Extremophilic Microorganisms in Central Europe
Abstract
1. Introduction
2. Caves and Karst Environments
2.1. Caves and Karst—Characteristics
2.2. Microorganisms in Caves and Karst Environments
2.3. Potential Uses of Cave Microorganisms
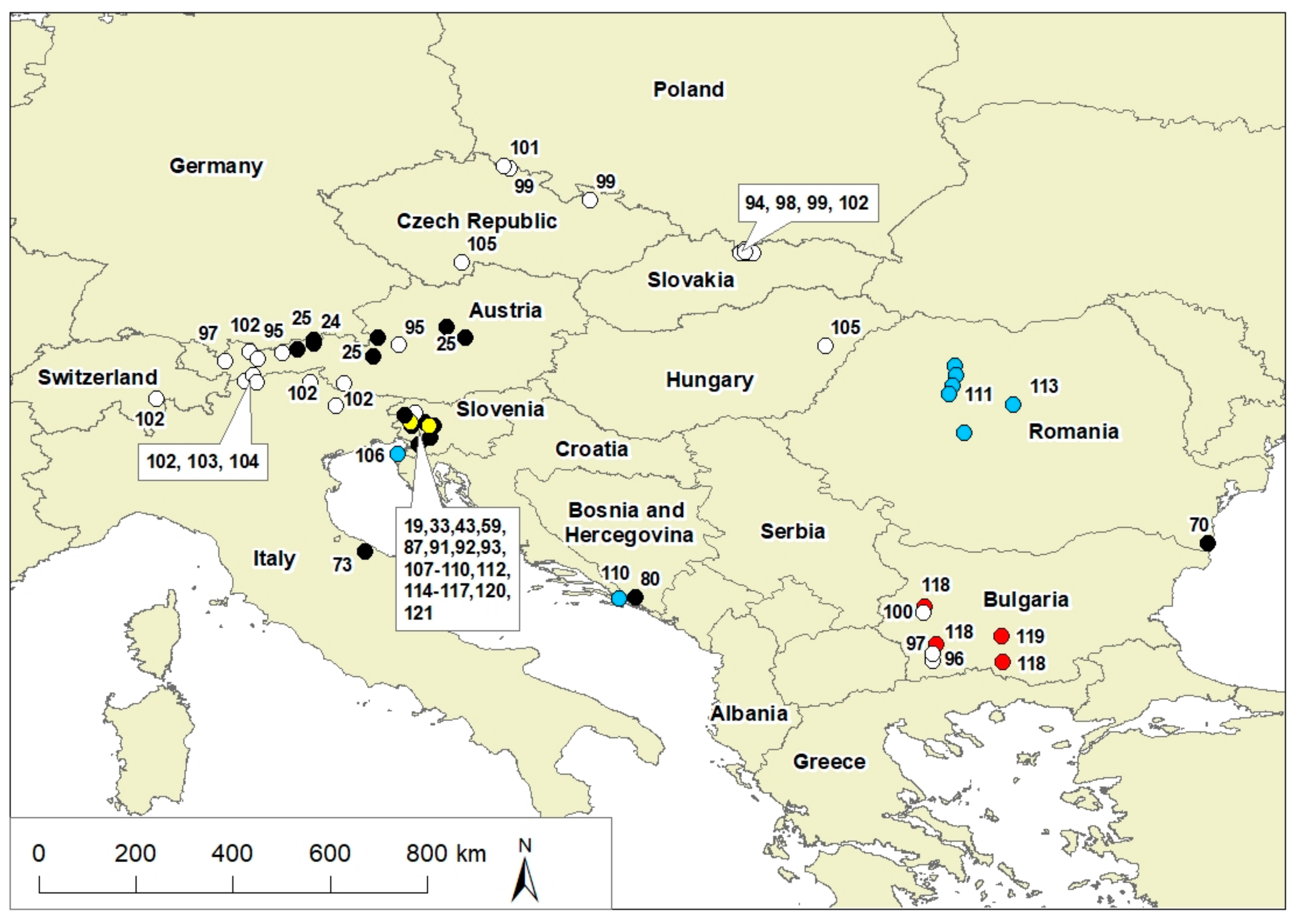
| Taxon | Country | References |
|---|---|---|
| Oligotrophs | ||
| Proteobacteria | Slovenia, Italy, Romania | [59,70,73,74] |
| Gammaproteobacteria Alphaproteobacteria Betaproteobacteria Deltaproteobacteria Acidobacteria Verrucomicrobia Planctomycetes Chloroflexi Gemmatimonadetes | Slovenia | [59] |
| Nitrospirae Betaproteobacteria Bacteriodetes Gammaproteobacteria Actinobacteria Alphaproteobacteria Planctomycetes | Bosnia and Herzegovina | [80] |
| Arthrobacter arilaitensis, Kocuria polaris, Paenibacillus amyolyticus P. polymixa Pseudomonas antarctica P. cedrina P. jessenii P. marginalis Staphylococcus equorum S. haemolyticus S. pasteuri S. warneri Streptomycesbadius | Slovenia | [91] |
| Nitrosopumilus maritimus, Nitrososphaera gargensis , Aciduliprofundum boonei , Methanomassiliicoccus luminyensis , and Thermogymnomonas acidicola | Austria, Italy | [24,25] |
| Mucor troglophilus ** | Slovenia | [43,87] |
| cyanobacteria and green microalgae * | Slovenia | [92] |
| cyanobacteria and green microalgae * | Slovenia | [19] |
| Psychrophiles | ||
| Pseudomonas | Slovenia | [93] |
| Lysobacter | Slovenia | [93] |
| Chloridella glacialis | Slovenia | [93] |
| Ellipsoidion perminimum | Slovenia | [93] |
| Chlamydomonas nivalis | Austria, Bulgaria, Slovakia | [94,95,96,97] |
| Chlainomonas | Austria, Slovakia | [95,98] |
| Chloromonas nivalis Chloromonas nivalis subsp. tatrae | Austria Slovakia | [94] |
| Chloromonas hindakii | Czech Republic, Slovakia, Poland, Bulgaria | [99] |
| Chloromonas brevispina | Czech Republic, Bulgaria | [100,101] |
| Chloromonas rosae | Czech Republic | [101] |
| Sanguina nivaloides | Austria, Italy, Slovenia, Slovakia, Switzerland | [102] |
| Mesotaenium berggrenii; Mesotaenium berggrenii var. alaskana | Austria | [103,104] |
| Haematococcus pluvialis (epitype); Haematococcus rubicundus; Haematococcus rubens | Europe | [105] |
