A Retrospective Study of Macropod Progressive Periodontal Disease (“Lumpy Jaw”) in Captive Macropods across Australia and Europe: Using Data from the Past to Inform Future Macropod Management
Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Case Definition
“Proliferative bony change of the maxilla/mandible or soft tissue inflammation (lumps), and/or radiographic/visual evidence of osteomyelitis/osteolysis; accompanied by dental disease. There may or may not be demonstrable bacterial involvement through microbial culture.”
2.2. Selection of Study Institutions
2.3. Extraction of Electronic Records
2.3.1. Taxon Reports
2.3.2. Medical Records (ZIMS and Filemaker)
2.3.3. Specimen Reports
2.3.4. Notes and Observations
2.4. Extraction of Paper-Based Records
2.5. Inter- and Intra-Zoo Transfers
2.6. Selection of Study Population
2.7. Statistical Analyses
2.7.1. Prevalence and Odds Ratio Calculations
2.7.2. Incidence Rate Calculations
3. Results
3.1. Risk Factors
3.1.1. Region
3.1.2. Sex
3.1.3. Age
3.1.4. Study Period
3.1.5. Institution
3.1.6. Genus
3.2. Inter-Zoo Transfers
3.3. Intra-Zoo Transfers
3.4. Outcome (First Incidence of Disease)
4. Discussion
4.1. Regional Prevalence and Incidence Rates
4.2. Risk Factor Analysis
4.2.1. Sex
4.2.2. Age
4.2.3. Genus
4.2.4. Institution
4.2.5. Stress (Intra- and Inter-Zoo Transfers)
4.2.6. Study Period
4.3. Outcome
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Butler, R.; Burton, J. Necrobacillosis of macropods–control and therapy. In Annual Proceedings of the American Association of Zoo Veterinarians; American Association of Zoo Veterinarians: Washington, DC, USA, 1980; pp. 137–140. [Google Scholar]
- Vogelnest, L.; Portas, T. Macropods. In Medicine of Australian Mammals; Vogelnest, L., Woods, R., Eds.; CSIRO Publishing: Collingwood, Victoria, Australia, 2008. [Google Scholar]
- Kido, N.; Chikuan, A.; Omiya, T.; Dan, M.; Nomura, M. Retrospective study of oral necrobacillosis in 54 swamp wallabies. Vet. Rec. 2013, 173, 118. [Google Scholar] [CrossRef] [PubMed]
- Sotohira, Y.; Okui, H.; Suzuki, K.; Asakawa, M.; Sano, T. Association between the levels of stress markers and the onset of kangaroo disease (lumpy jaw disease) in captive kangaroos. J. Zoo Biol. 2017, 1, 17–20. [Google Scholar] [CrossRef]
- Rendle, J.; Yeap, L.; Jackson, B.; Warren, K.; Ward, S.J.; Donaldson, R.; Mayberry, C.; Richardson, J.; Vaughan-Higgins, R. Prevalence of macropod progressive periodontal disease (“lumpy jaw”) in wild western grey kangaroos (Macropus fuliginosus). J. Vertebr. Biol. 2020, 69, 1–12. [Google Scholar] [CrossRef]
- Burton, J.D. Studies into Aspects of Lumpy Jaw in Macropods. Unpublished. Ph.D Thesis, University of Melbourne, Melbourne, Victoria, Australia, 1981. [Google Scholar]
- Bodley, K.; McCracken, H.E.; Lynch, M.J. Dental disease in macropod species at melbourne zoo. In Proceedings of the Joint Conference American Association of Zoo Veterinarians, American Association of Wildlife Veterinarians and Nutrition Advisory Group, Omaha, NE, USA, 14–21 October 2005; Baer, C., Ed.; American Association of Zoo Veterinarians: Omaha, NE, USA, 2005; pp. 243–246. [Google Scholar]
- Jackson, S. Australian Mammals: Biology and Captive Management; CSIRO Publishing: Collingwood, Victoria, Australia, 2003. [Google Scholar]
- Antiabong, J.F.; Boardman, W.; Moore, R.B.; Brown, M.H.; Ball, A.S. The oral microbial community of gingivitis and lumpy jaw in captive macropods. Res. Vet. Sci. 2013, 95, 996. [Google Scholar] [CrossRef]
- Finnie, E.P. Necrobacillosis in kangaroos. In Wildlife Diseases; Page, L.A., Ed.; Springer: Boston, MA, USA, 1976; pp. 511–518. [Google Scholar]
- Calaby, J.H.; Poole, W.E. Keeping kangaroos in captivity. Int. Zoo Yearb. 1971, 11, 5–12. [Google Scholar] [CrossRef]
