Faecal Glucocorticoid Metabolites and H/L Ratio Are Related Markers of Stress in Semi-Captive Asian Timber Elephants
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Outline
2.3. Physiological Markers of Stress
2.3.1. Faecal Sample Collection and FGM Analysis
2.3.2. Blood Smear Preparation and Heterophils/Lymphocytes Ratio
2.4. Body Weight
2.5. Statistical Analysis
3. Results
3.1. Minima, Maxima and Averages of FGM Concentrations and H/L Ratios
3.2. The General Correlation between H/L Ratio and FGM Concentrations
3.3. Correlations between H/L Ratio and FGM Concentrations Depending on Sex
3.4. Correlations between H/L Ratio and FGM Depending on Age Classes
3.5. Correlations between H/L Ratio and FGM Concentrations Depending on Season
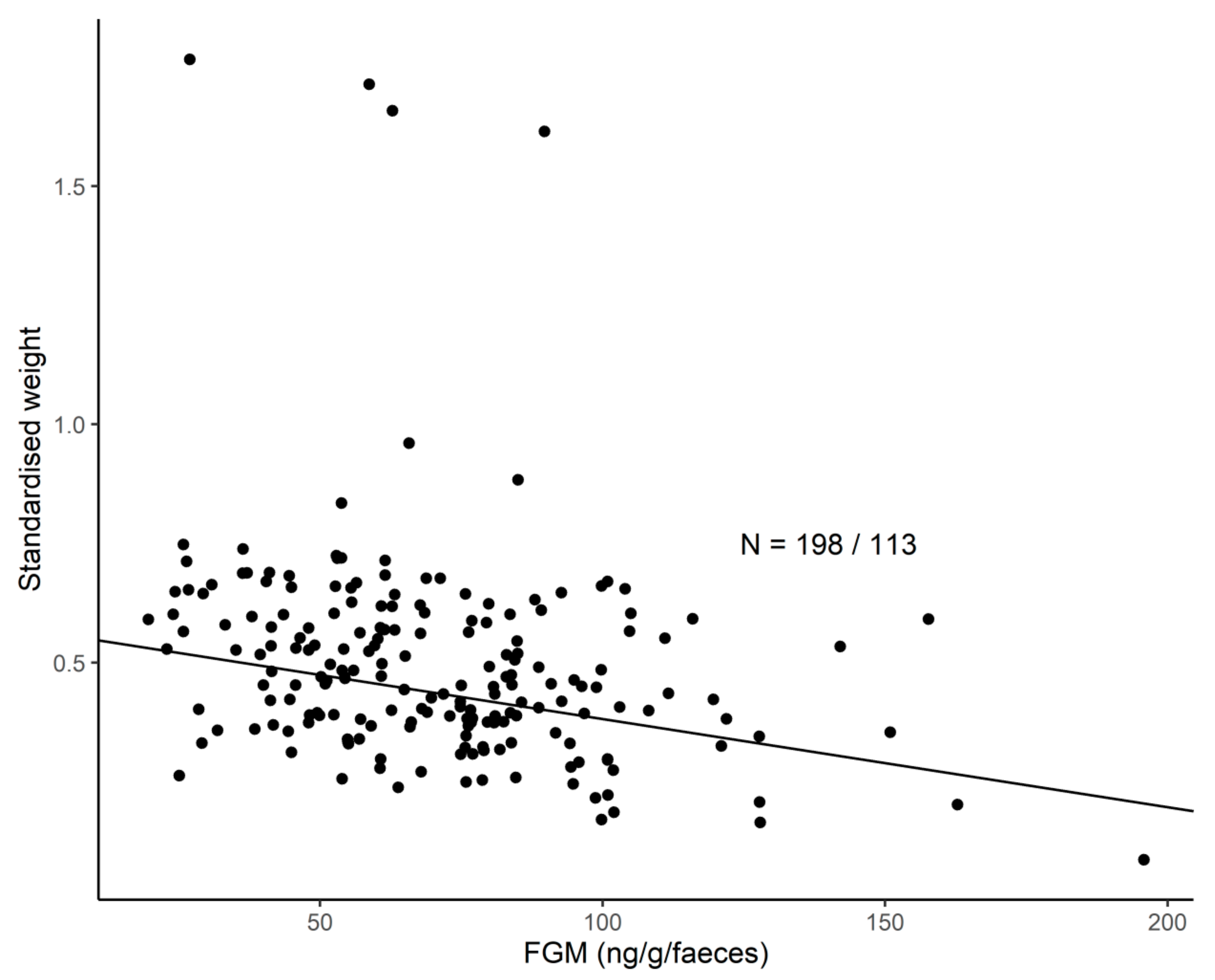
3.6. FGM Concentrations and H/L Ratio and Their Links to Body Weight
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tidière, M.; Gaillard, J.-M.; Berger, V.; Müller, D.W.H.; Lackey, L.B.; Gimenez, O.; Clauss, M.; Lemaître, J.-F. Comparative analyses of longevity and senescence reveal variable survival benefits of living in zoos across mammals. Sci. Rep. 2016, 6, 36361. [Google Scholar] [CrossRef]
- Morgan, K.N.; Tromborg, C.T. Sources of stress in captivity. Appl. Anim. Behav. Sci. 2007, 102, 262–302. [Google Scholar] [CrossRef]
- van Schaik, C.P.; Burkart, J.; Damerius, L.; Forss, S.I.F.; Koops, K.; van Noordwijk, M.A.; Schuppli, C. The reluctant innovator: Orangutans and the phylogeny of creativity. Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 20150183. [Google Scholar] [CrossRef] [PubMed]
- Sukumar, R. The Living Elephants: Evolutionary Ecology, Behavior, and Conservation; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Dawkins, M.S. A user’s guide to animal welfare science. Trends Ecol. Evol. 2006, 21, 77–82. [Google Scholar] [CrossRef]
- Dawkins, M.S. What is good welfare and how can we achieve it? In Future of Animal Farming: Renewing the Ancient Contract; Dawkins, M.S., Bonney, R., Eds.; BlackWell Publishing Ltd.: Malden, MA, USA, 2008. [Google Scholar]
- Hill, S.P.; Broom, D.M. Measuring zoo animal welfare: Theory and practice. Zoo Biol. 2009, 28, 531–544. [Google Scholar] [CrossRef] [PubMed]
- Busch, D.S.; Hayward, L.S. Stress in a conservation context: A discussion of glucocorticoid actions and how levels change with conservation-relevant variables. Biol. Conserv. 2009, 142, 2844–2853. [Google Scholar] [CrossRef]
- Romero, L.M. Physiological stress in ecology: Lessons from biomedical research. Trends Ecol. Evol. 2004, 19, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Clinchy, M.; Sheriff, M.J.; Zanette, L.Y. Predator-induced stress and the ecology of fear. Funct. Ecol. 2013, 27, 56–65. [Google Scholar] [CrossRef]
