Biofertilizers and Biocontrol Agents for Agriculture: How to Identify and Develop New Potent Microbial Strains and Traits
Abstract
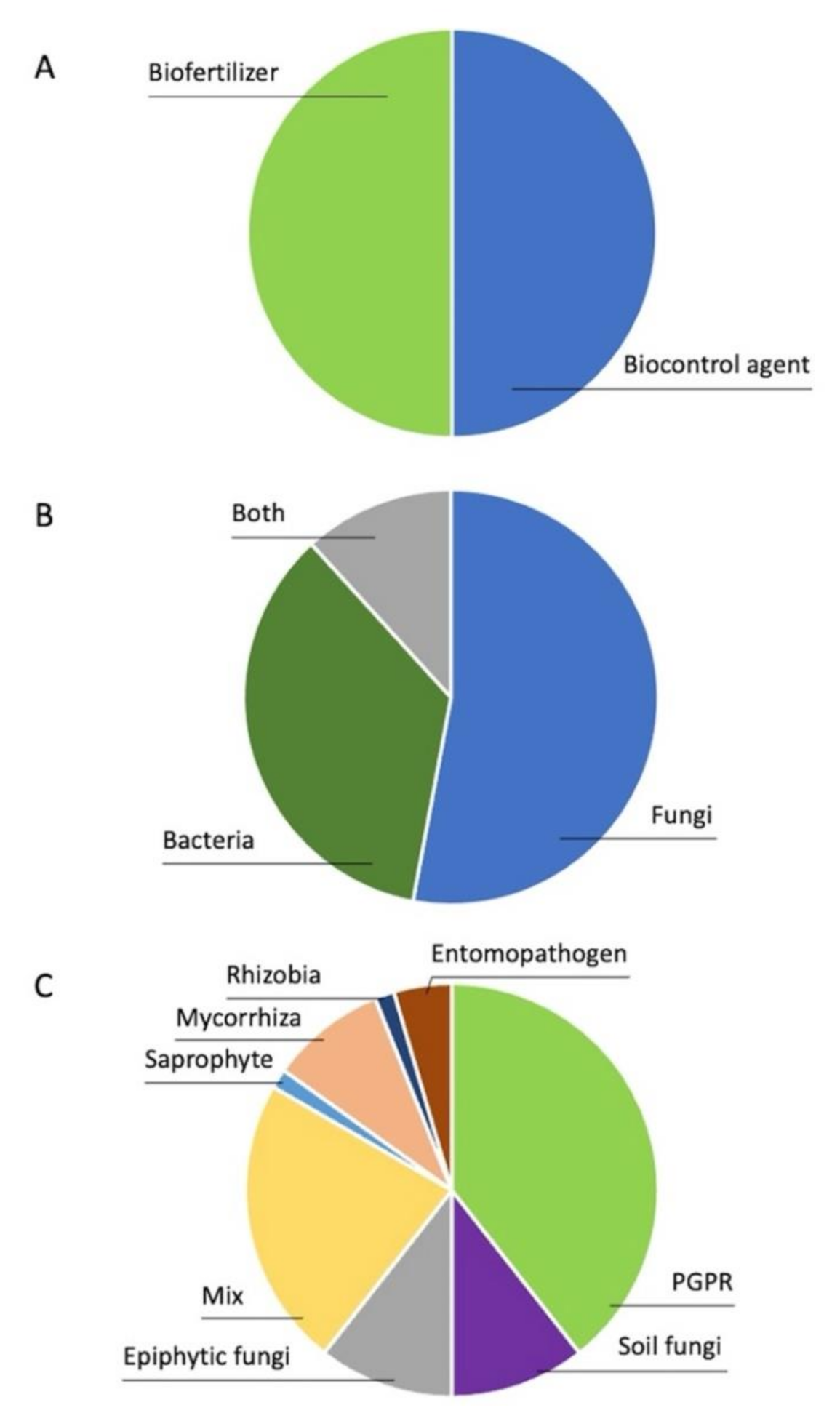
1. Introduction
2. Microorganisms Used in Biocontrol and Biofertilization
2.1. Plant-Growth-Promoting Rhizobacteria (PGPR)
2.2. Rhizobia
2.3. Mycorrhizal Fungi
2.4. Endophytic Fungi
2.5. Rhizospheric Fungi
2.6. Mycoparasitic and Entomopathogenic Fungi
3. Formulation
4. Current Trends
5. How to Identify New Potent Microbial Traits
5.1. Genome Mining
5.2. Molecular Plant–Microbe Interactions
5.3. Microbe–Microbe Interactions
5.4. Phage Interactions
6. How to Identify New Potent Microbial Strains
6.1. Modification of Microbial Genomes
6.2. Bacterial Endophytes and Endosymbionts
6.3. Microorganisms of Plants Adapted to Extreme Growth Conditions
6.4. Microorganisms of Diverse Environments
7. Conclusions and Future Prospects
- (1)
- A continuous search for new efficient microorganisms, enabling healthy plant cultivation in various field conditions, is pertinent. The potential new microbiological tools are likely to be found in plants adapted to extreme, in nutrient-poor growth conditions, in resistant plants under pathogen exposure, and in environments with high biodiversity, such as forests.
- (2)
- Studies on microbial traits beneficial for plants should be extended beyond mechanisms already known. The research for new microbial traits should cover not only plant–microbe interactions, but also microbe–microbe interactions, including phage effects and transmission.
- (3)
- The focus should be turned from the rhizosphere towards the plant holobiont, including strains occupying all plant organs, both bacteria and fungi. A high number of microbial inoculants should be combined in the biocontrol and biofertilizer products, with the aim of plant microbiome engineering.
- (4)
- For enabling the production of such complex biological products, the bottleneck of formulation needs to be solved. Therefore, more efforts should be placed on the development of technologies for microbial culture and live preservation. With the recent enormous advances in molecular and microbiological techniques, a technological jump enabling microbial live preservation in various conditions should be more than feasible.
- (5)
- A thorough analysis of the capacity of the strains to enhance plant health and growth under several growth conditions should become a routine in biofertilizer and biocontrol agent discovery. There are frequent reports in the scientific literature on new potent strains identified, but usually they are only tested in the laboratory conditions for few of the well-known microbial traits, and in one or two plant-growth conditions [144,145]. The effects of inoculants need to be tested under stressful conditions, along with an analysis of changes in the microbiome of the holobiont.
