Growth, Metabolite, Antioxidative Capacity, Transcriptome, and the Metabolome Response to Dietary Choline Chloride in Pacific White Shrimp Litopenaeus vannamei
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Diets
2.2. Growth Trial and Sampling
- (1)
- Survival (%) = 100% × final shrimp number/initial shrimp number.
- (2)
- Weight gain (%) = 100% × (final weight (g) − initial weight (g))/initial weight (g).
- (3)
- Specific growth rate (% day−1) = 100% × (ln (final weight) − ln (initial weight))/days.
- (4)
- Condition factor (%) = 100% × final weight (g)/(body length (cm))3.
- (5)
- Hepatosomatic index (%) = 100% × wet hepatopancreas weight (g)/wet body weight (g).
2.3. Proximate Composition Analysis
2.4. Biochemical Assay
2.5. Untargeted Metabolomic Analysis
2.5.1. Sample Preparation
2.5.2. LC-MS Analysis
2.5.3. Metabolomic Data Analysis
2.6. Transcriptomics Analysis
2.7. Statistical Analysis
3. Results
3.1. Growth Performance and Whole-Body Proximate Composition
3.2. Serum Metabolites and Hepatopancreatic Digestive Enzymes
3.3. MDA and Antioxidant Parameters
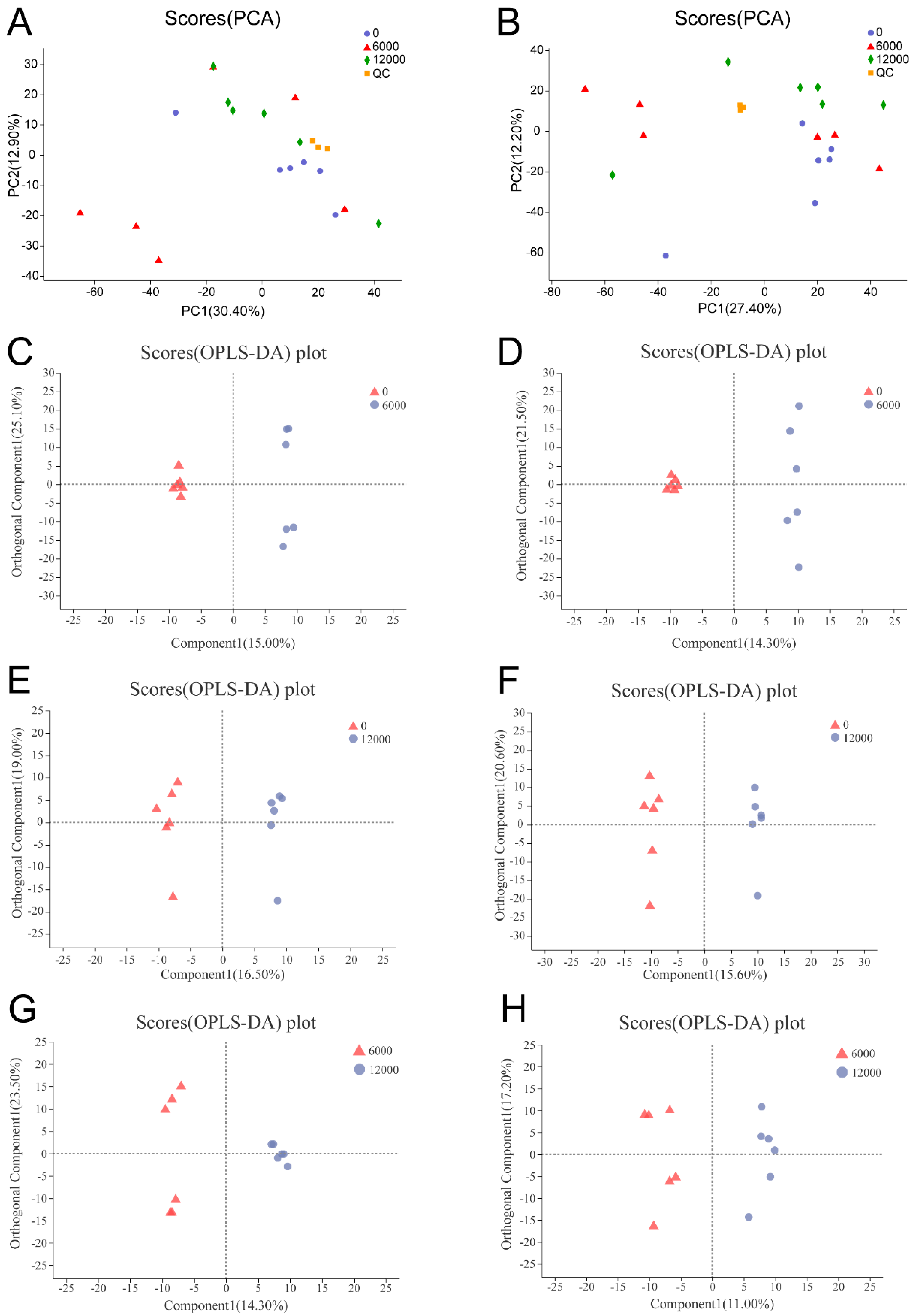
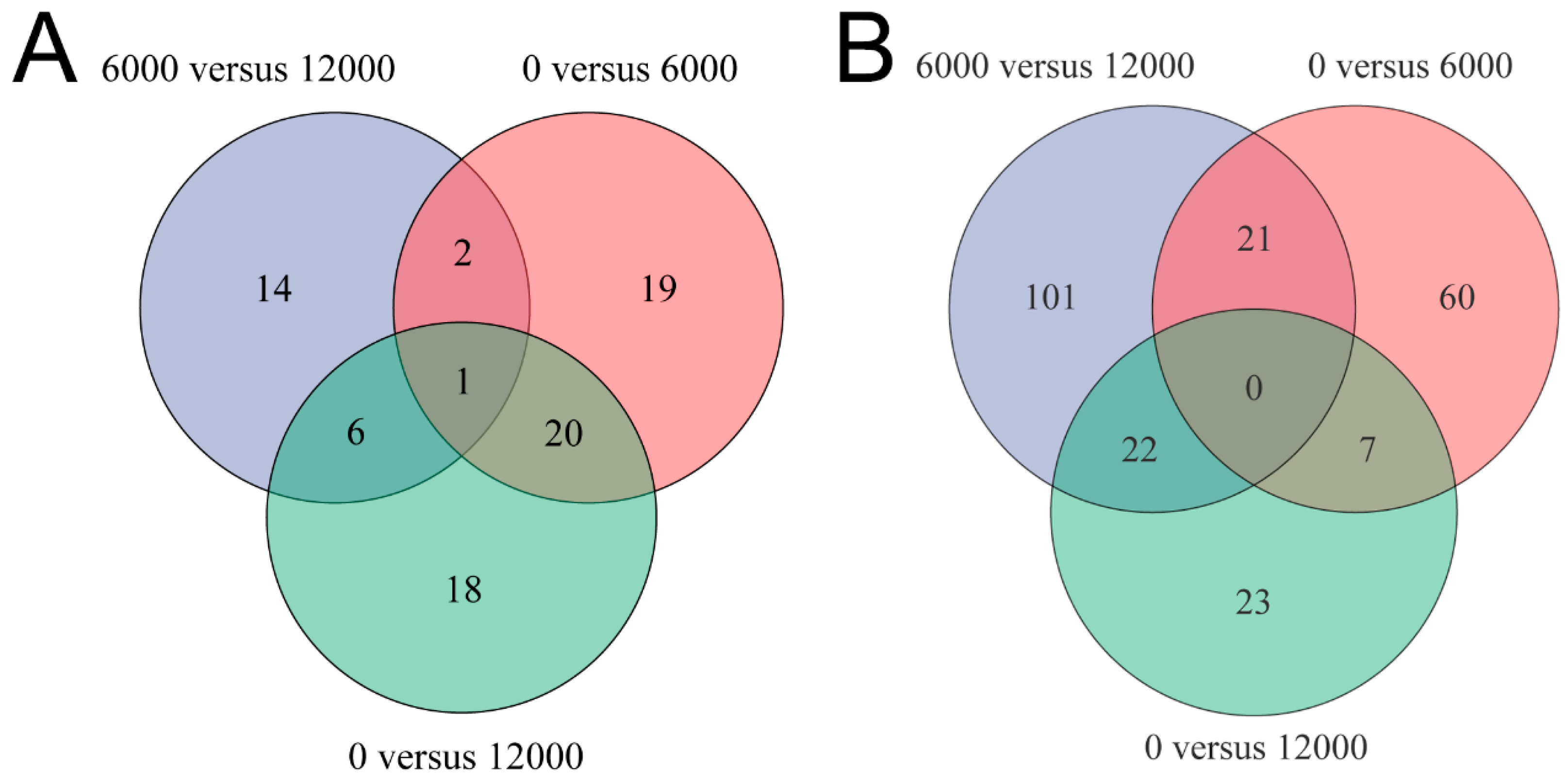
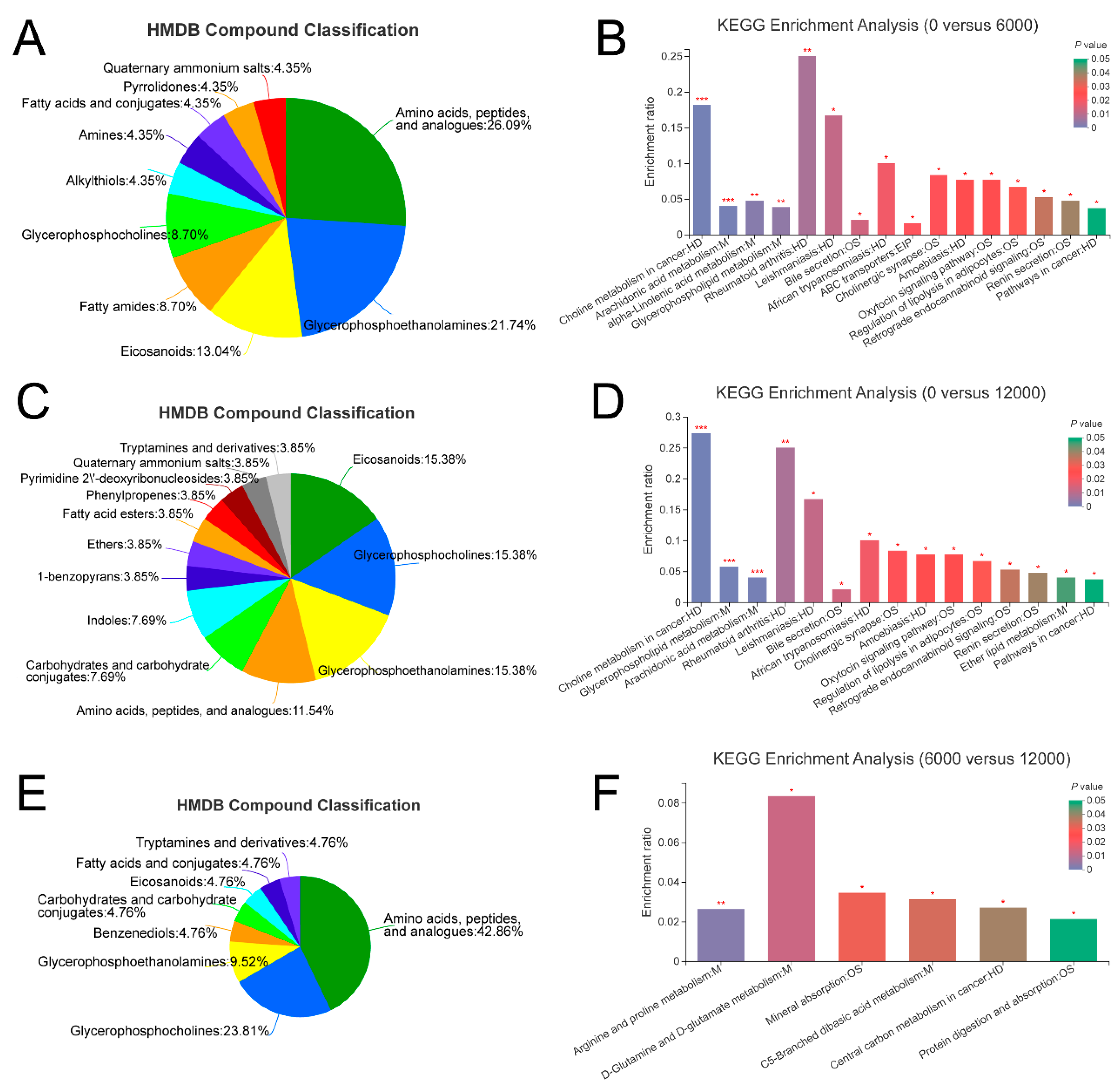
3.4. Metabolome Analysis
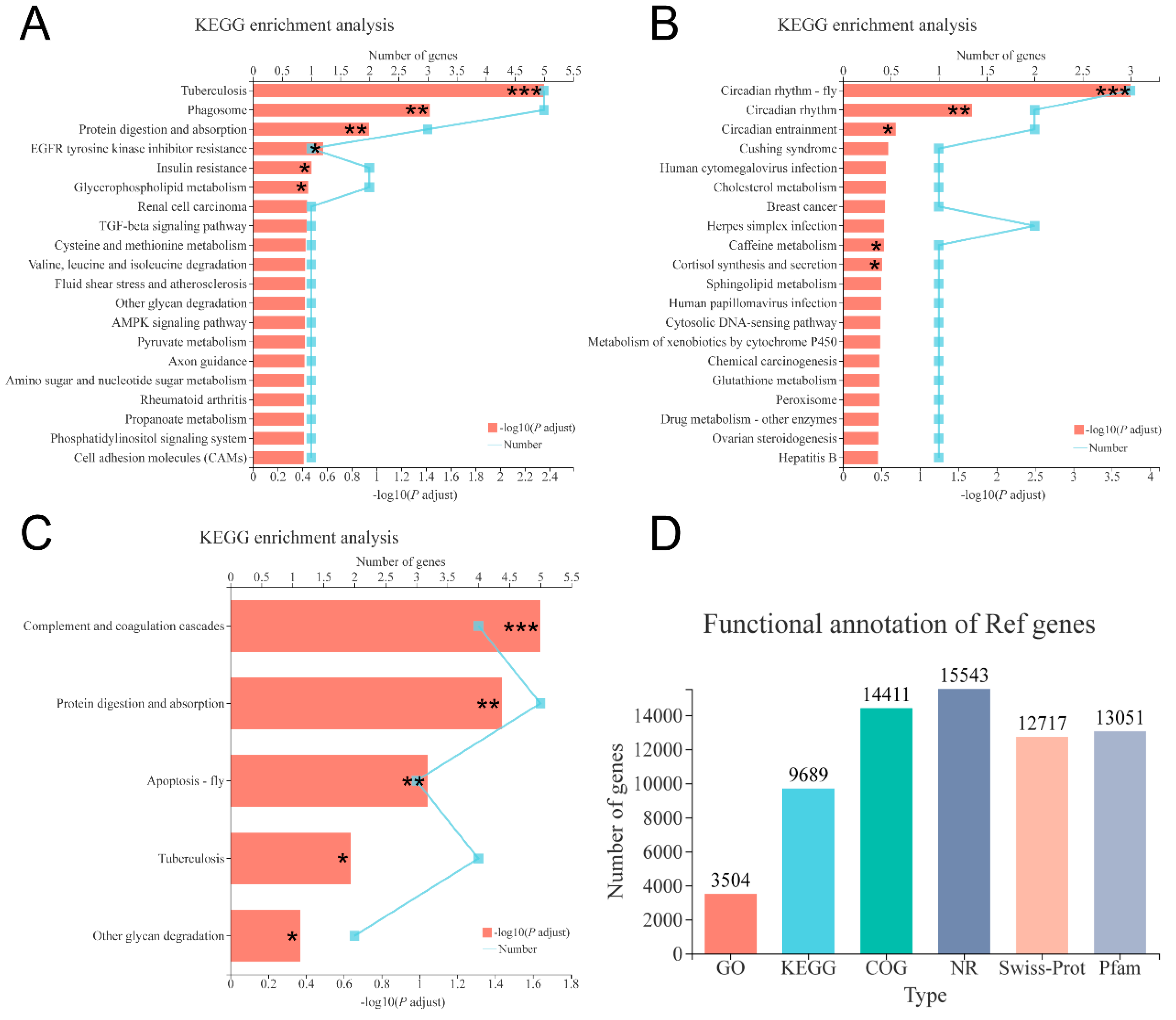
3.5. Transcriptome Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Craig, S.R.; Gatlin, D.M., III. Dietary choline requirement of juvenile red drum (Sciaenops ocellatus). J. Nutr. 1996, 126, 1696–1700. [Google Scholar] [CrossRef] [PubMed]
- Shiau, S.; Lo, P. Dietary choline requirement of juvenile grass shrimp (Penaeus monodon). Anim. Sci. 2001, 72, 477–482. [Google Scholar] [CrossRef]
- Shiau, S.Y.; Lo, P.S. Dietary choline requirements of juvenile hybrid tilapia, Oreochromis niloticus × O. aureus. J. Nutr. 2000, 130, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Michael, F.R.; Teshima, S.I.; Koshio, S.; Ishikawa, M.; Uyan, O.; Alam, M.S. Effects of water-soluble and fat-soluble choline sources on the performances of juvenile kuruma shrimp, Marsupenaeus japonicus Bate. Aquac. Res. 2005, 36, 1563–1571. [Google Scholar] [CrossRef]
