Vaginal Microbiota of the Sexually Transmitted Infections Caused by Chlamydia trachomatis and Trichomonas vaginalis in Women with Vaginitis in Taiwan
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sample Collection and Ethical Approval
2.2. Culture-Dependent Identification of Microorganisms by MALDI-TOF Analysis
2.3. BD Max CT/GC/TV Assay and DNA Extraction
2.4. Analysis of Microbiota Composition
2.4.1. PCR Amplification and Sequencing
2.4.2. Bioinformatics Analysis
2.5. In Vitro Interactions of Specific Bacteria with TV
2.6. Statistical Analyses
3. Results
3.1. The Prevalence of STIs and Their Differential Diagnosis in Women with Vaginitis
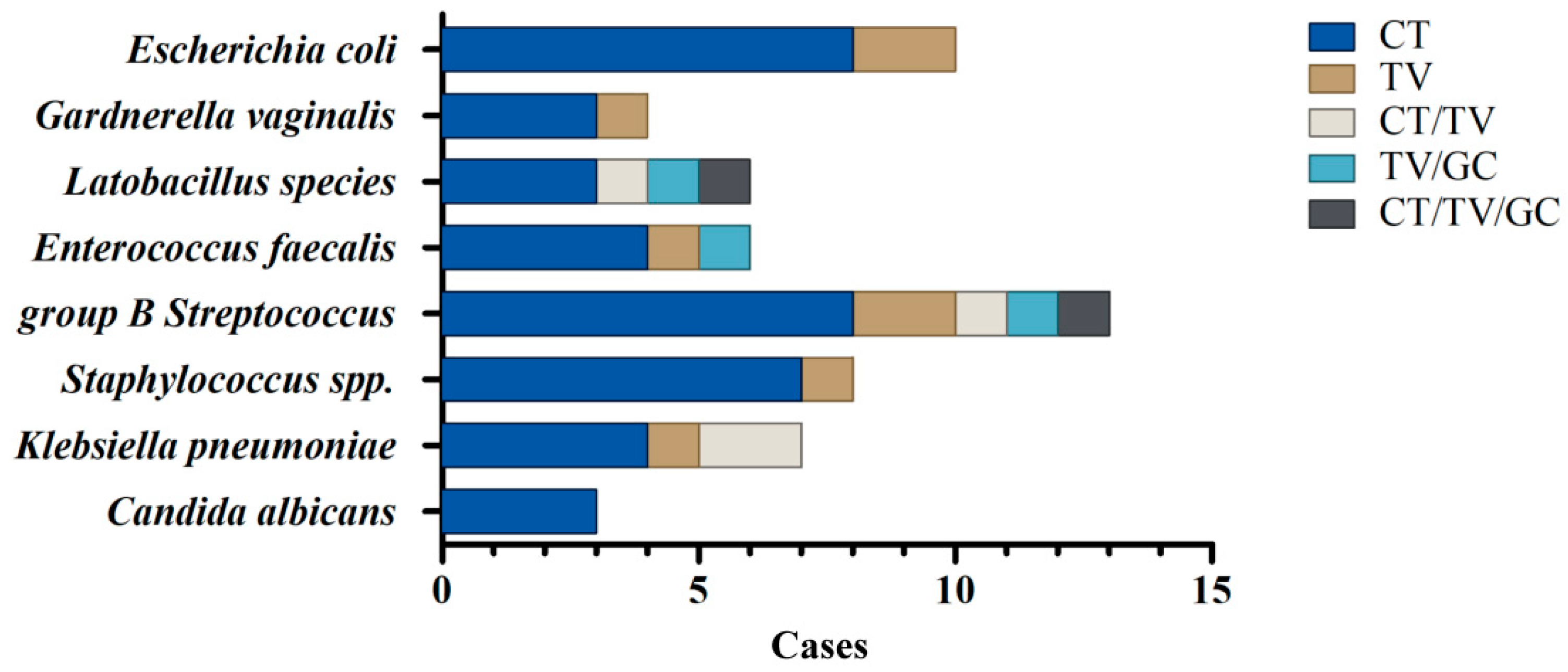
3.2. Identification of Bacterial Co-Infections in STI-Positive Patients by a Culture-Dependent Method Combined with MALDI-TOF Analysis
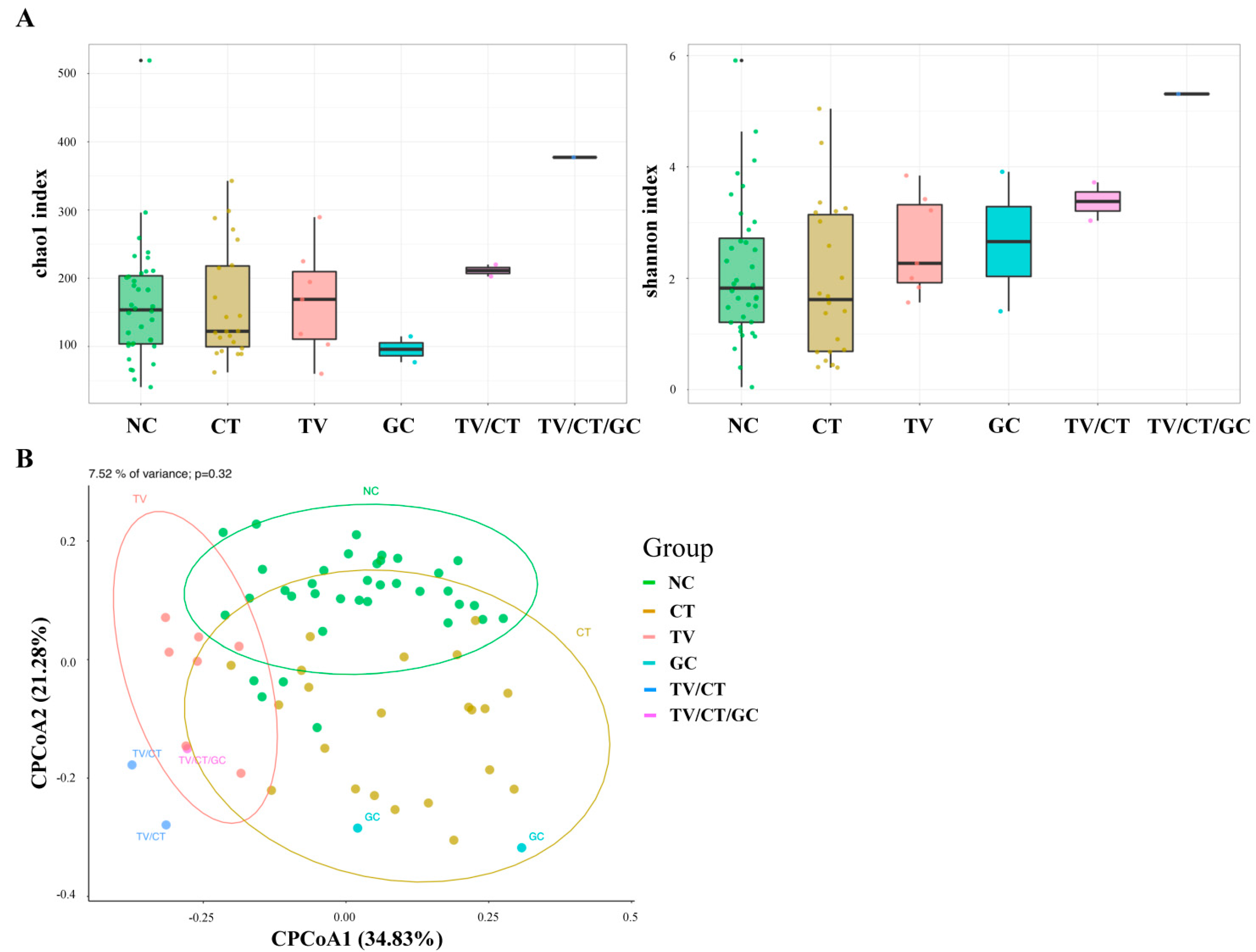
3.3. Vaginal Microbiota Profiling in STI-Positive Patients Using 16S rRNA Sequencing
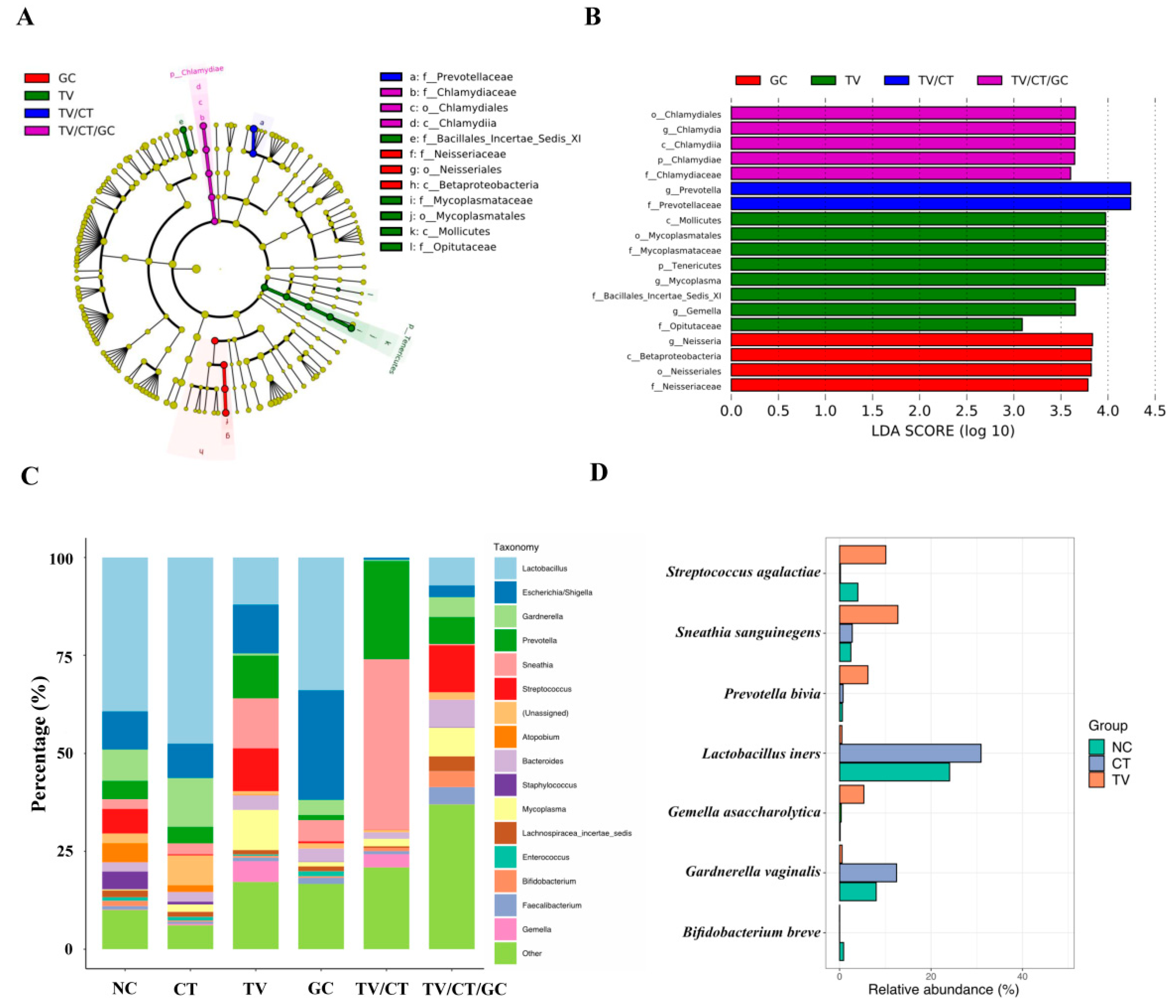
3.4. Vaginal Microbiota Revealed Specific Bacterial Taxa Associated with STI-Positive Patients
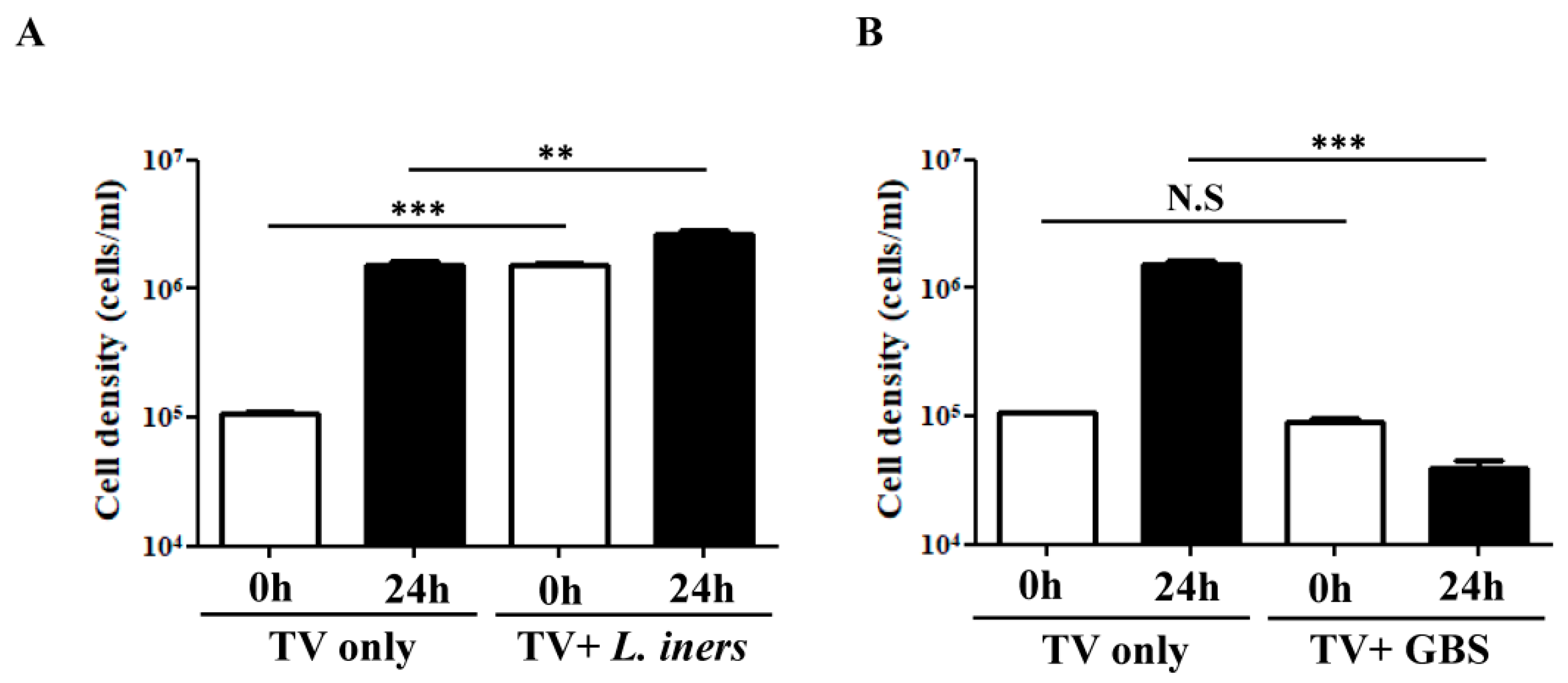
3.5. In Vitro TV-Bacteria Co-Culture Revealed Different Influence on the Growth of L. iners and GBS
3.6. Impact of the Crosstalk between TV and the Associated Bacteria on the Growth of Parasites
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Carr, P.L.; Felsenstein, D.; Friedman, R.H. Evaluation and management of vaginitis. J. Gen. Intern. Med. 1998, 13, 335–346. [Google Scholar] [CrossRef] [Green Version]
- Paladine, H.L.; Desai, U.A. Vaginitis: Diagnosis and Treatment. Am. Fam. Physician 2018, 97, 321–329. [Google Scholar]
- Roselletti, E.; Sabbatini, S.; Perito, S.; Mencacci, A.; Vecchiarelli, A.; Monari, C. Apoptosis of vaginal epithelial cells in clinical samples from women with diagnosed bacterial vaginosis. Sci. Rep. 2020, 10, 1978. [Google Scholar] [CrossRef] [PubMed]
- Gaydos, C.A.; Beqaj, S.; Schwebke, J.R.; Lebed, J.; Smith, B.; Davis, T.E.; Fife, K.H.; Nyirjesy, P.; Spurrell, T.; Furgerson, D.; et al. Clinical Validation of a Test for the Diagnosis of Vaginitis. Obstet. Gynecol. 2017, 130, 181–189. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jeanmonod, R.; Jeanmonod, D. Vaginal Candidiasis (Vulvovaginal Candidiasis). In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Sobel, J.D.; Subramanian, C.; Foxman, B.; Fairfax, M.; Gygax, S.E. Mixed vaginitis-more than coinfection and with therapeutic implications. Curr. Infect. Dis. Rep. 2013, 15, 104–108. [Google Scholar] [CrossRef]
