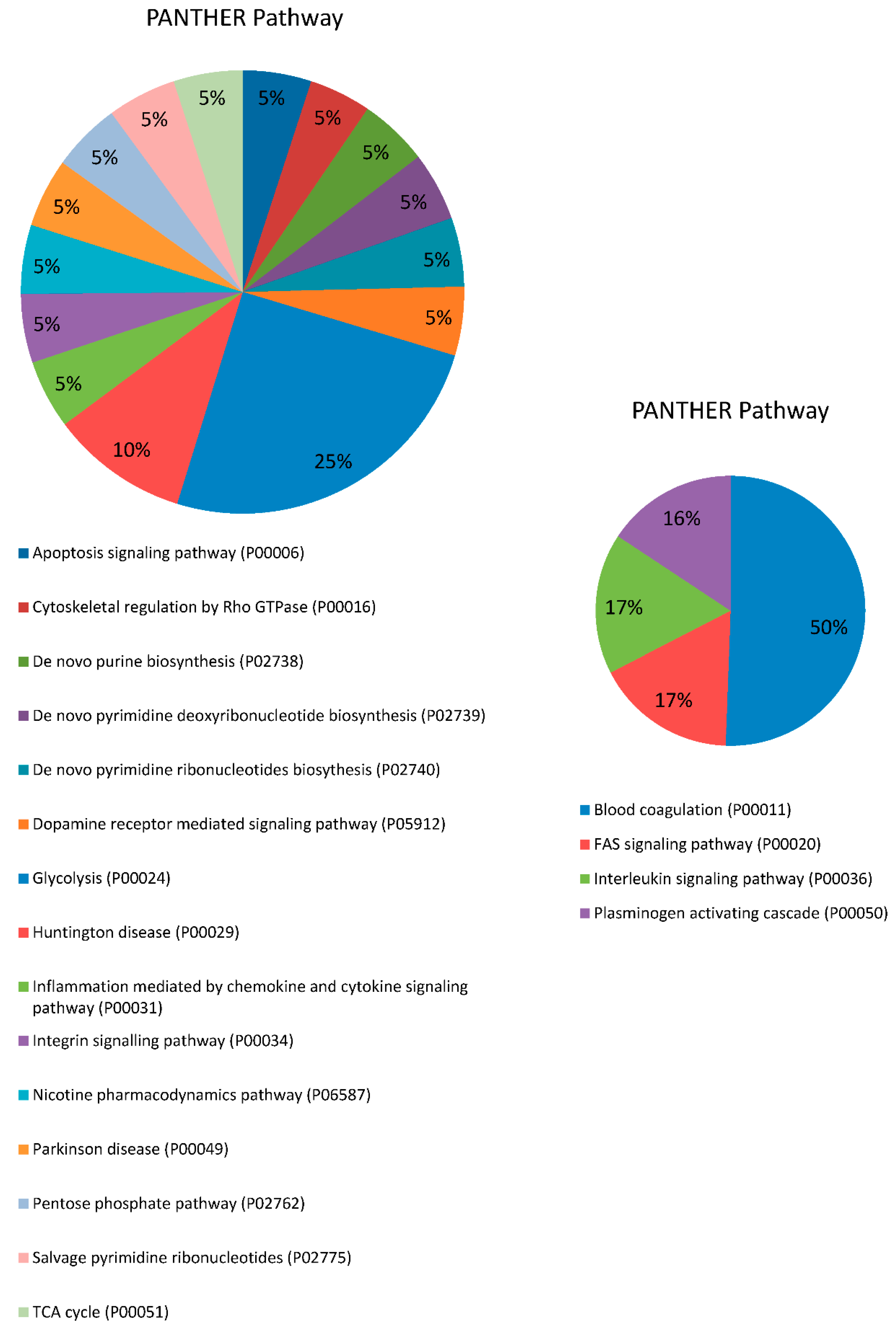
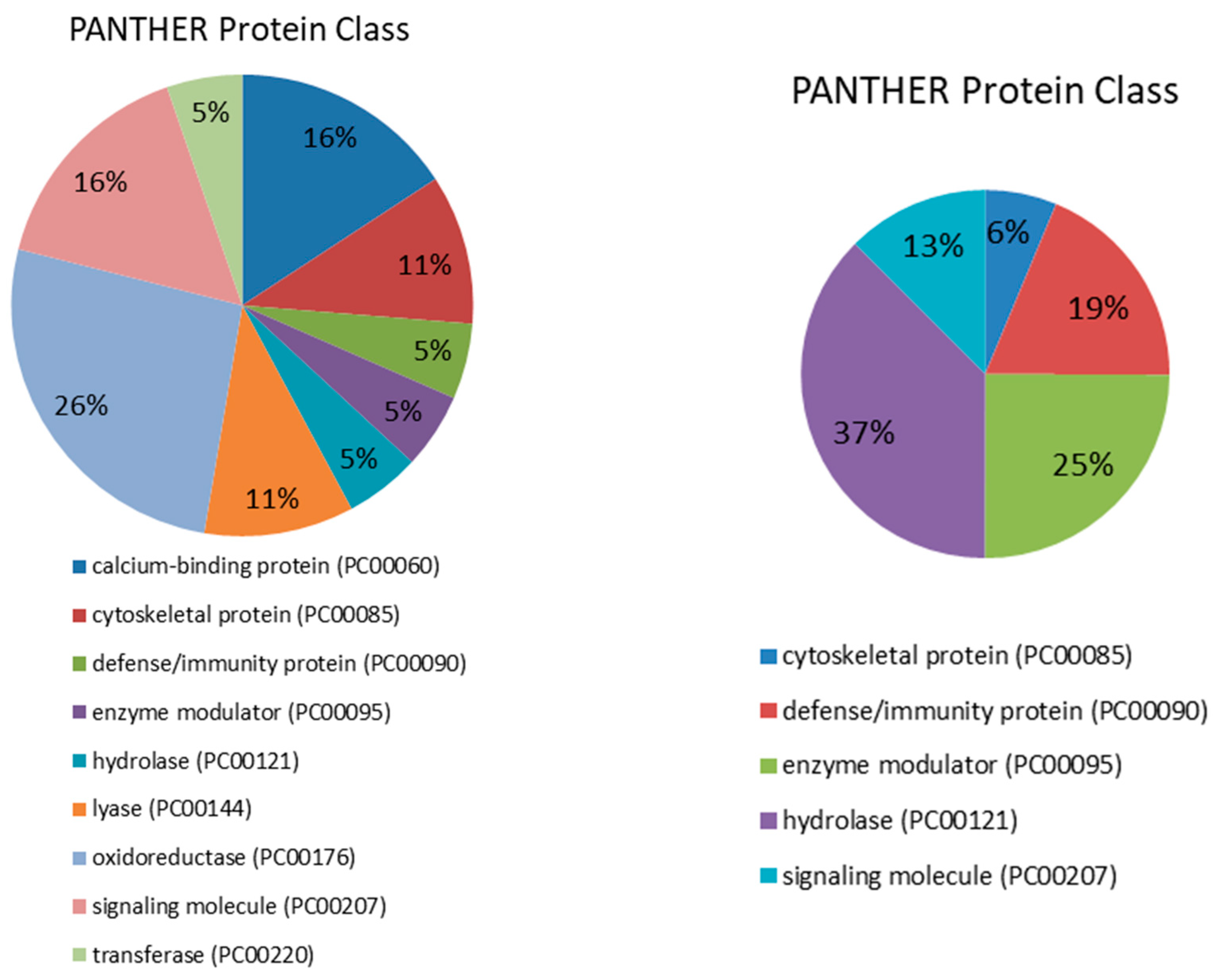
2.1. Proteomic Analysis in Serum
2.2. Proteomic Analysis in Saliva
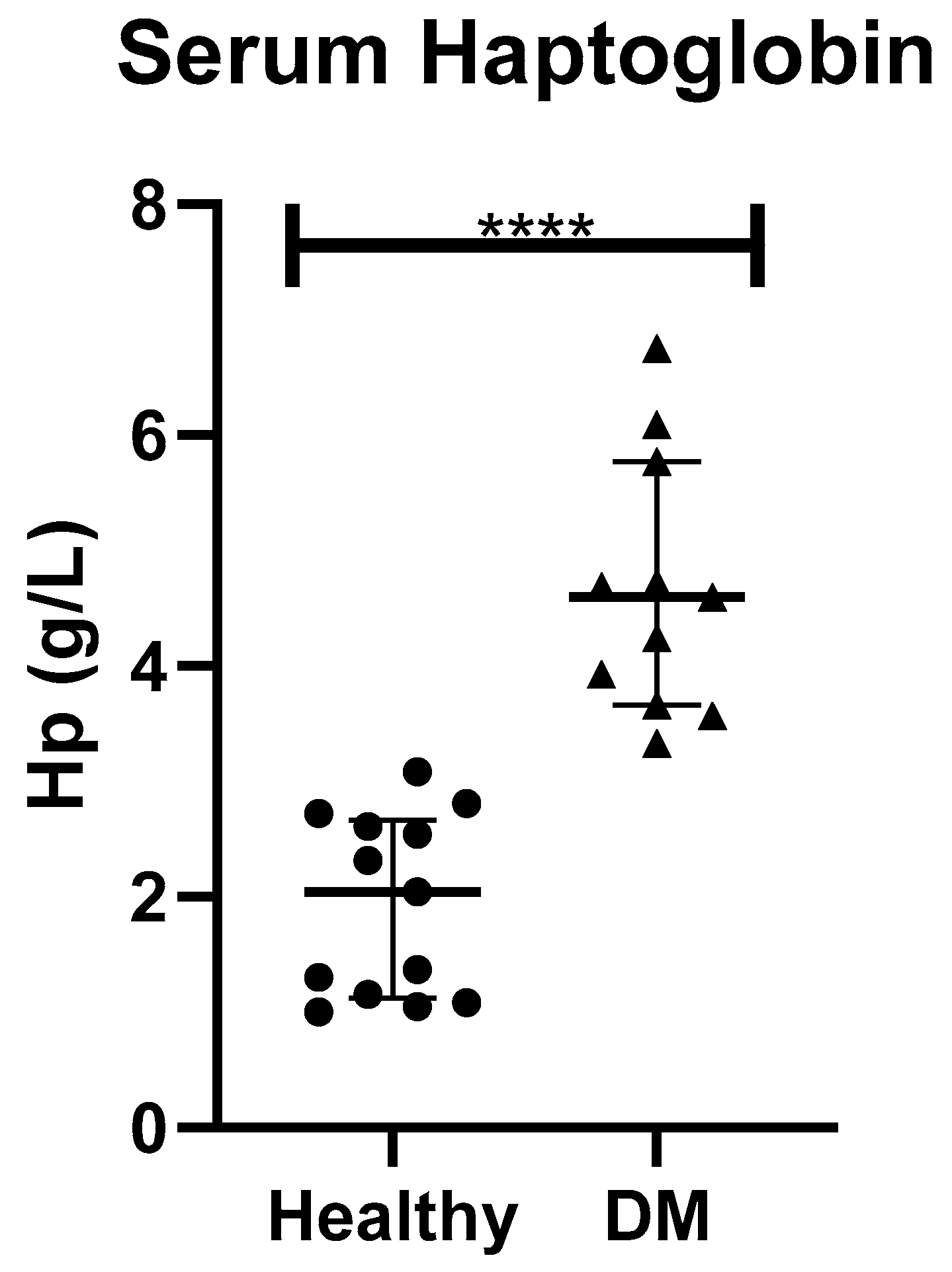
2.3. Validation of Proteomic Results
3. Discussion
4. Methods
4.1. Animals
4.2. Saliva and Serum Sampling
4.3. Proteomics Study of Saliva and Serum Samples and LC-MS/MS Analysis
4.4. Statistical Analysis
4.5. Validation of Proteomic Results
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Guptill, L.; Glickman, L. Time Trends and Risk Factors for Diabetes Mellitus in Dogs: Analysis of Veterinary Medical Data Base Records (1970–1999). Veter. J. 2003, 165, 240–247. [Google Scholar] [CrossRef]
- Davison, L.J.; Herrtage, M.E.; Catchpole, B. Study of 253 dogs in the United Kingdom with diabetes mellitus. Veter. Rec. 2005, 156, 467–471. [Google Scholar] [CrossRef] [PubMed]
- McAllister, M.; Breuninger, K.; Spofford, N.; Yang, M.; Trevejo, R. State of Pet Health 2016 Report. pp. 1–21. Available online: https://www.banfield.com/Banfield/media/PDF/Downloads/soph/Banfield-State-of-Pet-Health-Report-2016.pdf (accessed on 24 March 2020).
- Davison, L.J. Diabetes mellitus and pancreatitis-cause or effect? J. Small Anim. Pr. 2015, 56, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Kleinert, M.; Clemmensen, C.; Hofmann, S.M.; Moore, M.C.; Renner, S.; Woods, S.C.; Huypens, P.; Beckers, J.; De Angelis, M.H.; Schürmann, A.; et al. Animal models of obesity and diabetes mellitus. Nat. Rev. Endocrinol. 2018, 14, 140–162. [Google Scholar] [CrossRef] [PubMed]
- Moshref, M.; Tangey, B.; Gilor, C.; Papas, K.K.; Williamson, P.; Loomba-Albrecht, L.; Sheehy, P.; Kol, A. Concise Review: Canine Diabetes Mellitus as a Translational Model for Innovative Regenerative Medicine Approaches. Stem Cells Transl. Med. 2019, 8, 450–455. [Google Scholar] [CrossRef]
- Hess, R.S.; Saunders, H.M.; Van Winkle, T.J.; Ward, C.R. Concurrent disorders in dogs with diabetes mellitus: 221 cases (1993&1998). J. Am. Veter Med. Assoc. 2000, 217, 1166–1173. [Google Scholar] [CrossRef]
- Cerón, J.J. Acute phase proteins, saliva and education in laboratory science: An update and some reflections. BMC Veter Res. 2019, 15, 197. [Google Scholar] [CrossRef]
- Sivadasan, P.; Gupta, M.K.; Sathe, G.J.; Balakrishnan, L.; Palit, P.; Gowda, H.; Suresh, A.; Kuriakose, M.A.; Sirdeshmukh, R. Human salivary proteome—a resource of potential biomarkers for oral cancer. J. Proteom. 2015, 127, 89–95. [Google Scholar] [CrossRef]
- Kumar, S.; Padmashree, S.; Jayalekshmi, R. Correlation of salivary glucose, blood glucose and oral candidal carriage in the saliva of type 2 diabetics: A case-control study. Contemp. Clin. Dent. 2014, 5, 312. [Google Scholar] [CrossRef]