- Wu, P.; Jiang, W.D.; Liu, Y.; Chen, G.F.; Jiang, J.; Li, S.H.; Feng, L.; Zhou, X.Q. Effect of choline on antioxidant defenses and gene expressions of Nrf2 signaling molecule in the spleen and head kidney of juvenile Jian carp (Cyprinus carpio var. Jian). Fish Shellfish Immunol. 2014, 38, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Michael, F.; Teshima, S.; Koshio, S.; Ishikawa, M.; Uyan, O.; Ren, T. Effect of two choline sources on the performance of postlarval Marsupenaeus japonicus bate. Aquac. Nutr. 2007, 13, 59–64. [Google Scholar] [CrossRef]
- Michael, F.; Koshio, S. Effect of choline chloride as an osmoregulator as well as its role in growth and the biochemical content of postlarval kuruma shrimp; Marsupenaeus japonicus (Bate). Aquac. Nutr. 2016, 22, 597–605. [Google Scholar] [CrossRef]
- Deshimaru, Q.; Kuroki, K. Requirement of prawn for dietary thiamine, pyridoxine, and choline chloride. Nippon Suisan Gakk. 1979, 45, 363–367. [Google Scholar] [CrossRef]
- Shiau, S.Y.; Cho, W.H. Choline requirements of grass shrimp (Penaeus monodon) as affected by dietary lipid level. Anim. Sci. 2002, 75, 97–102. [Google Scholar] [CrossRef]
- Griffin, M.E.; Wilson, K.A.; White, M.R.; Brown, P.B. Dietary choline requirement of juvenile hybrid striped bass. J. Nutr. 1994, 124, 1685–1689. [Google Scholar] [CrossRef]
- Michael, F.R.; Koshio, S.; Teshima, S.I.; Ishikawa, M.; Uyan, O. Effect of choline and methionine as methyl group donors on juvenile kuruma shrimp, Marsupenaeus japonicus Bate. Aquaculture 2006, 258, 521–528. [Google Scholar] [CrossRef]
- Yeh, S.P.; Shiu, P.J.; Guei, W.C.; Lin, Y.H.; Liu, C.H. Improvement in lipid metabolism and stress tolerance of juvenile giant grouper, Epinephelus lanceolatus (Bloch), fed supplemental choline. Aquac. Res. 2015, 46, 1810–1821. [Google Scholar] [CrossRef]
- Baldissera, M.D.; Souza, C.F.; Baldisserotto, B.; Zimmer, F.; Paiano, D.; Petrolli, T.G.; Da Silva, A.S. Vegetable choline improves growth performance, energetic metabolism, and antioxidant capacity of fingerling Nile tilapia (Oreochromis niloticus). Aquaculture 2019, 501, 224–229. [Google Scholar] [CrossRef]
