The Simultaneous Administration of a Probiotic or Prebiotic with Live Salmonella Vaccine Improves Growth Performance and Reduces Fecal Shedding of the Bacterium in Salmonella-Challenged Broilers
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Salmonella Vaccine: Avipro® Salmonella Vac E
2.2. Probiotic and Prebiotic
2.3. The Challenge Bacterium
2.4. Experimental Design
2.5. Evaluated Parameters
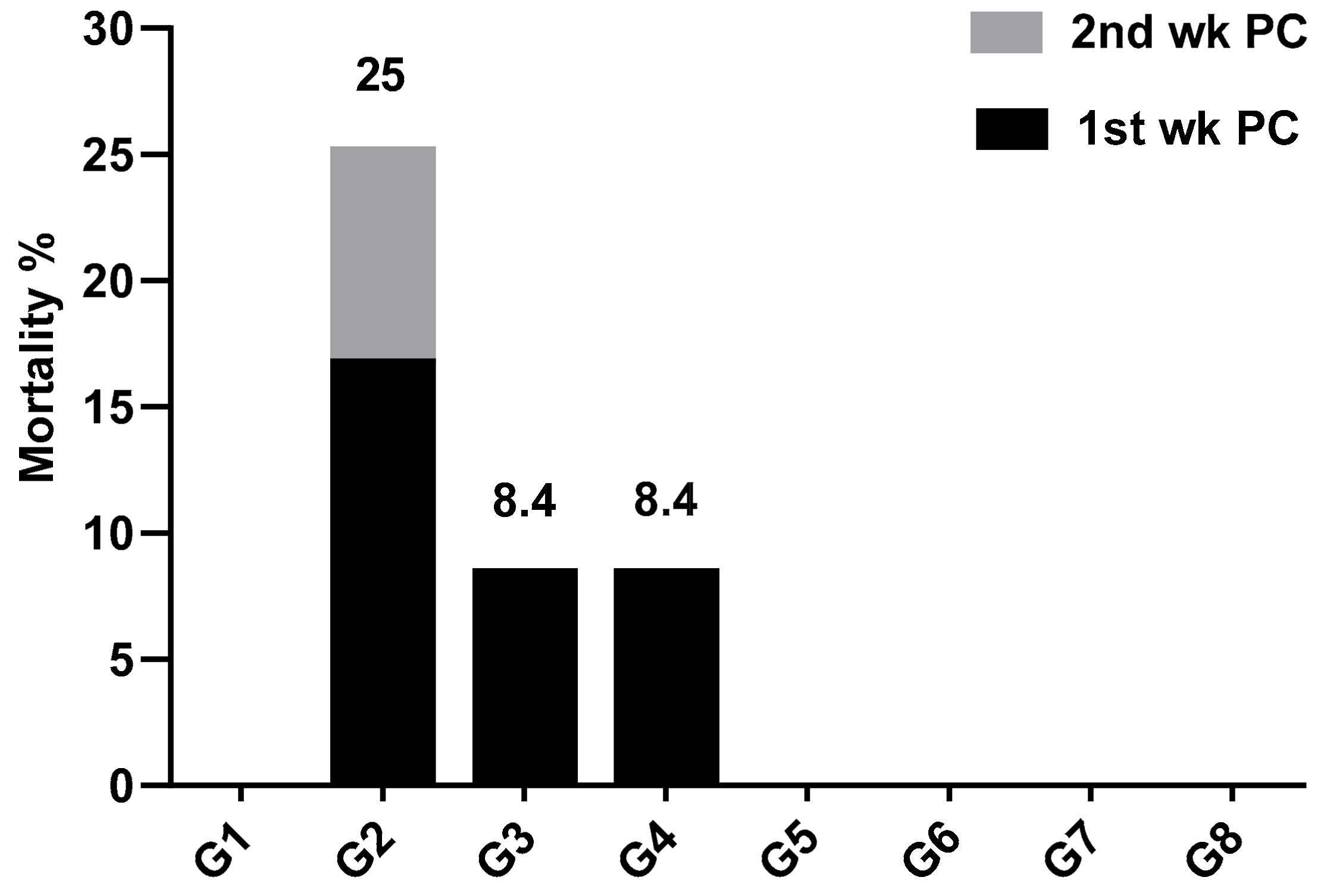
2.5.1. Clinical Investigation and Mortality Rate
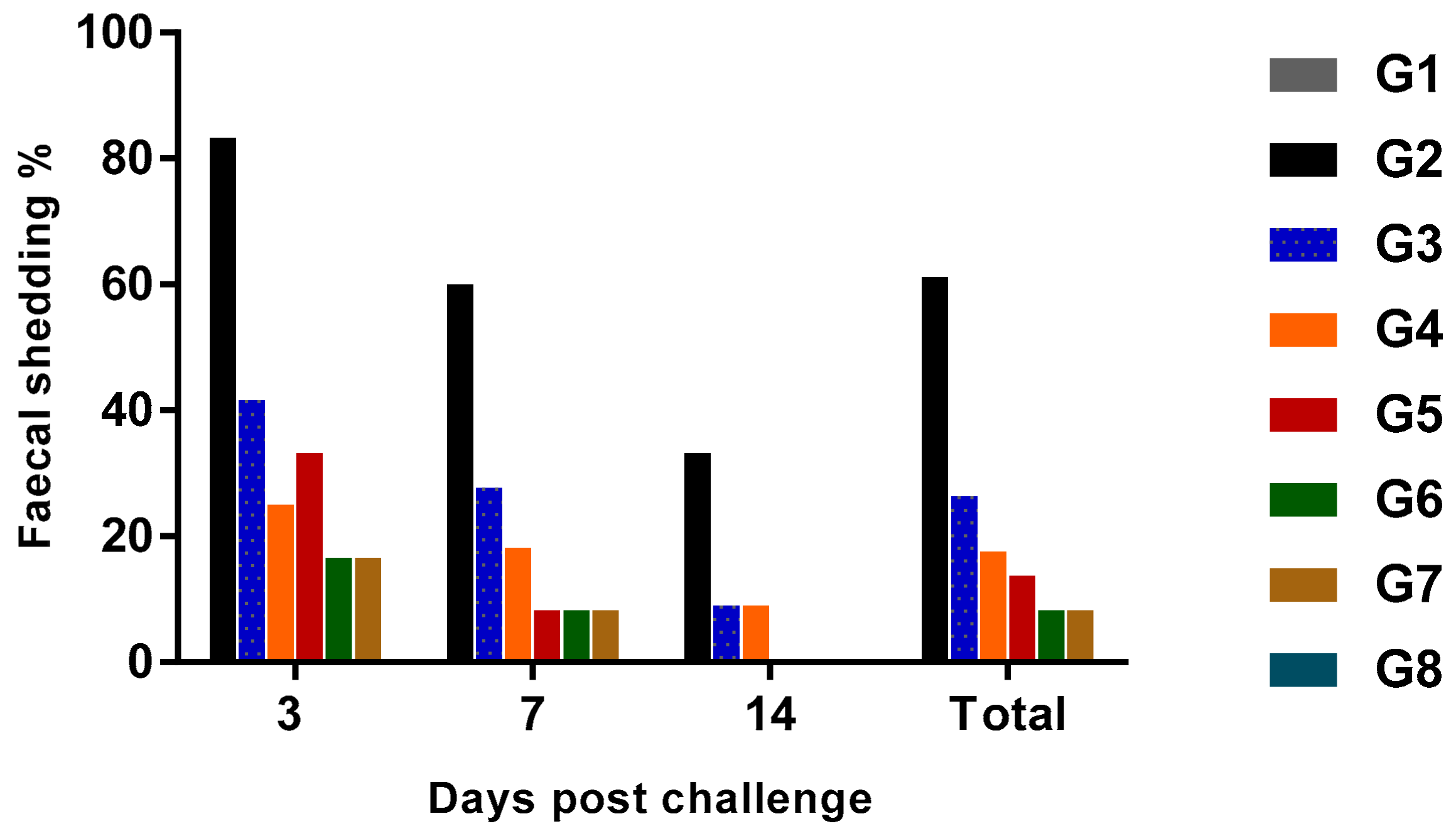
2.5.2. The Shedding Rate of SE on Cloacal Swabs
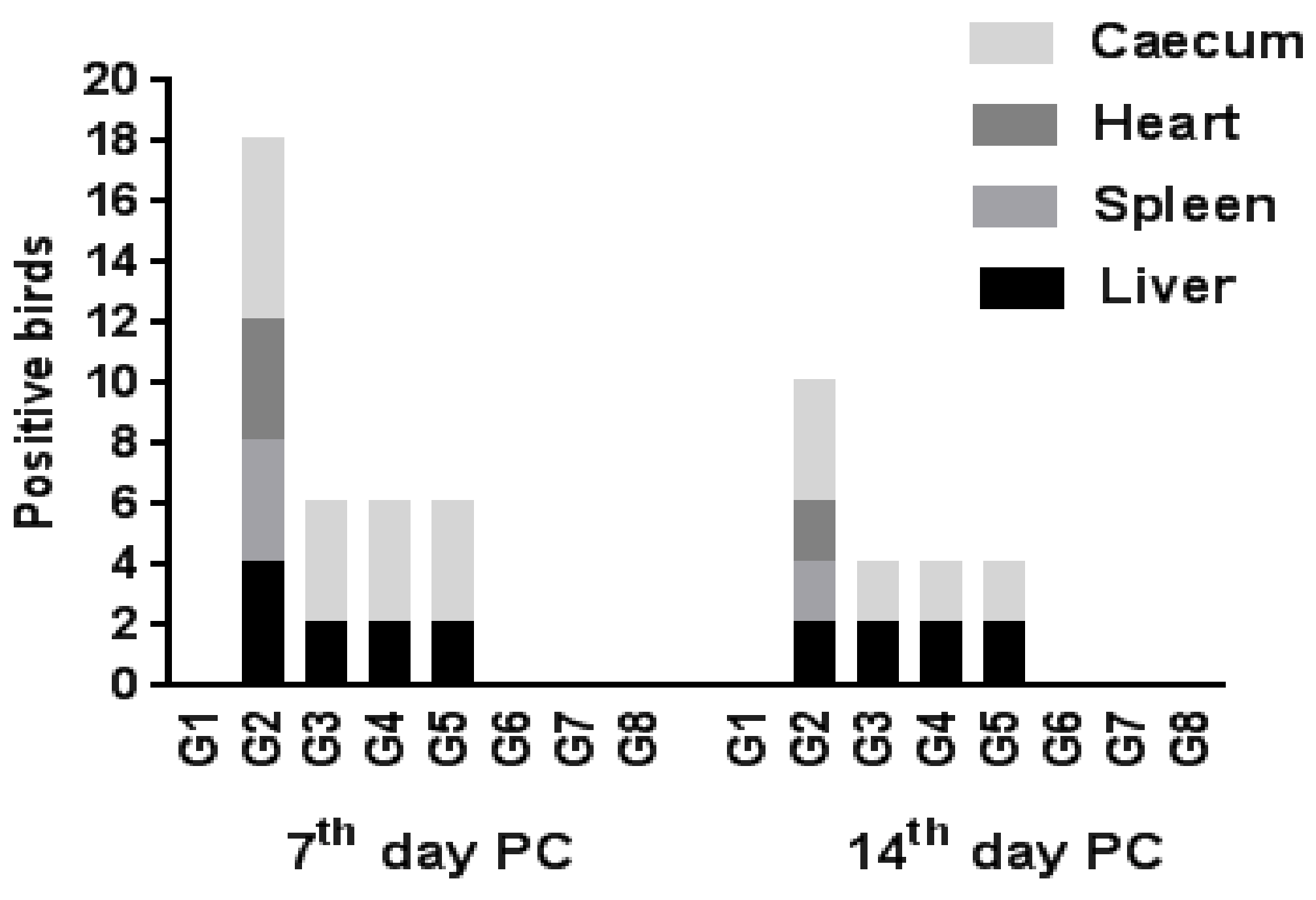
2.5.3. Re-Isolation Rate of SE from Different Organs
2.5.4. The Growth Performance
2.6. Statistical Analysis
3. Results and Discussion
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Góchez, D.; Raicek, M.; Ferreira, J.P.; Jeannin, M.; Moulin, G.; Erlacher-Vindel, E. OIE Annual Report on Antimicrobial Agents Intended for Use in Animals: Methods Used. Front. Vet. Sci. 2019, 6, 317. [Google Scholar] [CrossRef] [PubMed]
- Mezal, E.H.; Sabol, A.; Khan, M.A.; Ali, N.; Stefanova, R.; Khan, A.A. Isolation and molecular characterization of Salmonella enterica serovar Enteritidis from poultry house and clinical samples during 2010. Food Microbiol. 2014, 38, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Cha, S.Y.; Kang, M.; Yoon, R.H.; Park, C.K.; Moon, O.K.; Jang, H.K. Prevalence and antimicrobial susceptibility of Salmonella isolates in Pekin ducks from South Korea. Comp. Immunol. Microbiol. Infect. Dis. 2013, 36, 473–479. [Google Scholar]
- Chen, Z.; Jiang, X. Microbiological Safety of Chicken Litter or Chicken Litter-Based Organic Fertilizers: A Review. Agriculture 2014, 4, 1–29. [Google Scholar] [CrossRef]
- De Cort, W.; Ducatelle, R.; van Immerseel, F. Preharvest measures to improve the safety of eggs. In Producing Safe Eggs; Ricke, S.C., Gast, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 249–280. [Google Scholar]
- Cerquetti, M.C.; Gherardi, M.M. Vaccination of chickens with a temperature-sensitive mutant of Salmonella enteritidis. Vaccine 2000, 18, 1140–1145. [Google Scholar] [CrossRef]
