Sourdough Microbiome Comparison and Benefits
Abstract
1. Introduction
2. Microbial Communities in Different Sourdough Starters
2.1. Ingredients as Sources of Microorganisms
2.2. Microbial Succession along Fermentation Period
2.3. Carbohydrate Fermentation Patterns of Lactic Acid Bacteria (LAB)
3. Benefits of Sourdough
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Chavan, R.S.; Chavan, S.R. Sourdough Technology–A Traditional Way for Wholesome Foods: A Review. Compr. Rev. Food Sci. Food Saf. 2011, 10. [Google Scholar] [CrossRef]
- Hammes, W.P.; Gänzle, M.G. Sourdough breads and related products. In Microbiology of Fermented Foods; Springer: Boston, MA, USA, 1998. [Google Scholar]
- De Vuyst, L.; Van Kerrebroeck, S.; Harth, H.; Huys, G.; Daniel, H.M.; Weckx, S. Microbial ecology of sourdough fermentations: Diverse or uniform? Food Microbiol. 2014, 37. [Google Scholar] [CrossRef]
- Müller, M.R.A.; Wolfrum, G.; Stolz, P.; Ehrmann, M.A.; Vogel, R.F. Monitoring the growth of Lactobacillus species during a rye flour fermentation. Food Microbiol. 2001, 18. [Google Scholar] [CrossRef]
- De Vuyst, L.; Van Kerrebroeck, S.; Leroy, F. Microbial Ecology and Process Technology of Sourdough Fermentation. Adv. Appl. Microbiol. 2017, 100. [Google Scholar] [CrossRef]
- Decock, P.; Cappelle, S. Bread technology and sourdough technology. Trends Food Sci. Technol. 2005, 16. [Google Scholar] [CrossRef]
- Meroth, C.B.; Walter, J.; Hertel, C.; Brandt, M.J.; Hammes, W.P. Monitoring the bacterial population dynamics in sourdough fermentation processes by using PCR-denaturing gradient gel electrophoresis. Appl. Environ. Microbiol. 2003, 69. [Google Scholar] [CrossRef]
- De Vuyst, L.; Neysens, P. The sourdough microflora: Biodiversity and metabolic interactions. Trends Food Sci. Technol. 2005, 16. [Google Scholar] [CrossRef]
- Syrokou, M.K.; Themeli, C.; Paramithiotis, S.; Mataragas, M.; Bosnea, L.; Argyri, A.A.; Chorianopoulos, N.G.; Skandamis, P.N.; Drosinos, E.H. Microbial Ecology of Greek Wheat Sourdoughs, Identified by a Culture-Dependent and a Culture-Independent Approach. Foods 2020, 9, 1603. [Google Scholar] [CrossRef] [PubMed]
- Arena, M.P.; Russo, P.; Spano, G.; Capozzi, V. Exploration of the Microbial Biodiversity Associated with North Apulian Sourdoughs and the Effect of the Increasing Number of Inoculated Lactic Acid Bacteria Strains on the Biocontrol against Fungal Spoilage. Fermentation 2019, 5, 97. [Google Scholar] [CrossRef]
- Plessas, S. Innovations in Sourdough Bread Making. Fermentation 2021, 7, 29. [Google Scholar] [CrossRef]
- Garofalo, C.; Silvestri, G.; Aquilanti, L.; Clementi, F. PCR-DGGE analysis of lactic acid bacteria and yeast dynamics during the production processes of three varieties of Panettone. J. Appl. Microbiol. 2008, 105. [Google Scholar] [CrossRef]
- Lhomme, E.; Urien, C.; Legrand, J.; Dousset, X.; Onno, B.; Sicard, D. Sourdough microbial community dynamics: An analysis during French organic bread-making processes. Food Microbiol. 2016, 53. [Google Scholar] [CrossRef]
- Gobbetti, M.; Corsetti, A.; Rossi, J.; Rosa, F. la Identification and clustering of lactic acid bacteria and yeasts from wheat sourdoughs of central Italy. Ital. J. Food Sci. 1994, 6, 85–94. [Google Scholar]
- Salim-ur-Rehman; Paterson, A.; Piggott, J.R. Flavour in sourdough breads: A review. Trends Food Sci. Technol. 2006, 17. [Google Scholar] [CrossRef]
- Aysha Tai 18 Ways to Make Sourdough Bread More (or Less) Sour—True Sourdough—Sourdough Baking at Home. Available online: https://truesourdough.com/18-ways-to-make-sourdough-bread-more-or-less-sour/ (accessed on 21 March 2021).
- Gobbetti, M.; Rizzello, C.G.; Di Cagno, R.; De Angelis, M. How the sourdough may affect the functional features of leavened baked goods. Food Microbiol. 2014, 37. [Google Scholar] [CrossRef] [PubMed]
- Moroni, A.V.; Dal Bello, F.; Arendt, E.K. Sourdough in gluten-free bread-making: An ancient technology to solve a novel issue? Food Microbiol. 2009, 26. [Google Scholar] [CrossRef]
- Poutanen, K.; Flander, L.; Katina, K. Sourdough and cereal fermentation in a nutritional perspective. Food Microbiol. 2009, 26. [Google Scholar] [CrossRef]
- Thomas, B. Indigestible substances in bread. Bread Ind. 1976, 19, 340–346. [Google Scholar]
- Google’s Year in Search—Google Trends. Available online: https://trends.google.com/trends/yis/2020/GLOBAL/ (accessed on 21 March 2021).
- Gänzle, M.G.; Ehmann, M.; Hammes, W.P. Modeling of growth of Lactobacillus sanfranciscensis and Candida milleri in response to process parameters of sourdough fermentation. Appl. Environ. Microbiol. 1998, 64. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, N.; De Angelis, M.; Calasso, M.; Quinto, M.; Mentana, A.; Minervini, F.; Cappelle, S.; Gobbetti, M. Microbial cell-free extracts affect the biochemical characteristics and sensorial quality of sourdough bread. Food Chem. 2017, 237. [Google Scholar] [CrossRef] [PubMed]
- Pico, J.; Khomenko, I.; Capozzi, V.; Navarini, L.; Biasioli, F. Real-Time Monitoring of Volatile Compounds Losses in the Oven during Baking and Toasting of Gluten-Free Bread Doughs: A PTR-MS Evidence. Foods 2020, 9, 1498. [Google Scholar] [CrossRef]
- Pétel, C.; Onno, B.; Prost, C. Sourdough volatile compounds and their contribution to bread: A review. Trends Food Sci. Technol. 2017, 59, 105–123. [Google Scholar] [CrossRef]
- Laatikainen, R.; Koskenpato, J.; Hongisto, S.M.; Loponen, J.; Poussa, T.; Hillilä, M.; Korpela, R. Randomised clinical trial: Low-FODMAP rye bread vs. regular rye bread to relieve the symptoms of irritable bowel syndrome. Aliment. Pharmacol. Ther. 2016, 4. [Google Scholar] [CrossRef]
- Struyf, N.; Laurent, J.; Verspreet, J.; Verstrepen, K.J.; Courtin, C.M. Saccharomyces cerevisiae and kluyveromyces marxianus cocultures allow reduction of fermentable oligo-, Di-, and monosaccharides and polyols levels in whole wheat bread. J. Agric. Food Chem. 2017, 65, 8704–8713. [Google Scholar] [CrossRef]
- Di Cagno, R.; Rizzello, C.G.; De Angelis, M.; Cassone, A.; Giuliani, G.; Benedusi, A.; Limitone, A.; Surico, R.F.; Gobbetti, M. Use of selected sourdough strains of Lactobacillus for removing gluten and enhancing the nutritional properties of gluten-free bread. J. Food Prot. 2008, 71. [Google Scholar] [CrossRef]
- Leenhardt, F.; Levrat-Verny, M.A.; Chanliaud, E.; Rémésy, C. Moderate decrease of pH by sourdough fermentation is sufficient to reduce phytate content of whole wheat flour through endogenous phytase activity. J. Agric. Food Chem. 2005, 53. [Google Scholar] [CrossRef]
- Barbro, N.; Brittmarie, S.; ÅKE, C. Reduction of the phytate content of bran by leavening in bread and its effect on zinc absorption in man. Br. J. Nutr. 1985, 53. [Google Scholar] [CrossRef]
- Gupta, R.K.; Gangoliya, S.S.; Singh, N.K. Reduction of phytic acid and enhancement of bioavailable micronutrients in food grains. J. Food Sci. Technol. 2015, 52. [Google Scholar] [CrossRef]
- Campbell, J.; Hauser, M.; Hill, S. Nutritional Characteristics of Organic, Freshly Stone-ground Sourdough & Conventional Breads; Ecological Application Projects Publications: Quebec, QC, Canada, 1991. [Google Scholar]
- Vanessa, K. Sourdough and Digestibility”, Health, What to Eat. 2015. Available online: https://sustainablefoodtrust.org/articles/sourdough-and-digestibility/ (accessed on 21 March 2021).
