Bap-Independent Biofilm Formation in Staphylococcus xylosus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Conditions
2.2. DNA Manipulations and Bacterial Transformation: Mutagenesis of the Chromosomal Bap Gene by Allelic Exchange
2.3. Colony Morphology on CRA
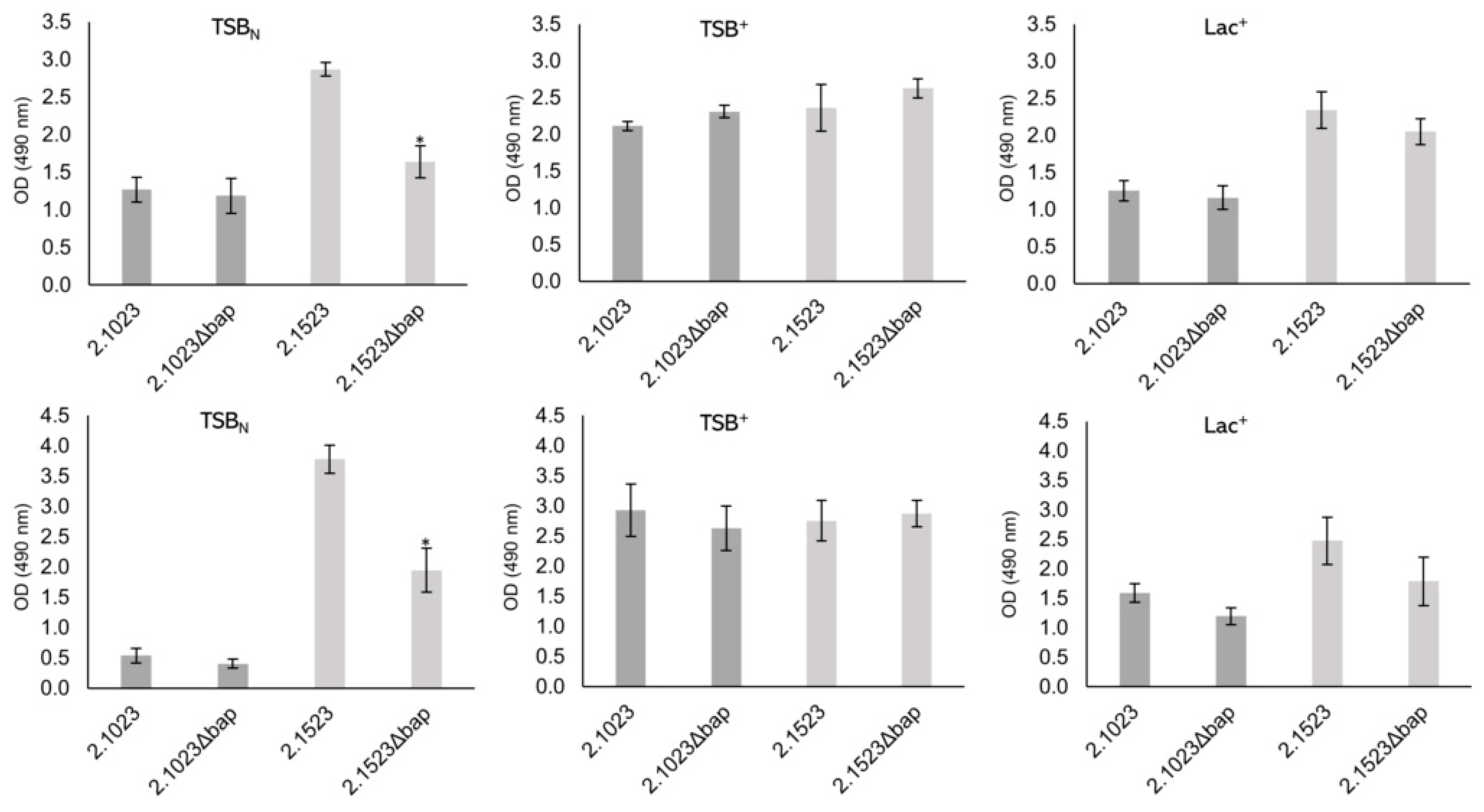
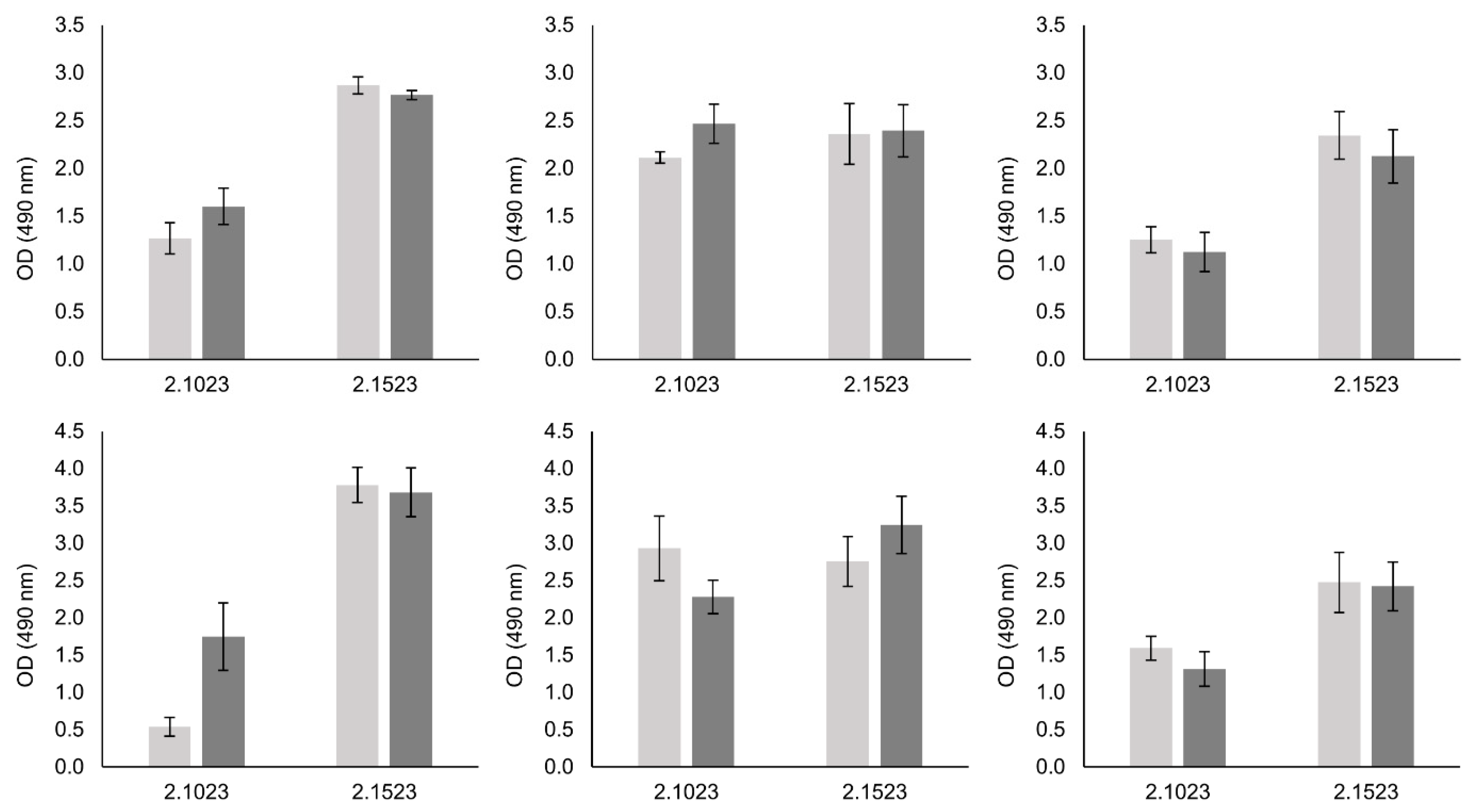
2.4. Biofilm Formation Assays
2.5. Bacterial Aggregation Assay
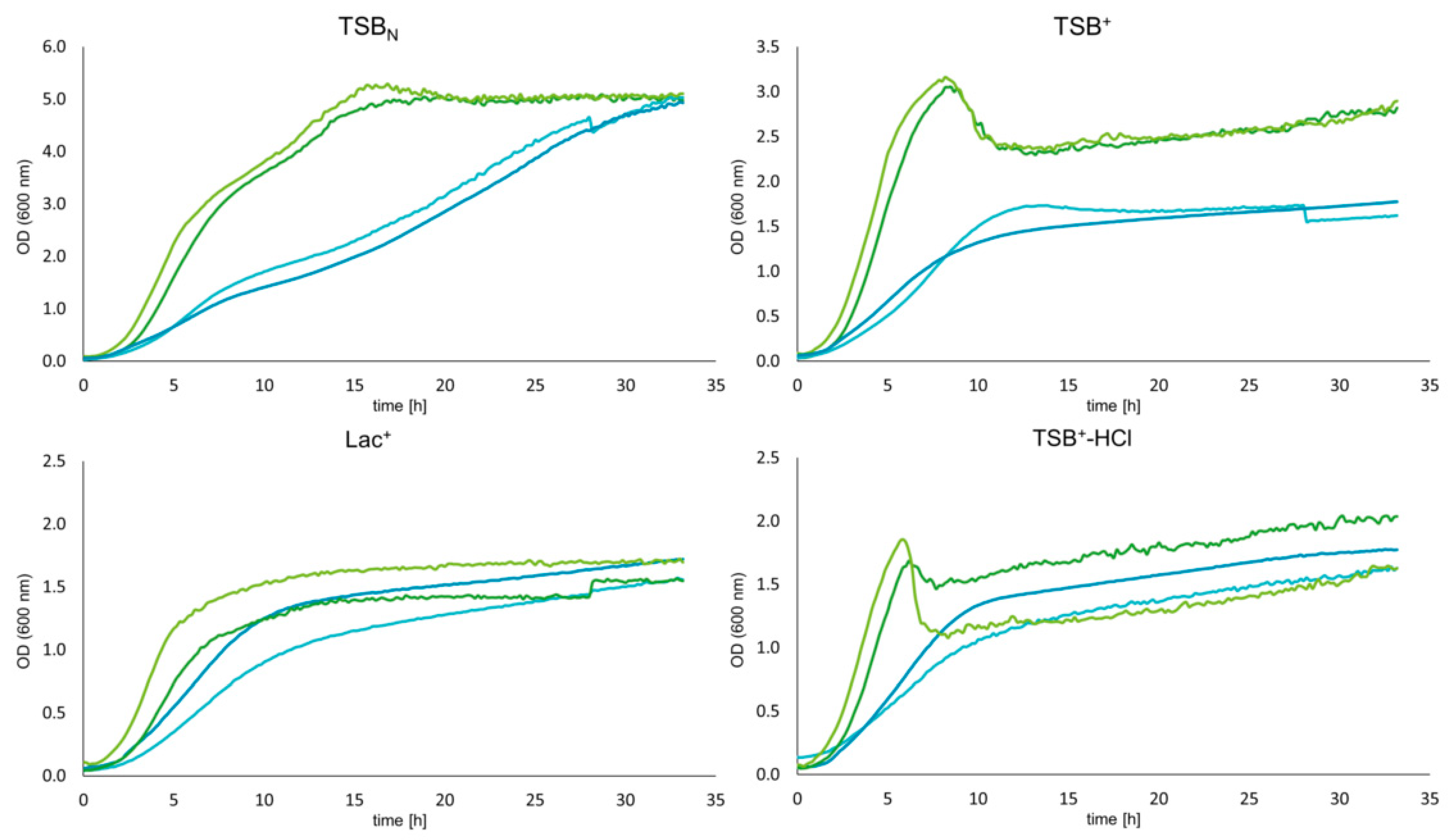
2.6. Growth and pH Dynamics
2.7. SDS Page of Protein Extracts
2.8. Full Proteome Analysis
2.9. Bioinformatic and Statistical Analysis
3. Results
3.1. Protein Motif Structural Organization of S. xylosus Bap
3.2. Mutagenesis of the Chromosomal Bap Gene
3.3. Growth Dynamics of Wildtype and Mutant Strains
3.4. Biofilm Formation of Bap Wildtypes and Mutants
3.5. Colony Morphology of Bap Wildtype and Mutant Strains on Congo Red Agar
3.6. pH Changes of S. xylosus during Growth in Glucose Supplemented Media
3.7. Calcium Does Not Impair Biofilm Formation of Bap Positive S. xylosus Wildtype Strains
3.8. Formation of Cell Aggregates in Wildtype and Mutant Strains
3.9. Proteomic Analysis of Expression Levels of Bap under Planktonic versus Sessile Conditions
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Leroy, S.; Vermassen, A.; Ras, G.; Talon, R. Insight into the genome of Staphylococcus xylosus, a ubiquitous species well adapted to meat products. Microorganisms 2017, 5, 52. [Google Scholar] [CrossRef] [Green Version]
- Flemming, H.-C.; Wingender, J. The biofilm matrix. Nat. Rev. Microbiol. 2010, 8, 623–633. [Google Scholar] [CrossRef] [PubMed]