| Halophiles | ||
| Salinibacter | Slovenia | [106] |
| Gammaproteobacteria (Acinetobacter spp.) | Slovenia | [107] |
| Dunaliella salina | Slovenia | [106] |
| Cladophora | Slovenia, France | [106,108] |
| Halorubrum | Slovenia | [109] |
| Haloquadratum | Slovenia | [106] |
| Haloquadratum spp. | Slovenia | [108] |
| Haloferax | Slovenia, Croatia | [110] |
| Haloarcula | Slovenia, Croatia | [110] |
| Haloterrigena | Slovenia, Croatia | [110] |
| Natrinema | Slovenia | [109] |
| Halobacterium | Croatia | [110] |
| Microcoelus chtonoplastes | Slovenia | [106] |
| Synechococcus | Romania | [111] |
| Coleofasciculus chthonoplastes | Slovenia | [112] |
| Phormidium | Slovenia | [112] |
| Lyngbya | Slovenia | [112] |
| Picochlorum oklahomense | Romania | [111] |
| Prosthecochloris vibrioformis | Romania | [113] |
| Mantionella, Picochlorum | Romania | [113] |
| Hortaea werneckii | Slovenia | [114,115] |
| Wallemia ichthyophaga | Slovenia | [116] |
| Phaeotheca triangularis | Slovenia | [117] |
| Trimmatostroma salinum | Slovenia | [117] |
| Aureobasidium pullulans | Slovenia | [114,115,116,117] |
| Cladosporium | Slovenia | [117] |
| Thermophiles | ||
| Bacillus | Bulgaria | [118,119] |
| Extremophiles in sulphidic springs | ||
| Proteobacteria: - Betaproteobacteria: Azospira, Iodobacter, Georgfuchsia, Pelomonas, Rhodoferax, Undibacterium, Thiobacillus - Gammaproteobacteria: Thiothrix - Epsilonproteobacteria: Dehalospirillum, Sulfuricurvum, Sulfurovum | Slovenia | [33] |
| Archaea (mainly Euryarchaeota) | Slovenia | [33] |
| Oscillatoria spp. | Slovenia | [120] |
| Caloneis tenui | Slovenia | [120] |
| Frustulla vulgaris | Slovenia | [120] |
| Gomphonema spp. | Slovenia | [120] |
| Navicula radiosa | Slovenia | [120] |
| Tribonema vulgare | Slovenia | [120] |
| diatoms | Slovenia | [120] |
| undifferentiated fungi | Slovenia | [33,121] |
| Ciliophora Fungi-Ascomycota | Slovenia | [33,121] |
3. Glaciers and Ice Caves
3.1. Characteristics of Frigid Environments
3.2. Psychrophiles in Glaciers and Ice Caves
3.3. Potential Uses of Psychrophiles
4. Adriatic Salterns
4.1. Characteristics of Hypersaline Habitats
4.2. Adriatic Halophiles