- Sashikumar, R.; Kannan, R. Salivary glucose levels and oral candidal carriage in type II diabetics. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontology 2010, 109, 706–711. [Google Scholar] [CrossRef]
- Gupta, S.; Sandhu, S.V.; Bansal, H.; Sharma, D. Comparison of Salivary and Serum Glucose Levels in Diabetic Patients. J. Diabetes Sci. Technol. 2014, 9, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.W.; Choi, J.-W.; Yun, J.W.; Chung, I.-S.; Cho, H.C.; Song, S.-E.; Im, S.-S.; Song, D.-K. Proteomics approach to identify serum biomarkers associated with the progression of diabetes in Korean patients with abdominal obesity. PLoS ONE 2019, 14, e0222032. [Google Scholar] [CrossRef] [PubMed]
- Pappa, E.; Vastardis, H.; Mermelekas, G.; Gerasimidi-Vazeou, A.; Zoidakis, J.; Vougas, K. Saliva Proteomics Analysis Offers Insights on Type 1 Diabetes Pathology in a Pediatric Population. Front. Physiol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Bencharit, S.; Baxter, S.S.; Carlson, J.; Byrd, W.C.; Mayo, M.V.; Border, M.B.; Kohltfarber, H.; Urrutia, E.; Howard-Williams, E.L.; Offenbacher, S.; et al. Salivary proteins associated with hyperglycemia in diabetes: A proteomic analysis. Mol. BioSyst. 2013, 9, 2785–2797. [Google Scholar] [CrossRef] [PubMed]
- Cabras, T.; Pisano, E.; Mastinu, A.; Denotti, G.; Pusceddu, P.P.; Inzitari, R.; Fanali, C.; Nemolato, S.; Castagnola, M.; Messana, I. Alterations of the Salivary Secretory Peptidome Profile in Children Affected by Type 1 Diabetes. Mol. Cell. Proteom. 2010, 9, 2099–2108. [Google Scholar] [CrossRef]
- Tvarijonaviciute, A.; Barić-Rafaj, R.; Horvatić, A.; Muñoz-Prieto, A.; Guillemin, N.; Lamy, E.; Tumpa, A.; Ceron, J.; Martínez-Subiela, S.; Mrljak, V. Identification of changes in serum analytes and possible metabolic pathways associated with canine obesity-related metabolic dysfunction. Veter. J. 2019, 244, 51–59. [Google Scholar] [CrossRef]
- Franco-Martínez, L.; Tvarijonaviciute, A.; Horvatić, A.; Guillemin, N.; Cerón, J.J.; Escribano, D.; Eckersall, D.; Kocaturk, M.; Yilmaz, Z.; Lamy, E.; et al. Changes in salivary analytes in canine parvovirus: A high-resolution quantitative proteomic study. Comp. Immunol. Microbiol. Infect. Dis. 2018, 60, 1–10. [Google Scholar] [CrossRef]
- Martínez-Subiela, S.; Horvatić, A.; Escribano, D.; Pardo-Marin, L.; Kocaturk, M.; Mrljak, V.; Burchmore, R.; Cerón, J.J.; Yilmaz, Z. Identification of novel biomarkers for treatment monitoring in canine leishmaniosis by high-resolution quantitative proteomic analysis. Veter. Immunol. Immunopathol. 2017, 191, 60–67. [Google Scholar] [CrossRef]
- Franco-Martínez, L.; Tvarijonaviciute, A.; Horvatić, A.; Guillemin, N.; Bernal, L.J.; Barić Rafaj, R.; Ceron, J.J.; Thomas, M.d.C.; Lopez, M.C.; Tecles, F.; et al. Changes in saliva of dogs with canine leishmaniosis: A proteomic approach. Vet. Parasitol. 2019, 272, 44–52. [Google Scholar] [CrossRef]