- Götz, F. Staphylococcus and biofilms. Mol. Microbiol. 2002, 43, 1367–1378. [Google Scholar] [CrossRef]
- Taglialegna, A.; Lasa, I.; Valle, J. Amyloid structures as biofilm matrix scaffolds. J. Bacteriol. 2016, 198, 2579–2588. [Google Scholar] [CrossRef] [Green Version]
- Valle, J.; Latasa, C.; Gil, C.; Toledo-Arana, A.; Solano, C.; Penadés, J.R.; Lasa, I. Bap, a biofilm matrix protein of Staphylococcus aureus prevents cellular internalization through binding to GP96 host receptor. PLoS Pathog. 2012, 8, e1002843. [Google Scholar] [CrossRef] [Green Version]
- Speziale, P.; Pietrocola, G.; Foster, T.J.; Geoghegan, J.A. Protein-based biofilm matrices in Staphylococci. Front. Cell. Infect. Microbiol. 2014, 4, 171. [Google Scholar] [CrossRef] [Green Version]
- Cucarella, C.; Solano, C.; Valle, J.; Amorena, B.; Lasa, I.; Penadés, J.R. Bap, a Staphylococcus aureus surface protein involved in biofilm formation. J. Bacteriol. 2001, 183, 2888–2896. [Google Scholar] [CrossRef] [Green Version]
- Taglialegna, A.; Matilla-Cuenca, L.; Dorado-Morales, P.; Navarro, S.; Ventura, S.; Garnett, J.A.; Lasa, I.; Valle, J. The biofilm-associated surface protein Esp of Enterococcus faecalis forms amyloid-like fibers. NPJ Biofilms Microbiomes 2020, 6, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taglialegna, A.; Navarro, S.; Ventura, S.; Garnett, J.A.; Matthews, S.; Penades, J.R.; Lasa, I.; Valle, J. Staphylococcal Bap proteins build amyloid scaffold biofilm matrices in response to environmental signals. PLoS Pathog. 2016, 12, e1005711. [Google Scholar] [CrossRef] [Green Version]