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Van Vliet, S.; Kronberg, S.L.; Provenza, F.D. Plant-based meats, human health, and climate change. Front. Sustain. Food Syst. 2020, 4, 128. [Google Scholar] [CrossRef]
- Buzek, J.; Malmström, C. Regulation (EC) No 1107/2009 of the European parliament and of the council of 21 October 2009 concerning the placing of plant protection products on the market and repealing council directives 79/117/EEC and 91/414/EEC. Off. J. Eur. Union 2009, 52, 1–50. [Google Scholar] [CrossRef]
- Rees, R.M.; Baddeley, J.A.; Bhogal, A.; Ball, B.C.; Chadwick, D.R.; Macleod, M.; Lilly, A.; Pappa, V.A.; Thorman, R.E.; Watson, C.A.; et al. Nitrous oxide mitigation in UK agriculture. Soil Sci. Plant Nutr. 2013, 59, 3–15. [Google Scholar] [CrossRef]
- Björk, P.; Kauppinen-Räisänen, H. Local food: A source for destination attraction. Int. J. Contemp. Hosp. Manag. 2016, 28, 177–194. [Google Scholar] [CrossRef]
- McGuire, S.; FAO; IFAD; WFP. The state of food insecurity in the world 2015: Meeting the 2015 international hunger targets: Taking stock of uneven progress. Rome: FAO. 2015. Adv. Nutr. 2015, 6, 623–624. [Google Scholar] [CrossRef]
- Ongley, E.D.; Xiaolan, Z.; Tao, Y. Current status of agricultural and rural non-point source Pollution assessment in China. Environ. Pollut. 2010, 158, 1159–1168. [Google Scholar] [CrossRef]
- Mahanty, T.; Bhattacharjee, S.; Goswami, M.; Bhattacharyya, P.; Das, B.; Ghosh, A.; Tribedi, P. Biofertilizers: A potential approach for sustainable agriculture development. Environ. Sci. Pollut. Res. 2016, 24, 3315–3335. [Google Scholar] [CrossRef]
- Sponsler, D.B.; Grozinger, C.M.; Hitaj, C.; Rundlöf, M.; Botías, C.; Code, A.; Lonsdorf, E.V.; Melathopoulos, A.P.; Smith, D.J.; Suryanarayanan, S.; et al. Pesticides and pollinators: A socioecological synthesis. Sci. Total. Environ. 2019, 662, 1012–1027. [Google Scholar] [CrossRef]
- Meena, R.S.; Kumar, S.; Datta, R.; Lal, R.; Vijayakumar, V.; Brtnicky, M.; Sharma, M.P.; Yadav, G.S.; Jhariya, M.K.; Jangir, C.K.; et al. Impact of Agrochemicals on Soil Microbiota and Management: A Review. Land 2020, 9, 34. [Google Scholar] [CrossRef]
- Donley, N. The USA lags behind other agricultural nations in banning harmful pesticides. Environ. Health 2019, 18, 1–12. [Google Scholar] [CrossRef]
- Fantke, P.; Friedrich, R.; Jolliet, O. Health impact and damage cost assessment of pesticides in Europe. Environ. Int. 2012, 49, 9–17. [Google Scholar] [CrossRef]
- Kauppinen, M.; Saikkonen, K.; Helander, M.; Pirttilä, A.M.; Wäli, P.R. Epichloë grass endophytes in sustainable agriculture. Nat. Plants 2016, 2, 1–7. [Google Scholar] [CrossRef]
- Yadav, A.N.; Singh, J.; Rastegari, A.A.; Yadav, N. (Eds.) Plant Microbiomes for Sustainable Agriculture; Springer: Cham, Switzerland, 2020. [Google Scholar]
- Verma, M.; Mishra, J.; Arora, N.K. Plant Growth-Promoting Rhizobacteria: Diversity and Applications. Environ. Biotechnol. Sustain. Future 2018, 2019, 129–173. [Google Scholar] [CrossRef]
- Parani, K.; Saha, B.K. Prospects of using phosphate solubilizing Pseudomonas as biofertilizer. Eur. J. Biol. Sci. 2012, 4, 40–44. [Google Scholar]
- Bhardwaj, D.; Ansari, M.W.; Sahoo, R.K.; Tuteja, N. Biofertilizers function as key player in sustainable agriculture by improving soil fertility, plant tolerance and crop productivity. Microb. Cell Factories 2014, 13, 66. [Google Scholar] [CrossRef]
- Köhl, J.; Kolnaar, R.; Ravensberg, W.J. Mode of Action of Microbial Biological Control Agents against Plant Diseases: Relevance beyond Efficacy. Front. Plant Sci. 2019, 10, 845. [Google Scholar] [CrossRef]
- Sharma, P.; Bora, L.; Puzari, K.; Baruah, A.; Baruah, R.; Talukdar, K.; Kataky, L.; Phukan, A. Review on bacterial blight of rice caused by Xanthomonas oryzae pv. oryzae: Different management approaches and role of Pseudomonas fluorescens as a potential biocontrol agent. Int. J. Curr. Microibol. Appl. Sci. 2017, 6, 982–1005. [Google Scholar]
- Kloepper, J.W.; Ryu, C.-M.; Zhang, S. Induced Systemic Resistance and Promotion of Plant Growth by Bacillus spp. Phytopathology 2004, 94, 1259–1266. [Google Scholar] [CrossRef]
- Adesemoye, A.; Torbert, H.; Kloepper, J. Enhanced plant nutrient use efficiency with PGPR and AMF in an integrated nutrient management system. Can. J. Microbiol. 2008, 54, 876–886. [Google Scholar] [CrossRef]
- Pii, Y.; Mimmo, T.; Tomasi, N.; Terzano, R.; Cesco, S.; Crecchio, C. Microbial interactions in the rhizosphere: Beneficial influences of plant growth-promoting rhizobacteria on nutrient acquisition process. A review. Biol. Fertil. Soils 2015, 51, 403–415. [Google Scholar] [CrossRef]
- Paterson, J.; Jahanshah, G.; Li, Y.; Wang, Q.; Mehnaz, S.; Gross, H. The contribution of genome mining strategies to the under-standing of active principles of PGPR strains. FEMS Microbiol. Ecol. 2017, 93, fiw249. [Google Scholar] [CrossRef]
- Adesemoye, A.O.; Kloepper, J.W. Plant–microbes interactions in enhanced fertilizer-use efficiency. Appl. Microbiol. Biotechnol. 2009, 85, 1–12. [Google Scholar] [CrossRef]