- Douglas, L.C.; Sanders, M.E. Probiotics and Prebiotics in Dietetics Practice. J. Am. Diet. Assoc. 2008, 108. [Google Scholar] [CrossRef]
- Beena Divya, J.; Kulangara Varsha, K.; Madhavan Nampoothiri, K.; Ismail, B.; Pandey, A. Probiotic fermented foods for health benefits. Eng. Life Sci. 2012, 12. [Google Scholar] [CrossRef]
- Schwab, C.; Mastrangelo, M.; Corsetti, A.; Gänzle, M. Formation of oligosaccharides and polysaccharides by Lactobacillus reuteri LTH5448 and Weissella cibaria 10M in sorghum sourdoughs. Cereal Chem. 2008, 85. [Google Scholar] [CrossRef]
- Pérez-Ramos, A.; Mohedano, M.L.; López, P.; Spano, G.; Fiocco, D.; Russo, P.; Capozzi, V. In Situ β-Glucan Fortification of Cereal-Based Matrices by Pediococcus parvulus 2.6: Technological Aspects and Prebiotic Potential. Int. J. Mol. Sci. 2017, 18, 1588. [Google Scholar] [CrossRef]
- Siepmann, F.B.; Ripari, V.; Waszczynskyj, N.; Spier, M.R. Overview of Sourdough Technology: From Production to Marketing. Food Bioprocess Technol. 2018, 11. [Google Scholar] [CrossRef]
- Novotni, D.; Ćurić, D.; Bituh, M.; Barić, I.C.; Škevin, D.; Čukelj, N. Glycemic index and phenolics of partially-baked frozen bread with sourdough. Int. J. Food Sci. Nutr. 2011, 62. [Google Scholar] [CrossRef]
- Bo, S.; Seletto, M.; Choc, A.; Ponzo, V.; Lezo, A.; Demagistris, A.; Evangelista, A.; Ciccone, G.; Bertolino, M.; Cassader, M.; et al. The acute impact of the intake of four types of bread on satiety and blood concentrations of glucose, insulin, free fatty acids, triglyceride and acylated ghrelin. A randomized controlled cross-over trial. Food Res. Int. 2017, 92. [Google Scholar] [CrossRef] [PubMed]
- Brand-Miller, J.; Hayne, S.; Petocz, P.; Colagiuri, S. Low-glycemic index diets in the management of diabetes: A meta-analysis of randomized controlled trials. Diabetes Care 2003, 26. [Google Scholar] [CrossRef]
- Rizkalla, S.W.; Taghrid, L.; Laromiguiere, M.; Huet, D.; Boillot, J.; Rigoir, A.; Elgrably, F.; Slama, G. Improved plasma glucose control, whole-body glucose utilization, and lipid profile on a low-glycemic index diet in type 2 diabetic men: A randomized controlled trial. Diabetes Care 2004, 27. [Google Scholar] [CrossRef]
- Salmerón, J.; Manson, J.E.; Stampfer, M.J.; Colditz, G.A.; Wing, A.L.; Willett, W.C. Dietary fiber, glycemic load, and risk of non-insulin-dependent diabetes mellitus in women. J. Am. Med. Assoc. 1997, 277. [Google Scholar] [CrossRef]
- Frost, G.; Leeds, A.; Trew, G.; Margara, R.; Dornhorst, A. Insulin sensitivity in women at risk of coronary heart disease and the effect of a low glycemic diet. Metabolism 1998, 47. [Google Scholar] [CrossRef]
- Kelly, S.A.; Frost, G.; Whittaker, V.; Summerbell, C.D. Low glycaemic index diets for coronary heart disease. In Cochrane Database of Systematic Reviews; John Wiley & Sons, Ltd.: West Sussex, UK, 2004. [Google Scholar]
- Ebbeling, C.B.; Leidig, M.M.; Sinclair, K.B.; Hangen, J.P.; Ludwig, D.S. A reduced-glycemic load diet in the treatment of adolescent obesity. Arch. Pediatr. Adolesc. Med. 2003, 157. [Google Scholar] [CrossRef]
- Liu, S.; Willett, W.C.; Stampfer, M.J.; Hu, F.B.; Franz, M.; Sampson, L.; Hennekens, C.H.; Manson, J.A.E. A prospective study of dietary glycemic load, carbohydrate intake, and risk of coronary heart disease in US women. Am. J. Clin. Nutr. 2000, 71. [Google Scholar] [CrossRef]
- Chiş, M.S.; Păucean, A.; Man, S.M.; Vodnar, D.C.; Teleky, B.-E.; Pop, C.R.; Stan, L.; Borsai, O.; Kadar, C.B.; Urcan, A.C.; et al. Quinoa Sourdough Fermented with Lactobacillus plantarum ATCC 8014 Designed for Gluten-Free Muffins—A Powerful Tool to Enhance Bioactive Compounds. Appl. Sci. 2020, 10, 7140. [Google Scholar] [CrossRef]
- De Angelis, M.; Gallo, G.; Corbo, M.R.; McSweeney, P.L.H.; Faccia, M.; Giovine, M.; Gobbetti, M. Phytase activity in sourdough lactic acid bacteria: Purification and characterization of a phytase from Lactobacillus sanfranciscensis CB1. Int. J. Food Microbiol. 2003, 87. [Google Scholar] [CrossRef]
- Selhub, E.M.; Logan, A.C.; Bested, A.C. Fermented foods, microbiota, and mental health: Ancient practice meets nutritional psychiatry. J. Physiol. Anthropol. 2014, 33, 1–12. [Google Scholar] [CrossRef]
- Lindenmeier, M.; Hofmann, T. Influence of Baking Conditions and Precursor Supplementation on the Amounts of the Antioxidant Pronyl-L-Lysine in Bakery Products. J. Agric. Food Chem. 2004, 52. [Google Scholar] [CrossRef]
- Galli, V.; Mazzoli, L.; Luti, S.; Venturi, M.; Guerrini, S.; Paoli, P.; Vincenzini, M.; Granchi, L.; Pazzagli, L. Effect of selected strains of lactobacilli on the antioxidant and anti-inflammatory properties of sourdough. Int. J. Food Microbiol. 2018, 286. [Google Scholar] [CrossRef]
- Rizzello, C.G.; Cassone, A.; Di Cagno, R.; Gobbetti, M. Synthesis of angiotensin I-converting enzyme (ACE)-inhibitory peptides and γ-aminobutyric acid (GABA) during sourdough fermentation by selected lactic acid bacteria. J. Agric. Food Chem. 2008, 56. [Google Scholar] [CrossRef]
- Zheng, J.; Wittouck, S.; Salvetti, E.; Franz, C.M.A.P.; Harris, H.M.B.; Mattarelli, P.; O’toole, P.W.; Pot, B.; Vandamme, P.; Walter, J.; et al. A taxonomic note on the genus Lactobacillus: Description of 23 novel genera, emended description of the genus Lactobacillus beijerinck 1901, and union of Lactobacillaceae and Leuconostocaceae. Int. J. Syst. Evol. Microbiol. 2020, 70, 2782–2858. [Google Scholar] [CrossRef]
- Özülkü, G. Rheological Behaviour of Type I Sourdough during Refreshment Procedure; Taylor & Francis: Oxfordshire, UK, 2019; Volume 2. [Google Scholar]
- Ercolini, D.; Pontonio, E.; De Filippis, F.; Minervini, F.; La Storia, A.; Gobbetti, M.; Di Cagno, R. Microbial ecology dynamics during rye and wheat sourdough preparation. Appl. Environ. Microbiol. 2013, 79. [Google Scholar] [CrossRef] [PubMed]
- Hammes, W.P.; Brandt, M.J.; Francis, K.L.; Rosenheim, J.; Seitter, M.F.H.; Vogelmann, S.A. Microbial ecology of cereal fermentations. Trends Food Sci. Technol. 2005, 16. [Google Scholar] [CrossRef]
- Reese, A.T.; Madden, A.A.; Joossens, M.; Lacaze, G.; Dunn, R.R. Influences of Ingredients and Bakers on the Bacteria and Fungi in Sourdough Starters and Bread. mSphere 2020, 5. [Google Scholar] [CrossRef] [PubMed]
- Ogunsakin, A.O.; Vanajakshi, V.; Anu-Appaiah, K.A.; Vijayendra, S.V.N.; Walde, S.G.; Banwo, K.; Sanni, A.I.; Prabhasankar, P. Evaluation of functionally important lactic acid bacteria and yeasts from Nigerian sorghum as starter cultures for gluten-free sourdough preparation. LWT Food Sci. Technol. 2017, 82. [Google Scholar] [CrossRef]
- Sekwati-Monang, B.; Valcheva, R.; Gänzle, M.G. Microbial ecology of sorghum sourdoughs: Effect of substrate supply and phenolic compounds on composition of fermentation microbiota. Int. J. Food Microbiol. 2012, 159. [Google Scholar] [CrossRef] [PubMed]
