Challenging Cosmetic Innovation: The Skin Microbiota and Probiotics Protect the Skin from UV-Induced Damage
Abstract
1. Introduction
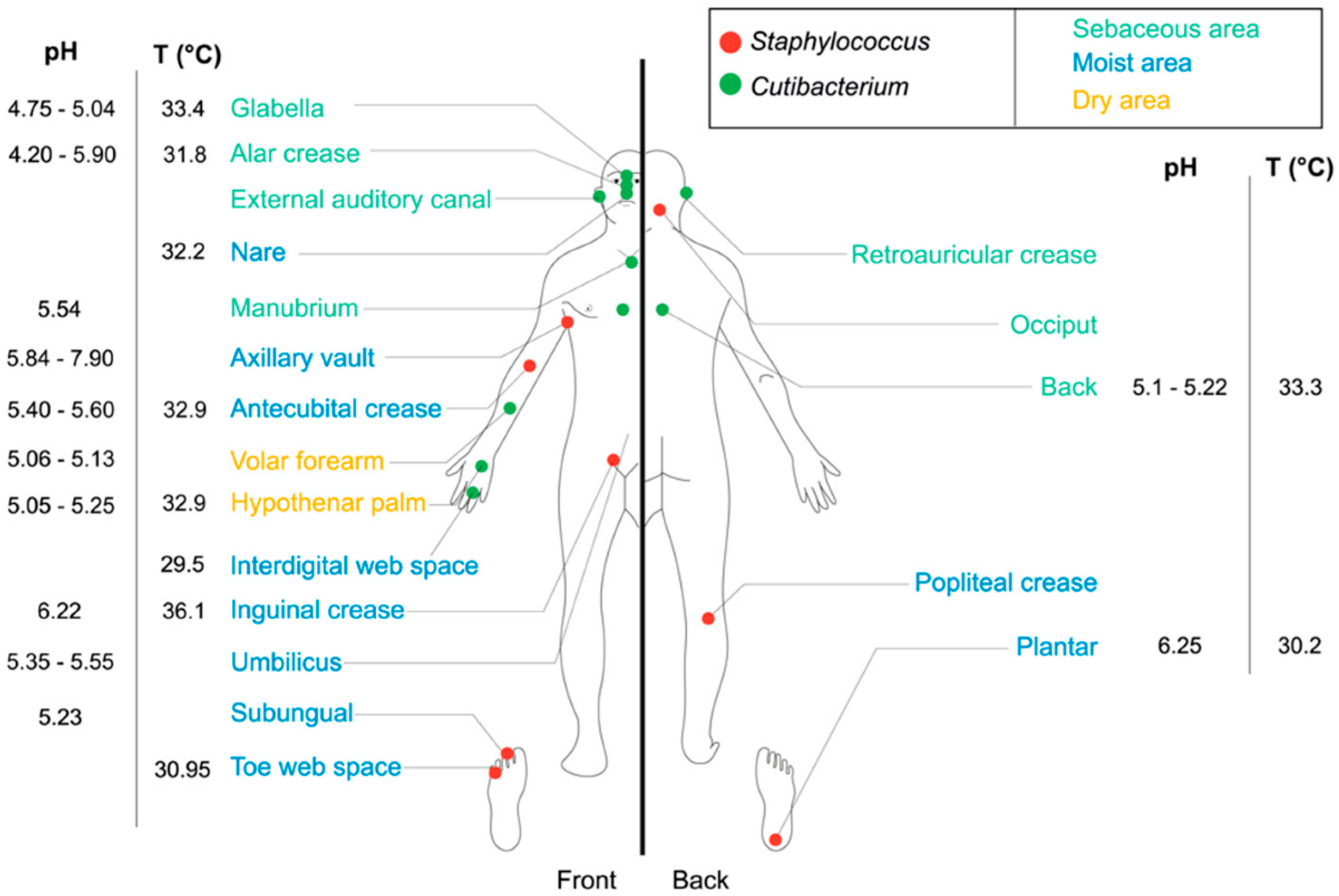
2. The Human Skin Microbiota
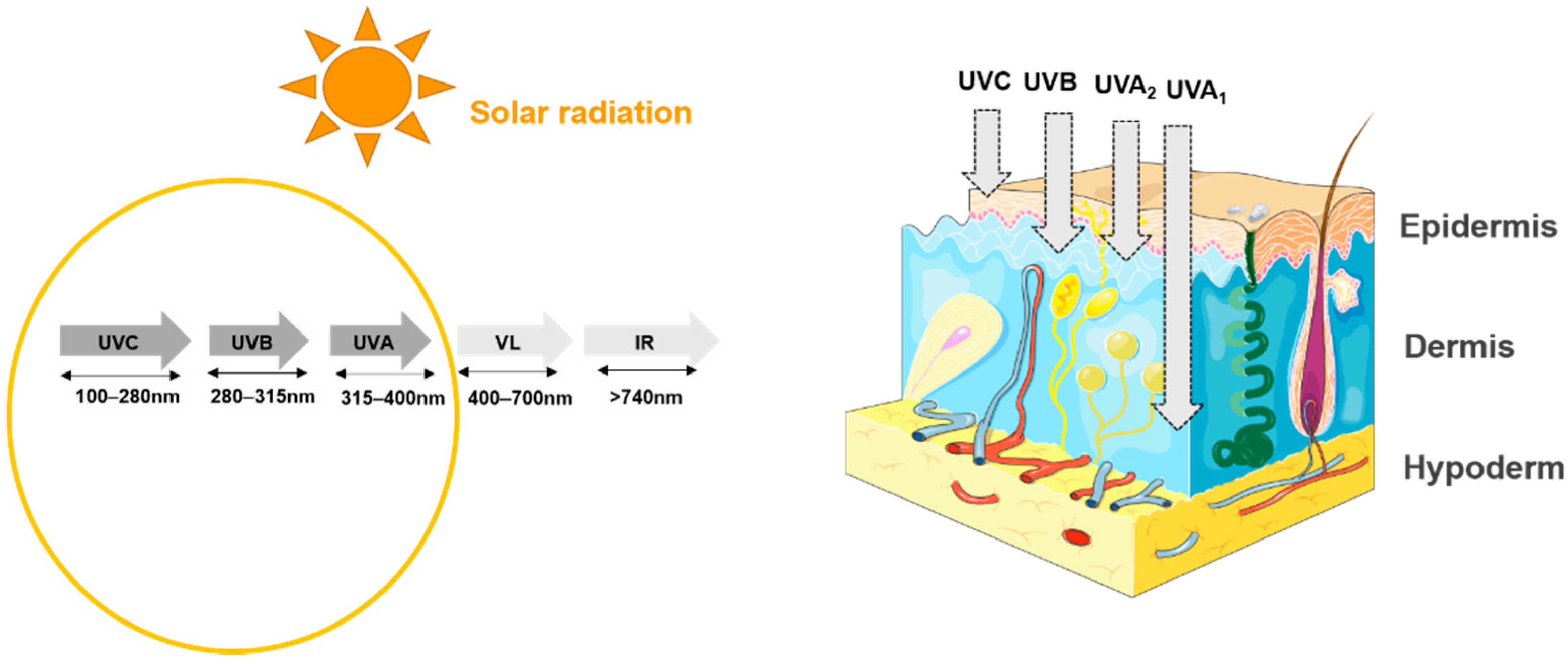
3. Impact of UVs on the Skin and Its Microbiota
3.1. Effect of UVs on the Skin
3.2. Effect of UVs on the Cutaneous Microbiota
4. Photoprotection
4.1. Classical Sun Protection
4.2. Bacterial-Based Cosmetic Approaches to Preventing UV-Induced Damages
4.3. Bacterial-Based Nutritional Approaches to Prevent UV-Induced Damages
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ngoc, L.T.N.; Van Tran, V.; Moon, J.-Y.; Chae, M.; Park, D.; Lee, Y.-C. Recent trends of sunscreen cosmetic: An update review. Cosmetics 2019, 6, 64. [Google Scholar] [CrossRef]
- Puebla-Barragan, S.; Reid, G. Probiotics in cosmetic and personal care products: Trends and challenges. Molecules 2021, 26, 1249. [Google Scholar] [CrossRef]
- Tsilingiri, K.; Barbosa, T.; Penna, G.; Caprioli, F.; Sonzogni, A.; Viale, G.; Rescigno, M. Probiotic and postbiotic activity in health and disease: Comparison on a novel polarised ex-vivo organ culture model. Gut 2012, 61, 1007–1015. [Google Scholar] [CrossRef] [PubMed]
- Grice, E.A.; Kong, H.H.; Renaud, G.; Young, A.C.; Bouffard, G.G.; Blakesley, R.W.; Wolfsberg, T.G.; Turner, M.L.; Segre, J.A.; Program, N.C.S. A diversity profile of the human skin microbiota. Genome Res. 2008, 18, 1043–1050. [Google Scholar] [CrossRef] [PubMed]
- Ngo, M.A.; Sinitsyna, N.N.; Qin, Q.; Rice, R.H. Oxygen-Dependent Differentiation of Human Keratinocytes. J. Investig. Dermatol. 2007, 127, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Grice, E.A.; Segre, J.A. The skin microbiome. Nat. Rev. Microbiol. 2011, 9, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Kong, H.H.; Segre, J.A. Skin microbiome: Looking back to move forward. J. Investig. Dermatol. 2012, 132, 933–939. [Google Scholar] [CrossRef]
- Capone, K.A.; Dowd, S.E.; Stamatas, G.N.; Nikolovski, J. Diversity of the human skin microbiome early in life. J. Investig. Dermatol. 2011, 131, 2026–2032. [Google Scholar] [CrossRef]
- Grice, E.A.; Kong, H.H.; Conlan, S.; Deming, C.B.; Davis, J.; Young, A.C.; Bouffard, G.G.; Blakesley, R.W.; Murray, P.R.; Green, E.D.; et al. Topographical and temporal diversity of the human skin microbiome. Science 2009, 324, 1190–1192. [Google Scholar] [CrossRef]