- Franco-Martínez, L.; Villar, M.; Tvarijonaviciute, A.; Escribano, D.; Bernal, L.J.; Cerón, J.J.; Thomas, M.D.C.; Mateos-Hernández, L.; Tecles, F.; De La Fuente, J.; et al. Serum proteome of dogs at subclinical and clinical onset of canine leishmaniosis. Transbound. Emerg. Dis. 2020, 67, 318–327. [Google Scholar] [CrossRef]
- Loo, J.; Yan, W.; Ramachandran, P.; Wong, D. Comparative Human Salivary and Plasma Proteomes. J. Dent. Res. 2010, 89, 1016–1023. [Google Scholar] [CrossRef] [PubMed]
- Franco-Martínez, L.; Gelemanović, A.; Horvatić, A.; Contreras-Aguilar, M.D.; Dabrowski, R.; Mrljak, V.; Ceron, J.J.; Martinez-Subiela, S.; Tvarijonaviciute, A. Changes in Serum and Salivary Proteins in Canine Mammary Tumors. Animals 2020, 10, 741. [Google Scholar] [CrossRef] [PubMed]
- Cerón, J.J.; Eckersall, P.D.; Martínez-Subiela, S. Acute phase proteins in dogs and cats: Current knowledge and future perspectives. Veter. Clin. Pathol. 2005, 34, 85–99. [Google Scholar] [CrossRef] [PubMed]
- Ricart, W.; López-Bermejo, A.; Ricart, W. Cross-Talk Between Iron Metabolism and Diabetes. Diabetes 2002, 51, 2348–2354. [Google Scholar] [CrossRef]
- McMillan, D.E. Increased levels of acute-phase serum proteins in diabetes. Metabolism 1989, 38, 1042–1046. [Google Scholar] [CrossRef]
- Awadallah, R.; El-Dessoukey, E.A.; Doss, H.; Khalifa, K.; Elhawary, Z. Blood-reduced glutathione, serum ceruloplasmin and mineral changes in juvenile diabetes. Eur. J. Nutr. 1978, 17, 79–83. [Google Scholar] [CrossRef]
- Golizeh, M.; Lee, K.; Ilchenko, S.; Ösme, A.; Bena, J.; Sadygov, R.G.; Kashyap, S.R.; Kasumov, T. Increased serotransferrin and ceruloplasmin turnover in diet-controlled patients with type 2 diabetes. Free. Radic. Biol. Med. 2017, 113, 461–469. [Google Scholar] [CrossRef]
- Lim, Y.K.; Jenner, A.; Ali, A.; Wang, Y.; Hsu, S.I.-H.; Chong, S.M.; Baumman, H.; Halliwell, B.; Lim, S.-K. Haptoglobin reduces renal oxidative DNA and tissue damage during phenylhydrazine-induced hemolysis. Kidney Int. 2000, 58, 1033–1044. [Google Scholar] [CrossRef]
- Kruger, A.; Yang, C.; Tam, S.W.; Hinerfeld, D.; Evans, J.E.; Green, K.M.; Leszyk, J.; Yang, K.; Guberski, D.L.; Mordes, J.P.; et al. Haptoglobin as an early serum biomarker of virus-induced autoimmune type 1 diabetes in biobreeding diabetes resistant and LEW1.WR1 rats. Exp. Biol. Med. 2010, 235, 1328–1337. [Google Scholar] [CrossRef]
- Sadrzadeh, S.H.; Bozorgmehr, J. Haptoglobin Phenotypes in Health and Disorders. Pathol. Patterns Rev. 2004, 121, S97–S104. [Google Scholar] [CrossRef]