- Ketz, V. Necrobacillosis in zoo kangaroos. Hypotheses of impact of management conditions. In Erkrankungen der Zootiere. Verhandlungsbericht des 38 Internationalen Symposiums über die Erkrankungen der Zoo und Wildiere; Hofmann, R.R., Schröder, H.D., Wisser, J., Jakob, W., Eds.; Institut für Zoo und Wildtierforschung: Zurich, Switzerland, 1997; Volume 38, pp. 137–142. [Google Scholar]
- Butler, R. Epidemiology and management of “lumpy jaw” in macropods. In Wildlife Diseases of the Pacific Basin and Other Countries; Fowler, M., Ed.; Wildlife Disease Association: Sydney, New South Wales, Australia, 1981; pp. 58–61. [Google Scholar]
- Oliphant, J.C.; Parsons, R.; Smith, G.R. Aetiological agents of necrobacillosis in captive wallabies. Res. Vet. Sci. 1984, 36, 382–384. [Google Scholar] [CrossRef]
- Staker, L. Macropod Husbandry, Healthcare and Medicinals, 1st ed.; Lynda Staker: Townsville, Australia, 2014; Volume 1 & 2. [Google Scholar]
- Broom, D.M. The effects of land transport on animal welfare. Rev. Sci. Et Tech. Off. Int. Des Epizoot. 2005, 24, 683. [Google Scholar] [CrossRef]
- Morgan, K.N.; Tromborg, C.T. Sources of stress in captivity. Appl. Anim. Behav. Sci. 2007, 102, 262–302. [Google Scholar] [CrossRef]
- Borland, D.; Coulson, G.; Beveridge, I. Oral necrobacillosis (‘lumpy jaw’) in a free-ranging population of eastern grey kangaroos (Macropus giganteus) in victoria. Aust. Mammal. 2012, 34, 29–35. [Google Scholar] [CrossRef]
- Clarke, D.E. Oral biology and disorders of chiroptera, insectivores, monotremes, and marsupials. Vet. Clin. N. Am. Exot. Anim. Pract. 2003, 6, 523–564. [Google Scholar] [CrossRef]
- Kirkpatrick, T. Molar progression and macropod age. Qld. J. Agric. Anim. Sci. 1964, 21, 163–165. [Google Scholar]
- Kido, N.; Tanaka, S.; Wada, Y.; Sato, S.; Omiya, T. Molar eruption and identification of the eastern grey kangaroo (Macropus giganteus) at different ages. J. Vet. Med. Sci. 2018, 80, 648–652. [Google Scholar] [CrossRef] [PubMed]
- Newsome, A.; Merchant, J.; Bolton, B.; Dudzinski, M. Sexual dimorphism in molar progression and eruption in the agile wallaby. Wildl. Res. 1977, 4, 1–5. [Google Scholar] [CrossRef]
- Mysterud, A. The relationship between ecological segregation and sexual body size dimorphism in large herbivores. Oecologia 2000, 124, 40–54. [Google Scholar] [CrossRef]
- Thrusfield, M.V.; Christley, R. Veterinary Epidemiology, 4th ed.; Wiley Blackwell: Chichester, UK, 2018. [Google Scholar]
- Noordzij, M.; Dekker, F.W.; Zoccali, C.; Jager, K.J. Measures of disease frequency: Prevalence and incidence. Nephron Clin. Pract. 2010, 115, 17–20. [Google Scholar] [CrossRef] [PubMed]
- McLelland, D. Macropod progressive periodontal disease (‘lumpy jaw’). In Current Therapy in Medicine of Australian Mammals; Vogelnest, L., Portas, T., Eds.; CSIRO Publishing: Clayton South, Victoria, Australia, 2019; pp. 451–462. [Google Scholar]
- Johnson, P.M. Reproduction in the bridled nailtail wallaby, Onychogalea fraenata gould (Marsupialia: Macropodidae), in captivity. Wildl. Res. 1997, 24, 411–415. [Google Scholar] [CrossRef]
- Ross, T.D. Accurate confidence intervals for binomial proportion and poisson rate estimation. Comput. Biol. Med. 2003, 33, 509–531. [Google Scholar] [CrossRef]
- Sergeant, E. Epitools Epidemiological Calculators; Ausvet Pty Ltd.: Fremantle, Australia, 2018; Available online: http://epitools.ausvet.com.au (accessed on 10 September 2020).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Griffith, J.E.; Dhand, N.K.; Krockenberger, M.B.; Higgins, D.P. A retrospective study of admission trends of koalas to a rehabilitation facility over 30 years. J. Wildl. Dis. 2013, 49, 18–28. [Google Scholar] [CrossRef]
- Burgdorf-Moisuk, A.; Pye, G.W.; Smith, J.A.; Papendick, R.; Ivy, J.A.; Hamlin-Andrus, C. A retrospective and prospective study of megaesophagus in the parma wallaby (Macropus parma) at the san diego zoo, california, USA. J. Zoo Wildl. Med. 2012, 43, 324–329. [Google Scholar] [CrossRef]
