Stress-Induced Phosphaturia in Weaned Piglets
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Animals and Experimental Design
2.3. Anesthesia Procedure and Blood Biochemical and Gasometric Analysis
2.4. Statistical Analysis
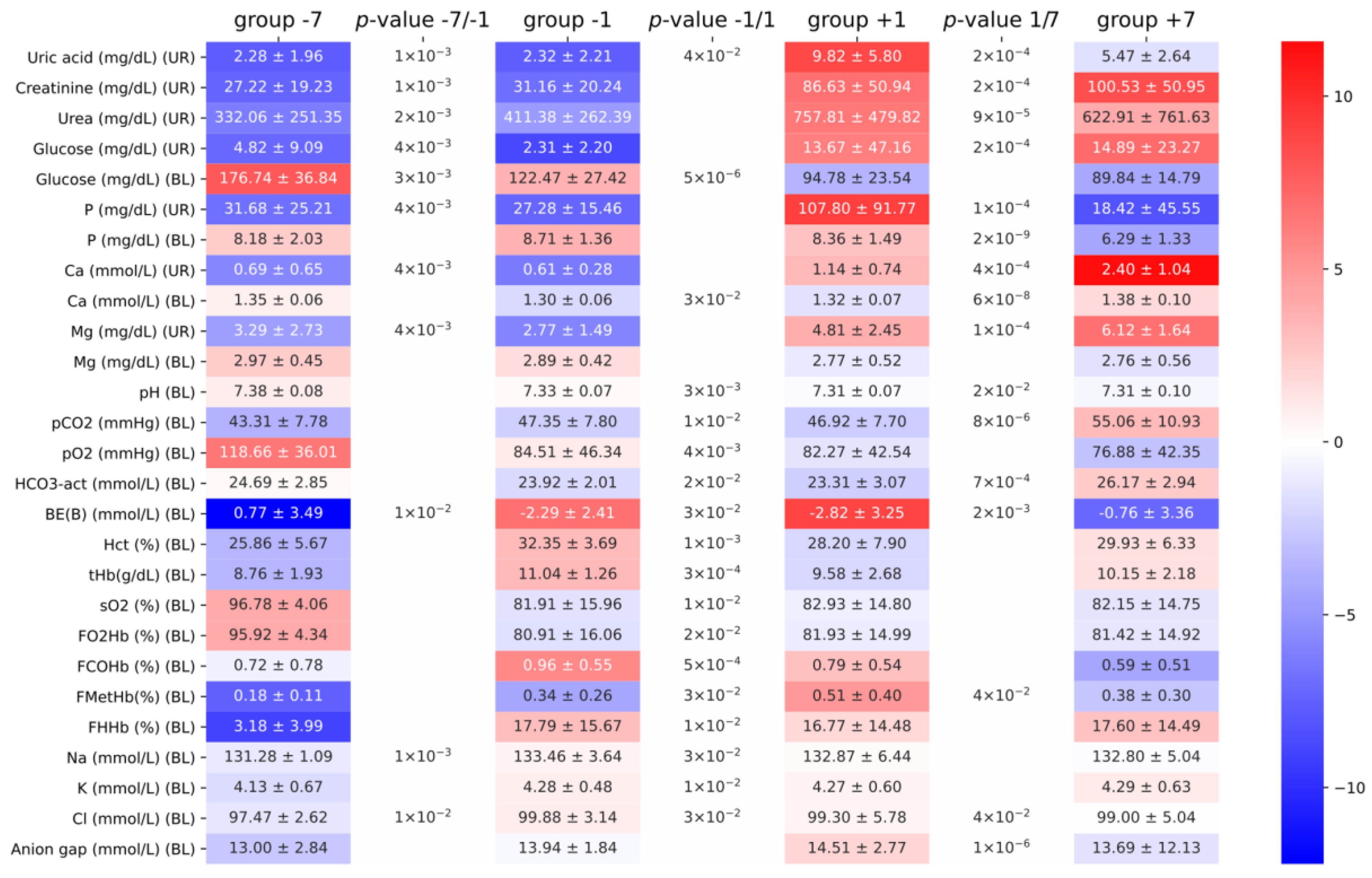
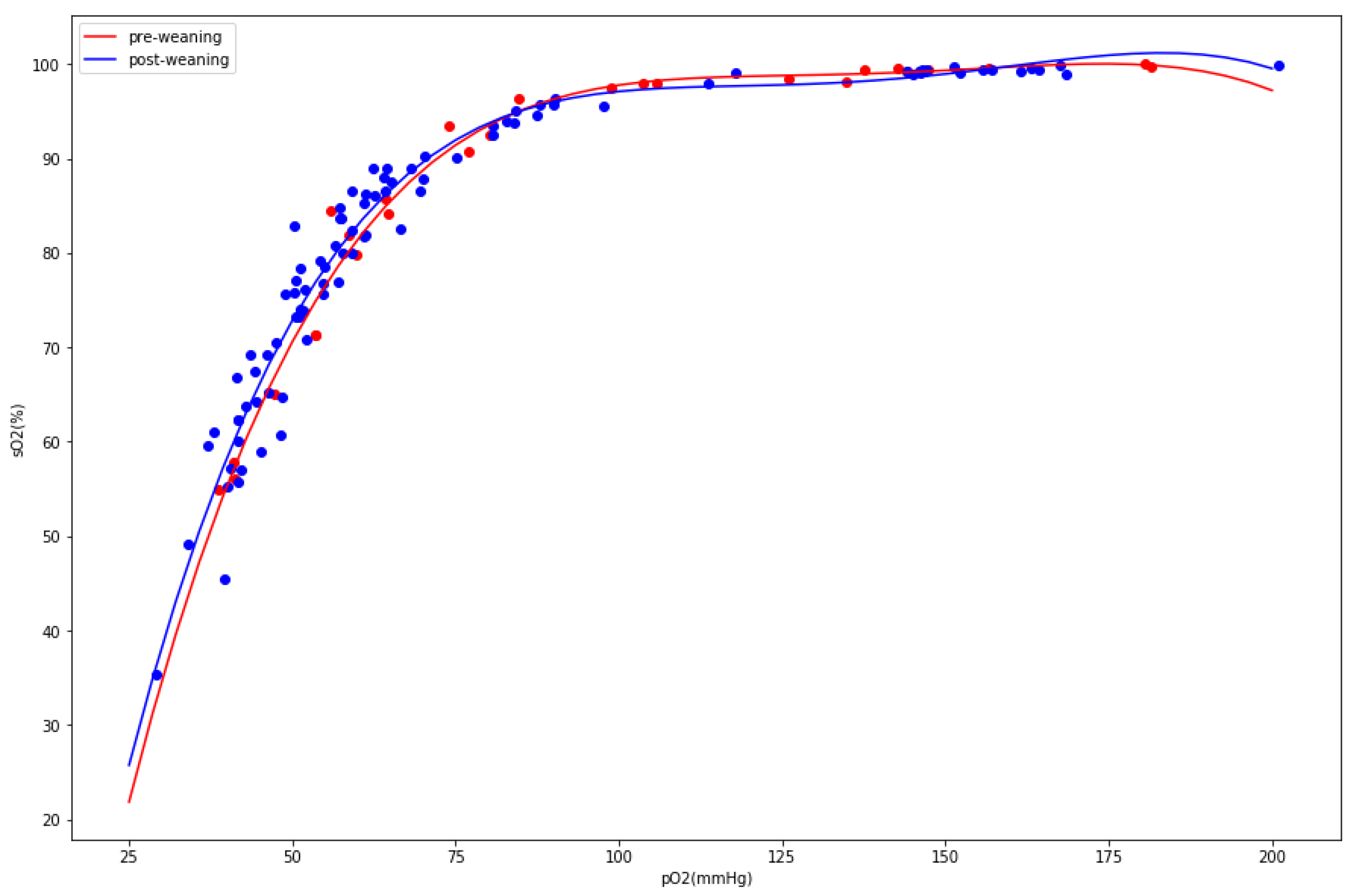
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Muns, R.; Nuntapaitoon, M.; Tummaruk, P. Non-infectious causes of pre-weaning mortality in piglets. Livest. Sci. 2016, 184, 46–57. [Google Scholar] [CrossRef]
- Martínez-Miró, S.; Tecles, F.; Ramón, M.; Escribano, D.; Hernández, F.; Madrid, J.; Orengo, J.; Martínez-Subiela, S.; Manteca, X.; Cerón, J.J. Causes, consequences and biomarkers of stress in swine: An update. BMC Vet. Res. 2016, 12, 171. [Google Scholar] [CrossRef] [PubMed]
- Schook, L.; Beattie, C.; Beever, J.; Donovan, S.; Jamison, R.; Zuckermann, F.; Niemi, S.; Rothschild, M.; Rutherford, M.; Smith, D. Swine in Biomedical Research: Creating the Building Blocks of Animal Models. Anim. Biotechnol. 2005, 16, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.; Canalias, F.; Solà-Oriol, D.; Arroyo, L.; Pato, R.; Saco, Y.; Terré, M.; Bassols, A. Age-Related Serum Biochemical Reference Intervals Established for Unweaned Calves and Piglets in the Post-weaning Period. Front. Vet. Sci. 2019, 6, 123. [Google Scholar] [CrossRef] [PubMed]
- Perri, A.M.; O’Sullivan, T.L.; Harding, J.C.S.; Wood, R.D.; Friendship, R.M. Hematology and biochemistry reference intervals for Ontario commercial nursing pigs close to the time of weaning. Can. Vet. J. 2017, 58, 371–376. [Google Scholar] [PubMed]
