Anaplasmosis Outbreak in Lambs: First Report Causing Carcass Condemnation
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Studied Lambs
2.2. Microbiological Analysis
2.3. Biomolecular Analysis
2.4. Clinical Examination
2.5. Haematological and Biochemical Analysis
2.6. Pathological Study
2.7. Statistical Analysis
3. Results and Discussion
3.1. Anaplasma ovis Detection by qPCR
3.2. Clinical Examination
3.3. Haematological Analysis
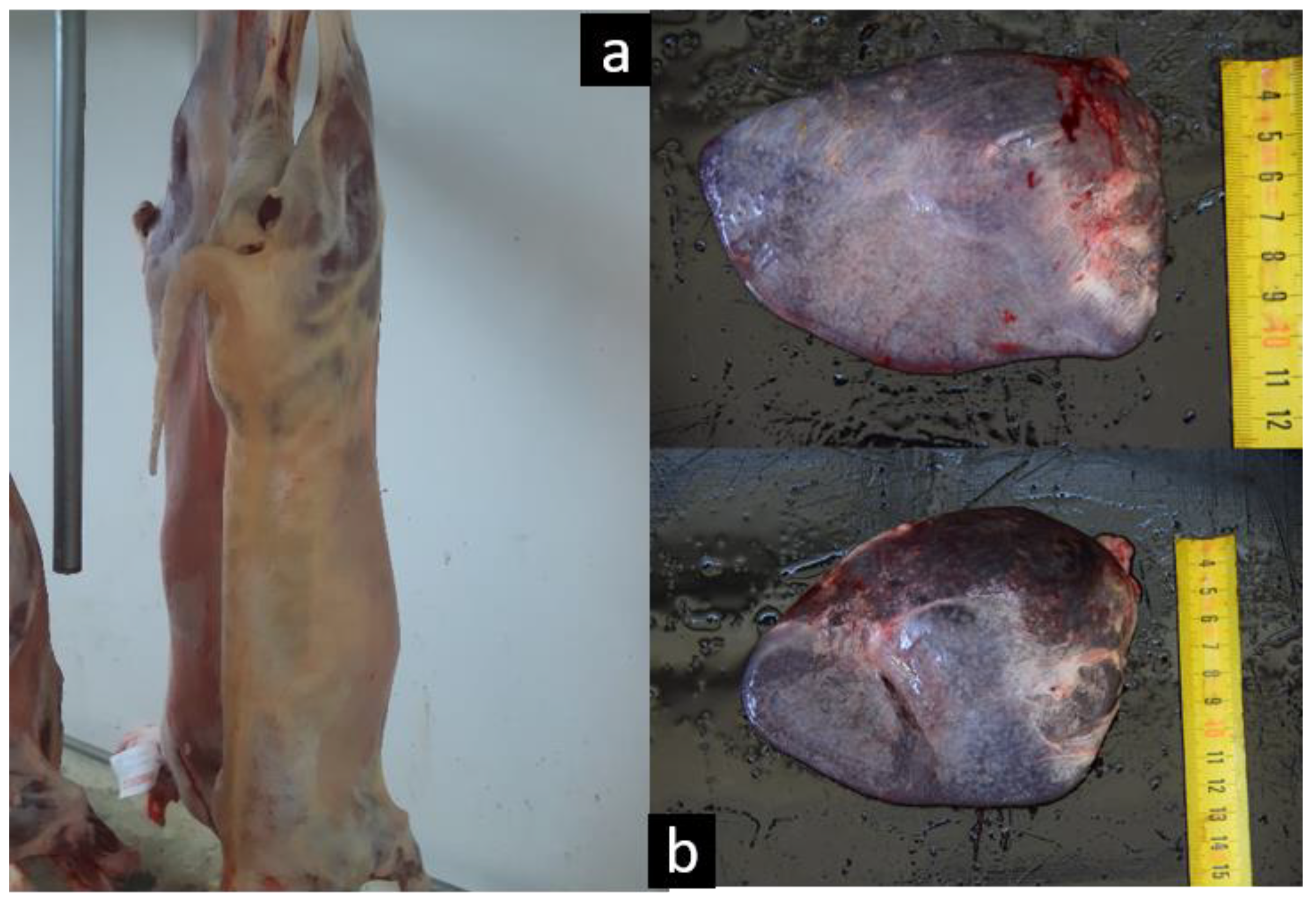
3.4. Pathological Findings
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rymaszewska, A.; Grenda, S. Bacteria of the genus Anaplasma–characteristics of Anaplasma and their vectors: A review. Vet. Med. 2008, 53, 573–584. [Google Scholar] [CrossRef] [Green Version]
- Gharbi, M.; Omri, H.; Jedidi, M.; Zorii, S.; Darghouth, M.A. Epidemiological study of sheep anaplasmosis (Anaplasma ovis infection) in Kairouan, Central Tunisia. J. Adv. Parasitol. 2015, 2, 30–33. [Google Scholar] [CrossRef]
- Hornok, S.; Elek, V.; de la Fuente, J.; Naranjo, V.; Farkas, R.; Majoros, G.; Foldvari, G. First serological and molecular evidence on the endemicity of Anaplasma ovis and A. marginale in Hungary. Vet. Microbial. 2007, 122, 316–322. [Google Scholar] [CrossRef] [PubMed]
- Stuen, S. Haemoparasites in small ruminants in European countries: Challenges and clinical relevance. Small Rumin. Res. 2016, 142, 22–27. [Google Scholar] [CrossRef]
- Renneker, S.; Abdo, J.; Salih, D.E.A.; Karagenç, T.; Bilgiç, H.; Torina, A.; Seitzer, U. Can Anaplasma ovis in small ruminants be neglected any longer? Transbound. Emerg. Dis. 2013, 60, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, C.; Benito, A.; Arnal, J.L.; Ortín, A.; Gómez, M.; López, A.; Villanueva-Saz, S.; Lacasta, D. Anaplasma ovis in sheep: Experimental infection, vertical transmission and colostral immunity. Small Rumin. Res. 2019, 178, 7–14. [Google Scholar] [CrossRef]
- Hashemi-Fesharki, R. Tick-borne diseases of sheep and goats and their related vectors in Iran. Parassitologia 1997, 39, 115–117. [Google Scholar] [PubMed]
- Cabezas-Cruz, A.; Gallois, M.; Fontugne, M.; Allain, E.; Denoual, M.; Moutailler, S.; Devillers, E.; Zientara, S.; Memmi, M.; Chauvin, A.; et al. Epidemiology and genetic diversity of Anaplasma ovis in goats in Corsica, France. Parasites Vectors 2019, 12. [Google Scholar] [CrossRef] [Green Version]