- Hess, R.S.; Kass, P.H.; Van Winkle, T.J. Association between Diabetes Mellitus, Hypothyroidism or Hyperadrenocorticism, and Atherosclerosis in Dogs. J. Vet. Intern. Med. 2003, 17, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Gleeson, J.M.; Hejazi, J.S.; Kwong, L.; Chan, I.F.; Le, T.; Alberts, A.W.; Wilson, D.E. Plasma apolipoprotein E, high density lipoprotein (HDL) and urinary mevalonate excretion in pancreatectomized diabetic dogs: Effects of insulin and lovastatin. Atherosclerosis 1990, 84, 1–12. [Google Scholar] [CrossRef]
- Seage, E.; Drobatz, K.; Hess, R.S. Spectrophotometry and Ultracentrifugation for Measurement of Plasma Lipids in Dogs with Diabetes Mellitus. J. Vet. Intern. Med. 2017, 32, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Rassart, E.; Bedirian, A.; Do Carmo, S.; Guinard, O.; Sirois, J.; Terrisse, L.; Milne, R. Apolipoprotein D. Biochim. Biophys. Acta. 2000, 1482, 185–198. [Google Scholar] [CrossRef]
- Safaei, A.; Tavirani, M.R.; Azodi, M.Z.; Lashay, A.; Mohammadi, S.-F.; Broumand, M.G.; Peyvandi, A.; Okhovatian, F.; Peyvandi, H.; Rostami-Nejad, M. Diabetic Retinopathy and Laser Therapy in Rats: A Protein-Protein Interaction Network Analysis. J. Lasers Med. Sci. 2017, 8, S20–S21. [Google Scholar] [CrossRef]
- Karthik, D.; Ilavenil, S.; Kaleeswaran, B.; Sunil, S.; Ravikumar, S. Proteomic Analysis of Plasma Proteins in Diabetic Rats by 2D Electrophoresis and MALDI-TOF-MS. Appl. Biochem. Biotechnol. 2012, 166, 1507–1519. [Google Scholar] [CrossRef]
- Yamada, Y.; Oguri, M.; Kato, K.; Yokoi, K.; Watanabe, S.; Metoki, N.; Yoshida, H.; Satoh, K.; Aoyagi, Y.; Nishigaki, Y.; et al. Association of polymorphisms of THBS2 and HSPA8 with hypertension in Japanese individuals with chronic kidney disease. Mol. Med. Rep. 2009, 2, 205–211. [Google Scholar] [CrossRef][Green Version]
- Priyanka, M.; Jeyaraja, K.; Thirunavakkarasu, P.S. Abnormal renovascular resistance in dogs with diabetes mellitus: Correlation with glycemic status and proteinuria. Iran. J. Veter. Res. 2018, 19, 304–309. [Google Scholar]
- Herring, I.; Panciera, D.; Werre, S. Longitudinal Prevalence of Hypertension, Proteinuria, and Retinopathy in Dogs with Spontaneous Diabetes Mellitus. J. Veter. Intern. Med. 2014, 28, 488–495. [Google Scholar] [CrossRef]
- Aluksanasuwan, S.; Sueksakit, K.; Fong-Ngern, K.; Thongboonkerd, V. Role of HSP60 (HSPD1) in diabetes-induced renal tubular dysfunction: Regulation of intracellular protein aggregation, ATP production, and oxidative stress. FASEB J. 2017, 31, 2157–2167. [Google Scholar] [CrossRef]
- Mayer, M.P.; Bukau, B. Hsp70 chaperones: Cellular functions and molecular mechanism. Cell. Mol. Life Sci. 2005, 62, 670–684. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, Q.; Lin, H.; Li, S.; Sun, L.; Yang, Y. HSP72 and gp96 in gastroenterological cancers. Clin. Chim. Acta 2013, 417, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-Y.; Chou, H.-C.; Chen, Y.-H.; Chan, H.-L. High glucose-induced proteome alterations in hepatocytes and its possible relevance to diabetic liver disease. J. Nutr. Biochem. 2013, 24, 1889–1910. [Google Scholar] [CrossRef] [PubMed]