- Creel, S. Social dominance and stress hormones. Trends Ecol. Evol. 2001, 16, 491–497. [Google Scholar] [CrossRef]
- Krause, J.S.; Pérez, J.H.; Chmura, H.E.; Meddle, S.L.; Hunt, K.E.; Gough, L.; Boelman, N.; Wingfield, J.C. The stress response is attenuated during inclement weather in parental, but not in pre-parental, Lapland longspurs (Calcarius lapponicus) breeding in the Low Arctic. Horm. Behav. 2016, 83, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Lindström, K.M.; Hawley, D.M.; Davis, A.K.; Wikelski, M. Stress responses and disease in three wintering house finch (Carpodacus mexicanus) populations along a latitudinal gradient. Gen. Comp. Endocrinol. 2005, 143, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Wolf, T.E.; Bennett, N.C.; Burroughs, R.; Ganswindt, A. The impact of age-class and social context on fecal glucocorticoid metabolite levels in free-ranging male giraffes. Gen. Comp. Endocrinol. 2018, 255, 26–31. [Google Scholar] [CrossRef]
- Heidinger, B.J.; Nisbet, I.C.T.; Ketterson, E.D. Older parents are less responsive to a stressor in a long-lived seabird: A mechanism for increased reproductive performance with age? Proc. R. Soc. B Biol. Sci. 2006, 273, 2227–2231. [Google Scholar] [CrossRef] [PubMed]
- Narayan, E.J.; Parnell, T.; Clark, G.; Martin-Vegue, P.; Mucci, A.; Hero, J.-M. Faecal cortisol metabolites in Bengal (Panthera tigris tigris) and Sumatran tigers (Panthera tigris sumatrae). Gen. Comp. Endocrinol. 2013, 194, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Kudielka, B.M.; Kirschbaum, C. Sex differences in HPA axis responses to stress: A review. Biol. Psychol. 2005, 69, 113–132. [Google Scholar] [CrossRef] [PubMed]
- Jimeno, B.; Briga, M.; Verhulst, S.; Hau, M. Effects of developmental conditions on glucocorticoid concentrations in adulthood depend on sex and foraging conditions. Horm. Behav. 2017, 93, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Mumby, H.S.; Mar, K.U.; Thitaram, C.; Courtiol, A.; Towiboon, P.; Min-Oo, Z.; Htut-Aung, Y.; Brown, J.L.; Lummaa, V. Stress and body condition are associated with climate and demography in Asian elephants. Conserv. Physiol. 2015, 3, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Hämäläinen, A.; Heistermann, M.; Kraus, C. The stress of growing old: Sex- and season-specific effects of age on allostatic load in wild grey mouse lemurs. Oecologia 2015, 178, 1063–1075. [Google Scholar] [CrossRef]
- Lynch, J.W.; Ziegler, T.E.; Strier, K.B. Individual and seasonal variation in fecal testosterone and cortisol levels of wild male tufted capuchin monkeys, Cebus apella nigritus. Horm. Behav. 2002, 41, 275–287. [Google Scholar] [CrossRef]
- Bonier, F.; Martin, P.R.; Moore, I.T.; Wingfield, J.C. Do baseline glucocorticoids predict fitness? Trends Ecol. Evol. 2009, 24, 634–642. [Google Scholar] [CrossRef] [PubMed]