- Nguyen, N.; Saggese, M.D.; Eng, C. Analysis of historical medical records of california condors (Gymnogyps californianus) admitted for lead exposure to the los angeles zoo and botanical gardens between 1997 and 2012: A case series study. J. Zoo Wildl. Med. 2018, 49, 902–911. [Google Scholar]
- Blyde, D. Advances in treating diseases in macropods. In Wildlife in Australia: Healthcare and Management; Post Graduate Foundation in Veterinary Science: Sydney, Dubbo, Australia, 1999; pp. 439–452. [Google Scholar]
- Clauss, M.; Franz-Odendaal, T.A.; Brasch, J.; Castell, J.C.; Kaiser, T. Tooth wear in captive giraffes (Giraffa camelopardalis): Mesowear analysis classifies free-ranging specimens as browsers but captive ones as grazers. J. Zoo Wildl. Med. 2007, 38, 433–445. [Google Scholar] [CrossRef]
- Brookins, M.D.; Rajeev, S.; Thornhill, T.D.; Kreinheder, K.; Miller, D.L. Mandibular and maxillary osteomyelitis and myositis in a captive herd of red kangaroos (Macropus rufus). J. Vet. Diagn. Investig. Off. Publ. Am. Assoc. Vet. Lab. Diagn. Inc. 2008, 20, 846–849. [Google Scholar] [CrossRef] [PubMed]
- Bakal-Weiss, M.; Steinberg, D.; Friedman, M.; Gati, I. Use of a sustained release chlorhexidine varnish as treatment of oral necrobacillosis in Macropus spp. J. Zoo Wildl. Med. 2010, 41, 371–373. [Google Scholar] [CrossRef] [PubMed]
- Arundel, J.; Barker, I.K.; Beveridge, I. Diseases of marsupials. In The Biology of Marsupials; Stonehouse, B., Gilmore, D., Eds.; Macmillan Press: London, UK, 1977; pp. 141–154. [Google Scholar]
- Miller, W.A.; Beighton, D.; Butler, R. Histological and osteological observations on the early stages of lumpy jaw. In The Comparative Pathology of Zoo Animals: Proceedings of a Symposium Held at the National Zoological Park; Montali, R.J., Migaki, G., Eds.; Smithsonian Institute: Washington, DC, USA, 1978; pp. 231–239. [Google Scholar]
- Mason, G.J. Species differences in responses to captivity: Stress, welfare and the comparative method. Trends Ecol. Evol. 2010, 25, 713–721. [Google Scholar] [CrossRef] [PubMed]
- Schulte-Hostedde, A.I.; Mastromonaco, G.F. Integrating evolution in the management of captive zoo populations. Evol. Appl. 2015, 8, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Hosey, G.R.A.; Melfi, V.A.; Pankhurst, S.A. Zoo Animals: Behaviour, Management and Welfare, 2nd ed.; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
- Rees, P.A. An Introduction to Zoo Biology and Management; Wiley-Blackwell: Chichester, UK, 2011. [Google Scholar]
- Gansloßer, U. Agonistic behaviour in macropodoids: A review. In Kangaroos, Wallabies and Rat-Kangaroos; Grigg, G., Jarman, P., Hume, I., Eds.; Surrey Beatty and Sons: Sydney, New South Wales, Australia, 1989; Volume 2, pp. 475–503. [Google Scholar]
- Dawson, T. Kangaroos: Biology of the Largest Marsupials; University of New South Wales Press: Sydney, New South Wales, Australia, 1995. [Google Scholar]
- Gansloßer, U. The importance of age/sex class relationships for captive-breeding: A review of Macropodoidea (kangaroos, wallabies and rat-kangaroos) as a case study. Anim. Welf. 1995, 4, 239–249. [Google Scholar]
- Rendle, J.; McCormick, W.; Ward, S. Behaviour and enclosure use of captive parma wallabies (Macropus parma): An assessment of compatibility within a mixed-species exhibit. J. Zoo Aquar. Res. 2018, 6, 137–142. [Google Scholar]
- Höhn, M.; Kronschnabl, M.; Gansloßer, U. Similarities and differences in activities and agonistic behavior of male eastern grey kangaroos (Macropus giganteus) in captivity and the wild. Zoo Biol. 2000, 19, 529–539. [Google Scholar] [CrossRef]
- Blyde, D. Common Diseases and Treatments in Macropods. In Proceedings of the American Association of Zoo Veterinarians, St Louis, MO, USA, 9–14 October 1993; American Association of Zoo Veterinarians: Yulee, FL, USA, 1993; pp. 168–170. [Google Scholar]
- Species360. Species360, ZIMS (Version 2.3 updated: 25th January 2016). Zoological Information Management System. Sponsoring Institution Perth Zoo, 2020. Available online: http://zims.Species360.Org (accessed on 16 April 2020).