- Rowley, J.; Vander Hoorn, S.; Korenromp, E.; Low, N.; Unemo, M.; Abu-Raddad, L.J.; Chico, R.M.; Smolak, A.; Newman, L.; Gottlieb, S.; et al. Chlamydia, gonorrhoea, trichomoniasis and syphilis: Global prevalence and incidence estimates, 2016. Bull. World Health Organ. 2019, 97, 548P–562P. [Google Scholar] [CrossRef]
- Wynn, A.; Bristow, C.C.; Cristillo, A.D.; Murphy, S.M.; van den Broek, N.; Muzny, C.; Kallapur, S.; Cohen, C.; Ingalls, R.R.; Wiesenfeld, H.; et al. Sexually Transmitted Infections in Pregnancy and Reproductive Health: Proceedings of the STAR Sexually Transmitted Infection Clinical Trial Group Programmatic Meeting. Sex. Transm. Dis. 2020, 47, 5–11. [Google Scholar] [CrossRef]
- Kirkcaldy, R.D.; Augostini, P.; Asbel, L.E.; Bernstein, K.T.; Kerani, R.P.; Mettenbrink, C.J.; Pathela, P.; Schwebke, J.R.; Secor, W.E.; Workowski, K.A.; et al. Trichomonas vaginalis antimicrobial drug resistance in 6 US cities, STD Surveillance Network, 2009–2010. Emerg. Infect. Dis. 2012, 18, 939–943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Unemo, M.; Del Rio, C.; Shafer, W.M. Antimicrobial Resistance Expressed by Neisseria gonorrhoeae: A Major Global Public Health Problem in the 21st Century. Microbiol. Spectr. 2016, 4. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.A.; Papp, J.R.; Stamm, W.E.; Peeling, R.W.; Martin, D.H.; Holmes, K.K. Evaluation of antimicrobial resistance and treatment failures for Chlamydia trachomatis: A meeting report. J. Infect. Dis. 2005, 191, 917–923. [Google Scholar] [CrossRef] [Green Version]
- Walker, J.; Tabrizi, S.N.; Fairley, C.K.; Chen, M.Y.; Bradshaw, C.S.; Twin, J.; Taylor, N.; Donovan, B.; Kaldor, J.M.; McNamee, K.; et al. Chlamydia trachomatis incidence and re-infection among young women--behavioural and microbiological characteristics. PLoS ONE 2012, 7, e37778. [Google Scholar] [CrossRef]
- Aghaizu, A.; Reid, F.; Kerry, S.; Hay, P.E.; Mallinson, H.; Jensen, J.S.; Kerry, S.; Kerry, S.; Oakeshott, P. Frequency and risk factors for incident and redetected Chlamydia trachomatis infection in sexually active, young, multi-ethnic women: A community based cohort study. Sex. Transm. Infect. 2014, 90, 524–528. [Google Scholar] [CrossRef] [Green Version]