- Arrizubieta, M.J.; Toledo-Arana, A.; Amorena, B.; Penadés, J.R.; Lasa, I. Calcium inhibits Bap-dependent multicellular behavior in Staphylococcus aureus. J. Bacteriol. 2004, 186, 7490–7498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tormo, M.A.; Knecht, E.; Götz, F.; Lasa, I.; Penades, J.R. Bap-dependent biofilm formation by pathogenic species of Staphylococcus: Evidence of horizontal gene transfer? Microbiology 2005, 151, 2465–2475. [Google Scholar] [CrossRef] [Green Version]
- Planchon, S.; Gaillard-Martinie, B.; Dordet-Frisoni, E.; Bellon-Fontaine, M.N.; Leroy, S.; Labadie, J.; Hébraud, M.; Talon, R. Formation of biofilm by Staphylococcus xylosus. Int. J. Food Microbiol. 2006, 109, 88–96. [Google Scholar] [CrossRef]
- Schiffer, C.; Hilgarth, M.; Ehrmann, M.; Vogel, R.F. Bap and cell surface hydrophobicity are important factors in Staphylococcus xylosus biofilm formation. Front. Microbiol. 2019, 10, 1387. [Google Scholar] [CrossRef] [PubMed]
- Monk, I.R.; Shah, I.M.; Xu, M.; Tan, M.-W.; Foster, T.J. Transforming the Untransformable: Application of Direct Transformation To Manipulate Genetically Staphylococcus aureus and Staphylococcus epidermidis. MBio 2012, 3, e00277-11. [Google Scholar] [CrossRef] [Green Version]
- Monk, I.R.; Stinear, T.P. From cloning to mutant in 5 days: Rapid allelic exchange in Staphylococcus aureus. Access Microbiol. 2021, 3, 1–7. [Google Scholar] [CrossRef]
- Schuster, C.F.; Howard, S.A.; Gründling, A. Use of the counter selectable marker PheS* for genome engineering in Staphylococcus aureus. Microbiology 2019, 165, 572–584. [Google Scholar] [CrossRef] [PubMed]
- Heilmann, C.; Götz, F. Further characterization of Staphylococcus epidermidis transposon mutants deficient in primary attachment or intercellular adhesion. Zent. Bakteriol. 1998, 287, 69–83. [Google Scholar] [CrossRef]