- Bonier, F.; Moore, I.T.; Martin, P.R.; Robertson, R.J. The relationship between fitness and baseline glucocorticoids in a passerine bird. Gen. Comp. Endocrinol. 2009, 163, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Jaatinen, K.; Seltmann, M.W.; Öst, M. Context-dependent stress responses and their connections to fitness in a landscape of fear. J. Zool. 2014, 294, 147–153. [Google Scholar] [CrossRef]
- Wikelski, M.; Cooke, S.J. Conservation physiology. Trends Ecol. Evol. 2006, 21, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Wasser, S.K.; Hunt, K.E.; Brown, J.L.; Cooper, K.; Crockett, C.M.; Bechert, U.; Millspaugh, J.J.; Larson, S.; Monfort, S.L. A generalized fecal glucocorticoid assay for use in a diverse array of nondomestic mammalian and avian species. Gen. Comp. Endocrinol. 2000, 120, 260–275. [Google Scholar] [CrossRef] [PubMed]
- Seltmann, M.W.; Öst, M.; Jaatinen, K.; Atkinson, S.; Mashburn, K.; Hollmén, T. Stress responsiveness, age and body condition interactively affect flight initiation distance in breeding female eiders. Anim. Behav. 2012, 84, 889–896. [Google Scholar] [CrossRef]
- Baugh, A.T.; Oers, K.; van Dingemanse, N.J.; Hau, M. Baseline and stress-induced glucocorticoid concentrations are not repeatable but covary within individual great tits (Parus major). Gen. Comp. Endocrinol. 2014, 208, 154–163. [Google Scholar] [CrossRef]
- Hudson, R.; Maqueda, B.; Velázquez Moctezuma, J.; Morales Miranda, A.; Rödel, H.G. Individual differences in testosterone and corticosterone levels in relation to early postnatal development in the rabbit Oryctolagus cuniculus. Physiol. Behav. 2011, 103, 336–341. [Google Scholar] [CrossRef]
- Romero, L.M.; Reed, J.M. Collecting baseline corticosterone samples in the field: Is under 3 min good enough? Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2005, 140, 73–79. [Google Scholar] [CrossRef]
- Wingfield, J.C.; Vleck, C.M.; Moore, M.C. Seasonal changes of the adrenocortical response to stress in birds of the Sonoran Desert. J. Exp. Zool. 1992, 264, 419–428. [Google Scholar] [CrossRef]
- Harper, J.M.; Austad, S.N. Fecal glucocorticoids: A noninvasive method of measuring adrenal activity in wild and captive rodents. Physiol. Biochem. Zool. 2000, 73, 12–22. [Google Scholar] [CrossRef]
- Davis, A.K.; Maney, D.L.; Maerz, J.C. The use of leukocyte profiles to measure stress in vertebrates: A review for ecologists. Funct. Ecol. 2008, 22, 760–772. [Google Scholar] [CrossRef]
- Salakij, J.; Salakij, C.; Narkkong, N.-A.; Apibal, S.; Suthunmapinuntra, P.; Rattanakukuprakarn, J.; Nunklang, G.; Yindee, M. Hematology, cytochemistry and ultrastructure of blood cells from Asian Elephant (Elephas maximus). Kasetsart J. (Nat. Sci.) 2005, 39, 482–493. [Google Scholar]