- Witkin, S.S.; Linhares, I.M. Why do lactobacilli dominate the human vaginal microbiota? BJOG 2017, 124, 606–611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romero, R.; Hassan, S.S.; Gajer, P.; Tarca, A.L.; Fadrosh, D.W.; Nikita, L.; Galuppi, M.; Lamont, R.F.; Chaemsaithong, P.; Miranda, J.; et al. The composition and stability of the vaginal microbiota of normal pregnant women is different from that of non-pregnant women. Microbiome 2014, 2, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ravel, J.; Gajer, P.; Abdo, Z.; Schneider, G.M.; Koenig, S.S.; McCulle, S.L.; Karlebach, S.; Gorle, R.; Russell, J.; Tacket, C.O.; et al. Vaginal microbiome of reproductive-age women. Proc. Natl. Acad. Sci. USA 2011, 108, 4680–4687. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- van der Veer, C.; Bruisten, S.M.; van der Helm, J.J.; de Vries, H.J.; van Houdt, R. The Cervicovaginal Microbiota in Women Notified for Chlamydia trachomatis Infection: A Case-Control Study at the Sexually Transmitted Infection Outpatient Clinic in Amsterdam, The Netherlands. Clin. Infect. Dis. 2017, 64, 24–31. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brotman, R.M.; Bradford, L.L.; Conrad, M.; Gajer, P.; Ault, K.; Peralta, L.; Forney, L.J.; Carlton, J.M.; Abdo, Z.; Ravel, J. Association between Trichomonas vaginalis and vaginal bacterial community composition among reproductive-age women. Sex. Transm. Dis. 2012, 39, 807–812. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jarrett, O.D.; Srinivasan, S.; Richardson, B.A.; Fiedler, T.; Wallis, J.M.; Kinuthia, J.; Jaoko, W.; Mandaliya, K.; Fredricks, D.N.; McClelland, R.S. Specific Vaginal Bacteria Are Associated with an Increased Risk of Trichomonas vaginalis Acquisition in Women. J. Infect. Dis. 2019, 220, 1503–1510. [Google Scholar] [CrossRef]
- Ketterer, M.R.; Rice, P.A.; Gulati, S.; Kiel, S.; Byerly, L.; Fortenberry, J.D.; Soper, D.E.; Apicella, M.A. Desialylation of Neisseria gonorrhoeae Lipooligosaccharide by Cervicovaginal Microbiome Sialidases: The Potential for Enhancing Infectivity in Men. J. Infect. Dis. 2016, 214, 1621–1628. [Google Scholar] [CrossRef]