- Montemurro, M.; Celano, G.; De Angelis, M.; Gobbetti, M.; Rizzello, C.G.; Pontonio, E. Selection of non-Lactobacillus strains to be used as starters for sourdough fermentation. Food Microbiol. 2020, 90. [Google Scholar] [CrossRef]
- Corsetti, A.; Settanni, L.; Chaves López, C.; Felis, G.E.; Mastrangelo, M.; Suzzi, G. A taxonomic survey of lactic acid bacteria isolated from wheat (Triticum durum) kernels and non-conventional flours. Syst. Appl. Microbiol. 2007, 30. [Google Scholar] [CrossRef] [PubMed]
- De Angelis, M.; Minervini, F.; Siragusa, S.; Rizzello, C.G.; Gobbetti, M. Wholemeal wheat flours drive the microbiome and functional features of wheat sourdoughs. Int. J. Food Microbiol. 2019, 302. [Google Scholar] [CrossRef] [PubMed]
- Rizzello, C.G.; Cavoski, I.; Turk, J.; Ercolini, D.; Nionelli, L.; Pontonio, E.; De Angelis, M.; De Filippis, F.; Gobbetti, M.; Di Cagno, R. Organic cultivation of Triticum turgidum subsp. durum is reflected in the flour-sourdough fermentation-bread axis. Appl. Environ. Microbiol. 2015, 81. [Google Scholar] [CrossRef]
- Vrancken, G.; Rimaux, T.; Weckx, S.; Leroy, F.; De Vuyst, L. Influence of temperature and backslopping time on the microbiota of a type I propagated laboratory wheat sourdough fermentation. Appl. Environ. Microbiol. 2011, 77. [Google Scholar] [CrossRef] [PubMed]
- Gobbetti, M.; Minervini, F.; Pontonio, E.; Di Cagno, R.; De Angelis, M. Drivers for the establishment and composition of the sourdough lactic acid bacteria biota. Int. J. Food Microbiol. 2016, 239. [Google Scholar] [CrossRef]
- Andrew, W. Bread Matters: The State of Modern Bread and a Definitive Guide to Baking Your Own; Andrews McMeel Publishing: Kansas City, MO, USA, 2011. [Google Scholar]
- Salovaara, H.; Valjakka, T. The effect of fermentation temperature, flour type, and starter on the properties of sour wheat bread. Int. J. Food Sci. Technol. 1987, 22. [Google Scholar] [CrossRef]
- Abedfar, A.; Sadeghi, A. Response surface methodology for investigating the effects of sourdough fermentation conditions on Iranian cup bread properties. Heliyon 2019, 5. [Google Scholar] [CrossRef]
- Mueen-ud-Din, G.; Salim-ur-Rehman; Anjum, F.M.; Nawaz, H. Studies on organic acids and minerals content of sourdough naans made from different extraction rate wheat flours and starter cultures. Pak. J. Nutr. 2009, 8. [Google Scholar] [CrossRef]
- Hui, Y.H.; Culbertson, J.D.; Duncan, S.E.; Legarreta, I.G.; Li-Chan, E.C.Y.; Ma, C.Y.; Manley, C.; McMeekin, T.; Nip, W.K.; Nollet, L.M.L.; et al. Handbook of Food Science, Technology, and Engineering; CRC Press: Boca Raton, FL, USA, 2005; ISBN 9781466507876. [Google Scholar]
- Kovacic, D. Why Not Rye? | Bay State Milling. 2011. Available online: http://www.baystatemilling.com/ingredients/why-not-rye/ (accessed on 21 March 2021).
- Fujimoto, A.; Ito, K.; Itou, M.; Narushima, N.; Ito, T.; Yamamoto, A.; Hirayama, S.; Furukawa, S.; Morinaga, Y.; Miyamoto, T. Microbial behavior and changes in food constituents during fermentation of Japanese sourdoughs with different rye and wheat starting materials. J. Biosci. Bioeng. 2018, 125. [Google Scholar] [CrossRef] [PubMed]
- Fraberger, V.; Unger, C.; Kummer, C.; Domig, K.J. Insights into microbial diversity of traditional Austrian sourdough. LWT 2020, 127. [Google Scholar] [CrossRef]
- Esteve, C.C.; De Barber, C.B.; Martinez-Anaya, M.A. Microbial Sour Doughs Influence Acidification Properties and Breadmaking Potential of Wheat Dough. J. Food Sci. 1994, 59. [Google Scholar] [CrossRef]
- Van Der Meulen, R.; Scheirlinck, I.; Van Schoor, A.; Huys, G.; Vancanneyt, M.; Vandamme, P.; De Vuyst, L. Population dynamics and metabolite target analysis of lactic acid bacteria during laboratory fermentations of wheat and spelt sourdoughs. Appl. Environ. Microbiol. 2007, 73. [Google Scholar] [CrossRef] [PubMed]
- Ispirli, H.; Demirbaş, F.; Yüzer, M.O.; Dertli, E. Identification of Lactic Acid Bacteria from Spontaneous Rye Sourdough and Determination of Their Functional Characteristics. Food Biotechnol. 2018, 32. [Google Scholar] [CrossRef]
- Rizzello, C.G.; Calasso, M.; Campanella, D.; De Angelis, M.; Gobbetti, M. Use of sourdough fermentation and mixture of wheat, chickpea, lentil and bean flours for enhancing the nutritional, texture and sensory characteristics of white bread. Int. J. Food Microbiol. 2014, 180. [Google Scholar] [CrossRef]
- Zhang, G.; Zhang, W.; Sadiq, F.A.; Arbab, S.H.; He, G. Microbiota succession and metabolite changes during the traditional sourdough fermentation of Chinese steamed bread. CYTA J. Food 2019, 17. [Google Scholar] [CrossRef]
- Ganchev, I.; Koleva, Z.; Kizheva, Y.; Moncheva, P.; Hristova, P. Lactic acid bacteria from spontaneosly fermented rye sourdough. Bulg. J. Agric. Sci. 2014, 20, 69–73. [Google Scholar]
- Syrokou, M.K.; Tziompra, S.; Psychogiou, E.-E.; Mpisti, S.-D.; Paramithiotis, S.; Bosnea, L.; Mataragas, M.; Skandamis, P.N.; Drosinos, E.H. Technological and Safety Attributes of Lactic Acid Bacteria and Yeasts Isolated from Spontaneously Fermented Greek Wheat Sourdoughs. Microorganisms 2021, 9, 671. [Google Scholar] [CrossRef]
- Oshiro, M.; Tanaka, M.; Zendo, T.; Nakayama, J. Impact of pH on succession of sourdough lactic acid bacteria communities and their fermentation properties. Biosci. Microbiota Food Health 2020, 39. [Google Scholar] [CrossRef]
- Weckx, S.; Van der Meulen, R.; Maes, D.; Scheirlinck, I.; Huys, G.; Vandamme, P.; De Vuyst, L. Lactic acid bacteria community dynamics and metabolite production of rye sourdough fermentations share characteristics of wheat and spelt sourdough fermentations. Food Microbiol. 2010, 27. [Google Scholar] [CrossRef]
- Kozlinskis, E.; Skudra, L.; Klava, D.; Kunkulberga, D. Characterization of Rye Sourdough Microflora. Latv. Univ. Agric. Fac. Food Technol. 2008, 89–93. [Google Scholar]
- Rosenquist, H.; Hansen, Å. The microbial stability of two bakery sourdoughs made from conventionally and organically grown rye. Food Microbiol. 2000, 17. [Google Scholar] [CrossRef]
- Edema, M.O.; Sanni, A.I. Functional properties of selected starter cultures for sour maize bread. Food Microbiol. 2008, 25. [Google Scholar] [CrossRef]
- Ben Omar, N.; Ampe, F. Microbial community dynamics during production of the Mexican fermented maize dough pozol. Appl. Environ. Microbiol. 2000, 66. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.B.; Tingirikari, J.M.R.; Kwon, Y.W.; Li, L.; Kim, G.E.; Han, N.S. Polyphasic microbial analysis of traditional korean jeung-pyun sourdough fermented with makgeolli. J. Microbiol. Biotechnol. 2017, 27. [Google Scholar] [CrossRef] [PubMed]
- Ogunsakin, O.A.; Banwo, K.; Ogunremi, O.R.; Sanni, A.I. Microbiological and physicochemical properties of sourdough bread from sorghum flour. Int. Food Res. J. 2015, 22, 2610–2618. [Google Scholar]
