The Role of Urine in Semiochemical Communication between Females and Males of Domestic Dog (Canis familiaris) during Estrus
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Statement
2.2. Animals
2.3. Collection of Urine Samples
2.4. Estrus Detection Methods
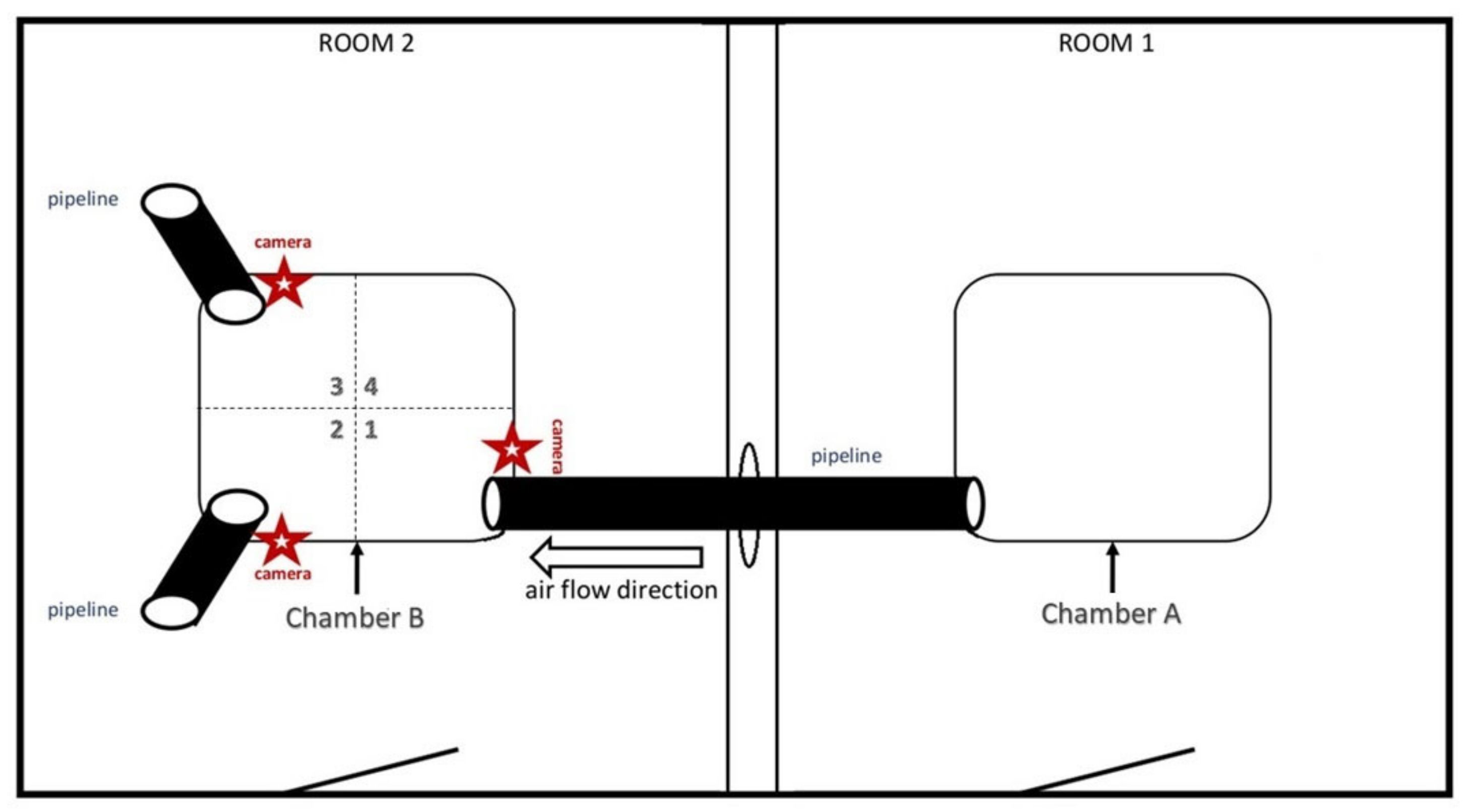
2.5. Experimental Design
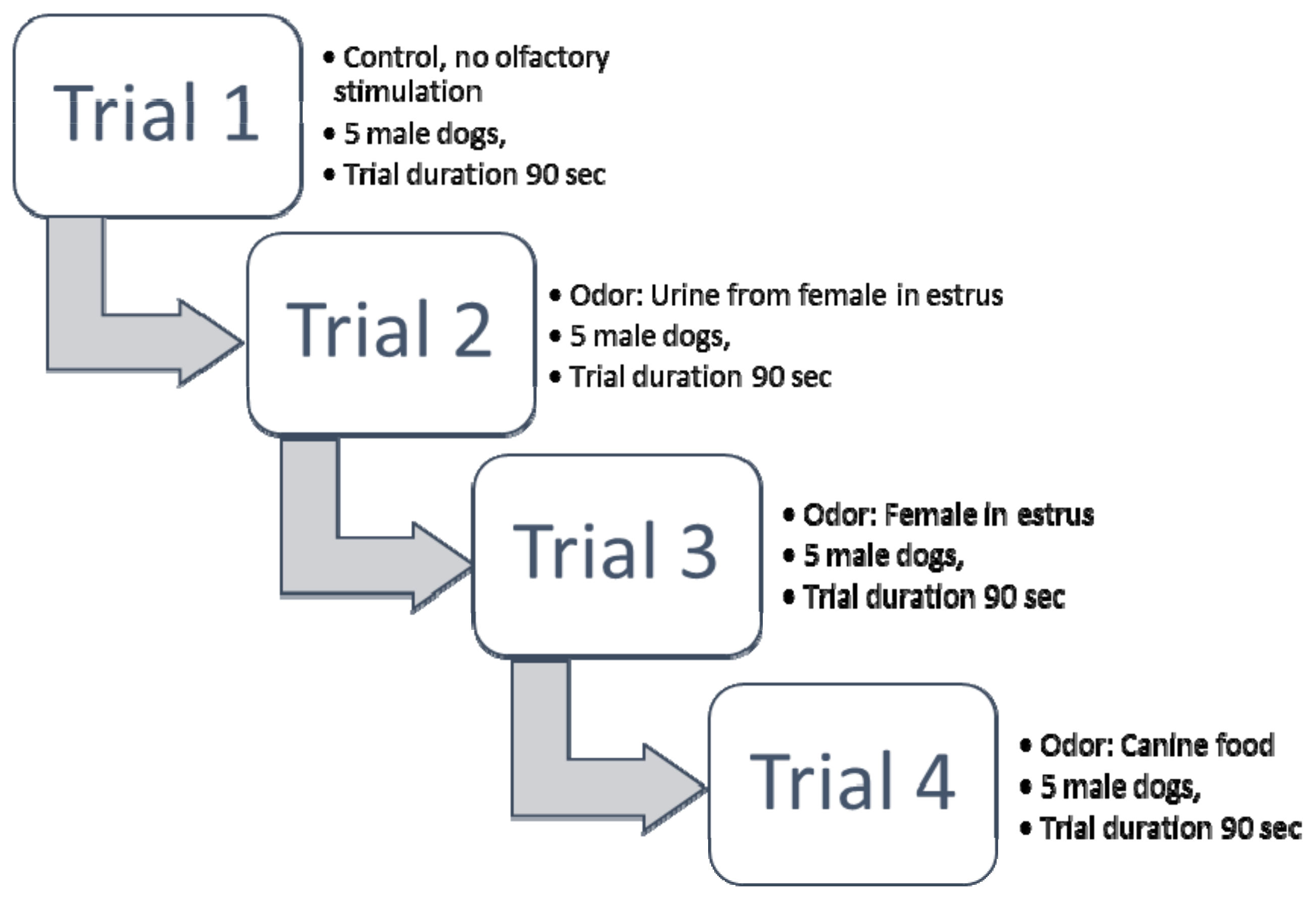
2.5.1. Experiment 1
Odor Sample Presentation to the Dogs
Behavioral Analyses
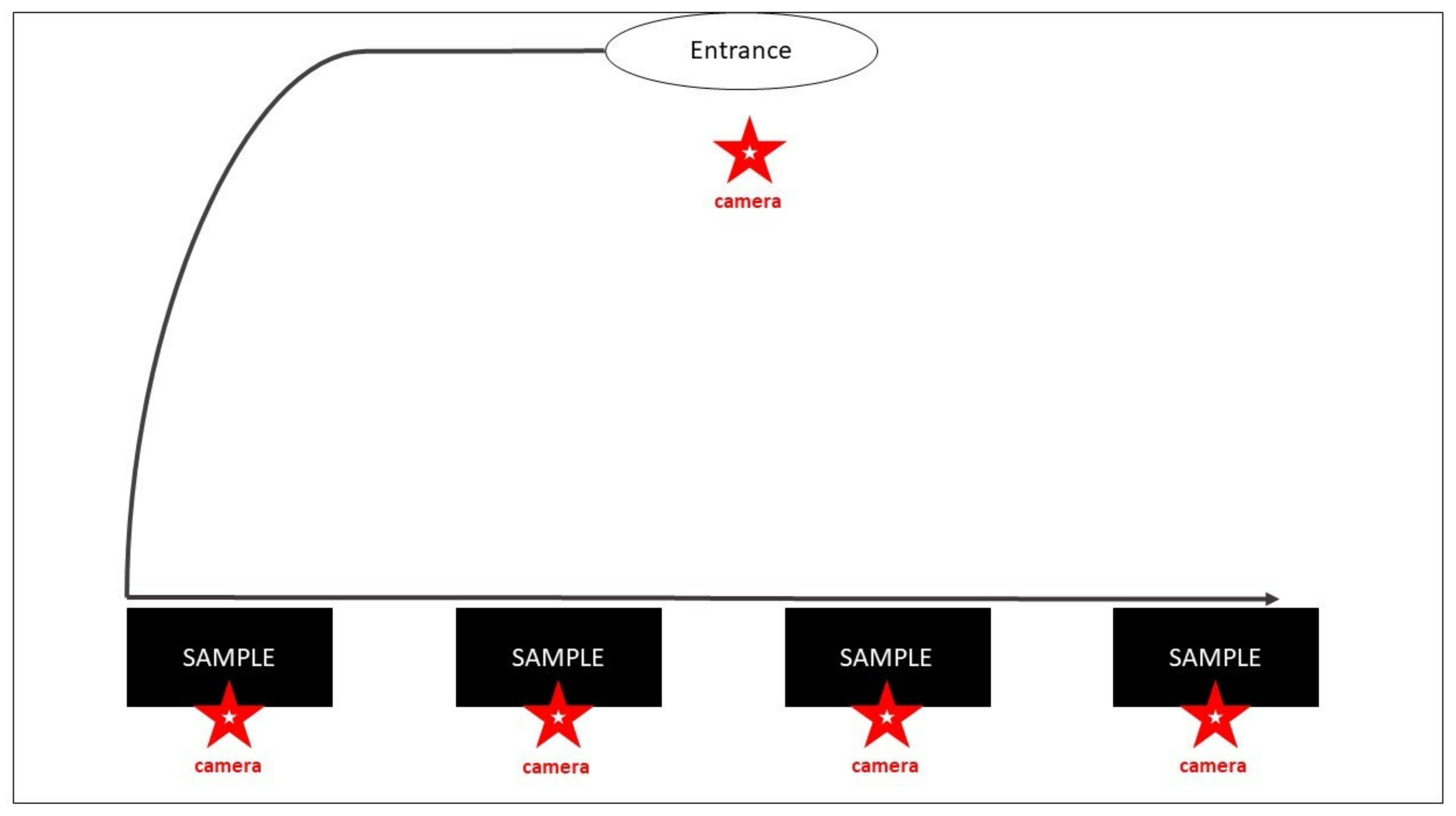
2.5.2. Experiment 2
Odor Sample Presentation to the Dogs
Behavioral Analyses
2.6. Statistical Methods
3. Results
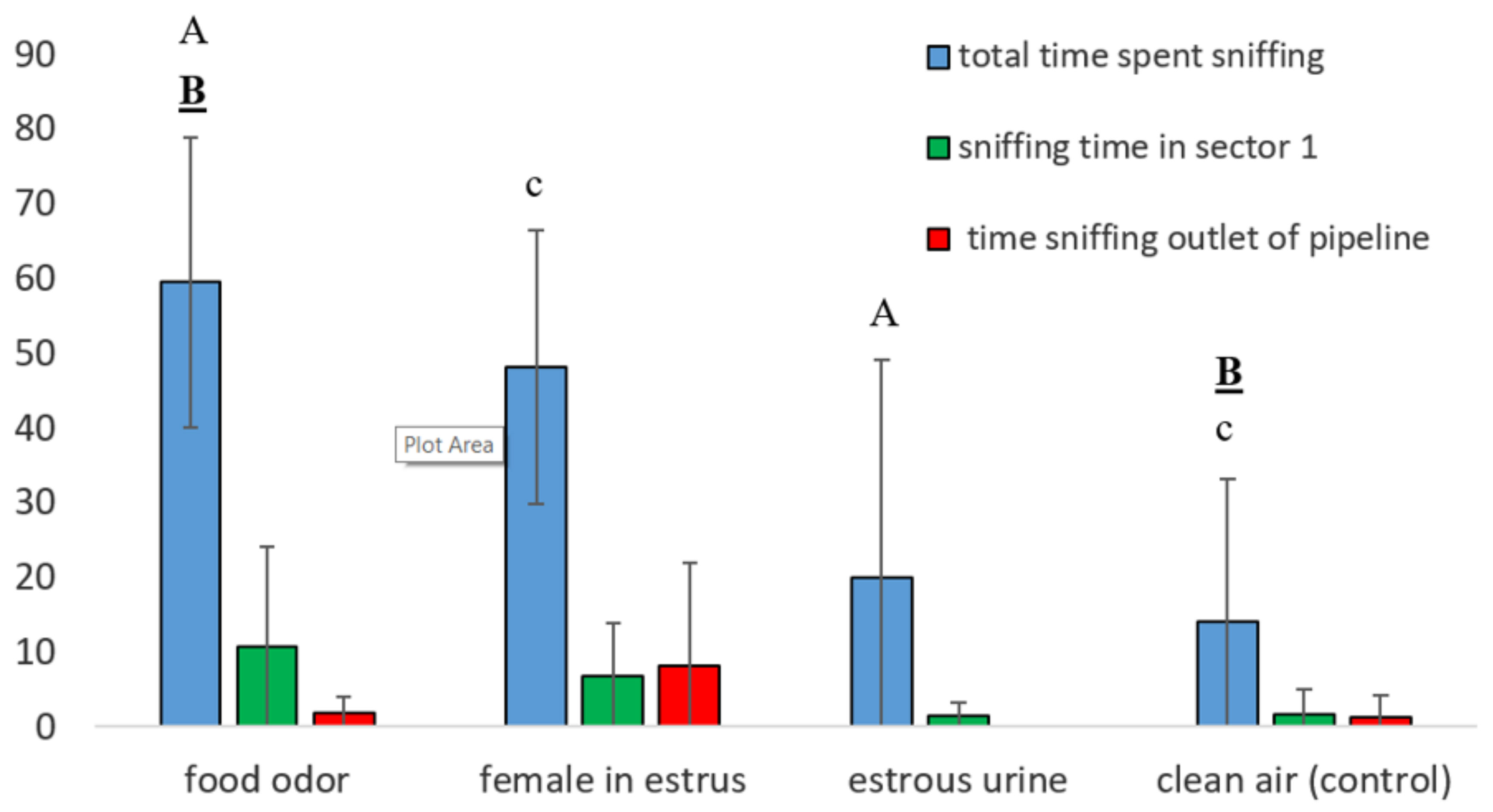
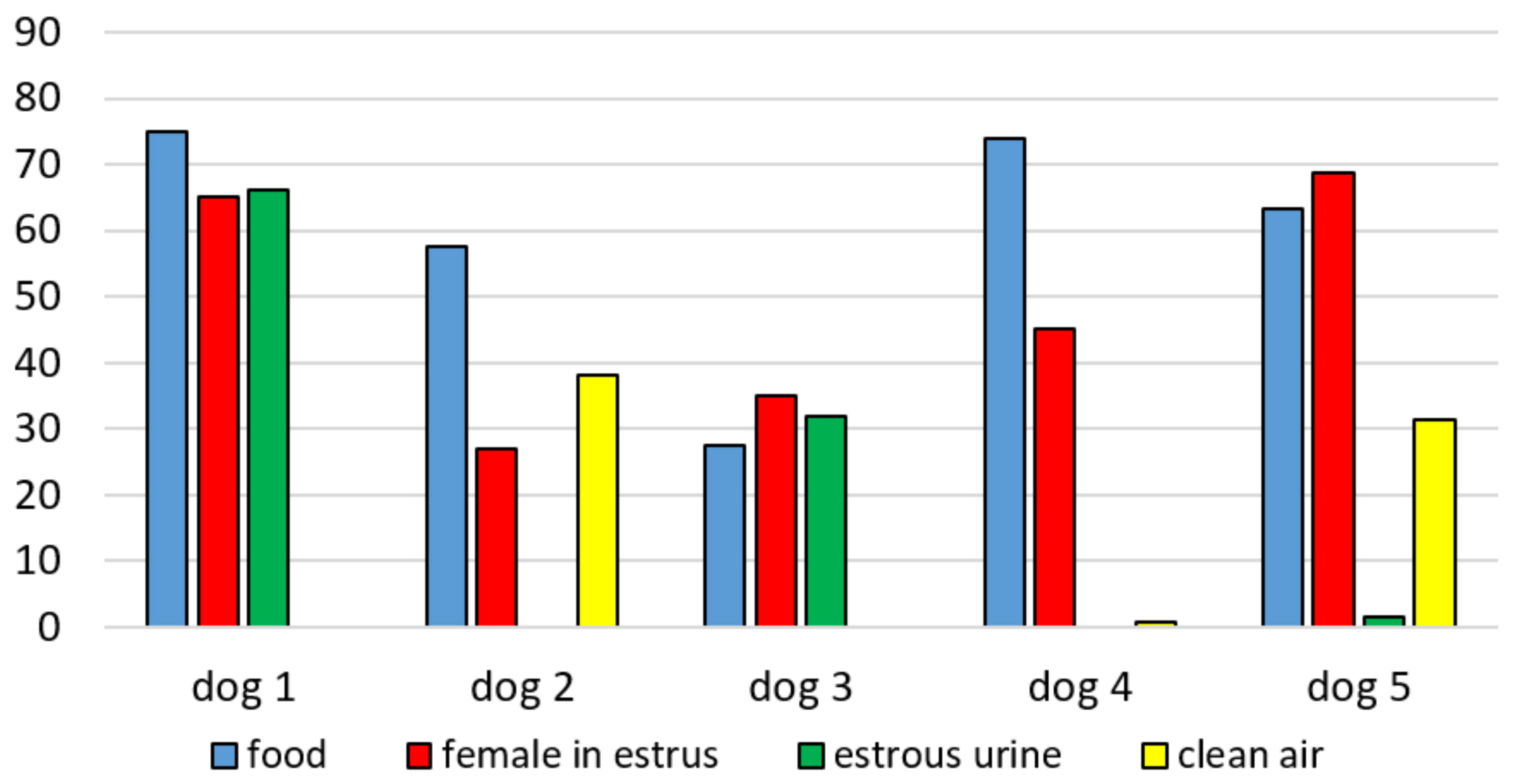
3.1. Experiment 1
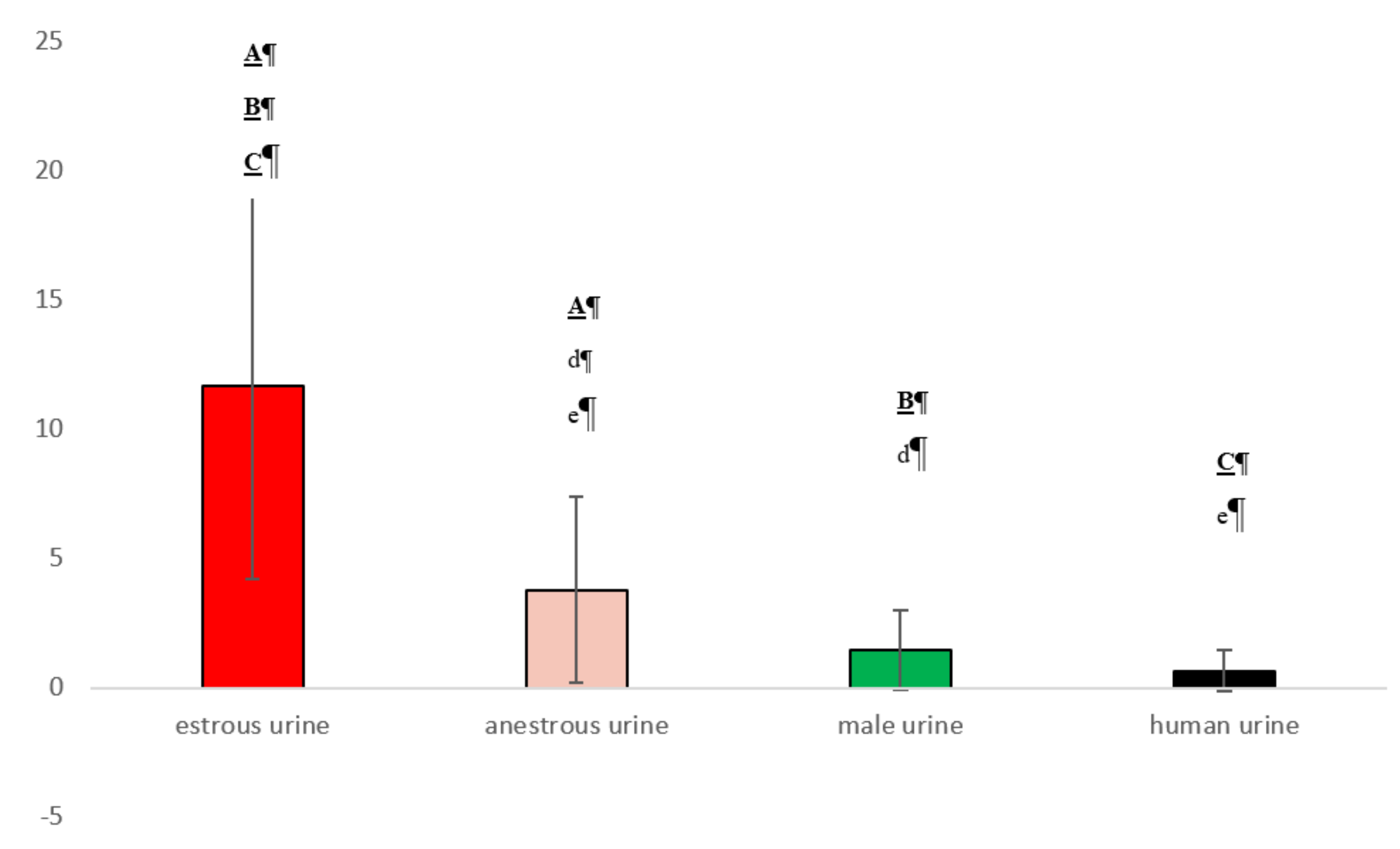
3.2. Experiment 2
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wyatt, T.D. Proteins and peptides as pheromone signals and chemical signatures. Anim. Behav. 2014, 97, 273–280. [Google Scholar] [CrossRef]
- Wyatt, T.D. Pheromones and Animal Behaviours; Cambridge University Press: Cambridge, UK, 2003. [Google Scholar]
- Chamero, P.; Leinders-Zufall, T.; Zufall, F. From genes to social communication: Molecular sensing by the vomeronasal organ. Trends Neurosci. 2012, 35, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Keverne, E.B. The vomeronasal organ. Science 1999, 286, 716–720. [Google Scholar] [CrossRef] [PubMed]
- Tirindelli, R.; Dibattista, M.; Pifferi, S.; Menini, A. From pheromones to behavior. Physiol. Rev. 2009, 89, 921–956. [Google Scholar] [CrossRef]