- Racine, P.; Janvier, X.; Clabaut, M.; Catovic, C.; Souak, D.; Boukerb, A.M.; Groboillot, A.; Konto-Ghiorghi, Y.; Duclairoir-Poc, C.; Lesouhaitier, O.; et al. Dialog between skin and its microbiota: Emergence of “Cutaneous Bacterial Endocrinology”. Exp. Dermatol. 2020, 29, 790–800. [Google Scholar] [CrossRef]
- Patra, V.; Byrne, S.N.; Wolf, P. The skin microbiome: Is it affected by UV-induced immune suppression? Front. Microbiol. 2016, 7, 1235. [Google Scholar] [CrossRef]
- Takahashi, T.; Gallo, R.L. The critical and multifunctional roles of antimicrobial peptides in dermatology. Dermatol. Clin. 2017, 35, 39–50. [Google Scholar] [CrossRef] [PubMed]
- Sanford, J.A.; Gallo, R.L. Functions of the skin microbiota in health and disease. Semin. Immunol. 2013, 25, 370–377. [Google Scholar] [CrossRef] [PubMed]
- Alekseyenko, A.V.; Perez-Perez, I.G.; De Souza, A.; Strober, B.; Gao, Z.; Bihan, M.; Li, K.; Methé, A.B.; Blaser, M.J. Community differentiation of the cutaneous microbiota in psoriasis. Microbiome 2013, 1, 31. [Google Scholar] [CrossRef] [PubMed]
- Musthaq, S.; Mazuy, A.; Jakus, J. The microbiome in dermatology. Clin. Dermatol. 2018, 36, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Paller, A.S.; Kong, H.H.; Seed, P.; Naik, S.; Scharschmidt, T.C.; Gallo, R.L.; Luger, T.; Irvine, A.D. The microbiome in patients with atopic dermatitis. J. Allergy Clin. Immunol. 2019, 143, 26–35. [Google Scholar] [CrossRef]
- Nakatsuji, T.; Gallo, R.L. The role of the skin microbiome in atopic dermatitis. Ann. Allergy Asthma Immunol. 2019, 122, 263–269. [Google Scholar] [CrossRef]
- Williams, M.R.; Nakatsuji, T.; Gallo, R.L. Staphylococcus aureus: Master Manipulator of the Skin. Cell Host Microbe 2017, 22, 579–581. [Google Scholar] [CrossRef]
- Schommer, N.N.; Gallo, R.L. Structure and function of the human skin microbiome. Trends Microbiol. 2013, 21, 660–668. [Google Scholar] [CrossRef]
- Burns, E.M.; Ahmed, H.; Isedeh, P.N.; Kohli, I.; Van Der Pol, W.; Shaheen, A.; Muzaffar, A.F.; Al-Sadek, C.; Foy, T.M.; Abdelgawwad, M.S.; et al. Ultraviolet radiation, both UVA and UVB, influences the composition of the skin microbiome. Exp. Dermatol. 2019, 28, 136–141. [Google Scholar] [CrossRef]
- Zhai, W.; Huang, Y.; Zhang, X.; Fei, W.; Chang, Y.; Cheng, S.; Zhou, Y.; Gao, J.; Tang, X.; Zhang, X.; et al. Profile of the skin microbiota in a healthy Chinese population. J. Dermatol. 2018, 45, 1289–1300. [Google Scholar] [CrossRef]