- Sharman, G.B.; Frith, H.J.; Calaby, J.H. Growth of the pouch young, tooth eruption and age determination in the red kangaroo. Megal. Rufa. Wildl. Res. 1964, 9, 20–49. [Google Scholar] [CrossRef]
- Jarman, P.J. The dietary ecology of macropod marsupials. Proc. Nutr. Soc. Austr. 1984, 9, 82–87. [Google Scholar]
- Garnick, S.; Di Stefano, J.; Moore, B.D.; Davis, N.E.; Elgar, M.A.; Coulson, G. Interspecific and intraspecific relationships between body mass and diet quality in a macropodid community. J. Mammal. 2018, 99, 428–439. [Google Scholar] [CrossRef]
- MacFarlane, A.; Coulson, G. Sexual segregation in australian marsupials. In Sexual Segregation in Vertebrates: Ecology of the Two Sexes; Ruckstuhl, K.E., Neuhaus, P., Eds.; Cambridge University Press: Cambridge, UK, 2005; pp. 254–279. [Google Scholar]
- Dudzinski, M.; Newsome, A.; Merchant, J.; Bolton, B. Comparing the two usual methods for aging Macropodidae on tooth-classes in the agile wallaby. Wildl. Res. 1977, 4, 219–221. [Google Scholar] [CrossRef]
- Blecha, F. Immune system response to stress. In TheBbiology of Animal Stress: Basic Principles and Implications for Animal Welfare; Moberg, G.P., Mench, J.A., Eds.; CABI Publishing: Wallingford, UK, 2000. [Google Scholar]
- Glatt, S.E.; Francl, K.E.; Scheels, J.L. A survey of current dental problems and treatments of zoo animals. Int. Zoo Yearb. 2008, 42, 206–213. [Google Scholar] [CrossRef]
- Learmonth, M.J.; Sherwen, S.; Hemsworth, P.H. The effects of zoo visitors on quokka (Setonix brachyurus) avoidance behavior in a walk-through exhibit. Zoo Biol. 2018, 37, 223–228. [Google Scholar] [CrossRef]
- Sanson, G. Morphological adaptations of teeth to diets and feeding in the macropodoidea. In Kangaroos, Wallabies and Rat-Kangaroos; Grigg, G., Jarman, P., Hume, I., Eds.; Surrey Beathy and Sons: Sydney, New South Wales, Australia, 1989; Volume 1, pp. 151–168. [Google Scholar]