- Hannon, J.P. Blood acid-base curve nomogram for immature domestic pigs. Am. J. Vet. Res. 1983, 44, 2385–2390. [Google Scholar] [PubMed]
- Harris, W.H. Hemoglobin, blood gases and serum electrolyte values in swine. Can. Vet. J. 1974, 15, 282–285. [Google Scholar] [PubMed]
- Milićević, D.; Jurić, V.; Stefanović, S.; Jovanović, M.; Janković, S. Survey of slaughtered pigs for occurrence of ochratoxin A and porcine nephropathy in Serbia. Int. J. Mol. Sci. 2008, 9, 2169–2183. [Google Scholar] [CrossRef] [PubMed]
- Gowda, S.N.; Gowda, J. Chapter-16 Fluid, Electrolyte and Acid-base Balance. In Advanced Critical Care in Medical, Surgical & Neonatal Nursing; Jaypee Brothers: New Delhi, India, 2017; pp. 278–294. [Google Scholar]
- Rotsztain, A.; Haddad, R.; Canter, H.G. Blood gas changes during voluntary hyperventilation in normal and disease states. Am. Rev. Respir. Dis. 1970, 102, 205–212. [Google Scholar] [PubMed]
- Gardner, W.N. The Pathophysiology of Hyperventilation Disorders. Chest 1996, 109, 516–534. [Google Scholar] [CrossRef] [PubMed]
- Lovén, L.; Jansson, I.; Larsson, L.; Lennquist, S. Posttraumatic hypophosphataemia and urinary phosphate excretion with and without phosphate supplementation. An experimental study in pigs. Acta Chir. Scand. 1983, 149, 233–238. [Google Scholar] [PubMed]
- Lovén, L.; Nordström, H.; Larsson, L.; Lindell, B.; Sjöberg, H.E.; Lennquist, S. Traumatically induced hypophosphataemia in anaesthetized pigs. Acta Chir. Scand. 1982, 148, 21–25. [Google Scholar] [PubMed]
- Kim, A.; Yamagishia, N.; Devkota, B.; Furuhama, K. Effects of cortisol secreted via a 12-h infusion of adrenocorticotropic hormone on mineral homeostatis and nome metabolism in ovariectomized cows. Domest. Anim. Endocrinol. 2012, 43, 264–269. [Google Scholar] [CrossRef] [PubMed]