- Fournière, M.; Latire, T.; Souak, D.; Feuilloley, M.; Bedoux, G. Staphylococcus epidermidis and Cutibacterium acnes: Two major sentinels of skin microbiota and the influence of cosmetics. Microorganisms 2020, 8, 1752. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, K.P.; Long, P.F.; Young, A.R. Mycosporine-like amino acids for skin photoprotection. Curr. Med. Chem. 2019, 25, 5512–5527. [Google Scholar] [CrossRef] [PubMed]
- Byrd, A.L.; Belkaid, Y.; Segre, J.A. The human skin microbiome. Nat. Rev. Microbiol. 2018, 16, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Percival, S.L.; Emanuel, C.; Cutting, K.F.; Williams, D.W. Microbiology of the skin and the role of biofilms in infection. Int. Wound J. 2012, 9, 14–32. [Google Scholar] [CrossRef] [PubMed]
- Feuilloley, M.G.J. Antidromic neurogenic activity and cutaneous bacterial flora. Semin. Immunopathol. 2018, 40, 281–289. [Google Scholar] [CrossRef]
- Lange-Asschenfeldt, B.; Marenbach, D.; Lang, C.; Patzelt, A.; Ulrich, M.; Maltusch, A.; Terhorst, D.; Stockfleth, E.; Sterry, W.; Lademann, J. Distribution of bacteria in the epidermal layers and hair follicles of the human skin. Ski. Pharmacol. Physiol. 2011, 24, 305–311. [Google Scholar] [CrossRef]
- Findley, K.; Oh, J.; Yang, J.; Conlan, S.; Deming, C.; Meyer, J.A.; Schoenfeld, D.; Nomicos, E.; Park, M.; Kong, H.H.; et al. Topographic diversity of fungal and bacterial communities in human skin. Nature 2013, 498, 367–370. [Google Scholar] [CrossRef]
- Probst, A.J.; Auerbach, A.K.; Moissl-Eichinger, C. Archaea on human skin. PLoS ONE 2013, 8, e65388. [Google Scholar] [CrossRef]
- Moissl-Eichinger, C.; Pausan, M.; Taffner, J.; Berg, G.; Bang, C.; Schmitz, R.A. Archaea are interactive components of complex microbiomes. Trends Microbiol. 2018, 26, 70–85. [Google Scholar] [CrossRef]
- Haynes, M.; Rohwer, F. The human virome. In Metagenomics of the Human Body; Springer Science and Business: Cham, Switzerland, 2011; pp. 63–77. [Google Scholar]
- Hannigan, G.D.; Meisel, J.S.; Tyldsley, A.S.; Zheng, Q.; Hodkinson, B.P.; SanMiguel, A.J.; Minot, S.; Bushman, F.D.; Grice, E.A. The human skin double-stranded DNA virome: Topographical and temporal diversity, genetic enrichment, and dynamic associations with the host microbiome. mBio 2015, 6, e01578-15. [Google Scholar] [CrossRef]