- Xu, F.; Greer, C.A.; Shepherd, G.M. Odor maps in the olfactory bulb. J. Comp. Neurol. 2000, 422, 489–495. [Google Scholar] [CrossRef]
- Kokocińska, A.; Ensminger, J.; Jezierski, T. Effects of novel odours on the mating behaviour in mice. Anim. Sci. Pap. Rep. 2017, 35, 301–316. [Google Scholar]
- O’Connell, R.J.; Meredith, M. Effects of volatile and nonvolatile chemical signals on male sex behaviors mediated by the main and accessory olfactory systems. Behav. Neurosci. 1984, 98, 1083–1093. [Google Scholar] [CrossRef]
- Stowers, L.; Marton, T.F. What is a Pheromone? Mammalian Pheromones Reconsidered. Neuron 2005, 46, 699–702. [Google Scholar] [CrossRef]
- Michael, R.P.; Keverne, E.B.; Bonsall, R.W. Pheromones: Isolation of male sex attractants from a female primate. Science 1971, 172, 964–966. [Google Scholar] [CrossRef]
- Kimura, R. Volatile substances in feces, urine and urine-marked feces of feral horses. Can. J. Anim. Sci. 2001, 81, 411–420. [Google Scholar] [CrossRef]
- Rekwot, P.I.; Ogwu, D.; Oyedipe, E.O.; Sekoni, V.O. The role of pheromones and biostimulation in animal reproduction. Anim. Reprod. Sci. 2001, 65, 157–170. [Google Scholar] [CrossRef]
- Sankar, R.; Archunan, G.; Habara, Y. Detection of oestrous-related odour in bovine (Bos taurus) saliva: Bioassay of identified compound. Animal 2007, 1, 1321–1327. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, L.E.; Lee, T.D.; Roelofs, W.L.; Zhang, A.; Daves, G.D., Jr. Insect pheromone in elephants. Nature 1996, 379, 684. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, B.L.; Jerôme, N.; Saint-Albin, A.; Christian Ouali, C.; Rochut, S.; Zins, E.-L.; Briant, C.; Guettier, E.; Reigner, F.; Couty, I.; et al. Oestrus odours from rats and mares: Behavioural responses of sexually naive and experienced rats to natural odours and odorants. Appl. Anim. Behav. Sci. 2016, 176, 128–135. [Google Scholar] [CrossRef]
- Rampin, O.; Jérôme, N.; Briant, C.; Boué, F.; Maurin, Y. Are oestrus odours species specific? Behav. Brain Res. 2006, 72, 169–172. [Google Scholar] [CrossRef]
- Buchinger, T.J.; Weiming, L. The evolution of (non)species specific pheromones. Evol. Ecol. 2020. [Google Scholar] [CrossRef]
- Doty, R.L.; Dunbar, I. Attraction of beagles to conspecific urine, vaginal and anal sac secretion odors. Physiol. Behav. 1974, 12, 825–833. [Google Scholar] [CrossRef]
- Dzięcioł, M.; Politowicz, J.; Szumny, A.; Niżański, W. Methyl paraben as a sex pheromone in canine urine—Is the question still open? Polish J. Vet. Sci. 2014, 17, 601–605. [Google Scholar] [CrossRef]
- Dzięcioł, M.; Niżański, W.; Jezierski, T.; Szumny, A.; Godzińska, E.J.; Ochota, M.; Stańczyk, E.; Najder-Kozdrowska, L.; Woszczyło, M.; Pieczewska, B. The efficiency of synthetic sex pheromones in sexual arousal stimulation in domestic dogs. Polish J. Vet. Sci. 2017, 20, 429–437. [Google Scholar] [CrossRef]