- Li, E.; Wang, X.; Chen, K.; Xu, C.; Qin, J.G.; Chen, L. Physiological change and nutritional requirement of Pacific white shrimp Litopenaeus vannamei at low salinity. Rev. Aquac. 2017, 9, 57–75. [Google Scholar] [CrossRef]
- Gong, H.; Lawrence, A.L.; Jiang, D.H.; Gatlin, D.M., III. Effect of dietary phospholipids on the choline requirement of Litopenaeus vannamei juveniles. J. World Aquac. Soc. 2003, 34, 289–299. [Google Scholar] [CrossRef]
- Gong, H.; Lawrence, A.L.; Jiang, D.H.; Castille, F.L.; Gatlin, D.M. Lipid nutrition of juvenile Litopenaeus vannamei: I. Dietary cholesterol and de-oiled soy lecithin requirements and their interaction. Aquaculture 2000, 190, 305–324. [Google Scholar] [CrossRef]
- Zeisel, S.H.; Mar, M.H.; Howe, J.C.; Holden, J.M. Concentrations of choline-containing compounds and betaine in common foods. J. Nutr. 2003, 133, 1302–1307. [Google Scholar] [CrossRef]
- Graham, S.F.; Hollis, J.H.; Migaud, M.; Browne, R.A. Analysis of betaine and choline contents of aleurone, bran, and flour fractions of wheat (Triticum aestivum L.) using 1H nuclear magnetic resonance (NMR) spectroscopy. J. Agric. Food Chem. 2009, 57, 1948–1951. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists. Official Methods of Analysis, 16th ed.; Association of Official Analytical Chemists: Arlington, VA, USA, 1995. [Google Scholar]
- McDonald, J.H. Handbook of Biological Statistics, 3rd ed.; Sparky House Publishing: Baltimore, MD, USA, 2014. [Google Scholar]
- Shi, B.; Hu, X.; Jin, M.; Xia, M.; Zhao, M.; Jiao, L.; Sun, P.; Zhou, Q. Dietary choline improves growth performance, antioxidant ability and reduces lipid metabolites in practical diet for juvenile Pacific white shrimp, Litopenaeus vannamei. Aquac. Nutr. 2020. [Google Scholar] [CrossRef]
- Kasper, C.S.; White, M.R.; Brown, P.B. Betaine can replace choline in diets for juvenile Nile Tilapia, Oreochromis niloticus. Aquaculture 2002, 205, 119–126. [Google Scholar] [CrossRef]
- Shi, X.Z.; Li, X.C.; Wang, S.; Zhao, X.F.; Wang, J.X. Transcriptome analysis of hemocytes and hepatopancreas in red swamp crayfish, Procambarus clarkii, challenged with white spot syndrome virus. ISJ Invert. Surviv. J. 2010, 7, 119–131. [Google Scholar]