- Antonissen, G.; Martel, A.; Pasmans, F.; Ducatelle, R.; Verbrugghe, E.; Vandenbroucke, V.; Li, S.; Haesebrouck, F.; Van Immerseel, F.; Croubels, S. The impact of Fusarium mycotoxins on human and animal host susceptibility to infectious diseases. Toxins 2014, 6, 430–452. [Google Scholar] [CrossRef] [PubMed]
- Al-Anati, L.; Petzinger, E. Immunotoxic activity of ochratoxin A. J. Vet. Pharmacol. Ther. 2006, 29, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Jongbloed, A.W. Phosphorus in the Feeding of Pigs: Effect of Diet on the Absorption and Retention of Phosphorus by Growing Pigs; Agricultural University: Wageningen, The Netherlands, 1987. [Google Scholar]
- Poulsen, H.D.; Jongbloed, A.W.; Latimier, P.; Fernández, J.A. Phosphorus consumption, utilisation and losses in pig production in France, The Netherlands and Denmark. Livest. Prod. Sci. 1999, 58, 251–259. [Google Scholar] [CrossRef]
- Cordell, D.; Rosemarin, A.; Schröder, J.J.; Smit, A.L. Towards global phosphorus security: A systems framework for phosphorus recovery and reuse options. Chemosphere 2011, 84, 747–758. [Google Scholar] [CrossRef] [PubMed]
- Heyer, C.M.E.; Weiss, E.; Schmucker, S.; Rodehutscord, M.; Hoelzle, L.E.; Mosenthin, R.; Stefanski, V. The impact of phosphorus on the immune system and the intestinal microbiota with special focus on the pig. Nutr. Res. Rev. 2015, 28, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Oster, M.; Gerlinger, C.; Heide, K.; Just, F.; Borgelt, L.; Wolf, P.; Polley, C.; Vollmar, B.; Muráni, E.; Ponsuksili, S.; et