- Sreekumar, R.; Halvatsiotis, P.; Schimke, J.C.; Nair, K.S. Gene Expression Profile in Skeletal Muscle of Type 2 Diabetes and the Effect of Insulin Treatment. Diabetes 2002, 51, 1913–1920. [Google Scholar] [CrossRef]
- Caseiro, A.; Ferreira, R.; Padrão, A.; Quintaneiro, C.; Pereira, A.; Marinheiro, R.; Vitorino, R.; Amado, F. Salivary Proteome and Peptidome Profiling in Type 1 Diabetes Mellitus Using a Quantitative Approach. J. Proteome Res. 2013, 12, 1700–1709. [Google Scholar] [CrossRef]
- Lim, R.R.; Vaidya, T.; Gadde, S.G.; Yadav, N.K.; Sethu, S.; Hainsworth, D.P.; Mohan, R.R.; Ghosh, A.; Chaurasia, S.S. Correlation between systemic S100A8 and S100A9 levels and severity of diabetic retinopathy in patients with type 2 diabetes mellitus. Diabetes Metab. Syndr. Clin. Res. Rev. 2019, 13, 1581–1589. [Google Scholar] [CrossRef]
- Dong, N.; Shi, H.; Xu, B.; Cai, Y. Increased Plasma S100A12 Levels Are Associated with Diabetic Retinopathy and Prognostic Biomarkers of Macrovascular Events in Type 2 Diabetic Patients. Investig. Opthalmology Vis. Sci. 2015, 56, 4177. [Google Scholar] [CrossRef]
- Shan, J.; Sun, Z.; Yang, J.; Xu, J.; Shi, W.; Wu, Y.; Fan, Y.; Li, H.-Q. Discovery and preclinical validation of proteomic biomarkers in saliva for early detection of oral squamous cell carcinomas. Oral Dis. 2018, 25, 97–107. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, J. Expression of S100A11 is a Prognostic Factor for Disease-free Survival and Overall Survival in Patients with High-grade Serous Ovarian Cancer. Appl. Immunohistochem. Mol. Morphol. 2017, 25, 110–116. [Google Scholar] [CrossRef]
- Gabril, M.; Girgis, H.; Scorilas, A.; Rotondo, F.; Wala, S.; Bjarnason, G.A.; Ding, Q.; Evans, A.; Tawedrous, E.; Pasic, M.; et al. S100A11 is a potential prognostic marker for clear cell renal cell carcinoma. Clin. Exp. Metastasis 2015, 33, 63–71. [Google Scholar] [CrossRef]
- Sabatini, P.V.; Speckmann, T.; Lynn, F.C. Friend and foe: Beta-cell Ca2+ signaling and the development of diabetes. Mol. Metab. 2019, 21, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Epstein, P.N.; Overbeek, P.A.; Means, A.R. Calmodulin-induced early-onset diabetes in transgenic mice. Cell 1989, 58, 1067–1073. [Google Scholar] [CrossRef]
- Qu, J.; Mei, Q.; Niu, R. Oxidative CaMKII as a potential target for inflammatory disease (Review). Mol. Med. Rep. 2019, 20, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Bartelt, D.C.; Wolff, D.J.; Scheele, G.A. Calmodulin-binding proteins and calmodulin-regulated enzymes in dog pancreas. Biochem. J. 1986, 240, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Westermeier, R.; Naven, T.; Hopker, H.R. Proteomics in Practice: A Guide to Successful Experimental Design; Wiley-VCH: Hoboken, NJ, USA, 2008. [Google Scholar]