- Van Der Pol, B.; Williams, J.A.; Fuller, D.; Taylor, S.N.; Hook, E.W., 3rd. Combined Testing for Chlamydia, Gonorrhea, and Trichomonas by Use of the BD Max CT/GC/TV Assay with Genitourinary Specimen Types. J. Clin. Microbiol. 2017, 55, 155–164. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.Y.; Yeh, Y.M.; Yu, H.Y.; Chin, C.Y.; Hsu, C.W.; Liu, H.; Huang, P.J.; Hu, S.N.; Liao, C.T.; Chang, K.P.; et al. Oral Microbiota Community Dynamics Associated with Oral Squamous Cell Carcinoma Staging. Front. Microbiol. 2018, 9, 862. [Google Scholar] [CrossRef] [Green Version]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glockner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. SINTAX: A simple non-Bayesian taxonomy classifier for 16S and ITS sequences. biorxiv 2016, 074161. [Google Scholar] [CrossRef] [Green Version]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Diamond, L.S.; Clark, C.G.; Cunnick, C.C. YI-S, a casein-free medium for axenic cultivation of Entamoeba histolytica, related Entamoeba, Giardia intestinalis and Trichomonas vaginalis. J. Eukaryot. Microbiol. 1995, 42, 277–278. [Google Scholar] [CrossRef]
- Pinheiro, J.; Biboy, J.; Vollmer, W.; Hirt, R.P.; Keown, J.R.; Artuyants, A.; Black, M.M.; Goldstone, D.C.; Simoes-Barbosa, A. The Protozoan Trichomonas vaginalis Targets Bacteria with Laterally Acquired NlpC/P60 Peptidoglycan Hydrolases. mBio 2018, 9, e01784-18. [Google Scholar] [CrossRef] [Green Version]
- Al-Awadhi, H.; Dashti, N.; Khanafer, M.; Al-Mailem, D.; Ali, N.; Radwan, S. Bias problems in culture-independent analysis of environmental bacterial communities: A representative study on hydrocarbonoclastic bacteria. SpringerPlus 2013, 2, 369. [Google Scholar] [CrossRef] [Green Version]
- Filardo, S.; Di Pietro, M.; Tranquilli, G.; Sessa, R. Biofilm in Genital Ecosystem: A Potential Risk Factor for Chlamydia trachomatis Infection. Can. J. Infect. Dis. Med. Microbiol. 2019, 2019, 1672109. [Google Scholar] [CrossRef] [Green Version]
- Giordani, B.; Melgoza, L.M.; Parolin, C.; Foschi, C.; Marangoni, A.; Abruzzo, A.; Dalena, F.; Cerchiara, T.; Bigucci, F.; Luppi, B.; et al. Vaginal Bifidobacterium breve for preventing urogenital infections: Development of delayed release mucoadhesive oral tablets. Int. J. Pharm. 2018, 550, 455–462. [Google Scholar] [CrossRef]