- Wang, W.N.; Zhou, J.; Wang, P.; Tian, T.T.; Zheng, Y.; Liu, Y.; Mai, W.J.; Wang, A.L. Oxidative stress, DNA damage and antioxidant enzyme gene expression in the Pacific white shrimp, Litopenaeus vannamei when exposed to acute pH stress. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2009, 150, 428–435. [Google Scholar] [CrossRef]
- Liu, G.; Zhu, S.; Liu, D.; Guo, X.; Ye, Z. Effects of stocking density of the white shrimp Litopenaeus vannamei (Boone) on immunities, antioxidant status, and resistance against Vibrio harveyi in a biofloc system. Fish Shellfish Immunol. 2017, 67, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.; Liu, Y.; Jiang, W.D.; Jiang, J.; Zhao, J.; Zhang, Y.A.; Zhou, X.Q.; Feng, L. A comparative study on antioxidant system in fish hepatopancreas and intestine affected by choline deficiency: Different change patterns of varied antioxidant enzyme genes and Nrf2 signaling factors. PLoS ONE 2017, 12, e0169888. [Google Scholar] [CrossRef] [PubMed]
- Grattagliano, I.; Vendemiale, G.; Caraceni, P.; Domenicali, M.; Nardo, B.; Cavallari, A.; Trevisani, F.; Bernardi, M.; Altomare, E. Starvation impairs antioxidant defense in fatty livers of rats fed a choline-deficient diet. J. Nutr. 2000, 130, 2131–2136. [Google Scholar] [CrossRef] [PubMed]
- Arun, S.; Subramanian, P. Antioxidant enzymes in freshwater prawn Macrobrachium malcolmsonii during embryonic and larval development. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1998, 121, 273–277. [Google Scholar] [CrossRef]
- Campa-Córdova, A.I.; Hernández-Saavedra, N.Y.; Ascencio, F. Superoxide dismutase as modulator of immune function in American white shrimp (Litopenaeus vannamei). Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2002, 133, 557–565. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, W.N.; Wang, A.L.; He, W.Y.; Zhou, Q.T.; Liu, Y.; Xu, J. Glutathione S-transferase in the white shrimp Litopenaeus vannamei: Characterization and regulation under pH stress. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2009, 150, 224–230. [Google Scholar] [CrossRef]