- Inoue, A.Y.; Berchieri, A.; Bernardino, A.; Paiva, J.B.; Sterzo, E. Passive immunity of progeny from broiler breeders vaccinated with oil-emulsion bacterin against Salmonella enteritidis. Avian Dis. 2008, 52, 567–571. [Google Scholar] [CrossRef]
- Darrell, T.; Thomas, H.; Richard, G. Integrated farm management to prevent Salmonella Enteritidis contamination of eggs. J. Appl. Poult. Res. 2014, 23, 353–365. [Google Scholar]
- Penha Filho, R.A.C.; Diaz, S.J.A.; da Silva Medina, T.; Chang, Y.F.; da Silva, J.S.; Berchieri, A., Jr. Evaluation of protective immune response against fowl typhoid in chickensvaccinated with the attenuatedstrain Salmonella Gallinarum ΔcobSΔcbiA. Res. Vet. Sci. 2016, 107, 220–227. [Google Scholar] [CrossRef]
- Berghaus, R.D.; Thayer, S.G.; Maurer, J.J.; Hofacre, C.L. Effect of Vaccinating Breeder Chickens with a Killed Salmonella Vaccine on Salmonella Prevalences and Loads in Breeder and Broiler Chicken Flocks. J. Food Protec. 2011, 74, 700–864. [Google Scholar] [CrossRef]
- Revolledo, L.; Ferreira, C.S.; Ferreira, A.J. Prevention of Salmonella typhimurium colonization and organ invasion by combination treatment in broiler chicks. Braz. J. Poult. Sci. 2009, 88, 734–743. [Google Scholar] [CrossRef]
- Crawford, J.S. Probiotics in Animal Nutrition. In Proceedings of the Arkansas Nutrition Conference, Arkansas, AR, USA, 27–28 September 1979; pp. 45–55. [Google Scholar]
- Jie-Lun, H.; Hai, Y.; Raveendra, K.; Steve, C.; Xie, M.-Y.; Nie, S.-P.; Joshua, G. Modulation of cytokine gene expression by selected Lactobacillus isolates in the ileum, caecal tonsils and spleen of Salmonella-challenged broilers. Avian Path. 2015, 44, 463–469. [Google Scholar]
- Charalampopoulos, D.; Rastall, R. Prebiotics and Probiotics. In Science and Technology; Springer: New York, NY, USA, 2009; Volume 1. [Google Scholar]