- De la Fuente, J.; Ruiz-Fons, F.; Naranjo, V.; Torina, A.; Rodríguez, O.; Gortázar, C. Evidence of Anaplasma infections in European roe deer (Capreolus capreolus) from Southern Spain. Res. Vet. Sci. 2008, 84, 382–386. [Google Scholar] [CrossRef]
- Masake, R.; Musoke, A. Blood Parasitic Diseases and Specific Immune Responses. Int. Livest. Res. Inst. 2010, OIE, 43–55. [Google Scholar]
- Bautista, G. La respuesta inmune celular en anaplasmosis bovina. Cienc. Vet. 1996, 7, 315–329. [Google Scholar]
- Brown, W.C. Adaptive immunity to Anaplasma pathogens and immune dysregulation: Implications for bacterial persistence. Comp. Immunol. Microbiol. Infect. Dis. 2012, 35, 241–252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torina, A.; Agnone, A.; Blanda, V.; Alongi, A.; D’Agostino, R.; Caracappa, S.; Marino, A.; Di Marco, V.; De la Fuente, J. Development and validation of two PCR tests for the detection of and differentiation between Anaplasma ovis and Anaplasma marginale. Ticks Tick Borne Dis. 2012, 3, 282–286. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Wittwer, C.T. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef] [Green Version]
- Otter, A.; Uzal, F.A. Clostridial diseases in farm animals: 1. Enterotoxaemias and other alimentary tract infections. Practice 2020, 42, 219–232. [Google Scholar] [CrossRef]
- Friedhoff, K.T. Tick-borne diseases of sheep and goats caused by Babesia, Theileria or Anaplasma spp. Parassitologia 1997, 39, 99–109. [Google Scholar]
- Urie, N.J.; Highland, M.A.; Knowles, D.P.; Branana, M.A.; Herndon, D.R.; Marshall, K.L. Mycoplasma ovis infection in domestic sheep (Ovis aries) in the United States: Prevalence, distribution, associated risk factors, and associated outcomes. Prev. Vet. Med. 2019, 171, 104750. [Google Scholar] [CrossRef]
- Katsogiannou, E.; Athanasiou, L.; Christodoulopoulos, G.; Polizopoulou, Z. Diagnostic approach of anemia in ruminants. J. Hell. Vet. Med. Soc. 2018, 69, 1033–1046. [Google Scholar] [CrossRef] [Green Version]
- Suarez, C.E.; Noh, S. Emerging perspectives in the research of bovine babesiosis and anaplasmosis. Vet. Parasitol. 2011, 180, 109–125. [Google Scholar] [CrossRef]
- Lepherd, M.L.; Canfield, P.J.; Hunt, G.B.; Bosward, K.L. Haematological, biochemical and selected acute phase protein reference intervals for weaned female Merino lambs. Aust. Vet. J. 2009, 87, 5–11. [Google Scholar] [CrossRef]