- Mącik, M.; Gryta, A.; Frąc, M. Biofertilizers in agriculture: An overview on concepts, strategies and effects on soil microorganisms. Adv. Agron. 2020, 162, 31–87. [Google Scholar] [CrossRef]
- Cabrefiga, J.; Bonaterra, A.; Montesinos, E. Mechanisms of antagonism of Pseudomonas fluorescens EPS62e against Erwinia amylo-vora, the causal agent of fire blight. Int. Microbiol. 2007, 10, 123. [Google Scholar]
- Johnsson, L.; Hökeberg, M.; Gerhardson, B. Performance of the Pseudomonas chlororaphis biocontrol agent MA 342 against cereal seed-borne diseases in field experiments. Eur. J. Plant Pathol. 1998, 104, 701–711. [Google Scholar] [CrossRef]
- Puopolo, G.; Raio, A.; Pierson, L.S., III; Zoina, A. Selection of a new Pseudomonas chlororaphis strain for the biological control of Fusarium oxysporum f. sp. radicis-lycopersici. Phytopathol. Mediterr. 2011, 50, 228–235. [Google Scholar]
- Han, Q.-Q.; Lü, X.-P.; Bai, J.-P.; Qiao, Y.; Paré, P.W.; Wang, S.-M.; Zhang, J.-L.; Wu, Y.-N.; Pang, X.-P.; Xu, W.-B.; et al. Beneficial soil bacterium Bacillus subtilis (GB03) augments salt tolerance of white clover. Front. Plant Sci. 2014, 5, 525. [Google Scholar] [CrossRef]
- Ngugi, H.; Dedej, S.; Delaplane, K.; Savelle, A.; Scherm, H. Effect of flower-applied Serenade biofungicide (Bacillus subtilis) on pollination-related variables in rabbiteye blueberry. Biol. Control. 2005, 33, 32–38. [Google Scholar] [CrossRef]
- Chowdhury, S.P.; Dietel, K.; Rändler, M.; Schmid, M.; Junge, H.; Borriss, R.; Hartmann, A.; Grosch, R. Effects of Bacillus amyloliquefaciens FZB42 on Lettuce Growth and Health under Pathogen Pressure and Its Impact on the Rhizosphere Bacterial Community. PLoS ONE 2013, 8, e68818. [Google Scholar] [CrossRef]
- Minuto, A.; Spadaro, D.; Garibaldi, A.; Gullino, M.L. Control of soilborne pathogens of tomato using a commercial formulation of Streptomyces griseoviridis and solarization. Crop. Prot. 2006, 25, 468–475. [Google Scholar] [CrossRef]
- Franche, C.; Lindström, K.; Elmerich, C. Nitrogen-fixing bacteria associated with leguminous and non-leguminous plants. Plant Soil 2009, 321, 35–59. [Google Scholar] [CrossRef]
- Vosátka, M.; Látr, A.; Gianinazzi, S.; Albrechtová, J. Development of arbuscular mycorrhizal biotechnology and industry: Cur-rent achievements and bottlenecks. Symbiosis 2012, 58, 29–37. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Elsevier: Amsterdam, The Netherlands, 2008; pp. 1–787. [Google Scholar]
- Smith, S.E.; Smith, F.A. Roles of arbuscular mycorrhizas in plant nutrition and growth: New paradigms from cellular to eco-system scales. Annu. Rev. Plant Biol. 2011, 62, 227–250. [Google Scholar] [CrossRef] [PubMed]
- Benami, M.; Isack, Y.; Grotsky, D.; Levy, D.; Kofman, Y. The Economic Potential of Arbuscular Mycorrhizal Fungi in Agriculture. Biotechnol. Extrem. 2020, 2020, 239–279. [Google Scholar] [CrossRef]
- Gianinazzi, S.; Gollotte, A.; Binet, M.-N.; Van Tuinen, D.; Redecker, D.; Wipf, D. Agroecology: The key role of arbuscular mycorrhizas in ecosystem services. Mycorrhiza 2010, 20, 519–530. [Google Scholar] [CrossRef]
- Albrechtova, J.; Latr, A.; Nedorost, L.; Pokluda, R.; Posta, K.; Vosatka, M. Dual Inoculation with Mycorrhizal and Saprotrophic Fungi Applicable in Sustainable Cultivation Improves the Yield and Nutritive Value of Onion. Sci. World J. 2012, 2012, 1–8. [Google Scholar] [CrossRef]
- Miller, R.M.; Jastrow, J.D. Mycorrhizal Fungi Influence Soil Structure. In Arbuscular Mycorrhizas: Physiology and Function; Kapulnik, Y., Douds, D.D., Eds.; Springer: Dordrecht, The Netherlands, 2000; pp. 3–18. [Google Scholar]
- Rillig, M.C.; Sosa-Hernández, M.A.; Roy, J.; Aguilar-Trigueros, C.A.; Vályi, K.; Lehmann, A. Towards an Integrated Mycorrhizal Technology: Harnessing Mycorrhiza for Sustainable Intensification in Agriculture. Front. Plant Sci. 2016, 7, 1625. [Google Scholar] [CrossRef]
- Zhang, S.; Lehmann, A.; Zheng, W.; You, Z.; Rillig, M.C. Arbuscular mycorrhizal fungi increase grain yields: A meta-analysis. New Phytol. 2019, 222, 543–555. [Google Scholar] [CrossRef]
- Affokpon, A.; Coyne, D.L.; Lawouin, L.; Tossou, C.; Agbèdè, R.D.; Coosemans, J. Effectiveness of native West African arbuscular mycorrhizal fungi in protecting vegetable crops against root-knot nematodes. Biol. Fertil. Soils 2011, 47, 207–217. [Google Scholar] [CrossRef]
- Hoeksema, J.D.; Chaudhary, V.B.; Gehring, C.A.; Johnson, N.C.; Karst, J.; Koide, R.T.; Pringle, A.; Zabinski, C.; Bever, J.D.; Moore, J.C.; et al. A meta-analysis of context-dependency in plant response to inoculation with mycorrhizal fungi. Ecol. Lett. 2010, 13, 394–407. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, V.B.; Rúa, M.A.; Antoninka, A.; Bever, J.D.; Cannon, J.; Craig, A.; Duchicela, J.; Frame, A.; Gardes, M.; Gehring, C.; et al. MycoDB, a global database of plant response to mycorrhizal fungi. Sci. Data 2016, 3, 160028. [Google Scholar] [CrossRef] [PubMed]
- Aamir, M.; Rai, K.K.; Zehra, A. Microbial bioformulation-based plant biostimulants: A plausible approach toward next generation of sustainable agriculture. In Microbial Endophytes; Elsevier: Amsterdam, The Netherlands, 2020; pp. 195–225. [Google Scholar]