- Pineiro, M.; Asp, N.G.; Reid, G.; Macfarlane, S.; Morelli, L.; Brunser, O.; Tuohy, K. FAO Technical Meeting on Prebiotics. J. Clin. Gastroenterol. 2008, 42, S156–S159. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Rubio, C.; Ordonez, C.; Abad-González, J.; Garcia-Gallego, A.; Honrubia, M.P.; Mallo, J.J.; Balana-Fouce, R. Butyric acid-based feed additives help protect broiler chickens from Salmonella Enteritidis infection. Poult. Sci. 2009, 88, 943–948. [Google Scholar] [CrossRef] [PubMed]
- Dhama, K.; Mahendran, M.; Tomar, S.; Chauhan, R.S. Beneficial effects of probiotics and prebiotics in livestock and poultry: The current perspectives. Intas Polivet 2008, 9, 1–12. [Google Scholar]
- Dhama, K.; Verma, V.; Sawant, P.M.; Tiwari, R.; Vaid, R.K.; Chauhan, R.S. Applications of Probiotics in Poultry: Enhancing Immunity and Beneficial Effects on Production Performances and Health—A Review. J. Immunol. Immunopathol. 2011, 13, 1–19. [Google Scholar]
- Alagawany, M.; El-Hack, M.E.A.; Farag, M.R.; Sachan, S.; Karthik, K.; Dhama, K. The use of probiotics as eco-friendly alternatives for antibiotics in poultry nutrition. Environ. Sci. Pollut. Res. Int. 2018, 25, 10611–10618. [Google Scholar] [CrossRef]
- Bérto, L.D.; Beirão, B.C.B.; Filho, T.F.; Ingberman, M.; Fávaro, C., Jr.; Tavella, R.; de Mesquita Silva, R.B.; Caron, L.F. Live and Inactivated Salmonella Enteritidis Vaccines: Immune Mechanisms in Broiler Breeders. World J. Vaccin. 2015, 5, 155–164. [Google Scholar] [CrossRef][Green Version]
- Sedeik, M.E.; Nahed, A.; Awad, A.M.; Elfeky, S.M.; El-Hack, M.E.A.; Hussein, E.O.; Alowaimer, A.N.; Swelum, A.A. Isolation, conventional and molecular characterization of Salmonella spp. from newly hatched broiler chicks. AMB Exp. 2019, 9, 136. [Google Scholar] [CrossRef]
- Timms, L.M.; Marshall, R.N.; Breslin, M.F. Laboratory assessment of protection given by an experimental S. Enteritidis PT4 inactivated adjuvant vaccine. Vet. Rec. 1990, 127, 611–614. [Google Scholar]
- Okamoto, A.S.; Andreatti Filho, R.L.; Lima, E.T.; Pereira, R.E.P.; Menconi, A.; Rocha, T.S.; Marietto-Gonçalves, G.A. Immunological evaluation of the intestinal mucosa of broiler chicks treated with lactobacillus spp. and challenged with Salmonella Enteritidis. Braz. J. Poult. Sci. 2007, 9, 259–262. [Google Scholar] [CrossRef]