- Coda, R.; Kianjam, M.; Pontonio, E.; Verni, M.; Di Cagno, R.; Katina, K.; Rizzello, C.G.; Gobbetti, M. Sourdough-type propagation of faba bean flour: Dynamics of microbial consortia and biochemical implications. Int. J. Food Microbiol. 2017, 248. [Google Scholar] [CrossRef]
- Gordún, E.; Del Valle, L.J.; Ginovart, M.; Carbó, R. Comparison of the microbial dynamics and biochemistry of laboratory sourdoughs prepared with grape, apple and yogurt. Food Sci. Technol. Int. 2015, 21. [Google Scholar] [CrossRef]
- Aplevicz, K.S.; Mazo, J.Z.; Ilha, E.C.; Dinon, A.Z.; Sant’Anna, E.S. Isolation and characterization of lactic acid bacteria and yeasts from the Brazilian grape sourdough. Braz. J. Pharm. Sci. 2014, 50. [Google Scholar] [CrossRef]
- Katsi, P.; Kosma, I.S.; Michailidou, S.; Argiriou, A.; Badeka, A.V.; Kontominas, M.G. Characterization of Artisanal Spontaneous Sourdough Wheat Bread from Central Greece: Evaluation of Physico-Chemical, Microbiological, and Sensory Properties in Relation to Conventional Yeast Leavened Wheat Bread. Foods 2021, 10, 635. [Google Scholar] [CrossRef]
- Arena, M.P.; Russo, P.; Spano, G.; Capozzi, V. From Microbial Ecology to Innovative Applications in Food Quality Improvements: The Case of Sourdough as a Model Matrix. J Multidiscip. Sci. J. 2020, 3, 9–19. [Google Scholar] [CrossRef]
- Gobbetti, M.; Corsetti, A.; Rossi, J. The sourdough microflora. Interactions between lactic acid bacteria and yeasts: Metabolism of carbohydrates. Appl. Microbiol. Biotechnol. 1994, 41. [Google Scholar] [CrossRef]
- Comasio, A.; Van Kerrebroeck, S.; De Vuyst, L. Lemon juice and apple juice used as source of citrate and malate, respectively, enhance the formation of buttery aroma compounds and/or organic acids during Type 2 and Type 3 sourdough productions performed with Companilactobacillus crustorum LMG 23699. Int. J. Food Microbiol. 2021, 339. [Google Scholar] [CrossRef] [PubMed]
- Jagelaviciute, J.; Cizeikiene, D. The influence of non-traditional sourdough made with quinoa, hemp and chia flour on the characteristics of gluten-free maize/rice bread. LWT 2021, 137. [Google Scholar] [CrossRef]
- Galli, V.; Venturi, M.; Coda, R.; Maina, N.H.; Granchi, L. Isolation and characterization of indigenous Weissella confusa for in situ bacterial exopolysaccharides (EPS) production in chickpea sourdough. Food Res. Int. 2020, 138. [Google Scholar] [CrossRef]
- Banwo, K.; Osagbemi, O.; Ajao, O.; Sanni, A. Sourdough Bread from the Blend of Cassava, Sweet Potato, and Soybean Flours Using Lactobacillus Plantarum and Pichia Kudriavzevii. Acta Aliment. 2020, 49, 441–450. [Google Scholar] [CrossRef]
- Minervini, F.; Dinardo, F.R.; De Angelis, M.; Gobbetti, M. Tap water is one of the drivers that establish and assembly the lactic acid bacterium biota during sourdough preparation. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhu, X.; Bi, Y.; Zhao, R.; Nie, Y.; Yuan, W. Dynamics of microbial community and changes of metabolites during production of type Ι sourdough steamed bread made by retarded sponge-dough method. Food Chem. 2020, 330. [Google Scholar] [CrossRef]
- Corsetti, A.; Settanni, L. Lactobacilli in sourdough fermentation. Food Res. Int. 2007, 40. [Google Scholar] [CrossRef]
- Zugic-Petrovic, T.; Jokovic, N.; Savic, D. The evolution of lactic acid bacteria community during the development of mature sourdough. Acta Period. Technol. 2009. [Google Scholar] [CrossRef]
- Reale, A.; Di Renzo, T.; Succi, M.; Tremonte, P.; Coppola, R.; Sorrentino, E. Identification of lactobacilli isolated in traditional ripe wheat sourdoughs by using molecular methods. World J. Microbiol. Biotechnol. 2011, 27. [Google Scholar] [CrossRef]
- Gänzle, M.G.; Vermeulen, N.; Vogel, R.F. Carbohydrate, peptide and lipid metabolism of lactic acid bacteria in sourdough. Food Microbiol. 2007, 24. [Google Scholar] [CrossRef] [PubMed]
- Damiani, P.; Gobbetti, M.; Cossignani, L.; Corsetti, A.; Simonetti, M.S.; Rossi, J. The sourdough microflora. Characterization of hetero- and homofermentative lactic acid bacteria, yeasts and their interactions on the basis of the volatile compounds produced. LWT Food Sci. Technol. 1996, 29. [Google Scholar] [CrossRef]
- Gänzle, M.G.; Zheng, J. Lifestyles of sourdough lactobacilli—Do they matter for microbial ecology and bread quality? Int. J. Food Microbiol. 2019, 302. [Google Scholar] [CrossRef] [PubMed]
- Axelsson, L.; Ahrné, S. Lactic Acid Bacteria. In Applied Microbial Systematics; Springer: Dordrecht, The Netherlands, 2000; pp. 367–388. [Google Scholar]
- Hansen, Å.; Hansen, B. Flavour of sourdough wheat bread crumb. Eur. Food Res. Technol. 1996, 202. [Google Scholar] [CrossRef]
- Salovaara, H.; Gänzle, M. Lactic acid bacteria in cereal-based products. In Lactic Acid Bacteria: Microbiological and Functional Aspects, 4th ed.; CRC Press: Boca Raton, FL, USA, 2011. [Google Scholar]
- Russo, P.; Fares, C.; Longo, A.; Spano, G.; Capozzi, V. Lactobacillus plantarum with Broad Antifungal Activity as a Protective Starter Culture for Bread Production. Foods 2017, 6, 110. [Google Scholar] [CrossRef]
- Corsetti, A.; Gobbetti, M.; Rossi, J.; Damiani, P. Antimould activity of sourdough lactic acid bacteria: Identification of a mixture of organic acids produced by Lactobacillus sanfrancisco CB1. Appl. Microbiol. Biotechnol. 1998, 50. [Google Scholar] [CrossRef]
- Jenkins, S. Baker Technology, Book 1, Bread; Lester and Orpen Limited: Toronto, ON, Canada, 1975. [Google Scholar]
- Torrieri, E.; Pepe, O.; Ventorino, V.; Masi, P.; Cavella, S. Effect of sourdough at different concentrations on quality and shelf life of bread. LWT Food Sci. Technol. 2014, 56. [Google Scholar] [CrossRef]
- Fernández-Peláez, J.; Paesani, C.; Gómez, M. Sourdough Technology as a Tool for the Development of Healthier Grain-Based Products: An Update. Agronomy 2020, 10, 1962. [Google Scholar] [CrossRef]
- Vermeulen, N.; Czerny, M.; Gänzle, M.G.; Schieberle, P.; Vogel, R.F. Reduction of (E)-2-nonenal and (E,E)-2,4-decadienal during sourdough fermentation. J. Cereal Sci. 2007, 45, 78–87. [Google Scholar] [CrossRef]
- De Angelis, M.; Mariotti, L.; Rossi, J.; Servili, M.; Fox, P.F.; Rollán, G.; Gobbetti, M. Arginine catabolism by sourdough lactic acid bacteria: Purification and characterization of the arginine deiminase pathway enzymes from Lactobacillus sanfranciscensis CB1. Appl. Environ. Microbiol. 2002, 68. [Google Scholar] [CrossRef]
- Thiele, C.; Gänzle, M.G.; Vogel, R.F. Contribution of sourdough lactobacilli, yeast, and cereal enzymes to the generation of amino acids in dough relevant for bread flavor. Cereal Chem. 2002, 79. [Google Scholar] [CrossRef]
- Hansen, A.; Schieberle, P. Generation of aroma compounds during sourdough fermentation: Applied and fundamental aspects. Trends Food Sci. Technol. 2005, 16. [Google Scholar] [CrossRef]