- Varma, A.; Verma, S.; Sahay, N.; Bütehorn, B.; Franken, P. Piriformospora indica, a cultivable plant-growth-promoting root endo-phyte. Appl. Environ. Microbiol. 1999, 65, 2741–2744. [Google Scholar] [CrossRef] [PubMed]
- Waller, F.; Achatz, B.; Baltruschat, H.; Fodor, J.; Becker, K.; Fischer, M.; Heier, T.; Hückelhoven, R.; Neumann, C.; Von Wettstein, D.; et al. The endophytic fungus Piriformospora indica reprograms barley to salt-stress tolerance, disease resistance, and higher yield. Proc. Natl. Acad. Sci. USA 2005, 102, 13386–13391. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Yadav, V.; Tuteja, N.; Johri, A.K. Antioxidant enzyme activities in maize plants colonized with Piriformospora indica. Microbiology 2009, 155, 780–790. [Google Scholar] [CrossRef]
- Oelmüller, R.; Sherameti, I.; Tripathi, S.; Varma, A. Piriformospora indica, a cultivable root endophyte with multiple biotechno-logical applications. Symbiosis 2009, 49, 1–17. [Google Scholar] [CrossRef]
- Yadav, V.; Kumar, M.; Deep, D.K.; Kumar, H.; Sharma, R.; Tripathi, T.; Tuteja, N.; Saxena, A.K.; Johri, A.K. A Phosphate Transporter from the Root Endophytic Fungus Piriformospora indica plays a Role in Phosphate Transport to the Host Plant. J. Biol. Chem. 2010, 285, 26532–26544. [Google Scholar] [CrossRef]
- Das, A.; Sherameti, I.; Varma, A. Contaminated soil: Physical, chemical and biological components. In Bio-Geo Interactions in Metal-Contaminated Soils; Springer: Cham, Switzerland, 2012; pp. 1–15. [Google Scholar]
- Rabiey, M.; Ullah, I.; Shaw, L.J.; Shaw, M.W. Potential ecological effects of Piriformospora indica, a possible biocontrol agent, in UK agricultural systems. Biol. Control. 2017, 104, 1–9. [Google Scholar] [CrossRef]
- Shrivastava, S.; Varma, A. From Piriformospora indica to Rootonic: A review. Afr. J. Microbiol. Res. 2014, 8, 2984–2992. [Google Scholar]
- Bouton, J.H.; Latch, G.C.M.; Hill, N.S.; Hoveland, C.S.; McCann, M.A.; Watson, R.H.; Parish, J.A.; Hawkins, L.L.; Thompson, F.N. Reinfection of Tall Fescue Cultivars with Non-Ergot Alkaloid–Producing Endophytes. Agron. J. 2002, 94, 567–574. [Google Scholar] [CrossRef]
- Bouton, J. The economic benefits of forage improvement in the United States. Euphytica 2006, 154, 263–270. [Google Scholar] [CrossRef]
- Johnson, L.J.; De Bonth, A.C.M.; Briggs, L.R.; Caradus, J.R.; Finch, S.C.; Fleetwood, D.J.; Fletcher, L.R.; Hume, D.E.; Johnson, R.D.; Popay, A.J.; et al. The exploitation of Epichloae endophytes for agricultural benefit. Fungal Divers. 2013, 60, 171–188. [Google Scholar] [CrossRef]
- Young, C.A.; Aiken, G.E.; McCulley, R.L.; Strickland, J.R.; Schardl, C.L. Epichloae, Endophytes of Cool Season Grasses: Implications, Utilization and Biology; Samuel Roberts Noble Foundation: Carter County, OK, USA, 2012. [Google Scholar]
- Harvey, P.R.; Warren, R.A.; Wakelin, S. Potential to improve root access to phosphorus: The role of non-symbiotic microbial inoculants in the rhizosphere. Crop. Pasture Sci. 2009, 60, 144–151. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y.; Di Mattia, E.; El-Nakhel, C.; Cardarelli, M. Co-inoculation of Glomus intraradices and Trichoderma atroviride acts as a biostimulant to promote growth, yield and nutrient uptake of vegetable crops. J. Sci. Food Agric. 2015, 95, 1706–1715. [Google Scholar] [CrossRef]
- Kakabouki, I.; Tataridas, A.; Mavroeidis, A.; Kousta, A.; Karydogianni, S.; Zisi, C.; Kouneli, V.; Konstantinou, A.; Folina, A.; Konstantas, A.; et al. Effect of Colonization of Trichoderma harzianum on Growth Development and CBD Content of Hemp (Cannabis sativa L.). Microorganisms 2021, 9, 518. [Google Scholar] [CrossRef]
- Perdikis, D.; Kapaxidi, E.; Papadoulis, G. Biological control of insect and mite pests in greenhouse solanaceous crops. Eur. J. Plant Sci. Biotechnol. 2008, 2, 125–144. [Google Scholar]
- Soytong, K.; Kanokmedhakul, S.; Kukongviriyapa, V.; Isobe, M. Application of Chaetomium species (Ketomium) as a new broad spectrum biological fungicide for plant disease control. Fungal Divers. 2001, 7, 1–15. [Google Scholar]
- Mercado-Blanco, J.; Lugtenberg, B.J.J. Biotechnological applications of bacterial endophytes. Curr. Biotechnol. 2014, 3, 60–75. [Google Scholar] [CrossRef]
- Schisler, D.A.; Slininger, P.J.; Behle, R.W.; Jackson, M.A. Formulation of Bacillus spp. for Biological Control of Plant Diseases. Phytopathology 2004, 94, 1267–1271. [Google Scholar] [CrossRef]
- Bashan, Y.; De-Bashan, L.E.; Prabhu, S.R.; Hernandez, J.-P. Advances in plant growth-promoting bacterial inoculant technology: Formulations and practical perspectives (1998–2013). Plant Soil 2014, 378, 1–33. [Google Scholar] [CrossRef]
- Kaur, R.; Kaur, J.; Singh, R.S. Nonpathogenic Fusarium as a Biological Control Agent. Plant Pathol. J. 2010, 9, 79–91. [Google Scholar] [CrossRef]
- Woo, S.L.; Ruocco, M.; Vinale, F.; Nigro, M.; Marra, R.; Lombardi, N.; Pascale, A.; Lanzuise, S.; Manganiello, G.; Lorito, M. Trichoderma-based Products and their Widespread Use in Agriculture. Open Mycol. J. 2014, 8, 71–126. [Google Scholar] [CrossRef]
