D-Fructose Assimilation and Fermentation by Yeasts Belonging to Saccharomycetes: Rediscovery of Universal Phenotypes and Elucidation of Fructophilic Behaviors in Ambrosiozyma platypodis and Cyberlindnera americana
Abstract
1. Introduction
2. Materials and Methods
2.1. Yeast Strains
2.2. Assimilation of Fructose
2.3. Fermentation of Fructose
2.4. Sugar Consumption during Fermentation
3. Results
3.1. Assimilation of Fructose
3.2. Fermentation of Fructose
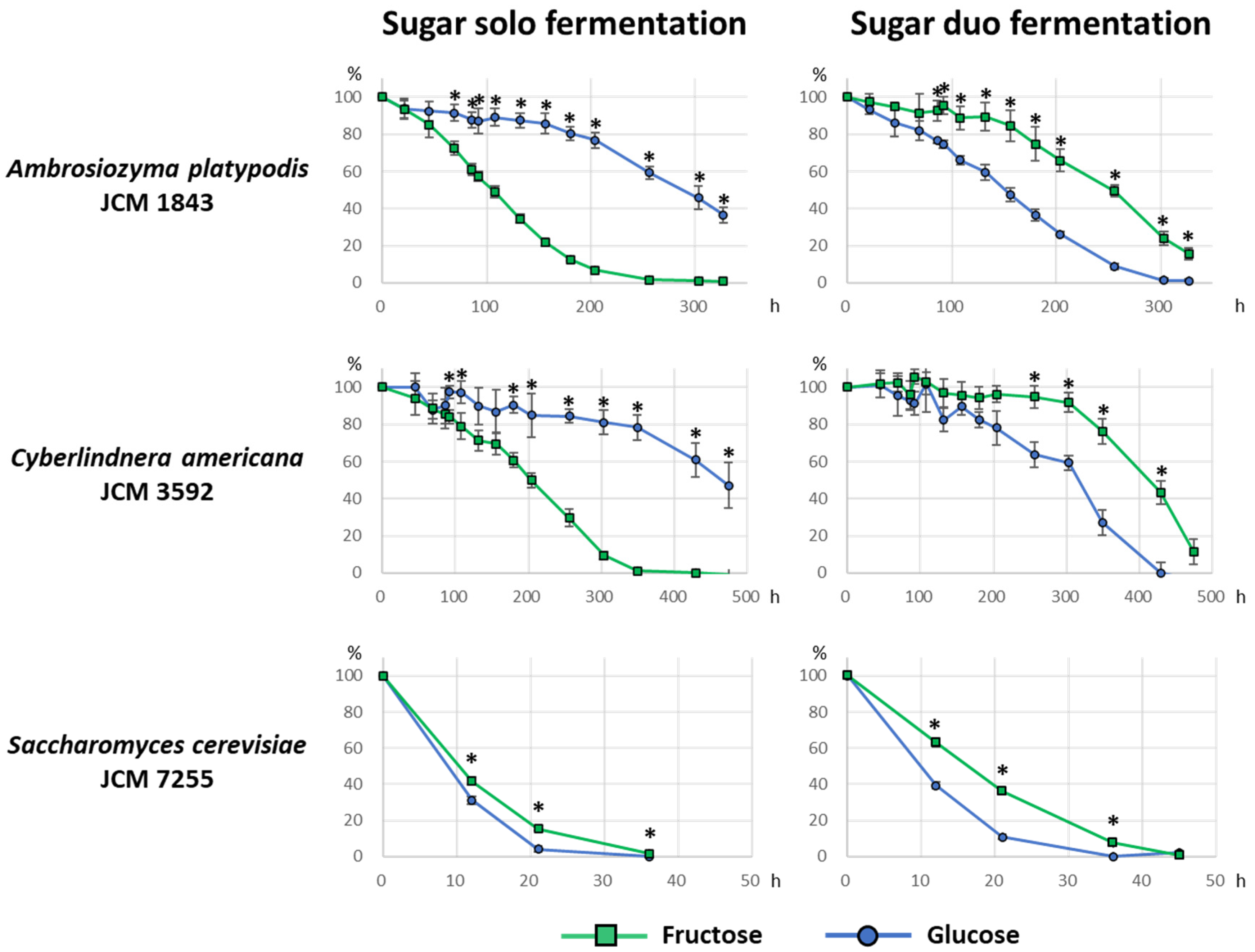
3.3. Sugar Consumption during Fermentation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wickerham, L.J.; Burton, K.A. Carbon assimilation tests for the classification of yeasts. J. Bacteriol. 1948, 56, 363–371. [Google Scholar] [CrossRef]
- Lodder, J.; Kreger-van Rij, N.J.W. The Yeasts, a Taxonomic Study; North-Holland Publishing Company: Amsterdam, The Netherlands, 1952. [Google Scholar]
- Kurtzman, C.P.; Fell, J.W.; Boekhout, T. The Yeasts, a Taxonomic Study, 5th ed.; Elsevier B.V.: London, UK, 2011. [Google Scholar]
- Guillaume, C.; Delobel, P.; Sablayrolles, J.-M.; Blondin, B. Molecular basis of fructose utilization by the wine yeast Saccharomyces cerevisiae: A mutated HXT3 allele enhances fructose fermentation. Appl. Environ. Microbiol. 2007, 73, 2432–2439. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, R.H. The selective fermentation of glucose and fructose by brewer’s yeast. Biochem. J. 1928, 22, 1145–1156. [Google Scholar] [CrossRef]
- Pinto, L.; Malfeito-Ferreira, M.; Quintieri, L.; Silva, A.C.; Baruzzi, F. Growth and metabolite production of a grape sour rot yeast-bacterium consortium on different carbon sources. Int. J. Food Microbiol. 2019, 296, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Pinto, L.; Caputo, L.; Quintieri, L.; de Candia, S.; Baruzzi, F. Efficacy of gaseous ozone to counteract postharvest table grape sour rot. Food Microbiol. 2017, 66, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Seixas, I.; Barbosa, C.; Mendes-Faia, A.; Güldener, U.; Tenreiro, R.; Mendes-Ferreira, A.; Mira, N.P. Genome sequence of the non-conventional wine yeast Hanseniaspora guilliermondii UTAD222 unveils relevant traits of this species and of the Hanseniaspora genus in the context of wine fermentation. DNA Res. 2019, 26, 67–83. [Google Scholar] [CrossRef] [PubMed]
- Kuanyshev, N.; Adamo, G.M.; Porro, D.; Branduardi, P. The spoilage yeast Zygosaccharomyces bailii: Foe or friend? Yeast 2017, 34, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Cabral, S.; Prista, C.; Loureiro-Dias, M.C.; Leandro, M.J. Occurrence of FFZ genes in yeasts and correlation with fructophilic behaviour. Microbiology 2015, 161, 2008–2018. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.W.; Phaff, H.J. A comparative study of the apiculate yeasts. Mycopathol. Mycol. Appl. 1958, 10, 113–141. [Google Scholar] [CrossRef] [PubMed]
- Beijerinck, M.W. L’auxanographie, ou la méthode de l’hydrodiffusion dans la gélatine appliquée aux recherches microbiologiques. Arch. Néerl. Sci. Exactes Nat. 1889, 23, 367–372. [Google Scholar]
- Kluyver, A.J. Biochemische Suikerbepalingen. Ph.D. Thesis, Delft University of Technology, Delft, The Netherland, 1914. [Google Scholar]
- Konno, H.; van Dijken, J.P.; Scheffers, W.A. Fructose fermentation in yeasts. Anton. Leeuwen. 1985, 51, 559. [Google Scholar] [CrossRef]
- Neumann, N.P.; Lampen, J.O. Purification and properties of yeast invertase. Biochemistry 1967, 6, 468–475. [Google Scholar] [CrossRef]
- Gottschalk, A. The mechanism of selective fermentation of d-fructose from invert sugar by Sauternes yeast. Biochem. J. 1946, 40, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Poomtien, J.; Jindamorakot, S.; Limtong, S.; Pinphanichakarn, P.; Thaniyavarn, J. Two new anamorphic yeasts species, Cyberlindnera samutprakarnensis sp. nov. and Candida thasaenensis sp. nov., isolated from industrial wastes in Thailand. Anton. Leeuwen. 2013, 103, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Brysch-Herzberg, M.; Wohlmann, E.; Fischer, R. Zygosaccharomyces seidelii sp. nov. a new yeast species from the Maldives, and a revisit of the single-strain species debate. Anton. Leeuwen. 2020, 113, 427–436. [Google Scholar] [CrossRef]