al. Lower dietary phosphorus supply in pigs match both animal welfare aspects and resource efficiency. Ambio 2018, 47, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Gerlinger, C.; Oster, M.; Borgelt, L.; Reyer, H.; Muráni, E.; Ponsuksili, S.; Polley, C.; Vollmar, B.; Reichel, M.; Wolf, P.; et al. Physiological and Transcriptional Responses in Weaned Piglets Fed Diets with Varying Phosphorus and Calcium Levels. Nutrients 2019, 11, 436. [Google Scholar] [CrossRef] [PubMed]
- Kebreab, E. Sustainable Animal Agriculture; CABI: Wallingford, UK, 2013; ISBN 9781780640426. [Google Scholar]
| Group | Time to Weaning | N (Numbers) |
|---|---|---|
| Group I | Seven days before | 14 (♀ = 0, ♂ = 14) |
| Group II | At the day | 16 (♀ = 3, ♂ = 13) |
| Group III | One day after | 46 (♀ = 10, ♂ = 36) |
| Group IV | Seven days after | 44 (♀ = 4, ♂ = 40) |
| Biomarker | Hypophosphatemic vs. Normophosphatemic | Postweaning vs. Preweaning | ||
|---|---|---|---|---|
| p-Value | Change | p-Value | Change | |
| Uric acid (mg/dL) (UR) | 0.02 | ↑ | 6.39 × 10−8 | ↑ |
| Creatinine (mg/dL) (UR) | 0.005 | ↑ | 8.23 × 10−8 | ↑ |
| Urea (mg/dL) (UR) | 0.01 | ↑ | 0.0001 | ↑ |
| Glucose (mg/dL) (UR) | 0.002 | ↓ | 0.002 | ↑ |
| Glucose (mg/dL) (BL) | 0.002 | ↓ | 2.44 × 10−15 | ↓ |
| P (mg/dL) (UR) | 0.02 | ↑ | 3.48 × 10−6 | ↑ |
| P (mg/dL) (BL) | NA | NA | 2.14 × 10−5 | ↓ |
| Ca (mmol/L) (UR) | 0.02 | ↑ | 0.0001 | ↑ |
| Ca (mmol/L) (BL) | 0.003 | ↑ | 0.15 | N.S. |
| Mg (mg/dL) (UR) | 0.03 | ↑ | 3.73 × 10−5 | ↑ |
| Mg (mg/dL) (BL) | 7.53 × 10−6 | ↓ | 0.29 | N.S. |
| pH (BL) | 0.02 | ↓ | 5.11 × 10−7 | ↓ |
| pCO2 (mmHg) (BL) | 0.02 | ↑ | 0.003 | ↑ |