- Saito, H.; Ishihara, K. Antioxidant activity and active sites of phospholipids as antioxidants. J. Am. Oil Chem. Soc. 1997, 74, 1531–1536. [Google Scholar] [CrossRef]
- Corbin, K.D.; Zeisel, S.H. Choline metabolism provides novel insights into non-alcoholic fatty liver disease and its progression. Curr. Opin. Gastroenterol. 2012, 28, 159. [Google Scholar] [CrossRef]
- Yao, Z.; Vance, D.E. The active synthesis of phosphatidylcholine is required for very low density lipoprotein secretion from rat hepatocytes. J. Biol. Chem. 1988, 263, 2998–3004. [Google Scholar] [PubMed]
- Ma, P.T.; Gil, G.; Südhof, T.C.; Bilheimer, D.W.; Goldstein, J.L.; Brown, M.S. Mevinolin, an inhibitor of cholesterol synthesis, induces mRNA for low density lipoprotein receptor in livers of hamsters and rabbits. Proc. Natl. Acad. Sci. USA 1986, 83, 8370. [Google Scholar] [CrossRef]
- Athamena, A.; Brichon, G.; Trajkovic-Bodennec, S.; Péqueux, A.; Chapelle, S.; Bodennec, J.; Zwingelstein, G. Salinity regulates N-methylation of phosphatidylethanolamine in euryhaline crustaceans hepatopancreas and exchange of newly-formed phosphatidylcholine with hemolymph. J. Comp. Physiol. B 2011, 181, 731–740. [Google Scholar] [CrossRef] [PubMed]
- Cabral, J.H.M.; Atkins, G.L.; Sánchez, L.M.; López-Boado, Y.S.; López-Otin, C.; Sawyer, L. Arachidonic acid binds to apolipoprotein D: Implications for the protein’s function. FEBS Lett. 1995, 366, 53–56. [Google Scholar] [CrossRef]
- Walker, D.W.; Muffat, J.; Rundel, C.; Benzer, S. Overexpression of a Drosophila homolog of apolipoprotein D leads to increased stress resistance and extended lifespan. Curr. Biol. 2006, 16, 674–679. [Google Scholar] [CrossRef] [PubMed]
- Ganfornina, M.D.; Do Carmo, S.; Lora, J.M.; Torres-Schumann, S.; Vogel, M.; Allhorn, M.; González, C.; Bastiani, M.J.; Rassart, E.; Sanchez, D. Apolipoprotein D is involved in the mechanisms regulating protection from oxidative stress. Aging Cell 2008, 7, 506–515. [Google Scholar] [CrossRef] [PubMed]