- Jacobsen, S.; Adler, D.; Bundgaard, L.; Sørensen, M.A.; Andersen, P.H.; Bendixen, E. The use of liquid chromatography tandem mass spectrometry to detect proteins in saliva from horses with and without systemic inflammation. Veter. J. 2014, 202, 483–488. [Google Scholar] [CrossRef]
- Horvatić, A.; Guillemin, N.; Kaab, H.; McKeegan, D.; O’Reilly, E.; Bain, M.; Kuleš, J.; Eckersall, P.D. Quantitative proteomics using tandem mass tags in relation to the acute phase protein response in chicken challenged with Escherichia coli lipopolysaccharide endotoxin. J. Proteom. 2019, 192, 64–77. [Google Scholar] [CrossRef]
- Heeley, A.M.; O’Neill, D.G.; Davison, L.J.; Church, D.B.; Corless, E.K.; Brodbelt, D.C. Diabetes mellitus in dogs attending UK primary-care practices: Frequency, risk factors and survival. Canine Med. Genet. 2020, 7, 1–19. [Google Scholar] [CrossRef]
- Lucena, S.; Coelho, A.; Capela-Silva, F.; Tvarijonaviciute, A.; Lamy, E. The Effect of Breed, Gender, and Acid Stimulation in Dog Saliva Proteome. BioMed Res. Int. 2018, 2018, 1–12. [Google Scholar] [CrossRef]
- Hong, H.-R.; Oh, Y.; Kim, Y.J.; Seo, K.W. Salivary alpha-amylase as a stress biomarker in diseased dogs. J. Veter. Sci. 2019, 20, e46. [Google Scholar] [CrossRef]
- Iacopetti, I.; Perazzi, A.; Badon, T.; Bedin, S.; Contiero, B.; Ricci, R. Salivary pH, calcium, phosphorus and selected enzymes in healthy dogs: A pilot study. BMC Veter. Res. 2017, 13, 330. [Google Scholar] [CrossRef]
- Durocher, L.L.; Hinchcliff, K.; DiBartola, S.P.; Johnson, S.E. Acid-base and hormonal abnormalities in dogs with naturally occurring diabetes mellitus. J. Am. Veter. Med Assoc. 2008, 232, 1310–1320. [Google Scholar] [CrossRef]
- Parra, M.D.; Tecles, F.; Subiela, S.M.; Cerón, J.J. C-Reactive Protein Measurement in Canine Saliva. J. Veter. Diagn. Investig. 2005, 17, 139–144. [Google Scholar] [CrossRef]
- Komsta, L. Package Outliers. 2015. Available online: https://www.google.com.hk/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwiQ3P365avtAhVQ1VkKHUxcAIMQFjAAegQIBBAC&url=https%3A%2F%2Fcran.r-project.org%2Fweb%2Fpackages%2Foutliers%2Foutliers.pdf&usg=AOvVaw1OsjFhE2OldfePZSksPWSa (accessed on 26 October 2020).
- R Core Team. R: A Language and Environment for Statistical Computing; R Found Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2009. [Google Scholar]
- Kolde, R. Pheatmap: Pretty Heatmaps in R Package. R Packag Version 61. 2012. Available online: https://rdrr.io/cran/pheatmap/ (accessed on 26 October 2020).