- Emmerich, W.; Radler, F. The anaerobic metabolism of glucose and fructose by Saccharomyces bailii. J. Gen. Microbiol. 1983, 129, 3311–3318. [Google Scholar] [CrossRef]
- Sousa-Dias, S.; Gonçalves, T.; Leyva, J.S.; Peinado, J.M.; Loureiro-Dias, M.C. Kinetics and regulation of fructose and glucose transport systems are responsible for fructophily in Zygosaccharomyces bailii. Microbiology 1996, 142, 1733–1738. [Google Scholar] [CrossRef]
- Leandro, M.J.; Sychrová, H.; Prista, C.; Loureiro-Dias, M.C. The osmotolerant fructophilic yeast Zygosaccharomyces rouxii employs two plasma membrane fructose uptake systems belonging to a new family of yeast sugar transporters. Microbiology 2011, 157, 601–608. [Google Scholar] [CrossRef]
- Leandro, M.J.; Cabral, S.; Prista, C.; Loureiro-Dias, M.C.; Sychrová, H. The high-capacity specific fructose facilitator ZrFfz1 is essential for the fructophilic behavior of Zygosaccharomyces rouxii CBS 732T. Eukaryot. Cell 2014, 13, 1371–1379. [Google Scholar] [CrossRef] [PubMed]
- Wieczorke, R.; Krampe, S.; Weierstall, T.; Freidel, K.; Hollenberg, C.P.; Boles, E. Concurrent knock-out of at least 20 transporter genes is required to block uptake of hexoses in Saccharomyces cerevisiae. FEBS Lett. 1999, 464, 123–128. [Google Scholar] [CrossRef]
- Boles, E.; Hollenberg, C.P. The molecular genetics of hexose transport in yeasts. FEMS Microbiol. Rev. 1997, 21, 85–111. [Google Scholar] [CrossRef] [PubMed]
- Kruckeberg, A.L.; Dickinson, J.R. Carbon metabolism. In Metabolism and Molecular Physiology of Saccharomyces Cerevisiae, 2nd ed.; Dickinson, J.R., Schweizer, M., Eds.; CRC Press: Boca Raton, FL, USA, 2004; pp. 42–103. [Google Scholar] [CrossRef]
- Reifenberger, E.; Freidel, K.; Ciriacy, M. Identification of novel HXT genes in Saccharomyces cerevisiae reveals the impact of individual hexose transporters on glycolytic flux. Mol. Microbiol. 1995, 16, 157–167. [Google Scholar] [CrossRef] [PubMed]
- Entian, K.-D. Sugar phosphorylation in yeast. In Yeast Sugar Metabolism; Zimmerman, F.K., Entian, K.-D., Eds.; Technomic Publishing Company: Lancaster, PA, USA, 1997; pp. 67–79. [Google Scholar]
- Diezemann, A.; Boles, E. Functional characterization of the Frt1 sugar transporter and of fructose uptake in Kluyveromyces lactis. Curr. Genet. 2003, 43, 281–288. [Google Scholar] [CrossRef] [PubMed]
| Species | JCM no. | Assimilation | Fermentation | ||||
|---|---|---|---|---|---|---|---|
| Glucose | Fructose | Sucrose | Glucose | Fructose | Sucrose | ||
| Saccharomycetes, Saccharomycotina | |||||||
| Saccharomycetales | |||||||
| Cephaloascaceae | |||||||
| Cephaloascus fragrans | 7613 | + | + | - | - | - 1 | - 1 |
| Debaryomycetaceae | |||||||
| Candida aaseri | 1689 | + | + | +/s | - | - | - |
| Candida albicans | 1542 | +ST | +ST | +ST | +ST | +ST | - |
| Candida atlantica | 9548 | +ST | +ST | +ST | w/- | w/- | - |
| Candida atmosphaerica | 9549 | +ST | +ST | +ST | + | + | - |
| Candida boleticola | 1500 | + | + | - | +ST | +ST | - |
| Candida buinensis | 9453 | +ST | +ST | +ST | +ST | +ST | - |
| Candida conglobata | 2373 | + | + | - | +ST | +ST | - |
| Candida dendronema | 1803 | + | + | + | +ST | +ST | - |
| Candida diddensiae | 9598 | + | + | + | + | + | - |
| Candida fluviatilis | 9552 | + | + | + | + | + | - |
| Candida friedrichii | 9553 | + | + | + | + | + | - |
| Candida glaebosa | 1590 | +ST | +ST | + | - | - 1 | - 1 |
| Candida insectamans | 9611 | + | + | - | - | - | - |
| Candida insectorum | 9457 | + | + | + | + | + | - |
| Candida lyxosophila | 7532 | + | + | + | +ST | + | - |
| Candida maltosa | 1504 | +ST | +ST | + | +ST | +ST | +ST |
| Candida multigemmis | 9559 | + | + | + | + | + | - |
| Candida oleophila | 1620 | + | + | + | +ST | +ST | - |
| Candida palmioleophila | 5218 | + | + | + | - | - 1 | - 1 |
| Candida membranifaciens | 9450 | +ST | +ST | +ST | + | + | + |
| Candida naeodendra | 1509 | + | + | + | +ST | +ST | - |
| Candida neustonensis | 14892 | +ST | +ST | +ST | + | + | +/s |
| Candida parapsilosis | 1612 | +ST | + | + | +ST | +ST | - |