- Garg, T.K.; Chang, J.Y. 15-deoxy-delta 12, 14-Prostaglandin J 2 prevents reactive oxygen species generation and mitochondrial membrane depolarization induced by oxidative stress. BMC Pharmacol. 2004, 4, 6. [Google Scholar] [CrossRef] [PubMed]
- Behl, T.; Kaur, I.; Goel, H.; Kotwani, A. Implications of the endogenous PPAR-gamma ligand, 15-deoxy-delta-12, 14-prostaglandin J2, in diabetic retinopathy. Life Sci. 2016, 153, 93–99. [Google Scholar] [CrossRef]
- Bhakuni, D.; Rawat, D. Marine prostaglandins. In Bioactive Marine Natural Products; Springer: Dordrecht, The Netherlands, 2005; pp. 329–354. [Google Scholar] [CrossRef]
- Fernández-Tomé, M.d.C.; Speziale, E.H.; Sterin-Speziale, N.B. Phospholipase C inhibitors and prostaglandins differentially regulate phosphatidylcholine synthesis in rat renal papilla: Evidence of compartmental regulation of CTP: Phosphocholine cytidylyltransferase and CDP-choline: 1, 2-diacylglycerol cholinephosphotransferase. Biochim. Biophys. Acta 2002, 1583, 185–194. [Google Scholar] [CrossRef]
- Colacicco, G.; Basu, M.K.; Ray, A.K.; Wittner, M.; Rosenbaum, R.M. Effects of prostaglandins E2 and F2α on lecithin biosynthesis by cultured lung cells. Prostaglandins 1977, 14, 283–294. [Google Scholar] [CrossRef]
- Kozawa, O.; Suzuki, A.; Kotoyori, J.; Tokuda, H.; Watanabe, Y.; Ito, Y.; Oiso, Y. Prostaglandin F2α activates phospholipase D independently from activation of protein kinase C in osteoblast-like cells. J. Cell. Biochem. 1994, 55, 373–379. [Google Scholar] [CrossRef] [PubMed]

| Ingredients | g/kg of Dry Matter |
|---|---|
| Casein | 400 |
| Wheat flour | 250 |
| Gelatin | 90 |
| Fish oil | 30 |
| Soybean oil | 30 |
| Lecithin | 10 |
| Cholesterol | 5 |
| Choline-free vitamin premix 1 | 20 |
| Mineral premix 2 | 20 |
| Amino acid mixture 3 | 30 |
| Butylated hydroxytoluene (BHT) | 0.05 |
| Alpha—cellulose | 114.95 |
| Choline chloride | 0 |
| Proximate composition (% of wet diet) | |
| Moisture | 9.67 |
| Crude protein | 42.20 |
| Crude lipid | 7.99 |
| Ash | 2.04 |
| Total choline (mg/kg diet) | 1082 |
| Choline Chloride Level (mg/kg Diet) | Hepatosomatic Index (%) | Weight Gain (%) | Specific Growth Rate (% day−1) | Condition Factor (%) | Survival (%) |
|---|---|---|---|---|---|