- Doellinger, J.; Schneider, A.; Hoeller, M.; Lasch, P. Sample Preparation by Easy Extraction and Digestion (SPEED)—A Universal, Rapid, and Detergent-free Protocol for Proteomics Based on Acid Extraction. Mol. Cell. Proteom. 2020, 19, 209–222. [Google Scholar] [CrossRef]
- Kolbeck, S.; Ludwig, C.; Meng, C.; Hilgarth, M.; Vogel, R.F. Comparative Proteomics of Meat Spoilage Bacteria Predicts Drivers for Their Coexistence on Modified Atmosphere Packaged Meat. Front. Microbiol. 2020, 11, 209. [Google Scholar] [CrossRef] [PubMed]
- Bechtner, J.; Ludwig, C.; Kiening, M.; Jakob, F.; Vogel, R.F. Living the Sweet Life: How Liquorilactobacillus hordei TMW 1.1822 Changes Its Behavior in the Presence of Sucrose in Comparison to Glucose. Foods 2020, 9, 1150. [Google Scholar] [CrossRef]
- Garbuzynskiy, S.O.; Lobanov, M.Y.; Galzitskaya, O.V. FoldAmyloid: A method of prediction of amyloidogenic regions from protein sequence. Bioinformatics 2010, 26, 326–332. [Google Scholar] [CrossRef]
- De Groot, N.S.; Castillo, V.; Graña-Montes, R.; Ventura, S. AGGRESCAN: Method, application, and perspectives for drug design. Methods Mol. Biol. 2012, 819, 199–220. [Google Scholar] [CrossRef]
- Louros, N.; Konstantoulea, K.; de Vleeschouwer, M.; Ramakers, M.; Schymkowitz, J.; Rousseau, F. WALTZ-DB 2.0: An updated database containing structural information of experimentally determined amyloid-forming peptides. Nucleic Acids Res. 2020, 48, D389–D393. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Escamilla, A.