- Dzięcioł, M.; Woszczylo, M.; Szumny, A.; Jezierski, T.; Kupczyński, R.; Godzińska, E.J.; Pieczewska, B.; Niżański, W. Identification of putative volatile sex pheromones in female domestic dogs (Canis familiaris). Anim. Reprod. Sci. 2018, 197, 87–92. [Google Scholar] [CrossRef]
- Goodwin, M.; Gooding, K.M.; Regnier, F. Sex pheromone in the dog. Science 1979, 203, 559–561. [Google Scholar] [CrossRef] [PubMed]
- Jorgenson, J.W.; Novotny, M.; Carmack, M.; Copland, G.B.; Wilson, S.R. Chemical scent constituents in the urine of the red fox (Vulpes vulpes L.) during the winter season. Science 1978, 199, 796–798. [Google Scholar] [CrossRef] [PubMed]
- Kruse, S.M.; Howard, W.E. Canid sex attractant studies. J. Chem. Ecol. 1983, 9, 1503–1510. [Google Scholar] [CrossRef] [PubMed]
- McKenna, S.M. Response of Male Beagles to “Canid Sex Pheromone,” Methyl p-hydroxy-benzoate. Master’s Thesis, University of California, Davis, CA, USA, 1981. [Google Scholar]
- Murphy, E.L.; Flath, R.A.; Black, D.R.; Mon, T.R.; Teranishi, R. Isolation, identification, and biological activity assay of chemical fractions from estrus urine attractive to the coyote. ACS Symposium Series 67; Bullard, R.W., Ed.; American Chemical Society: Washington, DC, USA, 1978; pp. 66–77. [Google Scholar]
- Schultz, T.H.; Kruse, S.M.; Flath, R.A. Some volatile constituents of female dog urine. J. Chem. Ecol 1985, 11, 169–175. [Google Scholar] [CrossRef]
- Būda, V.; Mozūraitis, R.; Kutra, J.; Borg-Karlson, A. p-Cresol: A Sex Pheromone Component Identified from the Estrous Urine of Mares. J. Chem. Ecol. 2012, 38, 811–813. [Google Scholar] [CrossRef]