- Kamilova, F.; Okon, Y.; De Weert, S.; Hora, K. Commercialization of Microbes: Manufacturing, Inoculation, Best Practice for Objective Field Testing, and Registration. Princ. Plant Microbe Interact. 2014, 2015, 319–327. [Google Scholar] [CrossRef]
- Young, S.D.; Townsend, R.J.; Swaminathan, J.; O’Callaghan, M. Serratia entomophila coated seed to improve ryegrass establishment in the presence of grass grubs. N. Zeal. Plant Prot. 2010, 63, 229–234. [Google Scholar] [CrossRef]
- Mejri, D.; Gamalero, E.; Souissi, T. Formulation development of the deleterious rhizobacterium Pseudomonas trivialis X33d for biocontrol of brome (Bromus diandrus) in durum wheat. J. Appl. Microbiol. 2013, 114, 219–228. [Google Scholar] [CrossRef]
- Arora, N.K.; Mishra, J. Prospecting the roles of metabolites and additives in future bioformulations for sustainable agriculture. Appl. Soil Ecol. 2016, 107, 405–407. [Google Scholar] [CrossRef]
- Podolich, O.; Ardanov, P.; Zaets, I.; Pirttilä, A.M.; Kozyrovska, N. Reviving of the endophytic bacterial community as a putative mechanism of plant resistance. Plant Soil 2015, 388, 367–377. [Google Scholar] [CrossRef]
- Latz, M.A.; Jensen, B.; Collinge, D.B.; Jørgensen, H.J. Endophytic fungi as biocontrol agents: Elucidating mechanisms in disease suppression. Plant Ecol. Divers. 2018, 11, 555–567. [Google Scholar] [CrossRef]
- Itelima, J.U.; Bang, W.J.; Onyimba, I.A.; Sila, M.D.; Egbere, O.J. Bio-Fertilizers as Key Player in Enhancing Soil Fertility and Crop Productivity: A Review. Direct Res. J. Agric. Food Sci. 2018, 6, 73–83. [Google Scholar] [CrossRef]
- Rahman, A.; Syed, S.F.; Singh, E.; Pieterse, C.M.; Schenk, P.M. Emerging microbial biocontrol strategies for plant pathogens. Plant Sci. 2018, 267, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-K.; Jeong, H.; Kloepper, J.W.; Ryu, C.-M. Genome Sequence of Bacillus amyloliquefaciens GB03, an Active Ingredient of the First Commercial Biological Control Product. Genome Announc. 2014, 2. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.H.; Koumoutsi, A.; Scholz, R.; Eisenreich, A.; Schneider, K.; Heinemeyer, I.; Morgenstern, B.; Voss, B.; Hess, W.R.; Reva, O.; et al. Comparative analysis of the complete genome sequence of the plant growth–promoting bacterium Bacillus amyloliquefaciens FZB42. Nat. Biotechnol. 2007, 25, 1007–1014. [Google Scholar] [CrossRef]
- Frank, A.C. The Genomes of Endophytic Bacteria. In Endophytes of Forest Trees; Pirttilä, A.M., Frank, A.C., Eds.; Springer: Cham, Switzerland, 2018; pp. 141–176. [Google Scholar]
- Fan, B.; Wang, C.; Song, X.; Ding, X.; Wu, L.; Wu, H.; Gao, X.; Borriss, R. Bacillus velezensis FZB42 in 2018: The Gram-Positive Model Strain for Plant Growth Promotion and Biocontrol. Front. Microbiol. 2018, 9, 2491. [Google Scholar] [CrossRef]
- Wang, J.; Xing, J.; Lu, J.; Sun, Y.; Zhao, J.; Miao, S.; Xiong, Q.; Zhang, Y.; Zhang, G. Complete Genome Sequencing of Bacillus velezensis WRN014, and Comparison with Genome Sequences of other Bacillus velezensis Strains. J. Microbiol. Biotechnol. 2019, 29, 794–808. [Google Scholar] [CrossRef]
- Ma, J.; Wang, C.; Wang, H.; Liu, K.; Zhang, T.; Yao, L.; Zhao, Z.; Du, B.; Ding, Y. Analysis of the Complete Genome Sequence of Bacillus atrophaeus GQJK17 Reveals Its Biocontrol Characteristics as a Plant Growth-Promoting Rhizobacterium. BioMed Res. Int. 2018, 2018, 1–9. [Google Scholar] [CrossRef]
- Gerbore, J.; Brutel, A.; Lemainque, A.; Mairey, B.; Médigue, C.; Vallenet, D.; Lefort, F.; Grizard, D. Complete Genome Sequence of Bacillus methylotrophicus Strain B25, a Potential Plant Growth-Promoting Rhizobacterium. Genome Announc. 2016, 4. [Google Scholar] [CrossRef]
- Kim, J.F.; Jeong, H.; Park, S.-Y.; Kim, S.-B.; Park, Y.K.; Choi, S.-K.; Ryu, C.-M.; Hur, C.-G.; Ghim, S.-Y.; Oh, T.K.; et al. Genome Sequence of the Polymyxin-Producing Plant-Probiotic Rhizobacterium Paenibacillus polymyxa E681. J. Bacteriol. 2010, 192, 6103–6104. [Google Scholar] [CrossRef]
- Ma, M.; Wang, C.; Ding, Y.; Li, L.; Shen, D.; Jiang, X.; Guan, D.; Cao, F.; Chen, H.; Feng, R.; et al. Complete Genome Sequence of Paenibacillus polymyxa SC2, a Strain of Plant Growth-Promoting Rhizobacterium with Broad-Spectrum Antimicrobial Activity. J. Bacteriol. 2010, 193, 311–312. [Google Scholar] [CrossRef]
- Paulsen, I.T.; Press, C.M.; Ravel, J.; Kobayashi, D.Y.; Myers, G.S.A.; Mavrodi, D.V.; DeBoy, R.T.; Seshadri, R.; Ren, Q.; Madupu, R.; et al. Complete genome sequence of the plant commensal Pseudomonas fluorescens Pf-5. Nat. Biotechnol. 2005, 23, 873–878. [Google Scholar] [CrossRef]
- Gross, H.; Stockwell, V.O.; Henkels, M.D.; Nowak-Thompson, B.; Loper, J.E.; Gerwick, W.H. The genomisotopic approach: A systematic method to isolate products of orphan biosynthetic gene clusters. Chem. Biol. 2007, 14, 53–63. [Google Scholar] [CrossRef]
- Van Der Voort, M.; Meijer, H.; Schmidt, Y.; Watrous, J.; Dekkers, E.; Mendes, R.; Dorrestein, P.C.; Gross, H.; Raaijmakers, J.M. Genome mining and metabolic profiling of the rhizosphere bacterium Pseudomonas sp. SH-C52 for antimicrobial compounds. Front. Microbiol. 2015, 6, 693. [Google Scholar] [CrossRef]