- Kratochvil, J.; Holas, J. Study of proteolytic processes, rye sour. Prog. Cereal Chem. Technol. 1983, 791–798. [Google Scholar]
- Schieberle, P. Intense aroma compounds: Useful tools to monitor the influence of processing and storage on bread aroma. Adv. Food Sci. 1996, 18, 237–244. [Google Scholar]
- Pico, J.; Bernal, J.; Gómez, M. Wheat bread aroma compounds in crumb and crust: A review. Food Res. Int. 2015, 75, 200–215. [Google Scholar] [CrossRef]
- Di Monaco, R.; Torrieri, E.; Pepe, O.; Masi, P.; Cavella, S. Effect of Sourdough with Exopolysaccharide (EPS)-Producing Lactic Acid Bacteria (LAB) on Sensory Quality of Bread during Shelf Life. Food Bioprocess Technol. 2015, 8, 691–701. [Google Scholar] [CrossRef]
- Taglieri, I.; Macaluso, M.; Bianchi, A.; Sanmartin, C.; Quartacci, M.F.; Zinnai, A.; Venturi, F. Overcoming bread quality decay concerns: Main issues for bread shelf life as a function of biological leavening agents and different extra ingredients used in formulation. A review. J. Sci. Food Agric. 2021, 101, 1732–1743. [Google Scholar] [CrossRef] [PubMed]
- Tamani, R.J.; Goh, K.K.T.; Brennan, C.S. Physico-Chemical Properties of Sourdough Bread Production Using Selected Lactobacilli Starter Cultures. J. Food Qual. 2013, 36, 245–252. [Google Scholar] [CrossRef]
- Bockwoldt, J.A.; Fellermeier, J.; Steffens, E.; Vogel, R.F.; Ehrmann, M.A. β-Glucan Production by Levilactobacillus brevis and Pediococcus claussenii for In Situ Enriched Rye and Wheat Sourdough Breads. Foods 2021, 10, 547. [Google Scholar] [CrossRef]
- Capozzi, V.; Fragasso, M.; Bimbo, F. Microbial Resources, Fermentation and Reduction of Negative Externalities in Food Systems: Patterns toward Sustainability and Resilience. Fermentation 2021, 7, 54. [Google Scholar] [CrossRef]
- Catzeddu, P. Sourdough breads. In Flour and Breads and Their Fortification in Health and Disease Prevention; Elsevier: Cambridge, MA, USA, 2019; pp. 177–188. ISBN 9780128146392. [Google Scholar]
- Kopeć, A.; Pysz, M.; Borczak, B.; Sikora, E.; Rosell, C.M.; Collar, C.; Sikora, M. Effects of sourdough and dietary fibers on the nutritional quality of breads produced by bake-off technology. J. Cereal Sci. 2011, 54. [Google Scholar] [CrossRef]
- Laatikainen, R.; Koskenpato, J.; Hongisto, S.M.; Loponen, J.; Poussa, T.; Huang, X.; Sontag-Strohm, T.; Salmenkari, H.; Korpela, R. Pilot study: Comparison of sourdough wheat bread and yeast-fermented wheat bread in individuals with wheat sensitivity and irritable bowel syndrome. Nutrients 2017, 9, 1215. [Google Scholar] [CrossRef]
- Huynh, B.L.; Palmer, L.; Mather, D.E.; Wallwork, H.; Graham, R.D.; Welch, R.M.; Stangoulis, J.C.R. Genotypic variation in wheat grain fructan content revealed by a simplified HPLC method. J. Cereal Sci. 2008, 48. [Google Scholar] [CrossRef]
- Gralnek, I.M.; Hays, R.D.; Kilbourne, A.A.; Naliboff, B.; Mayer, E.A. The impact of irritable bowel syndrome on health-related quality of life. Gastroenterology 2000, 119. [Google Scholar] [CrossRef]
- Lydiard, R.B. Irritable bowel syndrome, anxiety, and depression: What are the links? J. Clin. Psychiatry 2001, 62, 38–47. [Google Scholar]
- Tilley, M.; Chen, Y.R.; Miller, R.A. Wheat breeding and quality evaluation in the US. In Breadmaking: Improving Quality, 2nd ed.; Elsevier Inc.: Sawston, Cambridge, UK, 2012; pp. 216–236. ISBN 9780857090607. [Google Scholar]
- Di Sabatino, A.; Lancet, G.C. Coeliac disease. Lancet 2019, 373, 1480–1493. [Google Scholar] [CrossRef]
- Green, P.H.R.; Jabri, B. Coeliac disease. Lancet 2003, 362, 383–391. [Google Scholar] [CrossRef]
- Smith, F.; Pan, X.; Bellido, V.; Toole, G.A.; Gates, F.K.; Wickham, M.S.J.; Shewry, P.R.; Bakalis, S.; Padfield, P.; Mills, E.N.C. Digestibility of gluten proteins is reduced by baking and enhanced by starch digestion. Mol. Nutr. Food Res. 2015, 59. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Leffler, D.A.; Bai, J.C.; Biagi, F.; Fasano, A.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Kelly, C.P.; Leonard, J.N.; et al. The Oslo definitions for coeliac disease and related terms. Gut 2013, 62. [Google Scholar] [CrossRef] [PubMed]
- Lamacchia, C.; Camarca, A.; Picascia, S.; Di Luccia, A.; Gianfrani, C. Cereal-based gluten-free food: How to reconcile nutritional and technological properties of wheat proteins with safety for celiac disease patients. Nutrients 2014, 6, 575. [Google Scholar] [CrossRef]
- Catassi, C.; Gatti, S.; Fasano, A. The new epidemiology of celiac disease. J. Pediatr. Gastroenterol. Nutr. 2014, 59. [Google Scholar] [CrossRef] [PubMed]
- Lindfors, K.; Ciacci, C.; Kurppa, K.; Lundin, K.E.A.; Makharia, G.K.; Mearin, M.L.; Murray, J.A.; Verdu, E.F.; Kaukinen, K. Coeliac disease. Nat. Rev. Dis. Prim. 2019, 5, 1–18. [Google Scholar] [CrossRef]
- Kupper, C. Dietary guidelines and implementation for celiac disease. Gastroenterology 2005, 128. [Google Scholar] [CrossRef] [PubMed]
- Lohi, S.; Mustalahti, K.; Kaukinen, K.; Laurila, K.; Collin, P.; Rissanen, H.; Lohi, O.; Bravi, E.; Gasparin, M.; Reunanen, A.; et al. Increasing prevalence of coeliac disease over time. Aliment. Pharmacol. Ther. 2007, 26. [Google Scholar] [CrossRef]
- Cabrera-Chávez, F.; Calderón de la Barca, A.M. Trends in wheat technology and modification of gluten proteins for dietary treatment of coeliac disease patients. J. Cereal Sci. 2010, 52. [Google Scholar] [CrossRef]
- Rizzello, C.G.; De Angelis, M.; Di Cagno, R.; Camarca, A.; Silano, M.; Losito, I.; De Vincenzi, M.; De Bari, M.D.; Palmisano, F.; Maurano, F.; et al. Highly efficient gluten degradation by lactobacilli and fungal proteases during food processing: New perspectives for celiac disease. Appl. Environ. Microbiol. 2007, 73, 4499–4507. [Google Scholar] [CrossRef] [PubMed]
- Thiele, C.; Grassl, S.; Gänzle, M. Gluten Hydrolysis and Depolymerization during Sourdough Fermentation. J. Agric. Food Chem. 2004, 52, 1307–1314. [Google Scholar] [CrossRef]
- Fraberger, V.; Ladurner, M.; Nemec, A.; Grunwald-Gruber, C.; Call, L.M.; Hochegger, R.; Domig, K.J.; D’Amico, S. Insights into the Potential of Sourdough-Related Lactic Acid Bacteria to Degrade Proteins in Wheat. Microorganisms 2020, 8, 1689. [Google Scholar] [CrossRef] [PubMed]
- Admassu, S. Potential health benefits and problems associated with phytochemicals in food legumes. East Afr. J. Sci. 2010, 3, 116–133. [Google Scholar] [CrossRef]
- Afinah, S.; Yazid, A.M.; Anis Shobirin, M.H.; Shuhaimi, M. Phytase: Application in food industry. Int. Food Res. J. 2010, 17, 13–21. [Google Scholar]
- Kresser, C. Another Reason You Shouldn’t Go Nuts on Nuts Chris Kresser. 2019. Available online: https://chriskresser.com/another-reason-you-shouldnt-go-nuts-on-nuts/ (accessed on 21 March 2021).