- Jezierski, T.; Dzięcioł, M.; Szumny, A.; Niżański, W.; Woszczyło, M.; Pieczewska, B.; Godzińska, E.J. Discrimination of estrus odor in urine by male dogs in different experimental settings. J. Vet. Behav. 2019, 29, 25–30. [Google Scholar] [CrossRef]
- Kustritz, M.V.R. Reproductive behavior of small animals. Theriogenology 2005, 64, 734–746. [Google Scholar] [CrossRef]
- Kustritz, M.V.R. Collection of tissue and culture samples from the canine reproductive tract. Theriogenology 2006, 66, 567–574. [Google Scholar] [CrossRef]
- Brugger, N.; Otzdorff, C.; Walter, B.; Hoffmann, B.; Braun, J. Quantitative determination of progesterone (P4) in canine blood serum using an enzyme-linked fluorescence assay. Reprod. Domest. Anim. 2011, 46, 870–873. [Google Scholar] [CrossRef]
- Beauchamp, G.K. Attraction of male guinea pigs to conspecific urine. Physiol. Behav. 1973, 10, 589–594. [Google Scholar] [CrossRef]
- Lydell, K.; Doty, R.L. Male rat of odor preferences for female urine as a function of sexual experience, urine age, and urine source. Horm. Behav. 1972, 3, 205–212. [Google Scholar] [CrossRef]
- Scott, J.W.; Pfaff, D.W. Behavioral and electrophysiological responses of female mice to male urine odors. Physiol. Behav. 1970, 5, 407–411. [Google Scholar] [CrossRef]
- Powers, J.B.; Fields, R.B.; Winans, S.S. Olfactory and vomeronasal system participation in male hamsters’ attraction to female vaginal secretions. Physiol. Behav. 1979, 22, 77–84. [Google Scholar] [CrossRef]