- Foulongne, V.; Sauvage, V.; Hebert, C.; Dereure, O.; Cheval, J.; Gouilh, M.A.; Pariente, K.; Segondy, M.; Burguière, A.; Manuguerra, J.-C.; et al. Human skin microbiota: High diversity of DNA viruses identified on the human skin by high throughput sequencing. PLoS ONE 2012, 7, e38499. [Google Scholar] [CrossRef]
- Wylie, K.M.; Mihindukulasuriya, K.A.; Zhou, Y.; Sodergren, E.; Storch, A.G.; Weinstock, G.M. Metagenomic analysis of double-stranded DNA viruses in healthy adults. BMC Biol. 2014, 12, 71. [Google Scholar] [CrossRef]
- Krutmann, J.; Bouloc, A.; Sore, G.; Bernard, B.A.; Passeron, T. The skin aging exposome. J. Dermatol. Sci. 2017, 85, 152–161. [Google Scholar] [CrossRef]
- Krutmann, J. Phototherapy for atopic dermatitis. Clin. Exp. Dermatol. 2000, 25, 552–558. [Google Scholar] [CrossRef]
- Skotarczak, K.; Osmola-Mańkowska, A.; Lodyga, M.; Polańska, A.; Mazur, M.; Adamski, Z. Photoprotection: Facts and controversies. Eur. Rev. Med Pharmacol. Sci. 2015, 19, 98–112. [Google Scholar] [PubMed]
- Servier. Medical Art Skin. Available online: https://Smart.Servier.Com/Smart_image/Skin/ (accessed on 22 March 2021).
- IARC. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; IARC: Lyon, France, 2012; Volume 100D, ISBN 978-92-832-1321-5. [Google Scholar]
- Fondriest Environmental, Inc. Solar Radiation and Photosynethically Active Radiation. In Fundamentals of Environmental Measurements; Fondriest: Fairborn, OH, USA, 2014. [Google Scholar]
- D’Orazio, J.; Jarrett, S.; Amaro-Ortiz, A.; Scott, T. UV radiation and the skin. Int. J. Mol. Sci. 2013, 14, 12222–12248. [Google Scholar] [CrossRef] [PubMed]
- Juzeniene, A.; Brekke, P.; Dahlback, A.; Andersson-Engels, S.; Reichrath, J.; Moan, K.; Holick, M.F.; Grant, W.B.; Moan, J. Solar radiation and human health. Rep. Prog. Phys. 2011, 74, 066701. [Google Scholar] [CrossRef]
- Pérez-Sánchez, A.; Barrajón-Catalán, E.; Herranz-López, M.; Micol, V. Nutraceuticals for skin care: A comprehensive review of human clinical studies. Nutrients 2018, 10, 403. [Google Scholar] [CrossRef] [PubMed]
- Bernard, J.J.; Gallo, R.L.; Krutmann, J. Photoimmunology: How ultraviolet radiation affects the immune system. Nat. Rev. Immunol. 2019, 19, 688–701. [Google Scholar] [CrossRef] [PubMed]
- Patra, V.; Laoubi, L.; Nicolas, J.-F.; Vocanson, M.; Wolf, P. A Perspective on the interplay of ultraviolet-radiation, skin microbiome and skin resident memory TCRαβ+ Cells. Front. Med. 2018, 5, 166. [Google Scholar] [CrossRef]