- National Research Council. Nutrient Requirements of Poultry, 9th ed.; The National Academies Press: Washington, DC, USA, 1994. [Google Scholar]
- O’Brien, J.D.P. Salmonella Enteritidis infection in broiler chickens. Vet. Rec. 1988, 8, 214–218. [Google Scholar] [CrossRef]
- Cherry, W.; Davis, B.R.; Edwards, P.R.; Hogan, R. A simple procedure for the identification of the genus Salmonella by means of a specific bacteriophage. J. Lab. Clin. Med. 1954, 44, 51–55. [Google Scholar] [PubMed]
- Petrie, A.; Watson, P. Statistics for Veterinary and Animal Science, 1st ed.; Blackwell Publishing: Malden, MA, USA, 1999. [Google Scholar]
- Abd El-Ghany, A.W.; El-Shafii, S.A.S.; Hatem, M.E.; Dawood, E. A Trial to Prevent Salmonella Enteritidis Infection in Broiler Chickens Using Autogenous Bacterin Compared with Probiotic Preparation. J. Agric. Sci. 2012, 4, 91–108. [Google Scholar] [CrossRef]
- Hassan, J.O.; Curtiss, R. Development and evaluation of oralvaccination program using live avirulent Salmonella typhimurium strain to protect immunized chickens against challenge with homologous and heterologous Salmonella serotypes. Infect. Immun. 1994, 62, 5519–5527. [Google Scholar] [PubMed]
- Vandeplas, S.; Dauphin, R.D.; Beckers, Y.; Thonart, P.; Thewis, A. Salmonella in chicken: Current and developing strategies to reduce contamination at farm level. J. Food Prot. 2010, 73, 774–785. [Google Scholar] [CrossRef] [PubMed]
- Foley, S.L.; Lynne, A.M.; Nayak, R. Salmonella challenges: Prevalence in swine and poultry and potential pathogenicity of such isolates. J. Anim. Sci. 2008, 86, E149–E162. [Google Scholar] [CrossRef] [PubMed]
- Stern, N.J. Salmonella species and Campylobacter jejuni cecal colonization model in broilers. Poult. Sci. 2008, 87, 2399–2403. [Google Scholar] [CrossRef] [PubMed]
- Springer, S.; Lindner, T.; Ahrens, M. A new live Salmonella Enteritidis vaccine for chicken: Experimental evidence of its safety and efficacy. Berl. Munch. Tierarztl. Wochenschr. 2000, 113, 246–252. [Google Scholar] [PubMed]