- De Vero, L.; Iosca, G.; Gullo, M.; Pulvirenti, A. Functional and Healthy Features of Conventional and Non-Conventional Sourdoughs. Appl. Sci. 2021, 11, 3694. [Google Scholar] [CrossRef]
- Chow, J. Probiotics and prebiotics: A brief overview. J. Ren. Nutr. 2002, 12. [Google Scholar] [CrossRef] [PubMed]
- Ruan, Y.; Sun, J.; He, J.; Chen, F.; Chen, R.; Chen, H. Effect of probiotics on glycemic control: A systematic review and meta-analysis of randomized, controlled trials. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Couch, G.W. Effect of Sourdough Fermentation Parameters on Bread Properties. Master’s Thesis, Clemson University, Clemson, SC, USA, 2016. [Google Scholar]
- Gänzle, M.G. Enzymatic and bacterial conversions during sourdough fermentation. Food Microbiol. 2014, 37. [Google Scholar] [CrossRef] [PubMed]
- Fardet, A.; Leenhardt, F.; Lioger, D.; Scalbert, A.; Rémésy, C. Parameters controlling the glycaemic response to breads. Nutr. Res. Rev. 2006, 19. [Google Scholar] [CrossRef] [PubMed]
- Foster-Powell, K.; Holt, S.H.A.; Brand-Miller, J.C. International table of gylcemic index and glycemic load values: 2002. Am. J. Clin. Nutr. 2002, 76. [Google Scholar] [CrossRef]
- Romão, B.; Falcomer, A.L.; Palos, G.; Cavalcante, S.; Botelho, R.B.A.; Nakano, E.Y.; Raposo, A.; Shakeel, F.; Alshehri, S.; Mahdi, W.A.; et al. Glycemic Index of Gluten-Free Bread and Their Main Ingredients: A Systematic Review and Meta-Analysis. Foods 2021, 10, 506. [Google Scholar] [CrossRef] [PubMed]
- Demirkesen-Bicak, H.; Arici, M.; Yaman, M.; Karasu, S.; Sagdic, O. Effect of Different Fermentation Condition on Estimated Glycemic Index, In Vitro Starch Digestibility, and Textural and Sensory Properties of Sourdough Bread. Foods 2021, 10, 514. [Google Scholar] [CrossRef]
- Gobbetti, M.; De Angelis, M.; Corsetti, A.; Di Cagno, R. Biochemistry and physiology of sourdough lactic acid bacteria. Trends Food Sci. Technol. 2005, 16. [Google Scholar] [CrossRef]
- Polese, B.; Nicolai, E.; Genovese, D.; Verlezza, V.; La Sala, C.N.; Aiello, M.; Inglese, M.; Incoronato, M.; Sarnelli, G.; De Rosa, T.; et al. Postprandial gastrointestinal function differs after acute administration of sourdough compared with brewer’s yeast bakery products in healthy adults. J. Nutr. 2018, 148. [Google Scholar] [CrossRef] [PubMed]
- Scazzina, F.; Del Rio, D.; Pellegrini, N.; Brighenti, F. Sourdough bread: Starch digestibility and postprandial glycemic response. J. Cereal Sci. 2009, 49. [Google Scholar] [CrossRef]
- Liljeberg, H.G.M.; Björk, I.M.E. Delayed gastric emptying rate as a potential mechanism for lowered glycemia after eating sourdough bread: Studies in humans and rats using test products with added organic acids or an organic salt. Am. J. Clin. Nutr. 1996, 64. [Google Scholar] [CrossRef] [PubMed]
- Bondia-Pons, I.; Nordlund, E.; Mattila, I.; Katina, K.; Aura, A.M.; Kolehmainen, M.; Orešič, M.; Mykkänen, H.; Poutanen, K. Postprandial differences in the plasma metabolome of healthy Finnish subjects after intake of a sourdough fermented endosperm rye bread versus white wheat bread. Nutr. J. 2011, 10. [Google Scholar] [CrossRef]
- Östman, E.M.; Nilsson, M.; Liljeberg Elmståhl, H.G.M.; Molin, G.; Björck, I.M.E. On the effect of lactic acid on blood glucose and insulin responses to cereal products: Mechanistic studies in healthy subjects and in vitro. J. Cereal Sci. 2002, 36. [Google Scholar] [CrossRef]
- De Angelis, M.; Rizzello, C.G.; Alfonsi, G.; Arnault, P.; Cappelle, S.; Di Cagno, R.; Gobbetti, M. Use of sourdough lactobacilli and oat fibre to decrease the glycaemic index of white wheat bread. Br. J. Nutr. 2007, 98. [Google Scholar] [CrossRef]
- De Angelis, M.; Damiano, N.; Rizzello, C.G.; Cassone, A.; Di Cagno, R.; Gobbetti, M. Sourdough fermentation as a tool for the manufacture of low-glycemic index white wheat bread enriched in dietary fibre. Eur. Food Res. Technol. 2009, 229. [Google Scholar] [CrossRef]
- Lappi, J.; Selinheimo, E.; Schwab, U.; Katina, K.; Lehtinen, P.; Mykkänen, H.; Kolehmainen, M.; Poutanen, K. Sourdough fermentation of wholemeal wheat bread increases solubility of arabinoxylan and protein and decreases postprandial glucose and insulin responses. J. Cereal Sci. 2010, 51. [Google Scholar] [CrossRef]
- MacKay, K.A.; Tucker, A.J.; Duncan, A.M.; Graham, T.E.; Robinson, L.E. Whole grain wheat sourdough bread does not affect plasminogen activator inhibitor-1 in adults with normal or impaired carbohydrate metabolism. Nutr. Metab. Cardiovasc. Dis. 2012, 22. [Google Scholar] [CrossRef] [PubMed]
- Wolter, A.; Hager, A.S.; Zannini, E.; Arendt, E.K. Influence of sourdough on in vitro starch digestibility and predicted glycemic indices of gluten-free breads. Food Funct. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Freeland-Graves, J.H.; Peckham, G.C. Foundations of Food Preparation; Prentice Hall: Hoboken, NJ, USA, 1995. [Google Scholar]
- Bohn, L.; Meyer, A.S.; Rasmussen, S.K. Phytate: Impact on environment and human nutrition. A challenge for molecular breeding. J. Zhejiang Univ. Sci. B 2008, 9. [Google Scholar] [CrossRef] [PubMed]
- Katina, K.; Arendt, E.; Liukkonen, K.H.; Autio, K.; Flander, L.; Poutanen, K. Potential of sourdough for healthier cereal products. Trends Food Sci. Technol. 2005, 16, 104–112. [Google Scholar] [CrossRef]