- Wang, Y.; Zhu, W.; Shu, M.; Jiang, Y.; Gallo, R.L.; Liu, Y.-T.; Huang, C.-M. The response of human skin commensal bacteria as a reflection of UV radiation: UV-B decreases porphyrin production. PLoS ONE 2012, 7, e47798. [Google Scholar] [CrossRef]
- Hug, D.H.; Dunkerson, D.D.; Hunter, J.K. The degradation of L-histidine and tram- and cis-urocanic acid by bacteria from skin and the role of bacterial cis-urocanic acid isomerase. J. Photochem. Photobiol. B 1999, 8, 66–73. [Google Scholar] [CrossRef]
- Kullavanijaya, P.; Lim, H.W. Photoprotection. J. Am. Acad. Dermatol. 2005, 52, 937–958. [Google Scholar] [CrossRef] [PubMed]
- Rai, R.; Shanmuga, S.C.; Srinivas, C.R. Update on photoprotection. Indian J. Dermatol. 2012, 57, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Gabros, S.; Nessel, T.A.; Zito, P.M. Sunscreens and photoprotection. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. [Google Scholar]
- Beani, J. La photoprotection. Rev. Fr. d’Allergol. d’Immunol. Clin. 1999, 39, 311–323. [Google Scholar] [CrossRef]
- Al-Jamal, M.S.; Griffith, J.L.; Lim, H.W. Photoprotection in ethnic skin. Dermatol. Sin. 2014, 32, 217–224. [Google Scholar] [CrossRef]
- Beani, J.-C. Produits de protection solaire: Efficacité et risques. Ann. Dermatol. Vénéréologie 2012, 139, 261–272. [Google Scholar] [CrossRef] [PubMed]
- Saewan, N.; Jimtaisong, A. Natural products as photoprotection. J. Cosmet. Dermatol. 2015, 14, 47–63. [Google Scholar] [CrossRef] [PubMed]
- Soeur, J.; Belaïdi, J.-P.; Chollet, C.; Denat, L.; Dimitrov, A.; Jones, C.; Perez, P.; Zanini, M.; Zobiri, O.; Mezzache, S.; et al. Photo-pollution stress in skin: Traces of pollutants (PAH and particulate matter) impair redox homeostasis in keratinocytes exposed to UVA1. J. Dermatol. Sci. 2017, 86, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Grether-Beck, S.; Marini, A.; Jaenicke, T.; Krutmann, J. Photoprotection of human skin beyond ultraviolet radiation. Photodermatol. Photoimmunol. Photomed. 2014, 30, 167–174. [Google Scholar] [CrossRef]
- Jain, S.; Prajapat, G.; Abrar, M.; Ledwani, L.; Singh, A.; Agrawal, A. Cyanobacteria as efficient producers of mycosporine-like amino acids. J. Basic Microbiol. 2017, 57, 715–727. [Google Scholar] [CrossRef]
- Colabella, F.; Moline, M.; Libkind, D. UV sunscreens of microbial origin: Mycosporines and mycosporine- like aminoacids. Recent Patents Biotechnol. 2015, 8, 179–193. [Google Scholar] [CrossRef]