- Sharma, P.; Pande, V.V.; Moyle, T.S.; Mcwhorter, A.R.; Chousalkar, K.K. Correlating bacterial shedding with fecal corticosterone levels and serological responses from layer hens experimentally infected with Salmonella Typhimurium. Vet. Res. 2017, 48, 5. [Google Scholar] [CrossRef]
- McWhorter, A.R.; Chousalkar, K.K. A Long-Term Efficacy Trial of a Live, Attenuated Salmonella Typhimurium Vaccine in Layer Hens. Front. Microbiol. 2018, 9, 1380. [Google Scholar] [CrossRef]
- Pan, D.; Yu, Z. Intestinal microbiome of poultry and its interaction with host and diet. Gut Microb. 2014, 5, 108–119. [Google Scholar] [CrossRef] [PubMed]
- Azcarate-Peril, M.; Ritte, A.; Savaiano, D.; Monteagudo-Mera, A.; Anderson, C.; Magness, S.; Klaenhammer, T. Impact of short-chain galactooligosaccharides on the gut microbiome of lactose-intolerant individuals. Proc. Natl. Acad. Sci. USA 2017, 114, E367–E375. [Google Scholar] [CrossRef] [PubMed]
- Capozzo, A.V.E.; Cuberos, L.; Levine, M.M.; Pasetti, M.F. Mucosally delivered Salmonella live vector vaccines antigen in neonatal mice born to naive and immunemothers. Infect. Immun. 2004, 72, 4637–4646. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Methner, U.; Barrow, P.A.; Berndt, A.; Rychlik, I. Salmonella Enteritidis with double deletion in phoP fliC—A potential live Salmonella deletion in phoP fliC—A potential live Salmonella vaccine candidate with novel characteristics for use in chickens. Vaccine 2011, 29, 3248–3253. [Google Scholar] [CrossRef] [PubMed]
- Tellez, G.; Petrone, V.M.; Escorcia, M.; Mosrishita, T.Y.; Cobb, C.W.; Villasenor, L. Evaluation of Avian Specific Probiotic and Salmonella Enteritidis-, Salmonella Typhimurium-, and Salmonella Heidelberg Specific Antibodies on Cecal Colonization and Organ Invasion of Salmonella Enteritidis in Broilers. J. Food Prot. 2001, 64, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Zdunczyk, Z.; Juskiewicz, J.; Jankowski, J.; Biedrzycka, E. Metabolic response of the gastrointestinal tract of turkeys to diets with different levels of mannan-oligosaccharide. Poult. Sci. 2005, 84, 903–909. [Google Scholar] [CrossRef] [PubMed]