- Jing, T.; Zhou, D.; Zhang, M.; Yun, T.; Qi, D.; Wei, Y.; Chen, Y.; Zang, X.; Wang, W.; Xie, J. Newly Isolated Streptomyces sp. JBS5-6 as a Potential Biocontrol Agent to Control Banana Fusarium Wilt: Genome Sequencing and Secondary Metabolite Cluster Profiles. Front. Microbiol. 2020, 11. [Google Scholar] [CrossRef]
- Lopes, R.; Cerdeira, L.; Tavares, G.S.; Ruiz, J.C.; Blom, J.; Horácio, E.C.A.; Mantovani, H.C.; de Queiroz, M.V. Genome analysis reveals insights of the endophytic Bacillus toyonensis BAC3151 as a potentially novel agent for biocontrol of plant pathogens. World J. Microbiol. Biotechnol. 2017, 33, 1–15. [Google Scholar] [CrossRef]
- Eastman, A.W.; Heinrichs, D.E.; Yuan, Z.C. Comparative and genetic analysis of the four sequenced Paenibacillus polymyxa genomes reveals a diverse metabolism and conservation of genes relevant to plant-growth promotion and competitiveness. BMC Genom. 2014, 15, 1–22. [Google Scholar] [CrossRef]
- Crovadore, J.; Xu, S.; Chablais, R.; Cochard, B.; Lukito, D.; Calmin, G.; Lefort, F. Metagenome-assembled genome sequence of Rhodopseudomonas palustris strain eli 1980, commercialized as a biostimulant. Genome Announc. 2017, 5. [Google Scholar] [CrossRef]
- Luo, Y.; Zhou, M.; Zhao, Q.; Wang, F.; Gao, J.; Sheng, H.; An, L. Complete genome sequence of Sphingomonas sp. Cra20, a drought resistant and plant growth promoting rhizobacteria. Genomics 2020, 112, 3648–3657. [Google Scholar] [CrossRef]
- Chen, M.; Liu, Q.; Gao, S.; Young, A.E.; Jacobsen, S.E.; Tang, Y. Genome mining and biosynthesis of a polyketide from a biofertilizer fungus that can facilitate reductive iron assimilation in plant. Proc. Natl. Acad. Sci. USA 2019, 116, 5499–5504. [Google Scholar] [CrossRef]
- Koskimäki, J.J.; Pirttilä, A.M.; Ihantola, E.L.; Halonen, O.; Frank, A.C. The intracellular Scots pine shoot symbiont Methylobacterium extorquens DSM13060 aggregates around the host nucleus and encodes eukaryote-like proteins. MBio 2015, 6. [Google Scholar] [CrossRef]
- Lafi, F.F.; Ramirez-Prado, J.S.; Alam, I.; Bajic, V.B.; Hirt, H.; Saad, M.M. Draft genome sequence of plant growth–promoting Micrococcus luteus strain K39 isolated from Cyperus conglomeratus in Saudi Ara-bia. Genome Announc. 2017, 5, e01520-16. [Google Scholar] [CrossRef]
- Levy, A.; Gonzalez, I.S.; Mittelviefhaus, M.; Clingenpeel, S.; Paredes, S.H.; Miao, J.; Wang, K.; Devescovi, G.; Stillman, K.; Monteiro, F.; et al. Genomic features of bacterial adaptation to plants. Nat. Genet. 2018, 50, 138–150. [Google Scholar] [CrossRef]
- Fan, B.; Li, L.; Chao, Y.; Förstner, K.; Jörg Vogel, J.; Borriss, R.; Wu, X.-Q. dRNA-seq reveals genomewide TSSs and noncoding RNAs of plant beneficial rhizobacterium bacillus amyloliquefaciens FZB42. PLoS ONE 2015, 10, e0142002. [Google Scholar] [CrossRef]
- Koskimäki, J.J.; Kajula, M.; Hokkanen, J.; Ihantola, E.-L. Methyl-esterified 3-hydroxybutyrate oligomers protect bacteria from hydroxyl radicals. Nat. Chem. Biol. 2016, 12, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Müller-Santos, M.; Koskimäki, J.J.; Alves, L.P.S.; de Souza, E.M.; Jendrossek, D.; Pirttilä, A.M. The protective role of PHB and its degradation products against stress situations in bacteria. FEMS Microbiol. Rev. 2020. [Google Scholar] [CrossRef] [PubMed]
- Garbeva, P.; Silby, M.W.; Raaijmakers, J.M.; Levy, S.B.; De Boer, W. Transcriptional and antagonistic responses of Pseudomonas fluorescens Pf0-1 to phylogenetically different bacterial competitors. ISME J. 2011, 5, 973–985. [Google Scholar] [CrossRef] [PubMed]
- Tejesvi, M.V.; Segura, D.R.; Schnorr, K.M.; Sandvang, D.; Mattila, S.; Olsen, P.B.; Neve, S.; Kruse, T.; Kristensen, H.H.; Pirttilä, A.M. An antimicrobial peptide from endophytic Fusarium tricinctum of Rhododendron tomentosum Harmaja. Fungal Divers. 2013, 60, 153–159. [Google Scholar] [CrossRef]
- Tejesvi, M.V.; Andersen, B.; Antcheva, N.; Brinch, K.S.; Koskimäki, J.J.; Kristensen, H.H.; Tossi, A.; Pirttilä, A.M. MB1533 is a defensin-like antimicrobial peptide from the intracellular meristem endophyte of Scots pine Methylobacterium extorquens DSM13060. J. Microb. Biochem. Technol. 2016, 8, 1–5. [Google Scholar]
- Jung, H.J.; Kim, Y.; Lee, H.B.; Kwon, H.J. Antiangiogenic activity of the lipophilic antimicrobial peptides from an endophytic bacterial strain isolated from red pepper leaf. Mol. Cells 2014, 38, 273–278. [Google Scholar] [CrossRef]
- Reiter, B.; Pfeifer, U.; Schwab, H.; Sessitsch, A. Response of Endophytic Bacterial Communities in Potato Plants to Infection with Erwinia carotovora subsp. atroseptica. Appl. Environ. Microbiol. 2002, 68, 2261–2268. [Google Scholar] [CrossRef]
- Lian, J.; Wang, Z.; Zhou, S. Response of endophytic bacterial communities in banana tissue culture plantlets to Fusarium wilt pathogen infection. J. Gen. Appl. Microbiol. 2008, 54, 83–92. [Google Scholar] [CrossRef]
- Ardanov, P.; Sessitsch, A.; Häggman, H.; Kozyrovska, N.; Pirttilä, A.M. Methylobacterium-induced endophyte community changes correspond with protection of plants against pathogen attack. PLoS ONE 2012, 7, e46802. [Google Scholar] [CrossRef]