- Silva, I.D.; Kuruwita, V.Y. Hematology, plasma, and serum biochemistry values in free-ranging elephants (Elephas maximus ceylonicus) in Sri Lanka. J. Zoo Wildl. Med. 1993, 24, 434–439. [Google Scholar]
- Maxwell, M.H. Avian blood leucocyte responses to stress. World’s Poult. Sci. J. 1993, 49, 34–43. [Google Scholar] [CrossRef]
- Müller, C.; Jenni-Eiermann, S.; Jenni, L. Heterophils/Lymphocytes-ratio and circulating corticosterone do not indicate the same stress imposed on Eurasian kestrel nestlings. Funct. Ecol. 2011, 25, 566–576. [Google Scholar] [CrossRef]
- Obernier, J.A.; Baldwin, R.L. Establishing an appropriate period of acclimatization following transportation of laboratory animals. ILAR J. 2006, 47, 364–369. [Google Scholar] [CrossRef] [PubMed]
- Noda, K.; Akiyoshi, H.; Aoki, M.; Shimada, T.; Ohashi, F. Relationship between transportation stress and polymorphonuclear cell functions of bottlenose dolphins, Tursiops truncatus. J. Vet. Med. Sci. 2007, 69, 379–383. [Google Scholar] [CrossRef]
- Doherty, T.J.; Kattesh, H.G.; Adcock, R.J.; Welborn, M.G.; Saxton, A.M.; Morrow, J.L.; Dailey, J.W. Effects of a concentrated lidocaine solution on the acute phase stress response to dehorning in dairy calves. J. Dairy Sci. 2007, 90, 4232–4239. [Google Scholar] [CrossRef] [PubMed]
- McGlone, J.J.; Salak, J.L.; Lumpkin, E.A.; Nicholson, R.I.; Gibson, M.; Norman, R.L. Shipping stress and social status effects on pig performance, plasma cortisol, natural killer cell activity, and leukocyte numbers. J. Anim. Sci. 1992, 71, 888–896. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.-Y.; Han, J.S.; Suzuki, T.; Han, S.-S. Indirect indicator of transport stress in hematological values in newly acquired cynomolgus monkeys. J. Med. Primatol. 2005, 34, 188–192. [Google Scholar] [CrossRef]
- Gladbach, A.; Gladbach, D.J.; Koch, M.; Kuchar, A.; Möstl, E.; Quillfeldt, P. Can faecal glucocorticoid metabolites be used to monitor body condition in wild Upland geese Chloephaga picta leucoptera? Behav. Ecol. Sociobiol. 2011, 65, 1491–1498. [Google Scholar] [CrossRef]
- Jackson, J.; Childs, D.Z.; Mar, K.U.; Htut, W.; Lummaa, V. Long-term trends in wild-capture and population dynamics point to an uncertain future for captive elephants. Proc. R. Soc. B 2019, 286, 20182810. [Google Scholar] [CrossRef] [PubMed]
- Mar, K.U. The Demography and Life History Startegies of Timber Elephants in Myanmar. Ph.D. Thesis, University College London, London, UK, 2007. [Google Scholar]
- Lahdenperä, M.; Mar, K.U.; Courtiol, A.; Lummaa, V. Differences in age-specific mortality between wild-caught and captive-born Asian elephants. Nat. Commun. 2018, 9, 3023. [Google Scholar] [CrossRef] [PubMed]
- Reichert, S.; Berger, V.; dos Santos, D.; Lahdenperä, M.; Htut, W.; Lummaa, V. Age-related variation of molecular and physiological health markers in a long-lived mammal. Unpublished.