- Doncheva, N.T.; Morris, J.H.; Gorodkin, J.; Jensen, L.J. Cytoscape StringApp: Network Analysis and Visualization of Proteomics Data. J. Proteome Res. 2019, 18, 623–632. [Google Scholar] [CrossRef]
- Eckersall, P.D.; Duthie, S.; Safi, S.; Moffatt, D.; Horadagoda, N.U.; Doyle, S.; Perton, R.; Bennett, D.; Fitzpatrick, J.L. An automated biochemical assay for haptoglobin: Prevention of interference from albumin. Comp. Haematol. Int. 1999, 9, 117–124. [Google Scholar] [CrossRef]
| Gene Symbol | Description | p-Value | Fold Change | Mean (SD) Healthy | Mean (SD) Diseased |
|---|---|---|---|---|---|
| Proteins down-regulated in DM when compared to HC | |||||
| IL13RA2 | Interleukin-13 receptor subunit alpha-2 precursor [Canis lupus familiaris] | 0.016 | 0.426 | 0.941 (1.062) | 0.569 (0.032) |
| IGHG | Immunoglobulin gamma heavy chain D [Canis lupus familiaris] | 0.016 | 0.426 | 0.941 (1.062) | 0.569 (0.032) |
| IGLL | Immunoglobulin lambda-1 light chain [Canis lupus familiaris] | 0.016 | 0.519 | 1.235 (0.037) | 0.754 (0.173) |
| VPREB1 | Immunoglobulin iota chain-like [Canis lupus familiaris] | 0.016 | 0.538 | 1.179 (0.06) | 0.673 (0.138) |
| TFRC | Transferrin receptor [Canis lupus familiaris] | 0.016 | 0.664 | 1.149 (0.048) | 0.715 (0.2) |
| GSN | Gelsolin [Canis lupus familiaris] | 0.032 | 0.671 | 1.191 (0.117) | 0.795 (0.103) |
| IGHV | Immunoglobulin heavy chain variable region [Canis lupus familiaris] | 0.032 | 0.718 | 1.029 (0.085) | 0.884 (0.315) |
| ITIH2 | Inter-alpha-trypsin inhibitor heavy chain H2 [Canis lupus familiaris] | 0.008 | 0.735 | 1.076 (0.112) | 0.916 (0.15) |
| ITIH4 | Inter-alpha-trypsin inhibitor heavy chain H4 [Canis lupus familiaris] | 0.032 | 0.796 | 0.876 (0.071) | 0.704 (0.056) |
| ITIH1 | Inter-alpha-trypsin inhibitor heavy chain H1 [Canis lupus familiaris] | 0.032 | 0.809 | 1.124 (0.043) | 0.881 (0.064) |
| A2M | Alpha-2-macroglobulin [Canis lupus familiaris] | 0.008 | 0.813 | 1.031 (0.076) | 0.896 (0.157) |
| APOD | Apolipoprotein D [Canis lupus familiaris] | 0.032 | 0.841 | 0.999 (0.144) | 0.86 (0.072) |
| PLG | Plasminogen [Canis lupus familiaris] | 0.032 | 0.883 | 1.09 (0.071) | 0.944 (0.034) |
| Proteins up-regulated in DM when compared to HC | |||||
| C4BPB | C4b-binding protein beta chain [Canis lupus dingo] | 0.032 | 1.341 | 1.03 (0.126) | 1.343 (0.146) |
| MGAM | Maltase-glucoamylase, intestinal [Canis lupus familiaris] | 0.016 | 1.367 | 0.83 (0.026) | 1.061 (0.242) |
| HP | Haptoglobin heavy chain [dogs, Peptide, 245 aa] | 0.032 | 1.584 | 0.537 (0.15) | 0.972 (0.21) |
| Gene Symbol | Description | p-Value | Fold Change | Mean (IQR) Healthy | Mean (IQR) Diseased |
|---|---|---|---|---|---|
| Proteins down-regulated in DM when compared to HC | |||||
| A1BG | Alpha-1B-glycoprotein [Canis lupus familiaris] | 0.016 | 0.438 | 0.932 (0.541) | 0.498 (0.038) |
| KLK1 | Kallikrein-1-like [Canis lupus dingo] | 0.029 | 0.452 | 1.599 (0.094) | 0.753 (0.194) |
| ENO2 | Gamma-enolase [Canis lupus dingo] | 0.032 | 0.469 | 1.661 (1.274) | 1.121 (0.335) |
| CANF2 | Precursor Can f II [Canis lupus familiaris] | 0.031 | 0.47 | 2.459 (0.632) | 0.981 (0.367) |
| GAPDH | Glyceraldehyde-3-phosphate dehydrogenase [Canis lupus familiaris] | 0.016 | 0.667 | 1.058 (0.467) | 0.824 (0.004) |
| Proteins up-regulated in DM when compared to HC | |||||
| MDH1 | Malate dehydrogenase, cytoplasmic [Canis lupus familiaris] | 0.016 | 1.313 | 0.657 (0.118) | 0.91 (0.123) |
| IPSG | Double-headed protease inhibitor, submandibular gland [Canis lupus familiaris] | 0.029 | 1.39 | 0.698 (0.092) | 0.854 (0.206) |
| TPI1 | Triosephosphate isomerase | 0.032 | 1.445 | 0.851 (0.194) | 0.985 (0.113) |
| TKT | Transketolase [Canis lupus familiaris] | 0.032 | 1.463 | 0.648 (0.397) | 1.061 (0.086) |
| CALM3 | Calmodulin-3 [Canis lupus dingo] | 0.016 | 1.584 | 0.788 (0.199) | 1.088 (0.054) |