| Candida pseudoglaebosa | 2168 | + | + | + | + | + | - |
| Candida saitoana | 1438 | +ST | + | + | - | - | - |
| Candida santamariae | 1816 | + | + | - | +ST | +ST | - |
| Candida schatavii | 1778 | +ST | +ST | - | s | s | - |
| Candida sojae | 1644 | + | + | + | +ST | +ST | + |
| Candida tammaniensis | 10730 | + | + | + | +ST | +ST | - |
| Candida thasaenensis | 17817 | + | + | + | +ST | +ST | + |
| Candida tropicalis | 1541 | +ST | +ST | +ST | +ST | +ST | +ST |
| Candida trypodendroni | 10731 | + | + | + | + | + | - |
| Candida psychrophila | 2388 | + | + | - | - | - 1 | - 1 |
| Candida viswanathii | 9567 | + | + | + | +ST | +ST | - |
| Candida xestobii | 9569 | + | + | + | - | - | - |
| Candida zeylanoides | 1627 | + | + | - | - | - | - |
| Danielozyma ontarioensis | 10729 | + | + | + | + | + | + |
| Debaryomyces coudertii | 2387 | + | + | - | + | + | - |
| Debaryomyces hansenii | 1990 | +ST | +ST | +ST | +/s | +/s | +/s |
| Debaryomyces maramus | 1528 | + | + | + | w/- | w/- | w/- |
| Debaryomyces nepalensis | 2095 | +ST | +ST | +ST | + | + | + |
| Debaryomyces prosopidis | 9913 | + | + | + | +/s | s/w | s/w |
| Debaryomyces udenii | 7855 | +ST | +ST | +ST | +/d | + | d/w |
| Kurtzmaniella fragi | 1791 | +ST | +ST | +ST | +ST | +ST | - |
| Kurtzmaniella natalensis | 1445 | + | + | + | +ST | +ST | - |
| Kurtzmaniella quercitrusa | 9832 | + | + | + | +ST | +ST | - |
| Lodderomyces elongisporus | 1781 | +ST | +ST | +ST | + | + | - |
| Meyerozyma guilliermondii | 10735 | +ST | +ST | +ST | + | + | + |
| Millerozyma acaciae | 10732 | +ST | +ST | - | +ST | +ST | - |
| Millerozyma farinosa | 10734 | +ST | +ST | - | +ST | +ST | - |
| Millerozyma koratensis | 12576 | +ST | +ST | +ST | + | + | + |
| Priceomyces carsonii | 8121 | +ST | +ST | + | - | - 1 | - 1 |
| Priceomyces castillae | 10733 | + | +ST | - | - | - | - |
| Priceomyces fermenticarens | 9589 | +ST | +ST | - | - | - | - |
| Priceomyces haplophilus | 1635 | +ST | +ST | - | - | - 1 | - 1 |
| Priceomyces medius | 10737 | + | + | - | - | - | - |
| Priceomyces melissophilus | 1707 | +ST | +ST | +ST | - | - 1 | - 1 |
| Scheffersomyces coipomensis | 8916 | +ST | +ST | + | +ST | +ST | - |
| Scheffersomyces ergatensis | 9599 | +ST | +ST | + | +ST | + | - |
| Scheffersomyces insectosa | 9842 | + | + | + | +ST | +ST | - |
| Scheffersomyces lignosum | 9837 | +ST | + | + | +ST | +ST | - |
| Scheffersomyces segobiensis | 10740 | + | + | + | +ST | +ST | - |
| Scheffersomyces shehatae | 9840 | + | + | s | +ST | +ST | - |
| Scheffersomyces spartiniae | 10741 | + | + | + | +ST | +ST | - |
| Scheffersomyces stipitis | 10742 | + | + | + | +ST | +ST | - |
| Schwanniomyces capriottii | 6177 | +ST | +ST | +ST | +ST | +ST | +ST |
| Schwanniomyces etchellsii | 3656 | +ST | +ST | +ST | +ST | +ST | - |
| Schwanniomyces occidentalis var. occidentalis | 8123 | +ST | +ST | +ST | +ST | + | + |
| Schwanniomyces occidentalis var. persoonii | 8127 | + | + | + | + | + | + |
| Schwanniomyces polymorphus var. africanus | 7443 | +ST | +ST | +ST | +ST | +ST | +ST |
| Schwanniomyces polymorphus var. polymorphus | 3647 | +ST | +ST | +ST | +ST | +ST | + |
| Schwanniomyces pseudopolymorphus | 3652 | +ST | +ST | +ST | +ST | +ST | +ST |
| Schwanniomyces vanrijiae | 3657 | +ST | +ST | +ST | w | w | w |
| Schwanniomyces yamadae | 6191 | + | + | + | +ST | +ST | - |
| Wickerhamia fluorescens | 1821 | + | + | + | +ST | +ST | + |
| Yamadazyma akitaensis | 10738 | +ST | +ST | +ST | + | + | - |
| Yamadazyma kitorensis | 31005 | +ST | +ST | s | + | + | - |
| Yamadazyma mexicana | 1835 | +ST | +ST | +ST | + | + | - |
| Yamadazyma nakazawae | 7529 | +ST | +ST | +ST | +ST | +ST | - |
| Yamadazyma philogaea | 10739 | + | + | + | + | + | - |
| Yamadazyma scolyti | 3654 | + | + | + | + | + | - |
| Yamadazyma takamatsuzukensis | 15410 | + | + | + | + | + | - |
| Yamadazyma tenuis | 9827 | +ST | + | + | + | + | - |
| Yamadazyma triangularis | 9449 | + | + | + | w/- | w/- | - |
| Yamadazyma tumulicola | 15403 | +ST | +ST | + | + | + | - |
| Dipodascaceae | |||||||
| Dipodascus aggregatus | 31687 | + | + | - | - | - | - |
| Dipodascus australiensis | 31688 | + | + | - | - | - | - |