- Arman, S.D.; Prideaux, G.J. Dietary classification of extant kangaroos and their relatives (Marsupialia: Macropodoidea). Australa Ecol. 2015, 40, 909–922. [Google Scholar] [CrossRef]
- Schultz, D.J.; Whitehead, P.J.; Taggart, D.A. Review of surrogacy program for endangered victorian brush-tailed rock wallaby (Petrogale penicillata) with special reference to animal husbandry and veterinary considerations. J. Zoo Wildl. Med. 2006, 37, 33–39. [Google Scholar] [CrossRef]
- Sherwen, S.L.; Hemsworth, P.H.; Butler, K.L.; Fanson, K.V.; Magrath, M.J.L. Impacts of visitor number on kangaroos housed in free-range exhibits. Zoo Biol. 2015, 34, 287–295. [Google Scholar] [CrossRef]
- Takai, S.; Ohbushi, S.; Koike, K.; Tsubaki, S.; Oishi, H.; Kamada, M. Prevalence of virulent Rhodococcus equi in isolates from soil and feces of horses from horse-breeding farms with and without endemic infections. J. Clin. Microbiol. 1991, 29, 2887–2889. [Google Scholar] [CrossRef]
- Witcomb, L.A.; Green, L.E.; Kaler, J.; Ul-Hassan, A.; Calvo-Bado, L.A.; Medley, G.F.; Grogono-Thomas, R.; Wellington, E.M.H. A longitudinal study of the role of Dichelobacter nodosus and Fusobacterium necrophorum load in initiation and severity of footrot in sheep. Prev. Vet. Med. 2014, 115, 48–55. [Google Scholar] [CrossRef]
- Broom, D.M. Transport stress in cattle and sheep with details of physiological, ethological and other indicators. Dtsch. Tierarztl. Wochenschr. 2003, 110, 83–88. [Google Scholar]
- Dembiec, D.P.; Snider, R.J.; Zanella, A.J. The effects of transport stress on tiger physiology and behavior. Zoo Biol. 2004, 23, 335–346. [Google Scholar] [CrossRef]
- Grandin, T. Assessment of stress during handling and transport. J. Anim. Sci. 1997, 75, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Padalino, B.; Hall, E.; Raidal, S.; Celi, P.; Knight, P.; Jeffcott, L.; Muscatello, G. Health problems and risk factors associated with long haul transport of horses in Australia. Animals 2015, 5, 1296–1310. [Google Scholar] [CrossRef] [PubMed]