- Chang, Y.M.; Kelliher, K.R.; Baum, M.J. Steroidal modulation of scent investigation and marking behaviors in male and female ferrets. J. Comp. Psychol. 2000, 114, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Hurst, J.L. Female recognition and assessment of males through scent. Behav. Brain Res. 2009, 200, 295–303. [Google Scholar] [CrossRef]
- Martinez-Garcia, F.; Martinez-Ricos, J.; Agustin-Pavon, C.; Martinez-Hernandez, J.; Novejarque, A.; Lanuza, E. Refining the dual olfactory hypothesis: Pheromone reward and odour experience. Behav. Brain Res. 2009, 200, 277–286. [Google Scholar] [CrossRef]
- Cohen-Tannoudji, J.; Einhorn, J.; Signoret, J.P. Ram sexual pheromone: First approach of chemical identification. Physiol. Behav. 1994, 56, 955–961. [Google Scholar] [CrossRef]
- Fabre-Nys, C.; Kendrick, K.M.; Scaramuzzi, R.J. The “ram effect”: New insights into neural modulation of the gonadotropic axis by male odors and socio-sexual interactions. Front Neurosci. 2015, 9, 111. [Google Scholar] [CrossRef]
- Rasmussen, L.E.; Schmidt, M.J.; Henneous, R.; Groves, D.; Daves, G.D., Jr. Asian bull elephants: Flehmen-like responses to extractable components in female elephant estrous urine. Science 1982, 217, 159–162. [Google Scholar] [CrossRef]