- Clubb, R.; Rowcliffe, M.; Lee, P.; Mar, K.U.; Moss, C.; Mason, G.J. Compromised survivorship in zoo elephants. Science 2008, 322, 1649. [Google Scholar] [CrossRef] [PubMed]
- Lahdenperä, M.; Mar, K.U.; Lummaa, V. Reproductive cessation and post-reproductive lifespan in Asian elephants and pre-industrial humans. Front. Zool. 2014, 11, 54. [Google Scholar] [CrossRef] [PubMed]
- Lahdenperä, M.; Mar, K.U.; Lummaa, V. Nearby grandmother enhances calf survival and reproduction in Asian elephants. Sci. Rep. 2016, 6, 27213. [Google Scholar] [CrossRef]
- Watson, R.; Munro, C.; Edwards, K.L.; Norton, V.; Brown, J.L.; Walker, S.L. Development of a versatile enzyme immunoassay for non-invasive assessment of glucocorticoid metabolites in a diversity of taxonomic species. Gen. Comp. Endocrinol. 2013, 186, 16–24. [Google Scholar] [CrossRef]
- Reichert, S.; Berger, V.; Jackson, J.; Chapman, S.N.; Htut, W.; Mar, K.U.; Lummaa, V. Maternal age at birth shapes offspring life-history trajectory across generations in long-lived Asian elephants. J. Anim. Ecol. 2019, 1–12. [Google Scholar] [CrossRef]
- Mumby, H.S.; Chapman, S.N.; Crawley, J.A.H.; Mar, K.U.; Htut, W.; Thura Soe, A.; Aung, H.H.; Lummaa, V. Distinguishing between determinate and indeterminate growth in a long-lived mammal. BMC Evol. Biol. 2015, 15, 214. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018; Available online: https://www.R-project.org/ (accessed on 31 March 2018).
- Vleck, C.M. Comparison of corticosterone and heterophil to lymphocyte ratios as indicators of stress in free-living birds. In Avian Endocrinology; Dawson, A., Chaturvedi, C.M., Eds.; Narosa Publishing House: New Delhi, India, 2001; pp. 401–411. [Google Scholar]
- Barnett, J.L.; Cronin, G.M.; Winfield, C.G.; Dewar, A.M. The welfare of adult pigs: The effects of five housing treatments on behaviour, plasma corticosteroids and injuries. Appl. Anim. Behav. Sci. 1984, 12, 209–232. [Google Scholar] [CrossRef]
- Palme, R. Monitoring stress hormone metabolites as a useful, non-invasive tool for welfare assessment in farm animals. Anim. Welf. 2012, 21, 331–337. [Google Scholar] [CrossRef]
- Romero, L.M.; Wingfield, J.C. Tempests, Poxes, Predators, and People: Stress in Wild Animals and How They Cope; Oxford University Press: Oxford, UK, 2016. [Google Scholar]
- Horne, B.D.; Anderson, J.L.; John, J.M.; Weaver, A.; Bair, T.L.; Jensen, K.R.; Renlund, D.G. Which white blood cell subtypes predict increased cardiovascular risk? J. Am. Coll. Cardiol. 2005, 45, 1638–1643. [Google Scholar] [CrossRef] [PubMed]
- Al-Murrani, W.K.; Al-Rawi, I.K.; Raof, N.M. Genetic resistance to Salmonella typhimurium in two lines of chickens selected as resistant and sensitive on the basis of heterophil/lymphocyte ratio. Br. Poult. Sci. 2002, 43, 501–507. [Google Scholar] [CrossRef]