- Chen, K.T.; Chen, S.C.; Chiang, C.C.; Li, L.H.; Tang, L.H. Chlamydial infection among patients attending STD and genitourinary clinics in Taiwan. BMC Public Health 2007, 7, 120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Der Pol, B.; Daniel, G.; Kodsi, S.; Paradis, S.; Cooper, C.K. Molecular-based Testing for Sexually Transmitted Infections Using Samples Previously Collected for Vaginitis Diagnosis. Clin. Infect. Dis. 2019, 68, 375–381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bautista, C.T.; Wurapa, E.K.; Sateren, W.B.; Morris, S.M.; Hollingsworth, B.P.; Sanchez, J.L. Association of Bacterial Vaginosis with Chlamydia and Gonorrhea among Women in the U.S. Army. Am. J. Prev. Med. 2017, 52, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Edwards, V.L.; Smith, S.B.; McComb, E.J.; Tamarelle, J.; Ma, B.; Humphrys, M.S.; Gajer, P.; Gwilliam, K.; Schaefer, A.M.; Lai, S.K.; et al. The Cervicovaginal Microbiota-Host Interaction Modulates Chlamydia trachomatis Infection. mBio 2019, 10, e01548-19. [Google Scholar] [CrossRef] [Green Version]
- Gajer, P.; Brotman, R.M.; Bai, G.; Sakamoto, J.; Schutte, U.M.; Zhong, X.; Koenig, S.S.; Fu, L.; Ma, Z.S.; Zhou, X.; et al. Temporal dynamics of the human vaginal microbiota. Sci. Transl. Med. 2012, 4, 132ra152. [Google Scholar] [CrossRef] [Green Version]
- Torok, M.R.; Miller, W.C.; Hobbs, M.M.; Macdonald, P.D.; Leone, P.A.; Schwebke, J.R.; Sena, A.C. The association between Trichomonas vaginalis infection and level of vaginal lactobacilli, in nonpregnant women. J. Infect. Dis. 2007, 196, 1102–1107. [Google Scholar] [CrossRef] [Green Version]
- McClelland, R.S.; Lingappa, J.R.; Srinivasan, S.; Kinuthia, J.; John-Stewart, G.C.; Jaoko, W.; Richardson, B.A.; Yuhas, K.; Fiedler, T.L.; Mandaliya, K.N.; et al. Evaluation of the association between the concentrations of key vaginal bacteria and the increased risk of HIV acquisition in African women from five cohorts: A nested case-control study. Lancet Infect. Dis. 2018, 18, 554–564. [Google Scholar] [CrossRef]
- Hoogkamp-Korstanje, J.A.; Gerards, L.J.; Cats, B.P. Maternal carriage and neonatal acquisition of group B streptococci. J. Infect. Dis. 1982, 145, 800–803. [Google Scholar] [CrossRef] [PubMed]
- Vornhagen, J.; Adams Waldorf, K.M.; Rajagopal, L. Perinatal Group B Streptococcal Infections: Virulence Factors, Immunity, and Prevention Strategies. Trends Microbiol. 2017, 25, 919–931. [Google Scholar] [CrossRef]
- Zarate, G.; Nader-Macias, M.E. Influence of probiotic vaginal lactobacilli on in vitro adhesion of urogenital pathogens to vaginal epithelial cells. Lett. Appl. Microbiol. 2006, 43, 174–180. [Google Scholar] [CrossRef]