- Attia, Y.A.; Ellakany, H.F.; El-Hamid, A.A.; Bovera, F.; Ghazaly, S.A. Control of Salmonella enteritidis infection in male layer chickens by acetic acid and/or prebiotics, probiotics and antibiotics. Arch. Geflügelk. 2012, 76, 239–245. [Google Scholar]
- Fairchild, A.S.; Grimes, J.L.; Jones, F.T.; Wineland, M.J.; Edens, F.W.; Sefton, A.E. Effects of hen age, Bio-Mos and Flavomycin on poult susceptibility to oral Escherichia coli challenge. Poult. Sci. 2001, 80, 562–571. [Google Scholar] [CrossRef]
- Ellakany, H.F.; Rezk, M.S.H.; Awad, A.M.; El-hamid, H.S.A. The role of prebiotic and acidifier in the protection against experimental infection of Salmonella enteritidis in broiler chickens. In Proceedings of the 1st International Conference of The Veterinary Research Division, Cairo, Egypt, 15–17 February 2004; Egyptian National Research Center: Cairo, Egypt, 2004; pp. 181–194. [Google Scholar]
- Khan, R.U.; Naz, S.; Dhama, K.; Kathrik, K.; Tiwari, R.; Abdelrahman, M.M.; Alhdary, I.A.; Zahoor, A. Direct-fed microbial: Beneficial applications, modes of action and prospects as a safe tool for enhancing ruminant production and safeguarding health. Int. J. Pharmacol. 2016, 12, 220–231. [Google Scholar]
- FAO. Health and Nutritional Properties and guidelines for evaluation. Available online: http://www.fao.org/3/a-a0512e.pdf (accessed on 26 December 2019).
- Coates, M.E.; Fuller, R. The Genotobiotic Animal in the Study of Gut Microbiology. In Microbial Ecology of the Gut; Academic Press: London, UK, 1977; Volume 153, pp. 311–346. [Google Scholar]
- Bailey, J. Factors affecting microbial competitive exclusion in poultry overview outstanding symposia in food science and technology. Food Technol. 1987, 5, 88–92. [Google Scholar]
- Saarela, M.; Mogensen, G.; Fondens, R.; Matto, J.; Mattila-Sandholm, T. Probiotic bacteria: Safety, functional and technological properties. J. Biotechnol. 2000, 84, 197–215. [Google Scholar] [CrossRef]
- Nahashon, S.N.; Nakaue, H.S.; Mirosh, L.W. Effect of direct-fed microbials on nutrient retention and production parameters of laying pullets. Poult. Sci. 1992, 71 (Suppl. 1), 111. [Google Scholar]