- Goodwin, T.E.; Rasmussen, L.E.L.; Schulte, B.A.; Brown, P.A.; Davis, B.L.; Dill, W.M.; Dowdy, N.C.; Hicks, A.R.; Morshedi, R.G.; Mwanza, D.; et al. Chemical analysis of preovulatory female african elephant urine: A search for putative pheromones. In Chemical Signals in Vertebrates, 10th ed.; Mason, R.T., LeMaster, M.P., Müller-Schwarze, D., Eds.; Springer: Boston, MA, USA, 2005. [Google Scholar] [CrossRef]
- Schulte, B.A.; Bagley, K.; Correll, M.; Gray, A.; Heineman, S.M.; Loizi, H.; Malament, M.; Scott, N.L.; Slade, B.E.; Stanley, L.; et al. Assessing chemical communication in elephants. In Chemical Signals in Vertebrates, 10th ed.; Mason, R.T., LeMaster, M.P., Müller-Schwarze, D., Eds.; Springer: Boston, MA, USA, 2005. [Google Scholar] [CrossRef]
- Baum, M.J.; Bakker, J. Roles of sex and gonadal steroids in mammalian pheromonal communication. Front. Neuroendocr. 2013, 34, 268–284. [Google Scholar] [CrossRef]
- Baum, M.J.; Cherry, J.A. Processing by the main olfactory system of chemosignals that facilitate mammalian reproduction. Horm. Behav. 2015, 68, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Brennan, P.A.; Zufall, F. Pheromonal communication in vertebrates. Nature 2006, 444, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, G.M. Behaviour: Smells, brains and hormones. Nature 2006, 439, 149–151. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Nudelman, A.; Storm, D.R. Are pheromones detected through the main olfactory epithelium? Mol. Neurobiol. 2007, 35, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Slotnick, B.; Restrepo, D.; Schellinck, H.; Archbold, G.; Price, S.; Lin, W. Accessory olfactory bulb function is modulated by input from the main olfactory epithelium. Eur. J. Neurosci. 2010, 31, 1108–1116. [Google Scholar] [CrossRef] [PubMed]