| Dipodascus eriense | 3912 | + | + | - | +/s | w/- | - |
| Dipodascus fermentans | 2468 | + | + | - | +ST | +ST | - |
| Dipodascus ingens | 9471 | + | + | - | - | - | - |
| Dipodascus ovetensis | 3706 | + | + | - | - | - | - |
| Dipodascus reessii | 1943 | + | + | - | - | - 1 | - 1 |
| Dipodascus tetrasperma | 6361 | +ST | +ST | - | +ST | +ST | - |
| Geotrichum rectangulatum | 1750 | +ST | + | - | +ST | +ST | - |
| Lipomycetaceae | |||||||
| Babjevia anomala | 5988 | + | + | - | - | - 1 | - 1 |
| Lipomyces kononenkoae | 5989 | + | + | + | - | - | - |
| Lipomyces lipofer | 3769 | + | + | + | - | - | - |
| Lipomyces smithiae | 8928 | + | + | + | - | - 1 | - 1 |
| Lipomyces spencermartinsiae | 5990 | + | + | + | - | - | - |
| Lipomyces starkeyi | 5995 | + | + | + | - | - | - |
| Lipomyces suomiensis | 7660 | + | + | - | - | - 1 | - 1 |
| Lipomyces tetrasporus | 6000 | + | + | + | - | - 1 | - 1 |
| Myxozyma geophila | 5220 | + | + | - | - | - 1 | - 1 |
| Myxozyma kluyveri | 7661 | + | + | w | - | - 1 | - 1 |
| Myxozyma lipomycoides | 5198 | + | + | - | - | - 1 | - 1 |
| Myxozyma melibiosi | 5194 | + | + | - | - | - 1 | - 1 |
| Myxozyma mucilagina | 1834 | + | + | + | - | - 1 | - 1 |
| Myxozyma neglecta | 5197 | + | + | - | - | - 1 | - 1 |
| Myxozyma udenii | 8927 | + | + | + | - | - 1 | - 1 |
| Metschnikowiaceae | |||||||
| Aciculoconidium aculeatum | 13354 | + | + | + | s | s | - |
| Clavispora fructus | 1513 | + | + | - | +ST | +ST | - |
| Clavispora lusitaniae | 7533 | + | + | + | +ST | +ST | - |
| Candida akabanensis | 9115 | +ST | +ST | +ST | +ST | +ST | +ST |
| Candida auris | 15448 | +ST | +ST | +ST | + | + | + |
| Candida haemulonii | 3762 | +ST | +ST | +ST | +ST | +ST | + |
| Candida intermedia | 1607 | +ST | + | + | +ST | +ST | +ST |
| Candida melibiosica | 9558 | + | + | + | +ST | +ST | - |
| Candida mogii | 1611 | + | + | + | +ST | +ST | +ST |
| Candida pseudointermedia | 1592 | +ST | +ST | +ST | +ST | +ST | +ST |
| Candida fukazawae | 1641 | + | + | + | +ST | +ST | - |
| Candida fungicola | 10142 | +ST | +ST | +ST | - | - | - |
| Candida mesenterica | 2368 | + | + | + | - | - | - |
| Candida musae | 1598 | + | + | + | +ST | +ST | - |
| Candida oregonensis | 1811 | +ST | +ST | + | +ST | +ST | - |
| Candida pseudohaemulonii | 12453 | +ST | +ST | +ST | +ST | + | + |
| Candida tsuchiyae | 1638 | +ST | +ST | +ST | +ST | +ST | + |
| Hyphopichia burtonii | 3708 | + | + | + | + | + | + |
| Hyphopichia fennica | 9849 | + | + | + | +ST | +ST | + |
| Hyphopichia gotoi | 10145 | + | + | + | +ST | +ST | +ST |
| Hyphopichia homilentoma | 1507 | +ST | + | + | +ST | +ST | - |
| Hyphopichia khmerensis | 13262 | +ST | +ST | +ST | + | + | + |
| Hyphopichia pseudoburtonii | 16346 | + | + | + | +ST | +ST | + |
| Hyphopichia rhagii | 9839 | +ST | +ST | +ST | +ST | +ST | +ST |
| Metschnikowia agaves | 31832 | + | + | + | +/s | +/s | - |
| Metschnikowia kofuensis | 12563 | + | + | +/s | + | + | - |
| Metschnikowia lunata | 1798 | + | + | + | +ST | +ST | - |
| Metschnikowia reukaufii | 7534 | + | + | + | + | + | - |
| Metschnikowia torresii | 1845 | +ST | +ST | - | +ST | +ST | - |
| Metschnikowia viticola | 12561 | + | + | + | + | + | - |
| Phaffomycetaceae | |||||||
| Komagataella pastoris | 3650 | +ST | +ST | - | +ST | +ST | - |
| Phaffomyces opuntiae | 1836 | + | + | - | - | - 1 | - 1 |
| Phaffomyces thermotolerans | 1837 | + | + | - | - | - 1 | - 1 |
| Pichiaceae | |||||||
| Candida ethanolica | 9588 | + | + | - | s/- | s/- | - |
| Candida inconspicua | 9555 | + | + | - | + | + | - |
| Candida pseudolambica | 9830 | +ST | +ST | - | +ST | +ST | - |
| Candida rugopelliculosa | 1593 | + | + | - | +ST | +ST | - |
| Candida silvatica | 9828 | + | + | - | - | - | - |
| Dekkera anomala | 31686 | +ST | +ST | + | +ST | +ST | + |
| Dekkera bruxellensis | 11407 | + | + | + | +ST | +ST | +ST |
| Kregervanrija fluxuum | 3646 | + | + | - | +/s | +/s | - |
| Pichia cactophila | 1830 | + | + | - | +/s | +/w | - |
| Pichia exigua | 1829 | + | + | - | w | w | - |
| Pichia heedii | 1833 | + | + | - | +/w | +/w | - |
| Pichia kluyveri var. kluyveri | 11403 | + | + | - | +ST | +ST | - |