- Chrapusta, E.; Kaminski, A.; Duchnik, K.; Bober, B.; Adamski, M.; Bialczyk, J. Mycosporine-like amino acids: Potential health and beauty ingredients. Mar. Drugs 2017, 15, 326. [Google Scholar] [CrossRef]
- Yokoyama, H.; Mizutani, R. Structural biology of DNA (6-4) photoproducts formed by ultraviolet radiation and interactions with their binding proteins. Int. J. Mol. Sci. 2014, 15, 20321–20338. [Google Scholar] [CrossRef] [PubMed]
- Kageyama, H.; Waditee-Sirisattha, R. Antioxidative, anti-inflammatory, and anti-aging properties of mycosporine-like amino acids: Molecular and cellular mechanisms in the protection of skin-aging. Mar. Drugs 2019, 17, 222. [Google Scholar] [CrossRef] [PubMed]
- Sinha, R.P.; Häder, D.-P. UV-protectants in cyanobacteria. Plant Sci. 2008, 174, 278–289. [Google Scholar] [CrossRef]
- Nakatsuji, T.; Chen, T.H.; Butcher, A.M.; Trzoss, L.L.; Nam, S.-J.; Shirakawa, K.T.; Zhou, W.; Oh, J.; Otto, M.; Fenical, W.; et al. A commensal strain of Staphylococcus epidermidis protects against skin neoplasia. Sci. Adv. 2018, 4, eaao4502. [Google Scholar] [CrossRef]
- Bhatia, N.; Berman, B.; Ceilley, I.R.; Kircik, L.H. Understanding the role of photolyases: Photoprotection and beyond. J. Drugs Dermatol. 2017, 16, 61–66. [Google Scholar]
- Stege, H.; Roza, L.; Vink, A.A.; Grewe, M.; Ruzicka, T.; Grether-Beck, S.; Krutmann, J. Enzyme plus light therapy to repair DNA damage in ultraviolet-B-irradiated human skin. Proc. Natl. Acad. Sci. USA 2000, 97, 1790–1795. [Google Scholar] [CrossRef] [PubMed]
- Sondenheimer, K.; Krutmann, J. Novel means for photoprotection. Front. Med. 2018, 5, 162. [Google Scholar] [CrossRef]
- Allhorn, M.; Arve, S.; Brüggemann, H.; Lood, R. A novel enzyme with antioxidant capacity produced by the ubiquitous skin colonizer Propionibacterium acnes. Sci. Rep. 2016, 6, 36412. [Google Scholar] [CrossRef]
- Andersson, T.; Bergdahl, G.E.; Saleh, K.; Magnúsdóttir, H.; Stødkilde, K.; Andersen, C.B.F.; Lundqvist, K.; Jensen, A.; Brüggemann, H.; Lood, R. Common skin bacteria protect their host from oxidative stress through secreted antioxidant RoxP. Sci. Rep. 2019, 9, 3596. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Suárez, J.; Coy-Barrera, E.; Villamil, L.; Díaz, L. Streptomyces-derived metabolites with potential photoprotective properties—A systematic literature review and meta-analysis on the reported chemodiversity. Molecules 2020, 25, 3221. [Google Scholar] [CrossRef] [PubMed]