- De Gregorio, P.R.; Juarez Tomas, M.S.; Leccese Terraf, M.C.; Nader-Macias, M.E. Preventive effect of Lactobacillus reuteri CRL1324 on Group B Streptococcus vaginal colonization in an experimental mouse model. J. Appl. Microbiol. 2015, 118, 1034–1047. [Google Scholar] [CrossRef]
- Rosen, G.H.; Randis, T.M.; Desai, P.V.; Sapra, K.J.; Ma, B.; Gajer, P.; Humphrys, M.S.; Ravel, J.; Gelber, S.E.; Ratner, A.J. Group B Streptococcus and the Vaginal Microbiota. J. Infect. Dis. 2017, 216, 744–751. [Google Scholar] [CrossRef] [PubMed]
- Gosmann, C.; Anahtar, M.N.; Handley, S.A.; Farcasanu, M.; Abu-Ali, G.; Bowman, B.A.; Padavattan, N.; Desai, C.; Droit, L.; Moodley, A.; et al. Lactobacillus-Deficient Cervicovaginal Bacterial Communities Are Associated with Increased HIV Acquisition in Young South African Women. Immunity 2017, 46, 29–37. [Google Scholar] [CrossRef] [Green Version]
- Rendon-Maldonado, J.G.; Espinosa-Cantellano, M.; Gonzalez-Robles, A.; Martinez-Palomo, A. Trichomonas vaginalis: In vitro phagocytosis of lactobacilli, vaginal epithelial cells, leukocytes, and erythrocytes. Exp. Parasitol. 1998, 89, 241–250. [Google Scholar] [CrossRef] [PubMed]
- Petrin, D.; Delgaty, K.; Bhatt, R.; Garber, G. Clinical and microbiological aspects of Trichomonas vaginalis. Clin. Microbiol. Rev. 1998, 11, 300–317. [Google Scholar] [CrossRef] [Green Version]
- Nizet, V.; Kim, K.S.; Stins, M.; Jonas, M.; Chi, E.Y.; Nguyen, D.; Rubens, C.E. Invasion of brain microvascular endothelial cells by group B streptococci. Infect. Immun. 1997, 65, 5074–5081. [Google Scholar] [CrossRef] [Green Version]
- Doran, K.S.; Chang, J.C.; Benoit, V.M.; Eckmann, L.; Nizet, V. Group B streptococcal beta-hemolysin/cytolysin promotes invasion of human lung epithelial cells and the release of interleukin-8. J. Infect. Dis. 2002, 185, 196–203. [Google Scholar] [CrossRef]
- Tapsall, J.W.; Phillips, E.A. The hemolytic and cytolytic activity of group B streptococcal hemolysin and its possible role in early onset group B streptococcal disease. Pathology 1991, 23, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Fettucciari, K.; Rosati, E.; Scaringi, L.; Cornacchione, P.; Migliorati, G.; Sabatini, R.; Fetriconi, I.; Rossi, R.; Marconi, P. Group B Streptococcus induces apoptosis in macrophages. J. Immunol. 2000, 165, 3923–3933. [Google Scholar] [CrossRef] [Green Version]