| pO2 (mmHg) (BL) | 0.0002 | ↓ | 9.62 × 10−5 | ↓ |
| HCO3-act (mmol/L) (BL) | 0.19 | N.S. | 0.003 | ↑ |
| BE(B) (mmol/L) (BL) | 0.26 | N.S. | 5.48 × 10−5 | ↓ |
| Hct (%) (BL) | 0.14 | N.S. | 0.001 | ↓ |
| tHb(g/dL) (BL) | 0.14 | N.S. | 0.0004 | ↓ |
| sO2 (%) (BL) | 0.0001 | ↓ | 4.97 × 10−5 | ↓ |
| FO2Hb (%) (BL) | 0.0002 | ↓ | 2.77 × 10−5 | ↓ |
| FCOHb (%) (BL) | 0.44 | N.S. | 6.03 × 10−5 | ↓ |
| FMetHb (%) (BL) | 0.9 | N.S. | 0.003 | ↑ |
| FHHb (%) (BL) | 0.008 | ↑ | 0.002 | ↑ |
| Na (mmol/L) (BL) | 0.66 | N.S. | 0.15 | N.S. |
| K (mmol/L) (BL) | 0.06 | N.S. | 0.04 | ↑ |
| Cl (mmol/L) (BL) | 0.95 | N.S. | 0.15 | N.S. |
| >Anion gap (mmol/L) (BL) | >0.0003 | >↓ | >0.001 | >↑ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Habich, M.; Pawlinski, B.; Sady, M.; Siewruk, K.; Zielenkiewicz, P.; Gajewski, Z.; Szczesny, P. Stress-Induced Phosphaturia in Weaned Piglets. Animals 2020, 10, 2220. https://doi.org/10.3390/ani10122220
Habich M, Pawlinski B, Sady M, Siewruk K, Zielenkiewicz P, Gajewski Z, Szczesny P. Stress-Induced Phosphaturia in Weaned Piglets. Animals. 2020; 10(12):2220. https://doi.org/10.3390/ani10122220
Chicago/Turabian StyleHabich, Malgorzata, Bartosz Pawlinski, Maria Sady, Katarzyna Siewruk, Piotr Zielenkiewicz, Zdzislaw Gajewski, and Pawel Szczesny. 2020. "Stress-Induced Phosphaturia in Weaned Piglets" Animals 10, no. 12: 2220. https://doi.org/10.3390/ani10122220
APA StyleHabich, M., Pawlinski, B., Sady, M., Siewruk, K., Zielenkiewicz, P., Gajewski, Z., & Szczesny, P. (2020). Stress-Induced Phosphaturia in Weaned Piglets. Animals, 10(12), 2220. https://doi.org/10.3390/ani10122220