- Morifuji, M. The beneficial role of functional food components in mitigating ultraviolet-induced skin damage. Exp. Dermatol. 2019, 28, 28–31. [Google Scholar] [CrossRef] [PubMed]
- Krutmann, J. Pre- and probiotics for human skin. J. Dermatol. Sci. 2009, 54, 1–5. [Google Scholar] [CrossRef]
- Guéniche, A.; David, P.; Philippe, B.; Stephanie, B.; Elif, B.; Isabelle, C.-H. Probiotics for photoprotection. Dermato-Endocrinology 2009, 1, 275–279. [Google Scholar] [CrossRef]
- Ouwehand, A.; Båtsman, A.; Salminen, S. Probiotics for the skin: A new area of potential application? Lett. Appl. Microbiol. 2003, 36, 327–331. [Google Scholar] [CrossRef]
- Friedrich, A.D.; Campo, V.E.; Cela, E.M.; Morelli, A.E.; Shufesky, W.J.; Tckacheva, O.A.; Leoni, J.; Paz, M.L.; Larregina, A.T.; Maglio, D.H.G. Oral administration of lipoteichoic acid from Lactobacillus rhamnosus GG overcomes UVB-induced immunosuppression and impairs skin tumor growth in mice. Eur. J. Immunol. 2019, 49, 2095–2102. [Google Scholar] [CrossRef]
- Patra, V.; Sérézal, I.G.; Wolf, P. Potential of skin microbiome, pro- and/or pre-biotics to affect local cutaneous responses to UV exposure. Nutrients 2020, 12, 1795. [Google Scholar] [CrossRef]
- Nole, K.L.B.; Yim, E.; Keri, J.E. Probiotics and prebiotics in dermatology. J. Am. Acad. Dermatol. 2014, 71, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Suh, M.G.; Bae, G.Y.; Jo, K.; Kim, J.M.; Hong, K.-B.; Suh, H.J. Photoprotective effect of dietary Galacto-Oligosaccharide (GOS) in hairless mice via regulation of the MAPK signaling pathway. Molecules 2020, 25, 1679. [Google Scholar] [CrossRef]
- Kim, H.M.; Lee, N.E.; Park, S.D.; Kim, Y.-T.; Kim, Y.J.; Jeong, J.W.; Jang, S.S.; Ahn, Y.-T.; Sim, J.-H.; Huh, C.-S.; et al. Oral administration of Lactobacillus plantarum HY7714 protects hairless mouse against ultraviolet B-induced photoaging. J. Microbiol. Biotechnol. 2014, 24, 1583–1591. [Google Scholar] [CrossRef] [PubMed]
- Verschooten, L.; Claerhout, S.; Van Laethem, A.; Agostinis, P.; Garmyn, M. New strategies of photoprotection. Photochem. Photobiol. 2006, 82, 1016–1023. [Google Scholar] [CrossRef] [PubMed]
| Cosmetic Ingredient | Nom I.N.C.I. | Mechanism of Action |
|---|---|---|
| Inoveol® CAFA (Sederma) | Thermus Thermophilus Ferment (and) Glycerin | Protection against UV and Infrared (IR). Anti-aging protection. |