- Oliveira, L.; Madureira, P.; Andrade, E.B.; Bouaboud, A.; Morello, E.; Ferreira, P.; Poyart, C.; Trieu-Cuot, P.; Dramsi, S. Group B streptococcus GAPDH is released upon cell lysis, associates with bacterial surface, and induces apoptosis in murine macrophages. PLoS ONE 2012, 7, e29963. [Google Scholar] [CrossRef] [PubMed]
- Freitas, A.C.; Chaban, B.; Bocking, A.; Rocco, M.; Yang, S.; Hill, J.E.; Money, D.M.; Group, V.R. The vaginal microbiome of pregnant women is less rich and diverse, with lower prevalence of Mollicutes, compared to non-pregnant women. Sci. Rep. 2017, 7, 9212. [Google Scholar] [CrossRef] [PubMed]
- Si, J.; You, H.J.; Yu, J.; Sung, J.; Ko, G. Prevotella as a Hub for Vaginal Microbiota under the Influence of Host Genetics and Their Association with Obesity. Cell Host Microbe 2017, 21, 97–105. [Google Scholar] [CrossRef] [Green Version]
- Bassis, C.M.; Allsworth, J.E.; Wahl, H.N.; Sack, D.E.; Young, V.B.; Bell, J.D. Effects of intrauterine contraception on the vaginal microbiota. Contraception 2017, 96, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Achilles, S.L.; Austin, M.N.; Meyn, L.A.; Mhlanga, F.; Chirenje, Z.M.; Hillier, S.L. Impact of contraceptive initiation on vaginal microbiota. Am. J. Obstet. Gynecol. 2018, 218, 622.e1–622.e10. [Google Scholar] [CrossRef] [Green Version]
- Fosch, S.E.; Ficoseco, C.A.; Marchesi, A.; Cocucci, S.; Nader-Macias, M.E.F.; Perazzi, B.E. Contraception: Influence on Vaginal Microbiota and Identification of Vaginal Lactobacilli Using MALDI-TOF MS and 16S rDNA Sequencing. Open Microbiol. J. 2018, 12, 218–229. [Google Scholar] [CrossRef] [PubMed]

| Total Sample | Total STIs | CT | TV | GC | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| STI Results | n | % | n | % | n | % | n | % | n | % |
| 327 | 48 | 14.6 | 35 | 10.8 | 7 | 2.2 | 2 | 0.6 | ||
| Demographic characteristics | ||||||||||
| Age, Median | 39 | 29.1 | 31.6 | 37.1 | 25.2 | |||||
| 18 yrs and below | 20 | 6.1 | 6 | 12.5 | 5 | 14.2 | 0 | 0 | 1 | 50 |
| 19 to 29 yrs | 73 | 22.3 | 20 | 41.6 | 14 | 40 | 2 | 28.5 | 0 | 0 |
| 30 to 39 | 85 | 25.9 | 11 | 22.9 | 9 | 25.7 | 1 | 14.2 | 1 | 50 |
| 40 yrs. and above | 149 | 45.7 | 11 | 22.9 | 7 | 20 | 4 | 57.1 | 0 | 0 |
| Clinic type | ||||||||||
| ER | 62 | 18.7 | 8 | 16.6 | 7 | 20 | 1 | 14.2 | 0 | 0 |
| STD clinic | 98 | 29.8 | 15 | 31.2 | 11 | 31.4 | 1 | 14.2 | 1 | 50 |
| OB/GYN | 167 | 51.5 | 25 | 52 | 17 | 48.5 | 5 | 71.4 | 1 | 50 |
| Differential diagnosis | ||||||||||
| Vulval pruritus | 18 | 5.5 | 4 | 8.3 | 4 | 11.4 | 0 | 0 | 0 | 0 |