- Beirão, B.C.B.; Ingberman, M.; Fávaro, C., Jr.; Mesa, D.; Letícia, C. Effect of an Enterococcus faecium probiotic on specific IgA following live Salmonella Enteritidis vaccination of layer chickens. Avian Pathol. 2018, 47, 325–333. [Google Scholar] [CrossRef] [PubMed]
| Measurements | Treatments | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| G1 | G2 | G3 | G4 | G5 | G6 | G7 | G8 | ||
| 7th day PC | |||||||||
| Liver | +/total | 0/6 | 4/6 | 2/6 | 2/6 | 2/6 | 0/6 | 0/6 | 0/6 |
| % | 0 | 66.67 | 33.33 | 33.33 | 33.33 | 0 | 0 | 0 | |
| Spleen | +/total | 0/6 | 4/6 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 |
| % | 0 | 66.67 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Heart | +/total | 0/6 | 4/6 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 |
| % | 0 | 66.67 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Cecum | +/total | 0/6 | 6/6 | 4/6 | 4/6 | 4/6 | 0/6 | 0/6 | 0/6 |
| % | 0 | 100 | 66.67 | 66.67 | 66.67 | 0 | 0 | 0 | |
| 14th day PC | |||||||||
| Liver | +/total | 0/6 | 2/6 | 2/6 | 2/6 | 2/6 | 0/6 | 0/6 | 0/6 |
| % | 0 | 33.33 | 33.33 | 33.33 | 33.33 | 0 | 0 | 0 | |
| Spleen | +/total | 0/6 | 2/6 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 |
| % | 0 | 33.33 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Heart | +/total | 0/6 | 2/6 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 | 0/6 |
| % | 0 | 33.33 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Cecum | +/total | 0/6 | 4/6 | 2/6 | 2/6 | 2/6 | 0/6 | 0/6 | 0/6 |
| % | 0 | 66.67 | 33.33 | 33.33 | 33.33 | 0 | 0 | 0 | |
| Treatments | Measurements | |||||
|---|---|---|---|---|---|---|
| Body weight Gain (g)/bird | FCR (g/g) | |||||
| Weeks of Age | ||||||
| 0–4 (BC) * | 4–6 (PC) ** | 0–6 (Overall) | 0–4 (BC) * | 4–6 (PC) ** | 0–6 (Overall) | |
| G1 | 1385 ± 31.3 bc | 1526 ± 38.6 a | 2911 ± 57. 3 a | 1.38 ± 0.03 bc | 1.25 ± 0.03 bc | 1.30 ± 0.03 cd |
| G2 | 1411 ± 33.6 abc | 1083 ± 34.1 c | 2494 ± 65.4 c | 1.33 ± 0.03 bc | 1.60 ± 0.05 a | 1.46 ± 0.04 ab |
| G3 | 1444 ± 23.4 abc | 1408 ± 30.8 ab | 2852 ± 52.5 a | 1.25 ± 0.02 c | 1.21 ± 0.03 c | 1.23 ± 0.02 d |