- Romero, L.M.; Wikelski, M. Corticosterone levels predict survival probabilities of Galapagos marine iguanas during El Nino events. Proc. Natl. Acad. Sci. USA 2001, 98, 7366–7370. [Google Scholar] [CrossRef] [PubMed]
- Lobato, E.; Moreno, J.; Merino, S.; Sanz, J.J.; Arriero, E. Haematological variables are good predictors of recruitment in nestling pied flycatchers (Ficedula hypoleuca). Ecoscience 2005, 12, 27–34. [Google Scholar] [CrossRef]
- Alm, M.; Tauson, R.; Holm, L.; Wichman, A.; Kalliokoski, O.; Wall, H. Welfare indicators in laying hens in relation to nest exclusion. Poult. Sci. 2016, 95, 1238–1247. [Google Scholar] [CrossRef] [PubMed]
- Bilandzic, N.; Zuric, M.; Lojkic, M.; Simic, D.; Milic, D.; Barac, I. Cortisol and immune measures in boars exposed to three-day administration of exogenous adrenocorticotropic hormone. Veterninary Res. Commun. 2006, 30, 433–444. [Google Scholar] [CrossRef] [PubMed]
- Dickens, M.J.; Romero, L.M. General and Comparative Endocrinology A consensus endocrine profile for chronically stressed wild animals does not exist. Gen. Comp. Endocrinol. 2013, 191, 177–189. [Google Scholar] [CrossRef]
- Seltmann, M.W.; Helle, S.; Htut, W.; Lahdenperä, M. Males have more aggressive and less sociable personalities than females in semi-captive Asian elephants. Sci. Rep. 2019, 9, 2668. [Google Scholar] [CrossRef]
- Lynsdale, C.L.; Mumby, H.S.; Hayward, A.D.; Mar, K.U.; Lummaa, V. Parasite-associated mortality in a long-lived mammal: Variation with host age, sex, and reproduction. Ecol. Evol. 2017, 7, 10904–10915. [Google Scholar] [CrossRef]
- Kumar, V.; Pradheeps, M.; Kokkiligadda, A.; Niyogi, R.; Umapathy, G. Non-invasive assessment of physiological stress in captive Asian elephants. Animals 2019, 9, 553. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, H.B.; Ganswindt, A.; Douglas-Hamilton, I.; Vollrath, F. Endocrine and behavioral changes in male African elephants: Linking hormone changes to sexual state and reproductive tactics. Horm. Behav. 2008, 54, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Goymann, W. On the use of non-invasive hormone research in uncontrolled, natural environments: The problem with sex, diet, metabolic rate and the individual. Methods Ecol. Evol. 2012, 3, 757–765. [Google Scholar] [CrossRef]
- Jaatinen, K.; Seltmann, M.W.; Hollmén, T.; Atkinson, S.; Mashburn, K.; Öst, M. Context dependency of baseline glucocorticoids as indicators of individual quality in a capital breeder. Gen. Comp. Endocrinol. 2013, 191, 231–238. [Google Scholar] [CrossRef]