| Acute vaginitis | 9 | 2.8 | 2 | 4.1 | 2 | 5.7 | 0 | 0 | 0 | 0 |
| Subacute vaginitis | 65 | 19.9 | 8 | 16.6 | 5 | 14.2 | 2 | 28.5 | 1 | 50 |
| Cervicitis | 53 | 16.2 | 8 | 16.6 | 5 | 14.2 | 2 | 28.5 | 0 | 0 |
| PID | 40 | 12.2 | 8 | 16.6 | 7 | 20 | 0 | 9 | 0 | 0 |
| Vulvar vestibulitis | 32 | 9.8 | 4 | 8.3 | 2 | 5.7 | 1 | 14.2 | 0 | 0 |
| Gonorrhea | 2 | 0.6 | 1 | 2 | 1 | 2.8 | 0 | 0 | 0 | 0 |
| VVC | 12 | 3.7 | 2 | 4.1 | 1 | 2.8 | 1 | 14.2 | 0 | 0 |
| Leiomyoma | 6 | 1.8 | 2 | 4.1 | 2 | 5.7 | 0 | 0 | 0 | 0 |
| Trichomoniasis | 1 | 0.3 | 1 | 2 | 0 | 0 | 1 | 14.2 | 0 | 0 |
| Peritonitis | 5 | 1.5 | 2 | 4.1 | 0 | 0 | 0 | 0 | 1 | 50 |
| Atrophic vaginitis | 17 | 5.2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Battered rape | 20 | 6.1 | 6 | 12.5 | 6 | 17.1 | 0 | 0 | 0 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiu, S.-F.; Huang, P.-J.; Cheng, W.-H.; Huang, C.-Y.; Chu, L.J.; Lee, C.-C.; Lin, H.-C.; Chen, L.-C.; Lin, W.-N.; Tsao, C.-H.; et al. Vaginal Microbiota of the Sexually Transmitted Infections Caused by Chlamydia trachomatis and Trichomonas vaginalis in Women with Vaginitis in Taiwan. Microorganisms 2021, 9, 1864. https://doi.org/10.3390/microorganisms9091864
Chiu S-F, Huang P-J, Cheng W-H, Huang C-Y, Chu LJ, Lee C-C, Lin H-C, Chen L-C, Lin W-N, Tsao C-H, et al. Vaginal Microbiota of the Sexually Transmitted Infections Caused by Chlamydia trachomatis and Trichomonas vaginalis in Women with Vaginitis in Taiwan. Microorganisms. 2021; 9(9):1864. https://doi.org/10.3390/microorganisms9091864
Chicago/Turabian StyleChiu, Shu-Fang, Po-Jung Huang, Wei-Hung Cheng, Ching-Yun Huang, Lichieh Julie Chu, Chi-Ching Lee, Hsin-Chung Lin, Lih-Chyang Chen, Wei-Ning Lin, Chang-Huei Tsao, and et al. 2021. "Vaginal Microbiota of the Sexually Transmitted Infections Caused by Chlamydia trachomatis and Trichomonas vaginalis in Women with Vaginitis in Taiwan" Microorganisms 9, no. 9: 1864. https://doi.org/10.3390/microorganisms9091864
APA StyleChiu, S.-F., Huang, P.-J., Cheng, W.-H., Huang, C.-Y., Chu, L. J., Lee, C.-C., Lin, H.-C., Chen, L.-C., Lin, W.-N., Tsao, C.-H., Tang, P., Yeh, Y.-M., & Huang, K.-Y. (2021). Vaginal Microbiota of the Sexually Transmitted Infections Caused by Chlamydia trachomatis and Trichomonas vaginalis in Women with Vaginitis in Taiwan. Microorganisms, 9(9), 1864. https://doi.org/10.3390/microorganisms9091864