-M.; Rousseau, F.; Schymkowitz, J.; Serrano, L. Prediction of sequence-dependent and mutational effects on the aggregation of peptides and proteins. Nat. Biotechnol. 2004, 22, 1302–1306. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.; Neuhauser, N.; Michalski, A.; Scheltema, R.A.; Olsen, J.V.; Mann, M. Andromeda: A peptide search engine integrated into the MaxQuant environment. J. Proteome Res. 2011, 10, 1794–1805. [Google Scholar] [CrossRef]
- Tyanova, S.; Temu, T.; Cox, J. The MaxQuant computational platform for mass spectrometry-based shotgun proteomics. Nat. Protoc. 2016, 11, 2301–2319. [Google Scholar] [CrossRef]
- Tyanova, S.; Temu, T.; Sinitcyn, P.; Carlson, A.; Hein, M.Y.; Geiger, T.; Mann, M.; Cox, J. The Perseus computational platform for comprehensive analysis of (prote)omics data. Nat. Methods 2016, 13, 731–740. [Google Scholar] [CrossRef]
- Shah, A.D.; Goode, R.J.A.; Huang, C.; Powell, D.R.; Schittenhelm, R.B. LFQ-Analyst: An Easy-To-Use Interactive Web Platform To Analyze and Visualize Label-Free Proteomics Data Preprocessed with MaxQuant. J. Proteome Res. 2020, 19, 204–211. [Google Scholar] [CrossRef]
- Perez-Riverol, Y.; Csordas, A.; Bai, J.; Bernal-Llinares, M.; Hewapathirana, S.; Kundu, D.J.; Inuganti, A.; Griss, J.; Mayer, G.; Eisenacher, M.; et al. The PRIDE database and related tools and resources in 2019: Improving support for quantification data. Nucleic Acids Res. 2019, 47, D442–D450. [Google Scholar] [CrossRef] [PubMed]
- Cucarella, C.; Tormo, M.A.; Ubeda, C.; Trotonda, M.P.; Monzon, M.; Peris, C.; Amorena, B.; Lasa, I.; Penades, J.R. Role of biofilm-associated protein Bap in the pathogenesis of bovine Staphylococcus aureus. Infect. Immun. 2004, 72, 2177–2185. [Google Scholar] [CrossRef] [Green Version]