- Lopez, H.W.; Krespine, V.; Guy, G.; Messager, A.; Demigne, C.; Remesy, C. Prolonged fermentation of whole wheat sourdough reduces phytate level and increases soluble magnesium. J. Agric. Food Chem. 2001, 49. [Google Scholar] [CrossRef] [PubMed]
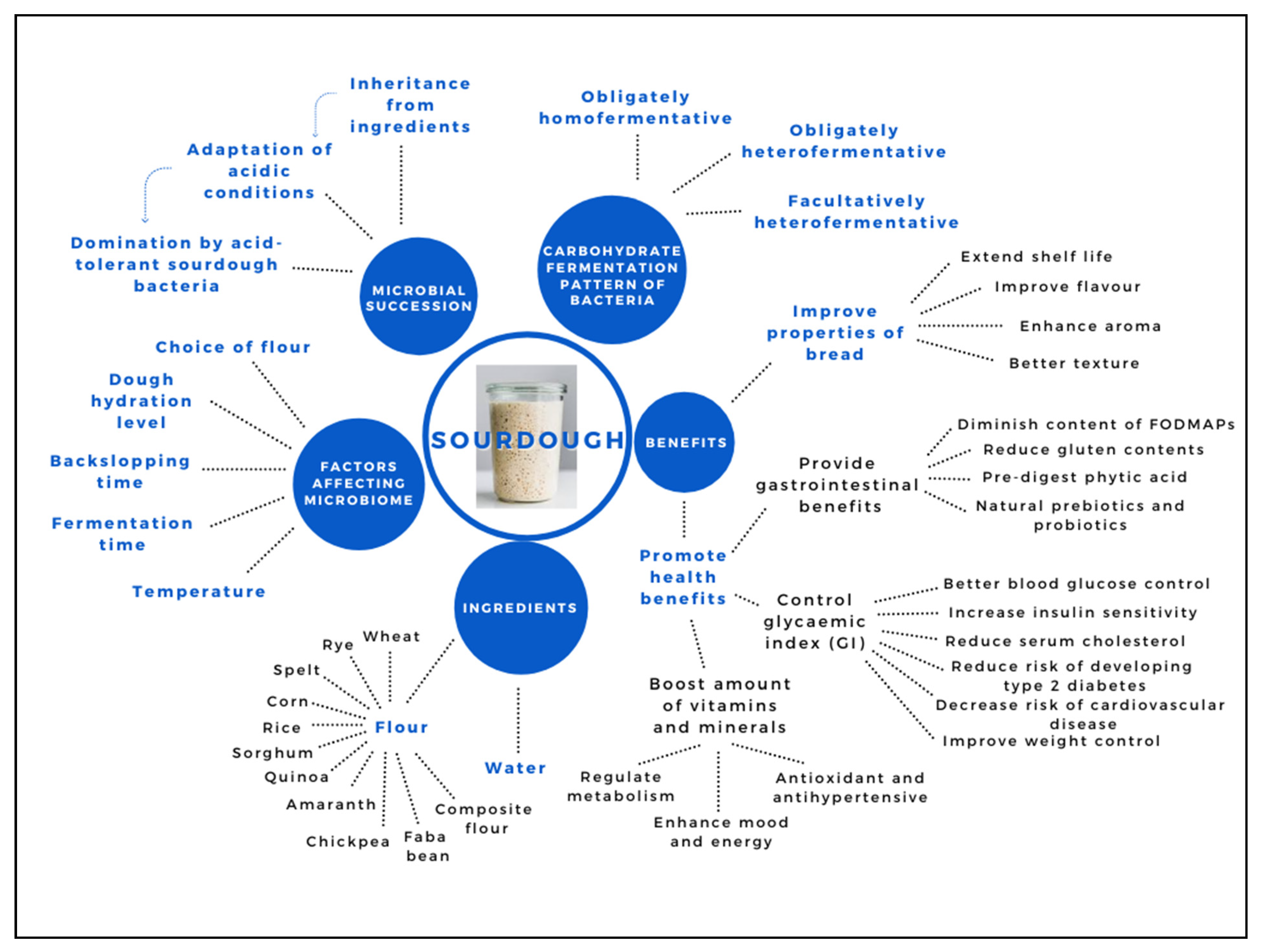
| Benefits | References |
|---|---|
| Improved properties of bread | |
| (a) Extend shelf life | [12,18,22] |
| (b) Improve flavour | [12,17,18,23] |
| (c) Enhance aroma | [18,23,24,25] |
| (d) Better texture | [12,18,23] |
| Promote health benefits | |
| (a) Provide gastrointestinal benefits | |
| -Diminish content of FODMAPs | [26,27] |
| -Reduce gluten contents | [28] |
| -Pre-digest phytic acid | [29,30,31,32,33] |
| -Natural prebiotics and probiotics | [34,35,36,37] |
| (b) Control glycaemic index (GI) | [38,39,40] |
| -Better blood glucose control | [41,42,43] |
| -Increase insulin sensitivity | [41,42,44] |
| -Reduce serum cholesterol | [45] |
| -Reduce risk of developing type 2 diabetes | [41,42,46] |
| -Decrease risk of cardiovascular disease | [41,42,45,46,47] |
| -Improve weight control | [46] |
| (c) Boost amount of vitamins and minerals | [38,48,49] |
| -Regulate metabolism | [50] |
| -Enhance mood and energy | [50] |
| -Antioxidant and antihypertensive | [51,52,53] |
| Flour | Dough (Prior to Fermentation) | Sourdough (After Fermentation) | ||
|---|---|---|---|---|
| LAB | References | LAB | References | |
| Cereals | ||||
| Wheat | Lb. acidophilus | [14] | ||
| Lb. alimentarius | ||||
| Lb. brevis | ||||
| Lb. farciminis | ||||
| Lb. fermentum | ||||
| Lb. fructivorans | ||||
| Lb. plantarum | ||||
| Lb. sanfranciscensis | ||||
| Aerococcus spp. | [56] | |||
| Lb. plantarum | Lb. plantarum | [56] | ||
| Lb. sakei | Lb. sakei | |||
| Lb. sanfranciscensis | ||||
| Lc. lactis | Lc. lactis | |||
| Leuconostoc spp. | ||||
| P. pentosaceus | P. pentosaceus | |||
| Weissella spp. | Weissella spp. | |||
| Ec. hermanniensis | [63] | |||
| Lb. brantae | [63] | |||
| Lb. lindneri | ||||
| Lb. nodensis | ||||
| Lb. plantarum | ||||
| L. carnosum | ||||
| L. kimchii | ||||
| L. mesenteroides | ||||
| P. argentinicus | ||||
| P. pentosaceus | ||||
| W. cibaria | ||||
| W. confusa | ||||
| W. salipiscis | ||||
| Ec. lactis | [64] | |||
| Lb. brevis | [64] | |||
| Lb. plantarum | Lb. plantarum | |||
| Lb. sanfranciscensis | ||||
| Lc. lactis | ||||
| L. citreum | L. citreum | |||
| P. pentosaceus | ||||
| W. cibaria | ||||
| Lb. fermentum | [65] | |||
| Lb. johnsonii | ||||
| Lb. plantarum | ||||
| Lb. sakei | ||||
| Lactococcus spp. | ||||
| L. citreum | ||||
| P. pentosaceus | ||||
| A. viridans | [76] | |||
| Lb. curvatus | [76] | |||
| Lb. fermentum | Lb. fermentum | |||
| Lb. plantarum | ||||
| Lc. lactis | ||||
| L. citreum | ||||
| P. pentosaceus | ||||
| W. cibaria | ||||
| A. viridans | [62] | |||
| Ec. casseliflavus | ||||
| Ec. durans | ||||
| Ec. faecalis | Ec. cecorum | [79] | ||
| Ec. faecium | Ec. faecium | |||
| Ec. mundtii | ||||
| Lb. coryniformis | Lb. crustorum | |||
| Lb. graminis | Lb. delbrueckii | |||
| Lb. plantarum | ||||
| Lb. sanfranciscensis | ||||
| Lc. garvieae | Lc. garvieae | |||
| P. pentosaceus | ||||
| Lb. brevis | [75] | |||
| Lb. plantarum | ||||
| Lb. acidophilus | [69] | |||
| Lb. brevis | ||||
| Lb. casei | ||||
| Lb. crispatus | ||||
| Lb. delbrueckii | ||||
| Lb. fructivorans | ||||
| Lb. kefirgranum | ||||
| Lb. paracasei | ||||
| Lb. pentosus | ||||
| Lb. plantarum | ||||
| Lb. rhamnosus | ||||
| Lb. sakei | ||||
| Lb. brevis | [81] | |||
| Lb. curvatus | ||||
| Lb. paralimentarius | ||||
| Lb. plantarum | ||||
| Lb. sakei | ||||
| Lb. sanfranciscensis | ||||
| Lb. zymae | ||||
| Lc. lactis | ||||
| L. citreum | ||||
| L. mesenteroides | ||||
| Lb. brevis | [82] | |||
| Lb. sanfranciscensis | ||||
| P. pentosaceus | ||||
| W. cibaria | ||||
| W. confusa | ||||
| Rye | Lb. kimchii | [80] | ||
| Lb. paralimentarius | ||||
| Lb. sanfranciscensis | ||||
| Lb. spicheri | ||||
| Aerococcus spp. | [56] | |||
| Lb. plantarum | Lb. plantarum | [56] | ||
| Lb. sakei | Lb. sakei | |||
| Lc. lactis | Lc. lactis | |||
| P. pentosaceus | P. pentosaceus | |||
| Weissella spp. | Weissella spp. | |||
| Ec. casseliflavus | [83] | |||
| Ec. faecalis | ||||
| Ec. mundtii | ||||
| Lb. brevis | [83] | |||
| Lb. fermentum | ||||
| Lb. plantarum | ||||
| L. citreum | ||||
| P. pentosaceus | ||||
| Lb. graminis | [77] | |||
| Lb. plantarum | ||||
| Lc. lactis subsp. cremoris | ||||
| L. citreum | ||||
| W. cibaria | ||||
| W. confusa | ||||
| Lb. brevis | [84] | |||
| Lb. fermentum | ||||
| Lb. amylovorus | [85] | |||
| Lb. panis | ||||
| Lb. reuteri | ||||
| Spelt | Ec. mundtii | [76] | ||
| Lb. brevis | [76] | |||
| Lb. curvatus | ||||
| Lb. paraplantarum | ||||
| Lb. plantarum | ||||
| Lb. rossiae | ||||
| L. pseudomesenteroides | L. citreum | |||
| P. pentosaceus | ||||
| Corn | Lb. curvatus | [62] | Lb. acidophilus | [86] |
| Lb. brevis | ||||
| Lb. fermentum | ||||
| Lb. plantarum | ||||
| L. mesenteroides | ||||
| L. dextranicum | ||||
| P. acidilactici | ||||
| Ec. saccharolyticus | [87] | |||
| Lb. casei | ||||
| Lb. delbrueckii | ||||
| Lb. fermentum | ||||
| Lb. plantarum | ||||
| Strep. bovis | ||||