| Pichia membranifaciens | 1442 | + | + | - | w | w | - |
| Pichia myanmarensis | 12922 | +ST | +ST | +ST | +ST | +ST | + |
| Pichia nakasei | 1699 | +ST | +ST | - | +ST | +ST | - |
| Pichia occidentalis | 1711 | +ST | + | - | +ST | +ST | - |
| Pichia rarassimilans | 14993 | + | + | - | s | s | - |
| Pichia terricola | 1709 | +ST | + | - | + | + | - |
| Saturnispora ahearnii | 10726 | + | +ST | - | +ST | +ST | - |
| Saturnispora besseyi | 1706 | +ST | + | - | +ST | +ST | - |
| Saturnispora dispora | 1795 | + | + | - | +ST | +ST | - |
| Saturnispora diversa | 1848 | + | + | - | +ST | +ST | - |
| Saturnispora saitoi | 1793 | +ST | +ST | - | +ST | +ST | - |
| Saturnispora silvae | 6352 | +ST | + | - | +/s | +/s | - |
| Saturnispora zaruensis | 1515 | +ST | + | - | +ST | +ST | - |
| Saccharomycetaceae | |||||||
| Candida castellii | 9550 | + | + | - | +ST | +ST | - |
| Candida glabrata | 3761 | + | + | - | +ST | +ST | - |
| Issatchenkia orientalis | 1710 | +ST | +ST | - | +ST | +ST | - |
| Kazachstania aerobia | 31691 | + | + | - | +ST | +ST | - |
| Kazachstania bulderi | 31689 | + | + | + | +ST | +ST | + |
| Kazachstania exigua | 1790 | + | + | + | +ST | +ST | + |
| Kazachstania humilis | 9852 | + | + | - | +ST | +ST | - |
| Kazachstania servazzii | 5179 | + | + | - | +ST | +ST | - |
| Kazachstania telluris | 5298 | + | + | - | +ST | +/w | - |
| Kazachstania transvaalensis | 5178 | + | + | - | +ST | +ST | - |
| Kazachstania unispora | 5180 | + | + | - | +ST | +ST | - |
| Kluyveromyces marxianus | 9556 | + | + | + | +ST | +ST | +ST |
| Kluyveromyces nonfermentans | 10232 | + | + | - | - | - | - |
| Lachancea kluyveri | 7257 | + | + | + | +ST | +ST | +ST |
| Lachancea thermotolerans | 19085 | + | + | + | +ST | +ST | + |
| Lachancea waltii | 10745 | + | + | + | +ST | +ST | +ST |
| Saccharomyces bayanus | 7258 | + | + | + | +ST | +ST | +ST |
| Saccharomyces cerevisiae | 7255 | + | + | + | +ST | +ST | +ST |
| Saccharomyces pastorianus | 7256 | + | + | + | +ST | +ST | +ST |
| Tetrapisispora arboricola | 10813 | + | + | - | +ST | +ST | - |
| Tetrapisispora iriomotensis | 10810 | + | + | - | +ST | +ST | - |
| Tetrapisispora namnaoensis | 12664 | + | + | - | +ST | +ST | - |
| Tetrapisispora nanseiensis | 10811 | + | + | - | +ST | +ST | - |
| Torulaspora delbrueckii | 31684 | + | + | - | +ST | +ST | - |
| Torulaspora pretoriensis | 3662 | + | + | + | +ST | +ST | + |
| Zygosaccharomyces rouxii | 7619 | + | + | w/- | +ST | +ST | s |
| Zygosaccharomyces rouxii | 22060 | + | + | - | +ST | +ST | - |
| Zygosaccharomyces siamensis | 16825 | + | + | - | +ST | +ST | s/w |
| Zygotorulaspora mrakii | 1800 | + | + | + | +ST | +ST | + |
| Saccharomycodaceae | |||||||
| Hanseniaspora opuntiae | 31690 | + | + | - | +ST | +ST | - |
| Saccharomycopsidaceae | |||||||
| Candida fragicola | 1589 | + | + | - | +ST | +ST | - |
| Saccharomycopsis capsularis | 7619 | + | + | w/- | +ST | +ST | s |
| Saccharomycopsis crataegensis | 1700 | + | + | - | + | + | - |
| Saccharomycopsis fibuligera | 7609 | + | + | + | +ST | +ST | + |
| Saccharomycopsis javanensis | 3707 | + | + | - | - | - 1 | - 1 |
| Saccharomycopsis malanga | 7620 | + | + | - | +/s | + | - |
| Saccharomycopsis selenospora | 7616 | + | + | - | - | - | - |
| Saccharomycopsis synnaedendra | 7607 | + | + | - | - | - 1 | - 1 |
| Saccharomycopsis vini | 7623 | + | + | + | + | + | + |
| Trichomonascaceae | |||||||
| Blastobotrys adeninivorans | 8914 | +ST | +ST | +ST | +ST | + | +/s |
| Blastobotrys arbuscula | 2926 | + | + | - | +ST | + | - |
| Blastobotrys aristata | 2929 | + | + | s | +ST | +ST | - |
| Blastobotrys capitulata | 2934 | + | + | - | +ST | +ST | - |
| Blastobotrys chiropterorum | 9597 | + | + | + | - | - | - |
| Blastobotrys elegans | 2931 | + | + | - | + | +/w | - |
| Blastobotrys gigas | 2927 | + | + | - | +ST | + | - |
| Blastobotrys nivea | 2933 | + | + | s | + | + | - |
| Blastobotrys parvus | 9487 | + | + | s | - | - | - |
| Blastobotrys proliferans | 2928 | + | + | + | + | + | - |
| Blastobotrys terrestris | 8913 | + | + | + | - | - | - |
| Candida santjacobensis | 8924 | + | + | + | + | + | - |
| Groenewaldozyma auringiensis | 9593 | + | + | - | +ST | + | - |