- Tormo, M.Á.; Úbeda, C.; Martí, M.; Maiques, E.; Cucarella, C.; Valle, J.; Foster, T.J.; Lasa, Í.; Penadés, J.R. Phase-variable expression of the biofilm-associated protein (Bap) in Staphylococcus aureus. Microbiology 2007, 153, 1702–1710. [Google Scholar] [CrossRef] [Green Version]
- Leroy, S.; Lebert, I.; Andant, C.; Micheau, P.; Talon, R. Investigating Extracellular DNA Release in Staphylococcus xylosus Biofilm In Vitro. Microorganisms 2021, 9, 2192. [Google Scholar] [CrossRef] [PubMed]
- You, Y.; Xue, T.; Cao, L.; Zhao, L.; Sun, H.; Sun, B. Staphylococcus aureus glucose-induced biofilm accessory proteins, GbaAB, influence biofilm formation in a PIA-dependent manner. Int. J. Med. Microbiol. 2014, 304, 603–612. [Google Scholar] [CrossRef]
- Bowden, M.G.; Chen, W.; Singvall, J.; Xu, Y.; Peacock, S.J.; Valtulina, V.; Speziale, P.; Höök, M. Identification and preliminary characterization of cell-wall-anchored proteins of Staphylococcus epidermidis. Microbiology 2005, 151, 1453–1464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCrea, K.W.; Hartford, O.; Davis, S.; Eidhin, D.N.; Lina, G.; Speziale, P.; Foster, T.J.; Höök, M. The serine-aspartate repeat (Sdr) protein family in Staphylococcus epidermidis. Microbiology 2000, 146, 1535–1546. [Google Scholar] [CrossRef]
- Bai, Q.; Ma, J.; Zhang, Z.; Zhong, X.; Pan, Z.; Zhu, Y.; Zhang, Y.; Wu, Z.; Liu, G.; Yao, H. YSIRK-G/S-directed translocation is required for Streptococcus suis to deliver diverse cell wall anchoring effectors contributing to bacterial pathogenicity. Virulence 2020, 11, 1539–1556. [Google Scholar] [CrossRef] [PubMed]