- Laule, G.E.; Bloomsmith, M.A.; Schapiro, S.J. The use of positive reinforcement training techniques to enhance the care, management, and welfare of primates in the laboratory. J. Appl. Anim. Welf. Sci. 2003, 6, 163–173. [Google Scholar] [CrossRef]
- Ward, S.J.; Melfi, V. The implications of husbandry training on zoo animal response rates. Appl. Anim. Behav. Sci. 2013, 147, 179–185. [Google Scholar] [CrossRef]
- Clauss, M.; Kaiser, T.; Hummel, J. The morphophysiological adaptations of browsing and grazing mammals. In The Ecology of Browsing and Grazing; Springer: Berlin, Germany, 2008; pp. 47–88. [Google Scholar]
- Kaiser, T.M.; Brasch, J.; Castell, J.C.; Schulz, E.; Clauss, M. Tooth wear in captive wild ruminant species differs from that of free-ranging conspecifics. Mamm. Biol. Z. Für Säugetierkunde 2009, 74, 425–437. [Google Scholar] [CrossRef]

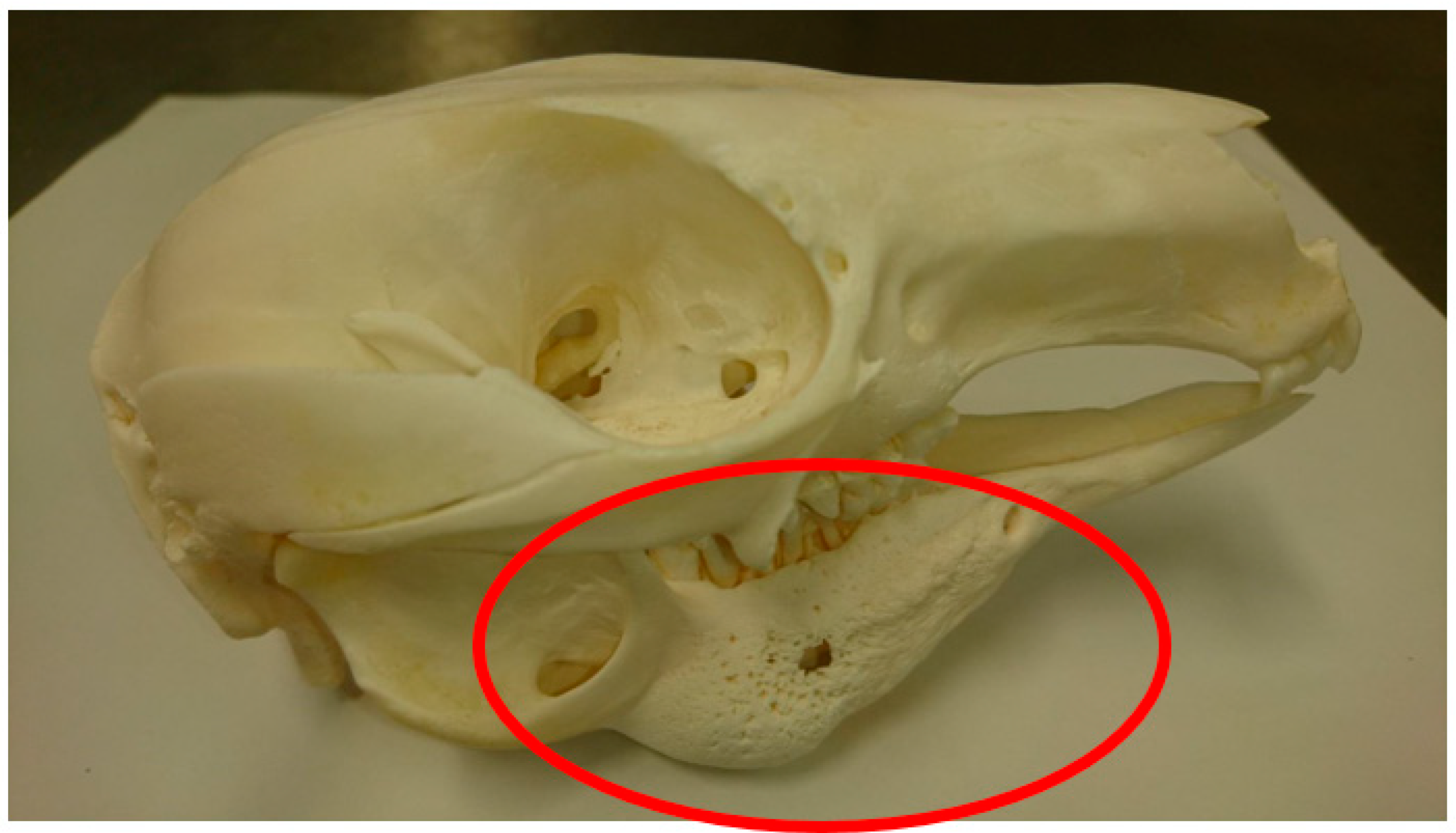
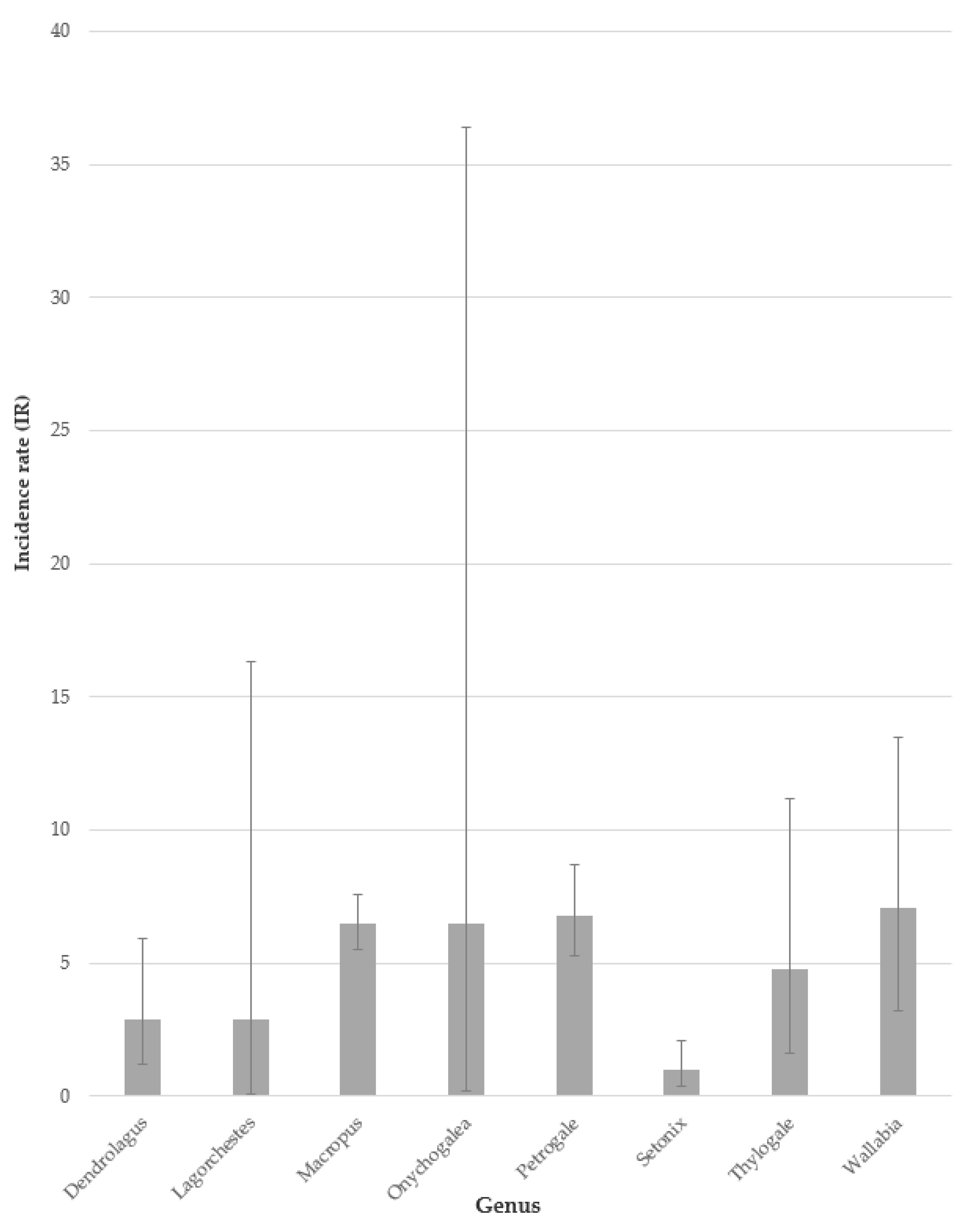
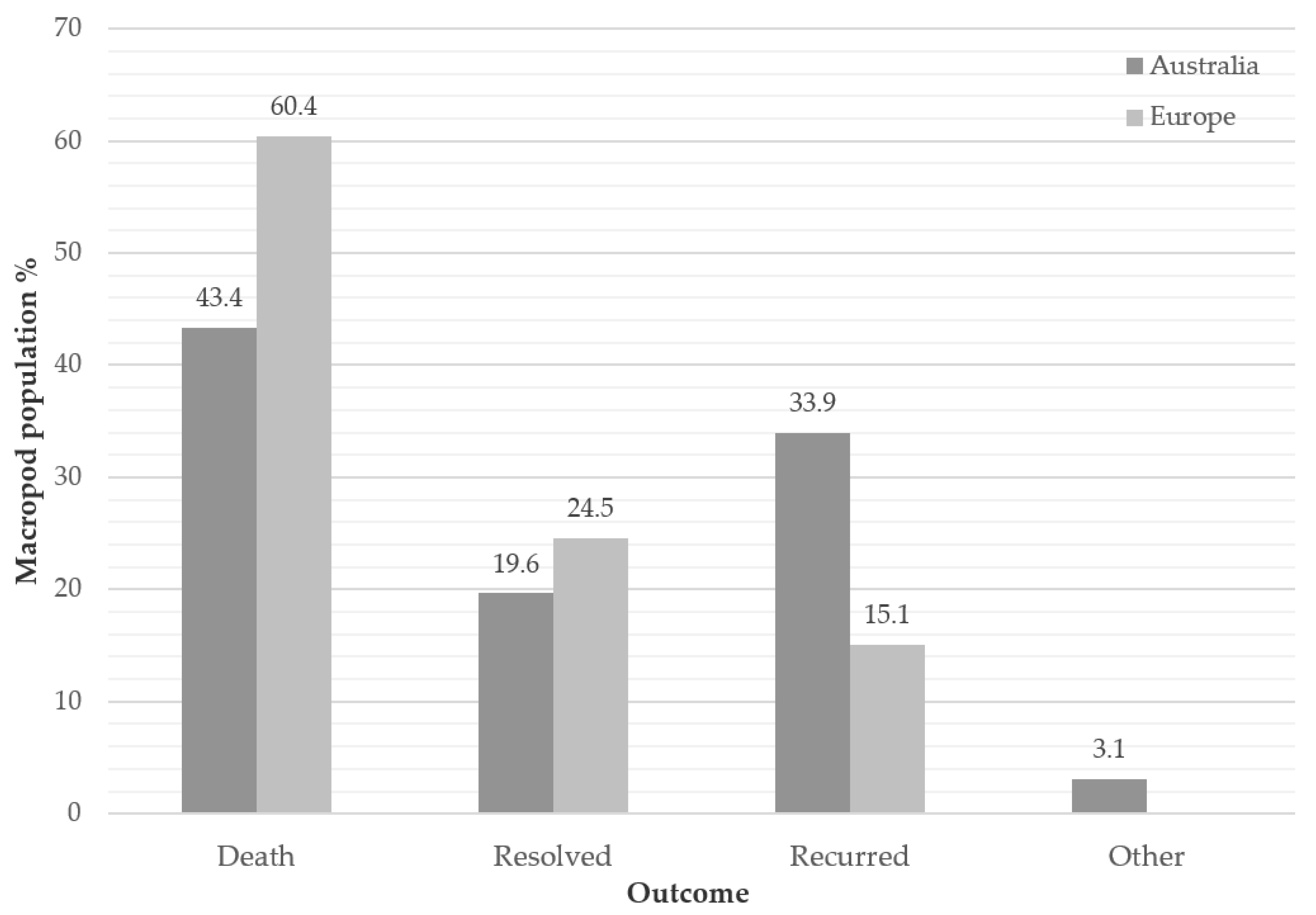
| Region | Institution | Total Population | Sex | No. of Genera | No. of Species | Total MPPD Cases |
|---|---|---|---|---|---|---|
| Australia | A1 | 643 | 257.359.27 | 5 | 11 | 89 |
| A2 | 354 | 151.190.13 | 7 | 17 | 66 | |
| A3 | 305 | 100.194.11 | 7 | 15 | 22 | |
| A4 | 318 | 135.165.18 | 8 | 19 | 47 | |
| Europe | E1 | 60 | 25.34.1 | 1 | 1 | 10 |
| E2 | 224 | 109.112.3 | 3 | 8 | 25 | |
| E3 | 84 | 41.35.8 | 2 | 5 | 11 | |
| E4 | 66 | 32.28.6 | 1 | 1 | 7 |
| Risk Factor | Australia IR (95% CI) | Europe IR (95% CI) |
|---|---|---|
| Sex | ||
| Male | 5.6 (4.3–7.1) | 6.2 (4.0–9.2) |
| Female | 5.9 (5.0–7.0) | 4.1 (2.7–6.1) |
| Age | ||
| <1 | 1.9 (1.0–3.2) | 2.2 (0.7–5.2) |
| 1–5 | 5.8 (4.7–7.1) | 5.4 (3.4–8.0) |
| 5–9 | 6.5 (5.0–8.4) | 4.4 (2.1–8.1) |
| 10+ | 12.3 (8.4–17.3) | 9.3 (4.6–16.7) |
| Study period | ||
| 1995–1999 | 5.5 (4.1–7.2) | 1.1 (0.1–3.9) |
| 2000–2004 | 6.6 (4.9–8.6) | 4.6 (2.0–9.1) |
| 2005–2009 | 6.8 (5.1–9.0) | 6.5 (3.7–10.5) |
| 2010–2016 | 4.6 (3.4–6.0) | 5.8 (3.7–8.7) |