| Groenewaldozyma salmanticensis | 8896 | + | + | + | +ST | +ST | +ST |
| Middelhovenomyces petrohuensis | 8922 | + | + | + | - | - | - |
| Middelhovenomyces tepae | 10265 | + | + | s | w | - | - |
| Sugiyamaella castrensis | 9585 | + | + | + | w/- | - | - |
| Sugiyamaella paludigena | 9614 | + | + | + | - | - | - |
| Sugiyamaella valdiviana | 9565 | + | + | + | +/w | w/- | - |
| Trichomonascus ciferrii | 7621 | + | + | + | - | - | - |
| Wickerhamiella azyma | 1691 | + | + | + | - | - | - |
| Wickerhamiella domercqiae | 9478 | + | + | +/w | - | - 1 | - 1 |
| Wickerhamiella galacta | 8257 | + | + | - | - | - | - |
| Wickerhamiella hasegawae | 12559 | + | + | - | + | +/w | - |
| Wickerhamiella kazuoi | 12558 | + | + | - | - | - | - |
| Wickerhamiella pararugosa | 1512 | + | + | - | - | - 1 | - 1 |
| Wickerhamiella sorbophila | 1514 | + | + | - | - | - | - |
| Wickerhamiella spandovensis | 9562 | + | + | + | +ST | w | w |
| Wickerhamiella vanderwaltii | 9615 | + | + | - | - | - | - |
| Wickerhamiella versatilis | 8065 | + | + | + | +ST | +ST | +ST |
| Zygoascus biomembranicola | 31007 | + | + | - | +ST | + | - |
| Wickerhamomycetaceae | |||||||
| Barnettozyma salicaria | 3653 | + | + | - | - | - | - 1 |
| Barnettozyma wickerhamii | 21961 | +ST | +ST | +ST | + | + | - |
| Candida berthetii | 9594 | + | + | - | + | + | - |
| Candida danieliae | 17247 | +ST | +ST | + | +ST | + | - |
| Candida dendrica | 9605 | + | + | - | + | +/s | - |
| Candida easanensis | 12476 | + | + | +/s | + | + | - |
| Candida eppingiae | 17241 | +ST | +ST | +ST | +ST | +ST | - |
| Candida freyschussii | 9850 | + | + | +/s | + | + | - |
| Candida maritima | 9612 | + | + | + | + | + | + |
| Candida montana | 2323 | + | + | - | - | - 1 | - 1 |
| Candida nakhonratchasimensis | 12474 | + | + | + | +ST | +ST | +ST |
| Candida norvegica | 8897 | + | + | - | - | - | - |
| Candida pattaniensis | 12475 | +ST | +ST | +ST | +ST | +ST | + |
| Candida pseudoflosculorum | 17242 | +ST | +ST | +ST | +ST | +ST | +ST |
| Candida quercuum | 1587 | +ST | + | + | +/w | + | - |
| Candida robnettiae | 17243 | +ST | +ST | +ST | +ST | +ST | - |
| Candida silvicultrix | 9831 | + | + | + | +ST | +ST | +ST |
| Candida solani | 2339 | + | + | + | +ST | +ST | - |
| Candida vartiovaarae | 3759 | + | + | + | +ST | +ST | + |
| Cyberlindnera americana | 3592 | + | + | + | - | +/s | - |
| Cyberlindnera americana | 3593 | + | + | + | w | s | - |
| Cyberlindnera amylophila | 1702 | + | + | + | +ST | +ST | - |
| Cyberlindnera bimundalis | 3591 | + | + | + | +/s | +/s | - |
| Cyberlindnera fabianii | 3601 | +ST | + | + | +ST | +ST | +ST |
| Cyberlindnera jadinii | 3617 | + | + | + | +ST | +ST | +ST |
| Cyberlindnera japonica | 11402 | + | + | + | s | s/w | - |
| Cyberlindnera mississippiensis | 1703 | +ST | + | + | +ST | +ST | - |
| Cyberlindnera mrakii | 3614 | + | + | - | +ST | +ST | - |
| Cyberlindnera petersonii | 3619 | + | + | + | + | + | + |
| Cyberlindnera rhizosphaerae | 16499 | + | + | s | + | + | + |
| Cyberlindnera rhodanensis | 3649 | +ST | +ST | +ST | +ST | +ST | - |
| Cyberlindnera samutprakarnensis | 17816 | + | + | + | +ST | +ST | +ST |
| Cyberlindnera subsufficiens | 3625 | + | + | + | + | + | + |
| Starmera amethionina | 1831 | + | + | - | s | s | - |
| Starmera pachycereana | 1832 | + | + | - | - | - | - |
| Starmera quercuum | 3659 | + | + | - | + | + | - |
| Starmera stellimalicola | 3546 | + | + | - | +ST | +ST | - |
| Wickerhamomyces anomalus | 3585 | +ST | +ST | +ST | +ST | +ST | +ST |
| Wickerhamomyces bisporus | 3590 | + | + | + | w | w | - |
| Wickerhamomyces bovis | 3640 | +ST | +ST | +ST | +ST | +ST | - |
| Wickerhamomyces canadensis | 3597 | +ST | +ST | +ST | - | - 1 | - 1 |
| Wickerhamomyces chaumierensis | 17246 | +ST | +ST | +ST | +ST | +ST | - |
| Wickerhamomyces ciferrii | 3599 | +ST | +ST | +ST | + | +ST | + |
| Wickerhamomyces mucosus | 6814 | + | + | + | +ST | +ST | - |
| Wickerhamomyces patagonicus | 16381 | + | + | - | - | - | - |
| Wickerhamomyces pijperi | 11406 | + | + | - | +ST | +ST | - |
| Wickerhamomyces silvicola | 3627 | +ST | +ST | + | +ST | +ST | - |