- Gesquiere, L.R.; Altmann, J.; Khan, M.Z.; Couret, J.; Yu, J.C.; Endres, C.S.; Lynch, J.W.; Ogola, P.; Fox, E.A.; Alberts, S.C.; et al. Coming of age: Steroid hormones of wild immature baboons (Papio cynocephalus). Am. J. Primatol. 2005, 67, 83–100. [Google Scholar] [CrossRef]
- Bansiddhi, P.; Brown, J.L.; Khonmee, J.; Norkaew, T.; Nganvongpanit, K.; Punyapornwithaya, V.; Angkawanish, T.; Somgird, C.; Thitaram, C. Management factors affecting adrenal glucocorticoid activity of tourist camp elephants in Thailand and implications for elephant welfare. PLoS ONE 2019, 14, e0221537. [Google Scholar] [CrossRef]
- Pokharel, S.S.; Seshagiri, P.B.; Sukumar, R. Assessment of season-dependent body condition scores in relation to faecal glucocorticoid metabolites in free-ranging Asian elephants. Conserv. Physiol. 2017, 5, 1–14. [Google Scholar] [CrossRef]
- Viljoen, J.J.; Ganswindt, A.; Palme, R.; Reynecke, H.C.; du Toit, J.T.; Langbauer, W.R.J. Measurement of concentrations of faecal glucocorticoid metabolites in free-ranging African elephants within the Kruger National Park. Koedoe 2008, 50, 18–21. [Google Scholar] [CrossRef]
- Huber, S.; Palme, R.; Arnold, G.W. Effects of season, sex and sample collection on concentrations of faecal cortisol metabolites in red deer (Cervus elaphus). Gen. Comp. Endocrinol. 2003, 130, 48–54. [Google Scholar] [CrossRef]
- Michael Romero, L. Seasonal changes in plasma glucocorticoid concentrations in free-living vertebrates. Gen. Comp. Endocrinol. 2002, 128, 1–24. [Google Scholar] [CrossRef]
- Moore, I.T.; Jessop, T.S. Stress, reproduction, and adrenocortical modulation in amphibians and reptiles. Horm. Behav. 2003, 43, 39–47. [Google Scholar] [CrossRef]
- Husak, J.F.; Moore, I.T. Stress hormones and mate choice. Trends Ecol. Evol. 2008, 23, 532–534. [Google Scholar] [CrossRef] [PubMed]
- Plotnik, J.M.; de Waal, F.B.M.; Moore, D.; Reiss, D. Self-Recognition in the Asian elephant and future directions for cognitive research with elephants in zoological settings. Zoo Biol. 2010, 29, 179–191. [Google Scholar] [CrossRef]
- Dale, R.; Plotnik, J.M. Elephants know when their bodies are obstacles to success in a novel transfer task. Sci. Rep. 2017, 7, 46309. [Google Scholar] [CrossRef] [PubMed]
- Nicol, C.J.; Caplen, G.; Edgar, J.; Richards, G.; Browne, W.J. Relationships between multiple welfare indicators measured in individual chickens across different time periods and environments. Anim. Welf. 2011, 20, 133–143. [Google Scholar]
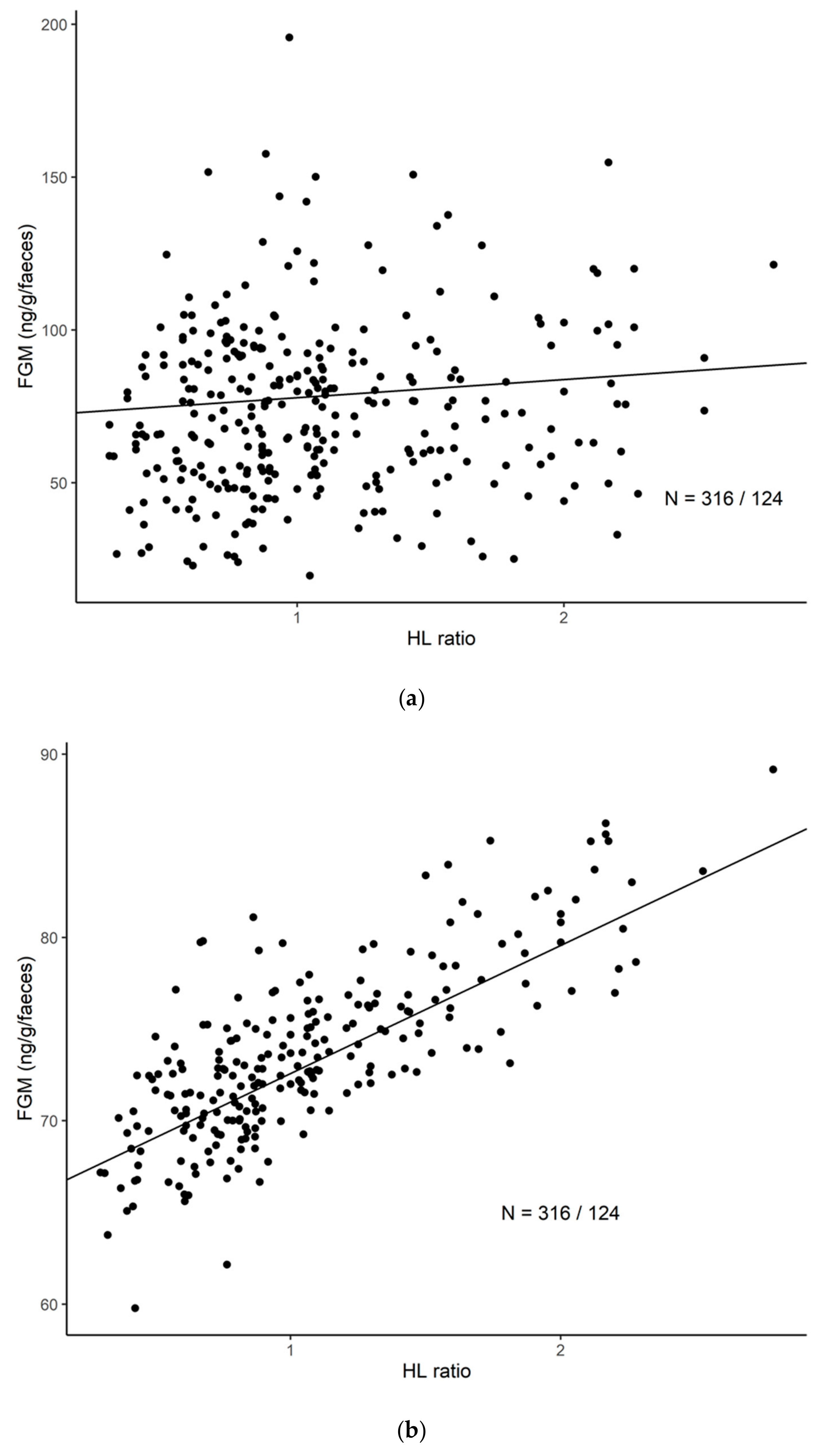
| FGM Concentration (ng/g)/H/L Ratio | ||||
|---|---|---|---|---|
| Minimum | Average | Maximum | ||
| Sex | male | 24.0/0.363 | 78.9/1.05 | 195/2.79 |
| female | 19.6/0.296 | 71.5/1.08 | 162/2.53 | |
| Age | taming | 24.30/0.363 | 80.0/1.05 | 195/2.79 |