- Alessandra, T.; Santo, C. Tick-borne diseases in sheep and goats: Clinical and diagnostic aspects. Small Rumin. Res. 2012, 106, S6–S11. [Google Scholar] [CrossRef]
- Boes, K.M.; Durham, A.C. Bone Marrow, Blood Cells, and the Lymphoid/Lymphatic System. In Pathologic Basis of Veterinary Diseases, 6th ed.; Elsevier: St. Louis, MO, USA, 2017. [Google Scholar]
| Haematological Parameters | PCR Negative (n = 6) | PCR Positive (<38) (n = 37) | Haematological Threshold Values |
|---|---|---|---|
| Erythrocytes (M/µL) | 13.12 a (12.395–13.440) | 8.63 b (6.800–11.540) | 9.49–15.12 M/μL |
| Haematocrit (%) | 36.50 a (34.75–38.00) | 30.53 b (25.50–36.00) | 27.0–42.0% |
| Haemoglobin (g/dL) | 11.80 a (11.45–12.00) | 9.90 b (7.70–11-10) | 10.0–14.9 g/dL |
| MCV (fL) | 27.70 a (25.30–30.73) | 35.10 b (29.20–39.50) | 24.4–32.5 fL |
| Reticulocytes (K/µL) | 4.45 (3.48–11.23) | 9.90 (4.85–24.40) | 0.0–15.0 K/μL |
| Haematological Parameters | Normal Carcass (n = 34) | Icteric Carcass (n = 37) | Haematological Threshold Values |
|---|---|---|---|
| Erythrocytes (M/µL) | 11.97 a (10.335–13.153) | 7.18 b (6.380–8.515) | 9.49–15.12 M/μL |
| Haematocrit (%) | 34.82 a (31.00–37.00) | 26.89 b (22.00–31.00) | 27.0–42.0% |
| Haemoglobin (g/dL) | 11.10 a (10.30–11.80) | 8.50 b (7.20–9.45) | 10.0–14.9 g/dL |
| MCV (fL) | 29.45 a (25.88–34.73) | 34.50 b (31.20–38.65) | 24.4–32.5 fL |
| Reticulocytes (K/µL) | 5.65 a (4.150–9.525) | 18.80 b (5.400–40.000) | 0.0–15.0 K/μL |
| Platelets (K/µL) | 467.5 ± 24.68 | 442.7 ± 24.28 | 301–922 K/μL |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lacasta, D.; Ferrer, L.M.; Sanz, S.; Labanda, R.; González, J.M.; Benito, A.Á.; Ruiz, H.; Rodríguez-Largo, A.; Ramos, J.J. Anaplasmosis Outbreak in Lambs: First Report Causing Carcass Condemnation. Animals 2020, 10, 1851. https://doi.org/10.3390/ani10101851
Lacasta D, Ferrer LM, Sanz S, Labanda R, González JM, Benito AÁ, Ruiz H, Rodríguez-Largo A, Ramos JJ. Anaplasmosis Outbreak in Lambs: First Report Causing Carcass Condemnation. Animals. 2020; 10(10):1851. https://doi.org/10.3390/ani10101851
Chicago/Turabian StyleLacasta, Delia, Luis Miguel Ferrer, Santiago Sanz, Raquel Labanda, José María González, Alfredo Ángel Benito, Héctor Ruiz, Ana Rodríguez-Largo, and Juan José Ramos. 2020. "Anaplasmosis Outbreak in Lambs: First Report Causing Carcass Condemnation" Animals 10, no. 10: 1851. https://doi.org/10.3390/ani10101851