| Wickerhamomyces subpelliculosus | 3631 | +ST | +ST | + | +ST | +ST | +ST |
| Wickerhamomyces sydowiorum | 9455 | +ST | +ST | +ST | + | +ST | + |
| Saccharomycetales incertae sedis | |||||||
| Ambrosiozyma cicatricosa | 7598 | +ST | + | + | +/s | +/s | +/s |
| Ambrosiozyma kamigamensis | 14990 | + | + | +/s | + | + | - |
| Ambrosiozyma kashinagicola | 15019 | + | + | - | + | + | - |
| Ambrosiozyma llanquihuensis | 8918 | + | + | - | + | + | - |
| Ambrosiozyma monospora | 7599 | + | + | + | +ST | +ST | +/w |
| Ambrosiozyma neoplatypodis | 14992 | + | + | - | + | + | - |
| Ambrosiozyma oregonensis | 1797 | +ST | + | + | + | + | - |
| Ambrosiozyma philentoma | 7600 | + | + | + | +/d | + | d/w |
| Ambrosiozyma platypodis | 1843 | + | + | + | w | + | - |
| Ambrosiozyma platypodis | 1796 | + | + | + | +/s | + | - |
| Ambrosiozyma pseudovanderkliftii | 15025 | + | + | s | + | + | - |
| Ambrosiozyma vanderkliftii | 15029 | +ST | +ST | + | +ST | +ST | w |
| Babjeviella inositovora | 10736 | + | + | + | - | - | - |
| Candida arabinofermentans | 10727 | + | + | - | + | + | - |
| Candida blankii | 8259 | + | + | + | +/s | w | w |
| Candida boidinii | 9604 | + | + | - | +ST | +ST | - |
| Candida chilensis | 1693 | + | + | + | + | + | - |
| Candida cylindracea | 9586 | + | + | - | + | + | - |
| Candida digboiensis | 12330 | + | + | + | - | - | - |
| Candida entomophila | 9607 | + | + | + | +ST | +ST | +ST |
| Candida incommunis | 8258 | + | + | + | + | + | - |
| Candida insectalens | 9610 | + | + | - | - | - | - |
| Candida krabiensis | 12266 | + | + | - | - | - | - |
| Candida maris | 9853 | + | + | - | - | - | - |
| Candida methanosorbosa | 9620 | + | + | - | + | + | - |
| Candida nanaspora | 9590 | + | + | - | +ST | +ST | - |
| Candida nemodendra | 9855 | + | + | - | s/- | w/- | - |
| Candida nitratophila | 9856 | + | + | - | + | + | - |
| Candida ovalis | 9444 | + | + | - | +ST | +ST | - |
| Candida pini | 9826 | + | +/s | - | +/s | +/s | - |
| Candida sake | 2951 | +ST | + | + | +ST | +ST | - |
| Candida savonica | 9561 | + | + | - | + | + | - |
| Candida sequanensis | 9841 | + | + | - | +ST | +ST | - |
| Candida silvanorum | 1804 | + | + | + | +ST | +ST | - |
| Candida sithepensis | 12265 | + | + | - | +ST | +ST | - |
| Candida sonorensis | 1827 | + | + | - | +ST | +ST | - |
| Candida sophiae-reginae | 8925 | +ST | +ST | + | +ST | +ST | - |
| Candida sorboxylosa | 1536 | +ST | +ST | - | + | + | - |
| Candida succiphila | 9445 | + | + | - | +ST | +ST | - |
| Citeromyces matritensis | 2333 | + | + | + | +ST | +ST | +ST |
| Citeromyces siamensis | 11522 | + | + | + | +ST | +ST | +ST |
| Diutina catenulata | 1604 | +ST | + | - | + | + | - |
| Diutina rugosa | 1619 | +ST | + | - | - | - 1 | - 1 |
| Kuraishia capsulata | 1991 | + | + | - | + | + | - |
| Nadsonia commutata | 10138 | + | + | - | - | - 1 | - 1 |
| Nadsonia fulvescens var. fulvescens | 9992 | + | + | - | +ST | +ST | - |
| Nadsonia starkeyi-henricii | 11408 | + | + | - | - | - | - |
| Nakazawaea anatomiae | 9547 | + | + | - | + | + | - |
| Nakazawaea holstii | 3608 | + | + | +/s | +ST | +ST | - |
| Nakazawaea ishiwadae | 9451 | + | + | + | +ST | +ST | - |
| Nakazawaea peltata | 9829 | + | + | + | +ST | +ST | - |
| Nakazawaea populi | 9833 | + | + | s | +ST | +ST | - |
| Nakazawaea wickerhamii | 9568 | + | + | - | +ST | +ST | - |
| Ogataea angusta | 3635 | + | + | + | +ST | +ST | - |
| Ogataea glucozyma | 3607 | + | + | - | +ST | +ST | - |
| Ogataea henricii | 3611 | + | + | - | - | - 1 | - 1 |
| Ogataea kodamae | 11404 | + | + | - | +/s | +/s | - |
| Ogataea methanolica | 10240 | + | + | - | +ST | +ST | - |
| Ogataea methylivora | 22142 | + | + | + | + | + | - |
| Ogataea minuta | 3622 | + | + | - | + | +ST | - |
| Ogataea naganishii | 22078 | + | + | + | + | + | - |
| Ogataea nonfermentans | 3615 | + | + | - | - | - | - 1 |
| Ogataea philodendri | 22070 | + | + | - | - | - | - |
| Ogataea pignaliae | 9836 | + | + | - | +ST | +ST | - |
| Ogataea pini | 3655 | + | + | - | s/- | s/- | - |
| Ogataea salicorniae | 10744 | + | + | - | +ST | +ST | - |
| Ogataea siamensis | 12264 | + | + | + | +/s | +/s | - |