| Rice | Ec. faecium | [62] | ||
| Lb. brevis | [88] | |||
| Lb. crustorum | ||||
| Lb. harbinensis | ||||
| Lb. pentosus | ||||
| Lb. plantarum | ||||
| L. pseudomesenteroides | ||||
| Sorghum | P. pentosaceus | [89] | ||
| P. pentosaceus | [59] | |||
| W. confusa | ||||
| Pseudocereals | ||||
| Quinoa | Ec. faecium | [62] | ||
| Ec. mundtii | ||||
| Lc. garvieae | ||||
| Amaranth | Ec. faecium | [62] | ||
| P. pentosaceus | ||||
| Non-conventional ingredients | ||||
| Faba bean | Lb. sakei | [90] | ||
| Lc. lactis | ||||
| L. mesenteroides | ||||
| P. pentosaceus | ||||
| W. cibaria | ||||
| W. koreensis | ||||
| Chickpea | Ec. faecium | [62] | ||
| Lb. graminis | ||||
| Composite ingredients | ||||
| Wheat | Lb. brevis | [78] | ||
| Chickpea | Lb. coryneformis | |||
| Lentil | Lb. fermentum | |||
| Bean | Lb. parabuchneri | |||
| Lb. paraplantarum | ||||
| Lb. plantarum | ||||
| Lb. pentosus | ||||
| Lb. rossiae | ||||
| Lb. sanfranciscensis | ||||
| L. mesenteroides | ||||
| W. cibaria | ||||
| Apple | Lb. plantarum | [91] | ||
| Honey | Lb. sakei | |||
| Wheat | P. pentosaceus | |||
| Yogurt | P. pentosaceus | [91] | ||
| Wheat | ||||
| White grape | Lb. brevis | [91] | ||
| Wheat | Lb. plantarum | |||
| Lb. sakei | ||||
| P. pentosaceus | ||||
| W. cibaria | ||||
| Brazilian grape | Lb. brevis | [92] | ||
| Wheat | Lb. casei | |||
| Rye | Lb. delbrueckii | |||
| Lb. paracasei | ||||
| Lb. rhamnosus | ||||
| Flour | Dough (Prior to Fermentation) | Sourdough (After Fermentation) | ||
|---|---|---|---|---|
| Yeast | References | Yeast | References | |
| Cereals | ||||
| Wheat | C. krusei | [14] | ||
| P. anomala | ||||
| S. cerevisiae | ||||
| S. exiguus | ||||
| C. humilis | [56] | |||
| K. barnettii | ||||
| S. bayanus | [56] | S. bayanus | ||
| S. cerevisiae | S. cerevisiae | |||
| W. anomalus | ||||
| C. humilis | [64] | |||
| K. barnettii | ||||
| R. glacialis | ||||
| S. bayanus | ||||
| S. cerevisiae | ||||
| C. humilis | [74] | |||
| K. unispora | ||||
| S. cerevisiae | ||||
| S. uvarum | ||||
| T. delbrueckii | ||||
| C. humilis | [79] | |||
| C. tropicalis | ||||
| Cyberlindnera jadinii | ||||
| S. cerevisiae | ||||
| T. delbrueckii | ||||
| W. anomalus | ||||
| S. cerevisiae | [95] | |||
| S. exiguus | ||||
| C. humilis | [81] | |||
| S. cerevisiae | ||||
| P. fermentans | ||||
| P. membranifaciens | ||||
| W. anomalus | ||||
| Rye | C. humilis | [56] | ||
| Cryptococcus sp. | [56] | |||
| K. barnettii | ||||
| S. bayanus | S. bayanus | |||
| S. cerevisiae | S. cerevisiae | |||
| W. anomalus | ||||
| C. humilis | [74] | |||
| P. fermentans | ||||
| S. cerevisiae | ||||
| S. cerevisiae | [84] | |||
| S. cerevisiae | [85] | |||
| Sorghum | S. cerevisiae | [59] | ||
| S. cerevisiae | [89] | |||
| Corn | S. cerevisiae | [86] | ||
| Rice | S. cerevisiae | [88] | ||
| Composite ingredients | ||||
| Brazilian grape | C. famata | [92] | ||
| Wheat | C. guilliermondii | |||
| Rye | C. pelliculosa | |||
| C. sphaerica | ||||
| S. cerevisiae | ||||
| Wheat | P. kudriavzevii | [96] | ||
| Lemon juice | P. myanmarensis | |||
| S. cariocanus | ||||
| Wheat | Candida spp. | [96] | ||
| Apple juice | P. myanmarensis | |||
| S. cariocanus | ||||
| Obligately Homofermentative | Obligately Heterofermentative | Facultatively Heterofermentative |
|---|---|---|
| Lb. acidophilus | Lb. acidifarinae | Lb. alimentarius |
| Lb. amylolyticus | Lb. brevis | Lb. casei |
| Lb. amylovorus | Lb. buchneri | Lb. curvatus |
| Lb. crispatus | Lb. cellobiosus | Lb. paralimentarius |
| Lb. delbrueckii subsp. bulgaricus | Lb. fermentum | Lb. plantarum |
| Lb. delbrueckii subsp. delbrueckii | Lb. fructivorans | Lb. pentosus |
| Lb. farciminis | Lb. frumenti | Lb. rhamnosus |
| Lb. helviticus | Lb. hilgardii | |
| Lb. johnsonii | Lb. panis | |
| Lb. lactis | Lb. pontis | |
| Lb. mindensis | Lb. reuteri | |
| Lb. rossiae | ||
| Lb. sanfranciscensis | ||
| Lb. siliginis | ||
| Lb. spicheri | ||
| Lb. viridescens | ||
| Lb. zymae |
| Biochemical Reactions | Obligately Homofermentative | Obligately Heterofermentative | Facultatively Heterofermentative | |
|---|---|---|---|---|
| Pentose fermentation | - 1 | + 2 | + | |
| CO2 released from | glucose | - | + | - |
| gluconate | - | + | + | |
| Carbohydrate digestion involving fructose-1,6-diphosphate aldolase | + | - | + | |
| Carbohydrate digestion involving phosphoketolase | - | + | + | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lau, S.W.; Chong, A.Q.; Chin, N.L.; Talib, R.A.; Basha, R.K. Sourdough Microbiome Comparison and Benefits. Microorganisms 2021, 9, 1355. https://doi.org/10.3390/microorganisms9071355
Lau SW, Chong AQ, Chin NL, Talib RA, Basha RK. Sourdough Microbiome Comparison and Benefits. Microorganisms. 2021; 9(7):1355. https://doi.org/10.3390/microorganisms9071355
Chicago/Turabian StyleLau, Siew Wen, Ann Qi Chong, Nyuk Ling Chin, Rosnita A. Talib, and Roseliza Kadir Basha. 2021. "Sourdough Microbiome Comparison and Benefits" Microorganisms 9, no. 7: 1355. https://doi.org/10.3390/microorganisms9071355
APA StyleLau, S. W., Chong, A. Q., Chin, N. L., Talib, R. A., & Basha, R. K. (2021). Sourdough Microbiome Comparison and Benefits. Microorganisms, 9(7), 1355. https://doi.org/10.3390/microorganisms9071355