- DeDent, A.; Bae, T.; Missiakas, D.M.; Schneewind, O. Signal peptides direct surface proteins to two distinct envelope locations of Staphylococcus aureus. EMBO J. 2008, 27, 2656–2668. [Google Scholar] [CrossRef] [Green Version]
- Lasa, I.; Penadés, J.R. Bap: A family of surface proteins involved in biofilm formation. Res. Microbiol. 2006, 157, 99–107. [Google Scholar] [CrossRef] [PubMed]
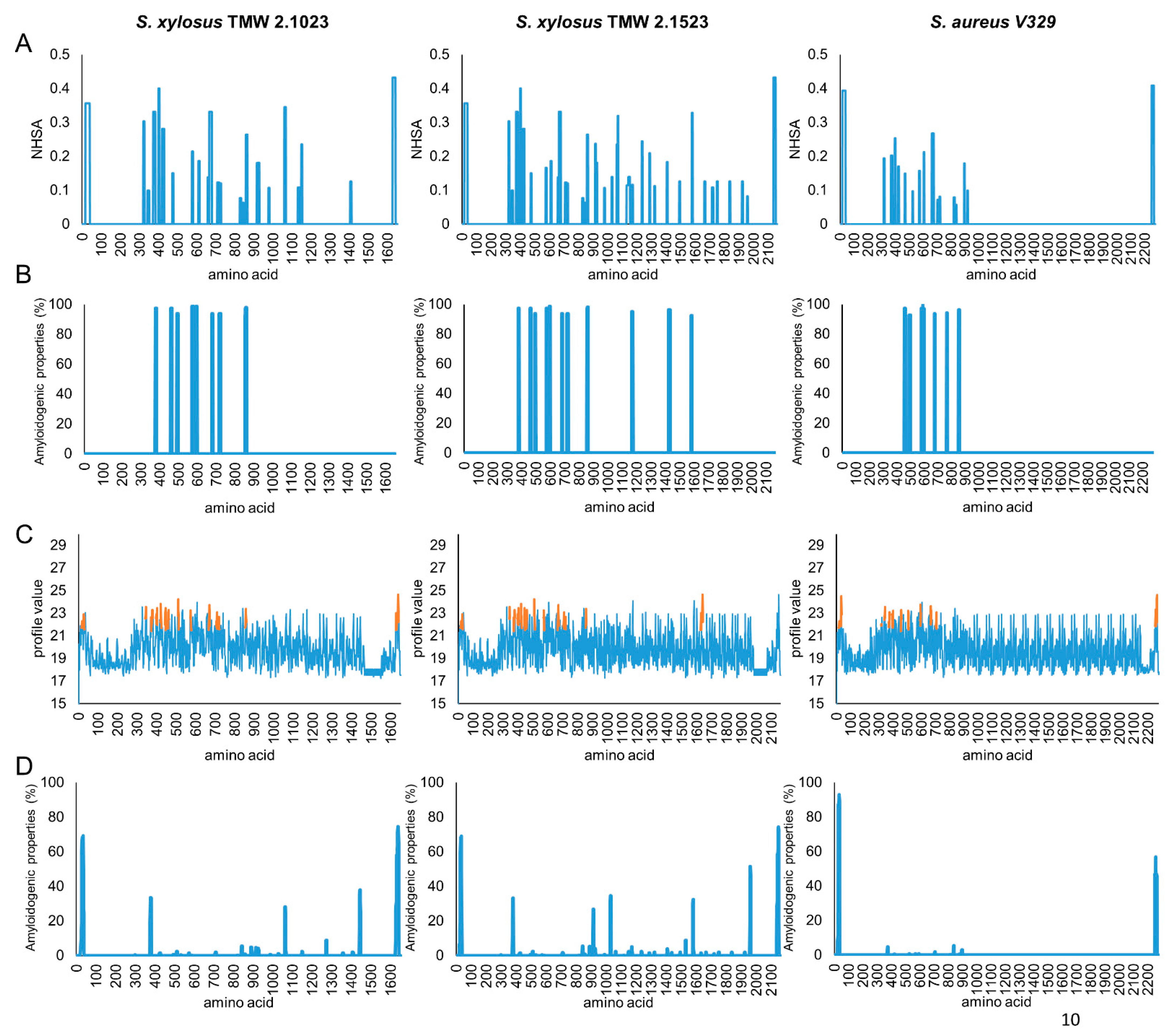


| Bap | S. xylosus 2.1023 | S. xylosus 2.1523 | S. aureus V329 |
|---|---|---|---|
| Accession | JGY91_02455 | JGY88_01140-45 | AAK38834 |
| Length total (aa) | 1651 | 2161 | 2276 |
| YSIRK Signal Peptide | 1–44 | 1–44 | 1–44 |
| Region A | 316 | 316 | 316 |
| Region B | 458 | 458 | 458 |
| Region C (incl. spacer) | 644 | 1160 | 1321 |
| Region D (incl LPXTG) | 189 | 183 | 137 |
| TM helix | 1624–1641 | 2134–2151 | 2249–2266 |
| MW (kDa) | 173.1 | 224.3 | 238.5 |
| pI_Bap | 4.01 | 3.90 | 3.90 |
| pI_BapB | 4.41 | 4.39 | 4.61 |
| # RepeatsA | 2 | 2 | 2 |
| sequence | TAEDN | TAEDN | AQDDDNIKEDSNTQEESTNTSSQSSEVPQTKK |
| # RepeatsC | 7 | 13 | 14 |
| type of C repeats | ig-like domain type 6 | ig-like domain type 6 | ig-like domain type 3 |
| # RepeatsD | 17 | 16 | 7 |
| sequence | 13× GTGENP, 1× GKGENP, 1× GGGENP, 1× GIGENP,1× GTGENT | 14× GTGENP, 1× GAGENP, 1× GTGENT | 2× SDDNSDNGNN 1× SDDNSGNGDN 1× SDDNSDN 1× SGAGDTSD 2× SGAGDNSD |
| %p.identity_Xyl vs Aur | 45.13 | 58.97 | - |
| %p.identity_B_Xyl vs Aur | 80.18 | 79.96 | - |
| # EF motifs | 7 | 7 | 4 |
| seq_EF2 | DYDKDGLLDRYER | DYDKDGLLDRYER | DYDKDGLLDRYER |
| seq_EF3 | DTDGDGKNDGDEV | DTDGDGKNDGDEV | DTDGDGKNDGDEV |
| %p.identity EF2_Xyl vs. Aur | 100 | 100 | - |