| G4 | 1474 ± 21.6 ab | 1477 ± 29.0 a | 2951 ± 31.9 a | 1.28 ± 0.02 c | 1.22 ± 0.03 c | 1.25 ± 0.01 cd |
| G5 | 1268 ± 37.0 d | 1327 ± 49.7 ab | 2595 ± 54.0 bc | 1.53 ± 0.05 a | 1.43 ± 0.05 abc | 1.48 ± 0.03 a |
| G6 | 1360 ± 34.3 c | 1445 ± 62.1 a | 2805 ± 35.8 ab | 1.42 ± 0.04 b | 1.25 ± 0.06 bc | 1.32 ± 0.02 cd |
| G7 | 1507 ± 23.8 a | 1227 ± 35.3 bc | 2734 ± 46.5 ab | 1.34 ± 0.02 bc | 1.37 ± 0.04 bc | 1.36 ± 0.02 bc |
| G8 | 1276 ± 22.3 d | 1366 ± 110 ab | 2642 ± 126 ab | 1.50 ± 0.02 a | 1.46 ± 0.13 ab | 1.48 ± 0.06 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Shall, N.A.; Awad, A.M.; El-Hack, M.E.A.; Naiel, M.A.E.; Othman, S.I.; Allam, A.A.; Sedeik, M.E. The Simultaneous Administration of a Probiotic or Prebiotic with Live Salmonella Vaccine Improves Growth Performance and Reduces Fecal Shedding of the Bacterium in Salmonella-Challenged Broilers. Animals 2020, 10, 70. https://doi.org/10.3390/ani10010070
El-Shall NA, Awad AM, El-Hack MEA, Naiel MAE, Othman SI, Allam AA, Sedeik ME. The Simultaneous Administration of a Probiotic or Prebiotic with Live Salmonella Vaccine Improves Growth Performance and Reduces Fecal Shedding of the Bacterium in Salmonella-Challenged Broilers. Animals. 2020; 10(1):70. https://doi.org/10.3390/ani10010070
Chicago/Turabian StyleEl-Shall, Nahed A., Ashraf M. Awad, Mohamed E. Abd El-Hack, Mohammed A. E. Naiel, Sarah I. Othman, Ahmed A. Allam, and Mahmoud E. Sedeik. 2020. "The Simultaneous Administration of a Probiotic or Prebiotic with Live Salmonella Vaccine Improves Growth Performance and Reduces Fecal Shedding of the Bacterium in Salmonella-Challenged Broilers" Animals 10, no. 1: 70. https://doi.org/10.3390/ani10010070
APA StyleEl-Shall, N. A., Awad, A. M., El-Hack, M. E. A., Naiel, M. A. E., Othman, S. I., Allam, A. A., & Sedeik, M. E. (2020). The Simultaneous Administration of a Probiotic or Prebiotic with Live Salmonella Vaccine Improves Growth Performance and Reduces Fecal Shedding of the Bacterium in Salmonella-Challenged Broilers. Animals, 10(1), 70. https://doi.org/10.3390/ani10010070