| 0 | 3.89 ± 0.08 | 499.24 ± 20.52 | 3.18 ± 0.05 | 0.79 ± 0.01 | 73.33 ± 4.51 |
| 2000 | 3.97 ± 0.05 | 476.31 ± 22.97 | 3.09 ± 0.09 | 0.77 ± 0.01 | 78.33 ± 2.89 |
| 4000 | 3.86 ± 0.08 | 495.21 ± 14.38 | 3.13 ± 0.06 | 0.79 ± 0.00 | 77.50 ± 4.79 |
| 6000 | 3.81 ± 0.17 | 559.51 ± 21.48 | 3.35 ± 0.07 | 0.80 ± 0.01 | 73.33 ± 4.71 |
| 8000 | 3.76 ± 0.17 | 522.74 ± 24.39 | 3.27 ± 0.08 | 0.77 ± 0.01 | 76.67 ± 2.72 |
| 12,000 | 3.86 ± 0.20 | 491.60 ± 28.01 | 3.15 ± 0.10 | 0.78 ± 0.01 | 69.17 ± 3.44 |
| p value | 0.928 | 0.173 | 0.235 | 0.592 | 0.587 |
| Choline Chloride Level (mg/kg Diet) | Moisture (%) | Crude Protein (%) | Crude Lipid (%) | Ash (%) |
|---|---|---|---|---|
| 0 | 76.38 ± 0.10 | 17.15 ± 0.20 ab | 2.04 ± 0.04 | 3.01 ± 0.03 |
| 2000 | 76.37 ± 0.40 | 17.39 ± 0.04 ab | 1.90 ± 0.10 | 3.14 ± 0.09 |
| 4000 | 76.40 ± 0.68 | 17.09 ± 0.31 ab | 1.78 ± 0.11 | 3.12 ± 0.02 |
| 6000 | 76.19 ± 0.29 | 17.52 ± 0.05 b | 2.01 ± 0.14 | 3.06 ± 0.04 |
| 8000 | 76.62 ± 0.20 | 16.73 ± 0.23 ab | 1.88 ± 0.06 | 3.01 ± 0.05 |
| 12,000 | 77.01 ± 0.13 | 16.61 ± 0.08 a | 1.85 ± 0.11 | 3.06 ± 0.10 |
| p value | 0.679 | 0.026 | 0.465 | 0.581 |
| Choline Chloride Level (mg/kg Diet) | Total Protein (g/L) | Glucose (mmol/L) | Triglyceride (mmol/L) | Total Cholesterol (mmol/L) |
|---|---|---|---|---|
| 0 | 39.55 ± 2.63 | 2.27 ± 0.19 | 1.01 ± 0.12 | 2.09 ± 0.29 ab |
| 2000 | 40.51 ± 1.90 | 2.34 ± 0.13 | 1.16 ± 0.16 | 2.77 ± 0.38 ab |
| 4000 | 40.67 ± 1.72 | 2.26 ± 0.12 | 1.23 ± 0.17 | 2.15 ± 0.18 a |
| 6000 | 37.35 ± 1.77 | 2.16 ± 0.11 | 1.44 ± 0.06 | 3.40 ± 0.31 b |
| 8000 | 41.67 ± 1.77 | 1.95 ± 0.06 | 1.33 ± 0.06 | 3.64 ± 0.62 ab |
| 12,000 | 40.84 ± 2.15 | 2.00 ± 0.06 | 1.42 ± 0.15 | 3.76 ± 0.48 ab |
| p value | 0.741 | 0.075 | 0.095 | 0.007 |
| Choline Chloride Level (mg/kg Diet) | Total Protein (g/L) | MDA (nmol/Mgprot) | SOD (U/Mgprot) | GSH-Px (U/Mgprot) | AMS (U/Mgprot) | LPS (U/Mgprot) |
|---|---|---|---|---|---|---|
| 0 | 28.43 ± 1.69 | 4.40 ± 0.22 b | 2287.84 ± 38.51 b | 517.49 ± 89.61 b | 2.67 ± 0.28 | 0.41 ± 0.05 |
| 2000 | 38.31 ± 1.88 | 2.95 ± 0.16 a | 1560.96 ± 208.59 a | 380.54 ± 29.39 ab | 2.31 ± 0.22 | 0.27 ± 0.06 |