| %p.identity EF3_Xyl vs. Aur | 100 | 100 | - |
| Biofilm Associated Protein (Bap) | |||
|---|---|---|---|
| TMW 2.1023 | JGY91_02455 | ||
| plankt. vs. sessile, Lac+ | log2 fold change | 0.901 | |
| p.val (adj.) | 0.158 | ||
| significant | FALSE | ||
| TMW 2.1523 | JGY88_01140-45 | ||
| plankt. vs. sessile, Lac+ | log2 fold change | 1.15 | |
| p.val (adj.) | 0.0189 | ||
| significant | TRUE | ||
| plankt. vs. sessile, TSBN | log2 fold change | 5.11 | |
| p.val (adj.) | 0.0000231 | ||
| significant | TRUE | ||
| Lac+ vs. TSBN (plankt.) | log2 fold change | −2.97 | |
| p.val (adj.) | 0.000139 | ||
| significant | TRUE | ||
| Lac+ vs. TSBN (sessil) | log2 fold change | 0.981 | |
| p.val (adj.) | 0.0356 | ||
| significant | FALSE | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schiffer, C.J.; Abele, M.; Ehrmann, M.A.; Vogel, R.F. Bap-Independent Biofilm Formation in Staphylococcus xylosus. Microorganisms 2021, 9, 2610. https://doi.org/10.3390/microorganisms9122610
Schiffer CJ, Abele M, Ehrmann MA, Vogel RF. Bap-Independent Biofilm Formation in Staphylococcus xylosus. Microorganisms. 2021; 9(12):2610. https://doi.org/10.3390/microorganisms9122610
Chicago/Turabian StyleSchiffer, Carolin J., Miriam Abele, Matthias A. Ehrmann, and Rudi F. Vogel. 2021. "Bap-Independent Biofilm Formation in Staphylococcus xylosus" Microorganisms 9, no. 12: 2610. https://doi.org/10.3390/microorganisms9122610
APA StyleSchiffer, C. J., Abele, M., Ehrmann, M. A., & Vogel, R. F. (2021). Bap-Independent Biofilm Formation in Staphylococcus xylosus. Microorganisms, 9(12), 2610. https://doi.org/10.3390/microorganisms9122610