| Sun’Alg® (Biosil Technologies Inc.) | Pongamia glabra seed oil (and) Dunaliella salina extract (and) Haematococcus pluvialis extract | Shield formation against UVA and UVB radiation (natural sunscreen thanks to its absorption). Two microalgal extracts: additional protective capacity against the oxidative stress (mixed carotenoids composition). |
| Arganyl® (BASF Beauty Care Solutions) | Argania Spinosa Leaf Extract (and) Maltodextrin | Anti-oxidative efficacy by inhibiting Reactive oxygen species production lipid peroxidation induced by pollutants (PM2.5, B[a]P) and UVA. |
| ChronogenTM (Ashland) | Water (aqua) (and) glycerin (and) hydrolyzed yeast protein | Repair of UV damage. Decrease in induced sunburned cells. |
| Ciste’M® (BASF Beauty Care Solutions) | Maltodextrin (and) Cistus Monspeliensis Flower/Leaf/Stem Extract | Protection against the deleterious effects of UVB, UVA radiation and benzo[a]pyrene. Anti-inflammatory. Antioxidants. DNA protective properties. |
| HelioguardTM365 (Mibelle AG Biochemistry) | Aqua/Water (and) Lecithin (and) Alcohol (and) Sodium Lactate (and) Porphyra Umbilicalis Extract (and) Phenoxyethanol | UVA bioprotector (UVA induced skin alterations). Prevents premature photoaging. Limits damage to DNA and lipids. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Souak, D.; Barreau, M.; Courtois, A.; André, V.; Duclairoir Poc, C.; Feuilloley, M.G.J.; Gault, M. Challenging Cosmetic Innovation: The Skin Microbiota and Probiotics Protect the Skin from UV-Induced Damage. Microorganisms 2021, 9, 936. https://doi.org/10.3390/microorganisms9050936
Souak D, Barreau M, Courtois A, André V, Duclairoir Poc C, Feuilloley MGJ, Gault M. Challenging Cosmetic Innovation: The Skin Microbiota and Probiotics Protect the Skin from UV-Induced Damage. Microorganisms. 2021; 9(5):936. https://doi.org/10.3390/microorganisms9050936
Chicago/Turabian StyleSouak, Djouhar, Magalie Barreau, Aurélie Courtois, Valérie André, Cécile Duclairoir Poc, Marc G. J. Feuilloley, and Manon Gault. 2021. "Challenging Cosmetic Innovation: The Skin Microbiota and Probiotics Protect the Skin from UV-Induced Damage" Microorganisms 9, no. 5: 936. https://doi.org/10.3390/microorganisms9050936
APA StyleSouak, D., Barreau, M., Courtois, A., André, V., Duclairoir Poc, C., Feuilloley, M. G. J., & Gault, M. (2021). Challenging Cosmetic Innovation: The Skin Microbiota and Probiotics Protect the Skin from UV-Induced Damage. Microorganisms, 9(5), 936. https://doi.org/10.3390/microorganisms9050936