| Institution | ||
| A1/E1 | 7.6 (6.0–9.4) | 5.3 (2.6–9.8) |
| A2/E2 | 7.4 (5.7–9.4) | 5.0 (3.1–7.4) |
| A3/E3 | 2.4 (1.4–3.9) | 5.9 (2.8–10.9) |
| A4/E4 | 4.5 (3.2–6.0) | 3.6 (1.4–7.3) |
| Risk Factor | Adjusted Incidence Rates | |||
|---|---|---|---|---|
| Australia | Europe | |||
| IRR (95% CI) | p-Value | IRR (95% CI) | p-Value | |
| Study period | ||||
| 2000–2004 | 1.20 (0.82–1.77) | 0.345 | 4.60 (1.14–30.56) | 0.055 |
| 2005–2009 | 1.10 (0.74–1.63) | 0.633 | 7.39 (2.06–47.21) | 0.008 ** |
| 2010–2016 | 0.75 (0.50–1.11) | 0.154 | 6.94 (1.96–44.18) | 0.010 ** |
| Age group | ||||
| 1–4 | 3.36 (1.95–6.31) | <0.001 *** | 2.46 (1.02–7.32) | 0.067 |
| 5–9 | 3.92 (2.22–7.47) | <0.001 *** | 2.72 (0.94–8.93) | 0.075 |
| 10+ | 7.63 (4.06–15.20) | <0.001 *** | 7.38 (2.50–24.85) | <0.001 *** |
| Institution | ||||
| Zoo A1/E1 | 3.88 (2.32–6.91) | <0.001 *** | 1.38 (0.52–3.86) | 0.524 |
| Zoo A2/E2 | 3.27 (1.94–5.86) | <0.001 *** | 1.12 (0.50–2.86) | 0.790 |
| Zoo A4/E3 | 2.09 (1.19–3.84) | 0.013 ** | 1.20 (0.44–3.45) | 0.723 |
| Sex | ||||
| Male | 1.08 (0.80–1.44) | 0.611 | 2.02 (1.08–3.83) | 0.029 * |
| Number of Inter-Zoo Transfers | Adjusted Incidence Rates | |||
|---|---|---|---|---|
| Australia | Europe | |||
| OR (95% CI) | p-Value | OR (95% CI) | p-Value | |
| 0 | - | - | - | - |
| 1 | 1.69 (1.23–2.32) | 0.001 *** | 0.91 (0.48–1.71) | 0.77 |
| 2 | 1.92 (1.19–3.10) | 0.01 ** | 2.4 (0.73–7.89) | 0.14 |
| 3 | 3.29 (1.48–7.30) | 0.002 ** | 2.37 (0.10–59.45) | 1 |
| 4 | 1.75 (0.20–12.18) | 0.48 | - | - |
| 5 | 8.76 (1.22–63.02) | 0.06 | - | - |
| 6 | 17.53 (1.57–195.35) | 0.03 * | - | - |
| 7 | 43.60 (2.08–915.60) | 0.01 ** | - | - |
| No. Intra-Zoo Transfers | Estimates | |||
|---|---|---|---|---|
| Australia | Europe | |||
| OR (95% CI) | p-Value | OR (95% CI) | p-Value | |
| 0 | - | - | - | - |
| 1 | 1.58 (0.78–3.22) | 0.20 | 1.67 (0.82–3.39) | 0.15 |
| 2 | 2.68 (1.36–5.30) | 0.003 ** | 0.83 (0.24–2.88) | 1 |
| 3 | 4.07 (2.06–8.04) | <0.0001 *** | 1.09 (0.06–21.52) | 1 |