- Haga-Yamanaka, S.; Ma, L.; He, J.; Qiu, Q.; Lavis, L.D.; Looger, L.L.; Yu, C.R. Integrated action of pheromone signals in promoting courtship behavior in male mice. eLife 2014, 3, e03025. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Woszczyło, M.; Jezierski, T.; Szumny, A.; Niżański, W.; Dzięcioł, M. The Role of Urine in Semiochemical Communication between Females and Males of Domestic Dog (Canis familiaris) during Estrus. Animals 2020, 10, 2112. https://doi.org/10.3390/ani10112112
Woszczyło M, Jezierski T, Szumny A, Niżański W, Dzięcioł M. The Role of Urine in Semiochemical Communication between Females and Males of Domestic Dog (Canis familiaris) during Estrus. Animals. 2020; 10(11):2112. https://doi.org/10.3390/ani10112112
Chicago/Turabian StyleWoszczyło, Martyna, Tadeusz Jezierski, Antoni Szumny, Wojciech Niżański, and Michał Dzięcioł. 2020. "The Role of Urine in Semiochemical Communication between Females and Males of Domestic Dog (Canis familiaris) during Estrus" Animals 10, no. 11: 2112. https://doi.org/10.3390/ani10112112
APA StyleWoszczyło, M., Jezierski, T., Szumny, A., Niżański, W., & Dzięcioł, M. (2020). The Role of Urine in Semiochemical Communication between Females and Males of Domestic Dog (Canis familiaris) during Estrus. Animals, 10(11), 2112. https://doi.org/10.3390/ani10112112