- Ardanov, P.; Lyastchenko, S.; Karppinen, K.; Häggman, H.; Kozyrovska, N.; Pirttilä, A.M. Effects of Methylobacterium sp. on emer-gence, yield, and disease prevalence in three cultivars of potato (Solanum tuberosum L.) were associated with the shift in en-dophytic microbial community. Plant Soil 2016, 405, 299–310. [Google Scholar] [CrossRef]
- Azevedo, J.L.; Araújo, W.L.; Lacava, P.T. The diversity of citrus endophytic bacteria and their interactions with Xylella fastidiosa and host plants. Genet. Mol. Biol. 2016, 39, 476–491. [Google Scholar] [CrossRef]
- Dy, R.L.; Rigano, L.A.; Fineran, P.C. Phage-based biocontrol strategies and their application in agriculture and aquaculture. Biochem. Soc. Trans. 2018, 46, 1605–1613. [Google Scholar] [CrossRef]
- Holtappels, D.; Fortuna, K.; Lavigne, R.; Wagemans, J. The future of phage biocontrol in integrated plant protection for sustainable crop production. Curr. Opin. Biotechnol. 2021, 68, 60–71. [Google Scholar] [CrossRef]
- Bulgarelli, D.; Garrido-Oter, R.; Münch, P.C.; Weiman, A.; Dröge, J.; Pan, Y.; McHardy, A.C.; Schulze-Lefert, P. Structure and function of the bacterial root microbiota in wild and domesticated barley. Cell Host Microbe 2015, 17, 392–403. [Google Scholar] [CrossRef]
- Vafadar, F.; Amooaghaie, R.; Otroshy, M. Effects of plant-growth-promoting rhizobacteria and arbuscular mycorrhizal fungus on plant growth, stevioside, NPK, and chlorophyll content of Stevia rebaudiana. J. Plant Interact. 2013, 9, 128–136. [Google Scholar] [CrossRef]
- Pohjanen, J.; Koskimäki, J.J.; Sutela, S.; Ardanov, P.; Suorsa, M.; Niemi, K.; Sarjala, T.; Häggman, H.; Pirttilä, A.M. Interaction with ectomycorrhizal fungi and endophytic Methylobacterium affects nutrient uptake and growth of pine seedlings in vitro. Tree Physiol. 2014, 34, 993–1005. [Google Scholar] [CrossRef]
- Chowdhury, S.P.; Hartmann, A.; Gao, X.; Borriss, R. Biocontrol mechanism by root-associated Bacillus amyloliquefaciens FZB42—A review. Front. Microbiol. 2015, 6, 780. [Google Scholar] [CrossRef]
- Wu, H.-J.; Wu, E. The role of gut microbiota in immune homeostasis and autoimmunity. Gut Microbes 2012, 3, 4–14. [Google Scholar] [CrossRef]
- Wei, Z.; Gu, Y.; Friman, V.-P.; Kowalchuk, G.A.; Xu, Y.; Shen, Q.; Jousset, A. Initial soil microbiome composition and functioning predetermine future plant health. Sci. Adv. 2019, 5, eaaw0759. [Google Scholar] [CrossRef]
- Hardoim, P.R.; Van Overbeek, L.S.; Berg, G.; Pirttilä, A.M.; Compant, S.; Campisano, A.; Döring, M.; Sessitsch, A. The hidden world within plants: Ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol. Mol. Biol. Rev. 2015, 79, 293–320. [Google Scholar] [CrossRef]
- Spadaro, D.; Gullino, M.L. Improving the efficacy of biocontrol agents against soilborne pathogens. Crop. Prot. 2005, 24, 601–613. [Google Scholar] [CrossRef]
- Leger, R.J.S.; Wang, C. Genetic engineering of fungal biocontrol agents to achieve greater efficacy against insect pests. Appl. Microbiol. Biotechnol. 2010, 85, 901–907. [Google Scholar] [CrossRef]
- Skirvin, R.M.; Kohler, E.; Steiner, H.; Ayers, D.; Laughnan, A.; Norton, M.A.; Warmund, M. The use of genetically engineered bacteria to control frost on strawberries and potatoes. Whatever happened to all of that research? Sci. Hortic. 2000, 84, 179–189. [Google Scholar] [CrossRef]
- Mark, G.L.; Morrissey, J.P.; Higgins, P.; O’gara, F. Molecular-based strategies to exploit Pseudomonas biocontrol strains for environmental biotechnology applications. FEMS Microbiol. Ecol. 2006, 56, 167–177. [Google Scholar] [CrossRef]
- Glandorf, D.C. Re-evaluation of biosafety questions on genetically modified biocontrol bacteria. Eur. J. Plant Pathol. 2019, 154, 43–51. [Google Scholar] [CrossRef]
- Shelake, R.M.; Pramanik, D.; Kim, J.-Y. Exploration of Plant-Microbe Interactions for Sustainable Agriculture in CRISPR Era. Microorganisms 2019, 7, 269. [Google Scholar] [CrossRef]
- Muñoz, I.V.; Sarrocco, S.; Malfatti, L.; Baroncelli, R.; Vannacci, G. CRISPR-Cas for fungal genome editing: A new tool for the management of plant diseases. Front. Plant Sci. 2019, 10, 135. [Google Scholar] [CrossRef]
- Ganley, R.J.; Brunsfeld, S.J.; Newcombe, G. A community of unknown, endophytic fungi in western white pine. Proc. Natl. Acad. Sci. USA 2004, 101, 10107–10112. [Google Scholar] [CrossRef]
- Chebotar, V.K.; Malfanova, N.V.; Shcherbakov, A.V.; Ahtemova, G.A.; Borisov, A.Y.; Lugtenberg, B.; Tikhonovich, I.A. Endophytic bacteria in microbial preparations that improve plant development. Appl. Biochem. Microbiol. 2015, 51, 271–277. [Google Scholar] [CrossRef]
- Liu, H.; Carvalhais, L.C.; Crawford, M.; Singh, E.; Dennis, P.G.; Pieterse, C.M.J.; Schenk, P.M. Inner Plant Values: Diversity, Colonization and Benefits from Endophytic Bacteria. Front. Microbiol. 2017, 8, 2552. [Google Scholar] [CrossRef]
- Adetunji, C.O.; Kumar, D.; Raina, M.; Arogundade, O.; Sarin, N.B. Endophytic Microorganisms as Biological Control Agents for Plant Pathogens: A Panacea for Sustainable Agriculture. Plant Biot. Interact. 2019, 2019, 1–20. [Google Scholar] [CrossRef]
- Pandey, P.K.; Samanta, R.; Yadav, R.N.S. Inside the plant: Addressing bacterial endophytes in biotic stress alleviation. Arch. Microbiol. 2019, 201, 415–429. [Google Scholar] [CrossRef] [PubMed]