| 4 | 6.61 (3.35–13.05) | <0.0001 *** | 2.53 (0.10–63.56) | 1 |
| 5 | 5.26 (2.45–11.3) | <0.0001 *** | - | - |
| 6–10 | 9.93 (5.57–17.73) | <0.0001 *** | - | - |
| 11+ | 16.18 (8.73–29.98) | <0.0001 *** | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rendle, J.; Jackson, B.; Hoorn, S.V.; Yeap, L.; Warren, K.; Donaldson, R.; Ward, S.J.; Vogelnest, L.; McLelland, D.; Lynch, M.; et al. A Retrospective Study of Macropod Progressive Periodontal Disease (“Lumpy Jaw”) in Captive Macropods across Australia and Europe: Using Data from the Past to Inform Future Macropod Management. Animals 2020, 10, 1954. https://doi.org/10.3390/ani10111954
Rendle J, Jackson B, Hoorn SV, Yeap L, Warren K, Donaldson R, Ward SJ, Vogelnest L, McLelland D, Lynch M, et al. A Retrospective Study of Macropod Progressive Periodontal Disease (“Lumpy Jaw”) in Captive Macropods across Australia and Europe: Using Data from the Past to Inform Future Macropod Management. Animals. 2020; 10(11):1954. https://doi.org/10.3390/ani10111954
Chicago/Turabian StyleRendle, Jessica, Bethany Jackson, Stephen Vander Hoorn, Lian Yeap, Kristin Warren, Rebecca Donaldson, Samantha J. Ward, Larry Vogelnest, David McLelland, Michael Lynch, and et al. 2020. "A Retrospective Study of Macropod Progressive Periodontal Disease (“Lumpy Jaw”) in Captive Macropods across Australia and Europe: Using Data from the Past to Inform Future Macropod Management" Animals 10, no. 11: 1954. https://doi.org/10.3390/ani10111954
APA StyleRendle, J., Jackson, B., Hoorn, S. V., Yeap, L., Warren, K., Donaldson, R., Ward, S. J., Vogelnest, L., McLelland, D., Lynch, M., Vitali, S., Sayers, G., Wyss, F., Webster, D., Snipp, R., & Vaughan-Higgins, R. (2020). A Retrospective Study of Macropod Progressive Periodontal Disease (“Lumpy Jaw”) in Captive Macropods across Australia and Europe: Using Data from the Past to Inform Future Macropod Management. Animals, 10(11), 1954. https://doi.org/10.3390/ani10111954