| 4000 | 39.05 ± 3.76 | 1.99 ± 0.24 a | 1345.69 ± 114.31 a | 214.99 ± 51.34 a | 2.56 ± 0.37 | 0.35 ± 0.07 |
| 6000 | 39.67 ± 4.11 | 2.95 ± 0.48 a | 1404.37 ± 87.10 a | 399.21 ± 59.51 ab | 2.45 ± 0.24 | 0.21 ± 0.05 |
| 8000 | 41.26 ± 3.81 | 2.84 ± 0.24 a | 1380.03 ± 88.90 a | 307.68 ± 34.89 ab | 2.26 ± 0.29 | 0.19 ± 0.04 |
| 12,000 | 39.60 ± 3.70 | 2.82 ± 0.43 a | 1612.94 ± 41.16 a | 409.22 ± 54.27 ab | 2.28 ± 0.37 | 0.19 ± 0.02 |
| p value | 0.103 | 0.000 | 0.000 | 0.016 | 0.910 | 0.076 |
| Choline Chloride Level (mg/kg Diet) | Clean Reads | Total Mapped (%) | Error Rate (%) | Q30 (%) | GC Content (%) |
|---|---|---|---|---|---|
| 0 | 48,699,065 | 88.70 | 0.02 | 95.67 | 50.81 |
| 6000 | 51,050,008 | 88.41 | 0.02 | 95.70 | 50.93 |
| 12,000 | 51,946,736 | 88.12 | 0.02 | 95.71 | 50.54 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, M.; Lin, H.; Xu, C.; Yu, Q.; Wang, X.; Qin, J.G.; Chen, L.; Han, F.; Li, E. Growth, Metabolite, Antioxidative Capacity, Transcriptome, and the Metabolome Response to Dietary Choline Chloride in Pacific White Shrimp Litopenaeus vannamei. Animals 2020, 10, 2246. https://doi.org/10.3390/ani10122246
Huang M, Lin H, Xu C, Yu Q, Wang X, Qin JG, Chen L, Han F, Li E. Growth, Metabolite, Antioxidative Capacity, Transcriptome, and the Metabolome Response to Dietary Choline Chloride in Pacific White Shrimp Litopenaeus vannamei. Animals. 2020; 10(12):2246. https://doi.org/10.3390/ani10122246
Chicago/Turabian StyleHuang, Maoxian, Hongxing Lin, Chang Xu, Qiuran Yu, Xiaodan Wang, Jian G. Qin, Liqiao Chen, Fenglu Han, and Erchao Li. 2020. "Growth, Metabolite, Antioxidative Capacity, Transcriptome, and the Metabolome Response to Dietary Choline Chloride in Pacific White Shrimp Litopenaeus vannamei" Animals 10, no. 12: 2246. https://doi.org/10.3390/ani10122246
APA StyleHuang, M., Lin, H., Xu, C., Yu, Q., Wang, X., Qin, J. G., Chen, L., Han, F., & Li, E. (2020). Growth, Metabolite, Antioxidative Capacity, Transcriptome, and the Metabolome Response to Dietary Choline Chloride in Pacific White Shrimp Litopenaeus vannamei. Animals, 10(12), 2246. https://doi.org/10.3390/ani10122246