- Koskimäki, J.J.; Nylund, S.; Suorsa, M.; Pirttilä, A.M. Mycobacterial endophytes are enriched during micropropagation of Pogonatherum paniceum. Environ. Microbiol. Rep. 2010, 2, 619–624. [Google Scholar] [CrossRef] [PubMed]
- van Hylckama Vlieg Johan, E.T.; Veiga, P.; Zhang, C.; Derrien, M.; Zhao, L. Impact of microbial transformation of food on health—From fermented foods to fermentation in the gastro-intestinal tract. Curr. Opin. Biotechnol. 2011, 22, 211–219. [Google Scholar] [CrossRef]
- Kumar, V.; Narula, N. Solubilization of inorganic phosphates and growth emergence of wheat as affected by Azotobacter chroococcum mutants. Biol. Fertil. Soils 1999, 28, 301–305. [Google Scholar] [CrossRef]
- Wisniewski-Dyé, F.; Borziak, K.; Khalsa-Moyers, G.; Alexandre, G.; Sukharnikov, L.O.; Wuichet, K.; Hurst, G.B.; McDonald, W.H.; Robertson, J.S.; Barbe, V.; et al. Azospirillum genomes reveal transition of bacteria from aquatic to terrestrial environ-ments. PLoS Genet. 2011, 7, e1002430. [Google Scholar] [CrossRef]
- Howell, C.R.; Stipanovic, R.D. Control of Rhizoctonia solani on cotton seedlings with Pseudomonas fluorescens and with an anti-biotic produced by the bacterium. Phytopathology 1979, 69, 480–482. [Google Scholar] [CrossRef]
- Etesami, H.; Glick, B.R. Halotolerant plant growth–promoting bacteria: Prospects for alleviating salinity stress in plants. Environ. Exp. Bot. 2020, 178, 104124. [Google Scholar] [CrossRef]
- Matías, L.; Jump, A.S. Interactions between growth, demography and biotic interactions in determining species range limits in a warming world: The case of Pinus sylvestris. For. Ecol. Manag. 2012, 282, 10–22. [Google Scholar] [CrossRef]
- Márquez, L.M.; Redman, R.S.; Rodriguez, R.J.; Roossinck, M.J. A Virus in a Fungus in a Plant: Three-Way Symbiosis Required for Thermal Tolerance. Science 2007, 315, 513–515. [Google Scholar] [CrossRef]
- Miller, J.D. Foliar Endophytes of Spruce Species Found in the Acadian Forest: Basis and Potential for Improving the Tolerance of the Forest to Spruce Budworm. In Endophytes of Forest Trees; Pirttilä, A.M., Frank, A.C., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 237–249. [Google Scholar]
- Muñoz-Arenas, L.C.; Fusaro, C.; Hernández-Guzmán, M.; Dendooven, L.; Estrada-Torres, A.; Navarro-Noya, Y.E. Soil microbial diversity drops with land-use change in a high mountain temperate forest: A metagenomics survey. Environ. Microbiol. Rep. 2020, 12, 185–194. [Google Scholar] [CrossRef]
- Ganley, R.J.; Newcombe, G. Fungal endophytes in seeds and needles of Pinus monticola. Mycol. Res. 2006, 110, 318–327. [Google Scholar] [CrossRef]
- Pancher, M.; Ceol, M.; Corneo, P.E.; Longa, C.M.O.; Yousaf, S.; Pertot, I.; Campisano, A. Fungal Endophytic Communities in Grapevines (Vitis vinifera L.) Respond to Crop Management. Appl. Environ. Microbiol. 2012, 78, 4308–4317. [Google Scholar] [CrossRef]
- Kãberl, M.; Dita, M.; Martinuz, A.; Staver, C.; Berg, G. Agroforestry leads to shifts within the gammaproteobacterial microbiome of banana plants cultivated in Central America. Front. Microbiol. 2015, 6, 91. [Google Scholar] [CrossRef]
- Pirttilä, A.M. Commentary: Agroforestry leads to shifts within the gammaproteobacterial microbiome of banana plants cultivated in Central America. Front. Microbiol. 2016, 7, 656. [Google Scholar] [CrossRef]
- Nian, J.; Yu, M.; Bradley, C.A.; Zhao, Y. Lysobacter enzymogenes strain C3 suppresses mycelium growth and spore germination of eight soybean fungal and oomycete pathogens and decreases disease incidences. Biol. Control. 2021, 152, 104424. [Google Scholar] [CrossRef]
- Mendoza-Arroyo, G.E.; Chan-Bacab, M.J.; Aguila-Ramírez, R.N.; Ortega-Morales, B.O.; Canché Solís, R.E.; Chab-Ruiz, A.O.; Cob-Rivera, K.I.; Dzib-Castillo, B.; Tun-Che, R.E.; Camacho-Chab, J.C. Inorganic phosphate solubilization by a novel isolated bacterial strain Enterobacter sp. ITCB-09 and its application potential as biofertilizer. Agriculture 2020, 10, 383. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pirttilä, A.M.; Mohammad Parast Tabas, H.; Baruah, N.; Koskimäki, J.J. Biofertilizers and Biocontrol Agents for Agriculture: How to Identify and Develop New Potent Microbial Strains and Traits. Microorganisms 2021, 9, 817. https://doi.org/10.3390/microorganisms9040817
Pirttilä AM, Mohammad Parast Tabas H, Baruah N, Koskimäki JJ. Biofertilizers and Biocontrol Agents for Agriculture: How to Identify and Develop New Potent Microbial Strains and Traits. Microorganisms. 2021; 9(4):817. https://doi.org/10.3390/microorganisms9040817
Chicago/Turabian StylePirttilä, Anna Maria, Habibollah Mohammad Parast Tabas, Namrata Baruah, and Janne J. Koskimäki. 2021. "Biofertilizers and Biocontrol Agents for Agriculture: How to Identify and Develop New Potent Microbial Strains and Traits" Microorganisms 9, no. 4: 817. https://doi.org/10.3390/microorganisms9040817
APA StylePirttilä, A. M., Mohammad Parast Tabas, H., Baruah, N., & Koskimäki, J. J. (2021). Biofertilizers and Biocontrol Agents for Agriculture: How to Identify and Develop New Potent Microbial Strains and Traits. Microorganisms, 9(4), 817. https://doi.org/10.3390/microorganisms9040817