| Ogataea thermomethanolica | 12984 | + | + | + | + | + | - |
| Ogataea trehalophila | 3651 | +ST | +ST | - | +ST | +ST | - |
| Pachysolen tannophilus | 31685 | + | + | - | +ST | +ST | - |
| Peterozyma toletana | 3658 | +ST | + | + | + | + | - |
| Saprochaete japonica | 2451 | +ST | +ST | - | +ST | +ST | - |
| Sporopachydermia cereana | 9480 | + | + | - | - | - | - |
| Sporopachydermia lactativora | 9485 | + | + | - | - | - 1 | - 1 |
| Sporopachydermia quercuum | 9486 | + | + | - | + | - | - |
| Starmerella apicola | 9592 | + | + | + | + | + | + |
| Starmerella apis | 8256 | +ST | +ST | + | - | w | - |
| Starmerella bombi | 9595 | + | + | + | +ST | +ST | + |
| Starmerella bombicola | 9596 | + | + | + | +ST | +ST | +ST |
| Starmerella etchellsii | 8066 | + | + | - | + | s/w | - |
| Starmerella floricola | 9439 | + | + | + | +ST | +ST | +ST |
| Starmerella geochares | 9851 | + | + | + | + | + | +/s |
| Starmerella gropengiesseri | 8255 | + | + | + | + | + | w |
| Starmerella lactis-condensi | 9472 | + | + | + | +ST | +ST | +ST |
| Starmerella magnoliae | 1446 | + | + | + | + | + | + |
| Starmerella stellata | 9476 | + | + | + | +ST | +ST | +ST |
| Starmerella vaccinii | 9446 | + | + | + | + | + | + |
| Suhomyces tanzawaensis | 1648 | + | + | + | s/w | +/w | - |
| Teunomyces kruisii | 1779 | + | + | + | +ST | +ST | - |
| Trigonopsis cantarellii | 8260 | + | + | - | +ST | +ST | - |
| Trigonopsis variabilis | 1823 | + | + | - | - | - 1 | - 1 |
| Trigonopsis vinaria | 1813 | + | + | - | - | - 1 | - 1 |
| Yarrowia deformans | 1694 | + | + | - | - | - 1 | - 1 |
| Yarrowia keelungensis | 14894 | +ST | +ST | - | - | - | - |
| Yarrowia lipolytica | 2320 | +ST | +ST | - | - | - 1 | - 1 |
| Yarrowia yakushimensis | 12782 | +ST | +ST | - | - | - | - |
| Taphrinomycotina | |||||||
| Saitoella complicata | 7358 | + | + | + | - | - | - |
| Schizosaccharomyces japonicus | 8264 | + | + | + | +ST | +ST | +ST |
| Schizosaccharomyces octosporus | 8261 | + | + | w | + | + | w |
| Schizosaccharomyces pombe | 8274 | + | + | + | +ST | +ST | +ST |
| Pezizomycotina | |||||||
| Trichosporiella flavificans | 1506 | + | + | - | +ST | +ST | - |
| Sugars | This Study | The Yeasts * | ||
|---|---|---|---|---|
| Assimilation | Fermentation | Assimilation | Fermentation | |
| Glucose | 100% (388/388) | 77.8% (302/388) | 100% (827/827) | 72.8% (602/827) |
| Fructose | 100% (388/388) | 77.8% (302/388) | nd | nd |
| Sucrose | 59.0% (229/388) | 25.5% (99/388) | 60.7% (502/827) | 24.2% (200/827) |
| Galactose | nt | nt | 65.4% (541/827) | 30.6% (253/827) |
| Trehalose | nt | nt | 70.0% (579/827) | 30.1% (249/827) |
| Maltose | nt | nt | 56.8% (470/827) | 18.3% (151/827) |
| Raffinose | nt | nt | 28.5% (236/827) | 13.8% (114/827) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Endoh, R.; Horiyama, M.; Ohkuma, M. D-Fructose Assimilation and Fermentation by Yeasts Belonging to Saccharomycetes: Rediscovery of Universal Phenotypes and Elucidation of Fructophilic Behaviors in Ambrosiozyma platypodis and Cyberlindnera americana. Microorganisms 2021, 9, 758. https://doi.org/10.3390/microorganisms9040758
Endoh R, Horiyama M, Ohkuma M. D-Fructose Assimilation and Fermentation by Yeasts Belonging to Saccharomycetes: Rediscovery of Universal Phenotypes and Elucidation of Fructophilic Behaviors in Ambrosiozyma platypodis and Cyberlindnera americana. Microorganisms. 2021; 9(4):758. https://doi.org/10.3390/microorganisms9040758
Chicago/Turabian StyleEndoh, Rikiya, Maiko Horiyama, and Moriya Ohkuma. 2021. "D-Fructose Assimilation and Fermentation by Yeasts Belonging to Saccharomycetes: Rediscovery of Universal Phenotypes and Elucidation of Fructophilic Behaviors in Ambrosiozyma platypodis and Cyberlindnera americana" Microorganisms 9, no. 4: 758. https://doi.org/10.3390/microorganisms9040758
APA StyleEndoh, R., Horiyama, M., & Ohkuma, M. (2021). D-Fructose Assimilation and Fermentation by Yeasts Belonging to Saccharomycetes: Rediscovery of Universal Phenotypes and Elucidation of Fructophilic Behaviors in Ambrosiozyma platypodis and Cyberlindnera americana. Microorganisms, 9(4), 758. https://doi.org/10.3390/microorganisms9040758