| training | 22.9/0.296 | 73.9/1.09 | 162/2.28 | |
| working | 25.8/0.323 | 65.6/1.01 | 142/2.23 | |
| retired | 19.6/0.371 | 63.3/1.23 | 143/2.53 | |
| Season | hot | 19.6/0.296 | 66.8/0.976 | 195/2.53 |
| monsoon | 44.9/0.488 | 91.6/0.975 | 158/2.17 | |
| cold | 24.3/0.425 | 81.5/1.31 | 150/2.79 | |
| Parameter | Variable | F | DF | p-Value |
|---|---|---|---|---|
| Model 1 | ||||
| FCM | H/L | 6.06 | 1, 186 | 0.0147 |
| sex | 8.38 | 1, 121 | <0.01 | |
| origin | 13.5 | 1, 121 | <0.001 | |
| season | 15.0 | 2, 186 | <0.0001 | |
| age | 0.163 | 3, 186 | 0.921 | |
| Model 2 | ||||
| FCM | H/L | 6.34 | 1, 185 | <0.01 |
| sex | 7.92 | 1, 121 | <0.01 | |
| origin | 13.3 | 1, 121 | <0.001 | |
| season | 15.0 | 2, 185 | <0.0001 | |
| age | 0.171 | 3, 185 | 0.916 | |
| H/L*sex | 0.570 | 1, 185 | 0.451 | |
| Model 3 | ||||
| FCM | H/L | 6.11 | 1, 183 | <0.01 |
| age | 4.28 | 3, 183 | <0.01 | |
| sex | 3.64 | 1, 121 | 0.0586 | |
| origin | 7.19 | 1, 121 | <0.01 | |
| season | 14.4 | 2, 183 | <0.0001 | |
| H/L*age | 0.817 | 3, 183 | 0.486 | |
| Model 4 | ||||
| FCM | H/L | 2.32 | 1, 184 | 0.130 |
| season | 19.9 | 2, 184 | <0.0001 | |
| origin | 8.28 | 1, 121 | <0.01 | |
| sex | 7.27 | 1, 121 | <0.01 | |
| age | 0.162 | 3, 184 | 0.922 | |
| H/L*season | 0.246 | 2, 184 | 0.782 | |
| Model 5 | ||||
| Standardised body weight | FCM | 7.14 | 1, 80 | 0.00920 |
| season | 2.02 | 2, 80 | 0.139 | |
| sex | 0.0927 | 1, 110 | 0.761 | |
| age | 0.876 | 3, 80 | 0.457 | |
| origin | 0.000800 | 1, 110 | 0.977 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seltmann, M.W.; Ukonaho, S.; Reichert, S.; Dos Santos, D.; Nyein, U.K.; Htut, W.; Lummaa, V. Faecal Glucocorticoid Metabolites and H/L Ratio Are Related Markers of Stress in Semi-Captive Asian Timber Elephants. Animals 2020, 10, 94. https://doi.org/10.3390/ani10010094
Seltmann MW, Ukonaho S, Reichert S, Dos Santos D, Nyein UK, Htut W, Lummaa V. Faecal Glucocorticoid Metabolites and H/L Ratio Are Related Markers of Stress in Semi-Captive Asian Timber Elephants. Animals. 2020; 10(1):94. https://doi.org/10.3390/ani10010094
Chicago/Turabian StyleSeltmann, Martin W., Susanna Ukonaho, Sophie Reichert, Diogo Dos Santos, U Kyaw Nyein, Win Htut, and Virpi Lummaa. 2020. "Faecal Glucocorticoid Metabolites and H/L Ratio Are Related Markers of Stress in Semi-Captive Asian Timber Elephants" Animals 10, no. 1: 94. https://doi.org/10.3390/ani10010094
APA StyleSeltmann, M. W., Ukonaho, S., Reichert, S., Dos Santos, D., Nyein, U. K., Htut, W., & Lummaa, V. (2020). Faecal Glucocorticoid Metabolites and H/L Ratio Are Related Markers of Stress in Semi-Captive Asian Timber Elephants. Animals, 10(1), 94. https://doi.org/10.3390/ani10010094