| NME2 | Nucleoside diphosphate kinase B [Canis lupus familiaris] | 0.016 | 1.624 | 0.547 (0.248) | 1.052 (0.139) |
| HSPA8 | Heat shock cognate 71 kDa protein [Canis lupus familiaris] | 0.032 | 1.666 | 0.585 (0.39) | 1.081 (0.175) |
| YWHAE | 14-3-3 protein epsilon [Canis lupus familiaris] | 0.032 | 1.671 | 0.672 (0.254) | 1.135 (0.17) |
| LCP1 | Plastin-2 [Canis lupus familiaris] | 0.016 | 1.684 | 0.538 (0.622) | 1.111 (0.161) |
| YWHAQ | 14-3-3 protein theta [Canis lupus dingo] | 0.032 | 1.702 | 0.785 (0.286) | 1.191 (0.193) |
| CALM2 | Calmodulin-2 [Canis lupus dingo] | 0.008 | 1.822 | 0.88 (0.298) | 1.341 (0.488) |
| S100A2 | Protein S100-A2 [Canis lupus dingo] | 0.016 | 1.840 | 0.541 (0.151) | 0.962 (0.03) |
| MDH2 | Malate dehydrogenase, mitochondrial-like [Canis lupus familiaris] | 0.008 | 1.969 | 0.447 (0.139) | 0.88 (0.281) |
| CANF1 | Major allergen Can f 1 | 0.016 | 1.978 | 0.492 (0.041) | 0.88 (0.461) |
| FLNA | Filamin-A [Canis lupus familiaris] | 0.032 | 2.102 | 0.464 (0.437) | 1.163 (0.117) |
| GSTA4 | Glutathione S-transferase A4-like [Canis lupus familiaris] | 0.032 | 2.105 | 0.43 (0.19) | 1.082 (0.258) |
| PTMA | Prothymosin alpha [Canis lupus familiaris] | 0.032 | 2.348 | 0.432 (0.155) | 1.056 (0.286) |
| S100A11 | Protein S100-A11 [Canis lupus familiaris] | 0.032 | 2.451 | 0.396 (0.246) | 1.136 (0.528) |
| ARPC4 | Actin-related protein 2/3 complex subunit 4 [Canis lupus familiaris] | 0.016 | 2.723 | 0.246 (0.384) | 1.161 (0.118) |
| S100A9 | Protein S100-A9 [Canis lupus familiaris] | 0.032 | 2.856 | 0.254 (0.442) | 1.327 (0.417) |
| S100A12 | Protein S100-A12-like [Canis lupus dingo] | 0.032 | 2.963 | 0.268 (0.526) | 1.199 (0.45) |
| ID | Sex | Age (y) | Breed | Body Weight (kg)/Diet | Glucose (mg/dL) | Fructosamine (µmol/L) | Clinical Signs |
|---|---|---|---|---|---|---|---|
| DM1 | Female | 12 | Mixed breed | 9.5/Dry food | 320 | 387 | Diabetic Retinopathy PU-PD-PF |
| DM2 | Female (neutered) | 10 | Mixed breed | 14.5/Dry food | 337 | 561 | Diabetic Retinopathy PU-PD-PF |
| DM3 | Male (neutered) | 6.5 | Mixed breed | 6/Dry food | 582 | 372 | PU-PD-PF |
| DM4 | Male | 10 | Mixed breed | 20/Dry food | 277 | 497 | PU-PD-PF |
| DM5 | Female | 6.5 | Yorkshire Terrier | 3/Dry food | 212 | 401 | PU-PD-PF |
| H1 | Male | 7 | Poodle | 5.7/mixed | 103 | 267 | - |
| H2 | Male (neutered) | 13 | Mixed breed | 14/Dry food | 98 | 181 | - |
| H3 | Female | 6 | German Shepherd | 28/Dry food | 78 | 239 | - |
| H4 | Female | 2 | Mixed breed | 15/Dry food | 82 | 152 | - |
| H5 | Female (neutered) | 5 | Mixed breed | 4/Dry food | 106 | 226 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franco-Martínez, L.; Gelemanović, A.; Horvatić, A.; Contreras-Aguilar, M.D.; Mrljak, V.; Cerón, J.J.; Martínez-Subiela, S.; Tvarijonaviciute, A. The Serum and Saliva Proteome of Dogs with Diabetes Mellitus. Animals 2020, 10, 2261. https://doi.org/10.3390/ani10122261
Franco-Martínez L, Gelemanović A, Horvatić A, Contreras-Aguilar MD, Mrljak V, Cerón JJ, Martínez-Subiela S, Tvarijonaviciute A. The Serum and Saliva Proteome of Dogs with Diabetes Mellitus. Animals. 2020; 10(12):2261. https://doi.org/10.3390/ani10122261
Chicago/Turabian StyleFranco-Martínez, Lorena, Andrea Gelemanović, Anita Horvatić, María Dolores Contreras-Aguilar, Vladimir Mrljak, José Joaquín Cerón, Silvia Martínez-Subiela, and Asta Tvarijonaviciute. 2020. "The Serum and Saliva Proteome of Dogs with Diabetes Mellitus" Animals 10, no. 12: 2261. https://doi.org/10.3390/ani10122261
APA StyleFranco-Martínez, L., Gelemanović, A., Horvatić, A., Contreras-Aguilar, M. D., Mrljak, V., Cerón, J. J., Martínez-Subiela, S., & Tvarijonaviciute, A. (2020). The Serum and Saliva Proteome of Dogs with Diabetes Mellitus. Animals, 10(12), 2261. https://doi.org/10.3390/ani10122261