4.3. Potential Uses of Halophiles
5. Thermal and Mineral Springs of South-Eastern Europe
5.1. Characteristics of High-Temperature Environments and Habitats with High Mineral Content
5.2. Thermophiles in Central-European Thermal Springs
5.3. Potential Uses of Thermophiles
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Macelroy, R.D. Some comments on the evolution of extremophiles. BioSystems 1974, 6, 74–75. [Google Scholar] [CrossRef]
- Rampelotto, P.H. Extremophiles and Extreme Environments. Life 2013, 3, 482–485. [Google Scholar] [CrossRef] [PubMed]
- Stan-Lotter, H. Physicochemical Boundaries of Life. In Adaption of Microbial Life to Environmental Extremes, 2nd ed.; Stan-Lotter, H., Fendrihan, S., Eds.; Springer: Vienna, Austria, 2017; pp. 1–21. [Google Scholar]
- Gupta, G.N.; Srivastava, S.; Khare, S.K.; Prakash, V. Extremophiles: An overview of microorganism from extreme environment. Int. J. Agric. Environ. Biotechnol. 2014, 7, 371. [Google Scholar] [CrossRef]
- Ashcroft, F. Life at the Extremes: The Science of Survival; University of California Press: Berkley, Los Angeles, CA, USA, 2002; p. 106. [Google Scholar]
- Castelle, C.J.; Wrighton, K.C.; Thomas, B.C.; Hug, L.A.; Brown, C.T.; Wilkins, M.J.; Frischkorn, K.R.; Tringe, S.G.; Singh, A.; Markillie, L.M.; et al. Genomic expansion of domain archaea highlights roles for organisms from new phyla in anaerobic carbon cycling. Curr. Biol. 2015, 25, 690–701. [Google Scholar] [CrossRef] [PubMed]
- Cavicchioli, R. Archaea—Timeline of the Third Domain. Nat. Rev. Microbiol. 2011, 9, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Emerson, J.B.; Thomas, B.C.; Alvarez, W.; Banfield, J.F. Metagenomic analysis of a high carbon dioxide subsurface microbial community populated by chemolithoautotrophs and bacteria and archaea from candidate phyla: High CO2 subsurface metagenomics. Environ. Microbiol. 2016, 18, 1686–1703. [Google Scholar] [CrossRef] [PubMed]
- Garima, D.; Bharati, K.; Usha, A.; Sneh, T.; Santosh, R.M. Significance of archaea in terrestrial biogeochemical cycles and global climate change. Afr. J. Microbiol. Res. 2015, 9, 201–208. [Google Scholar] [CrossRef]
- Ramanan, R.; Kannan, K.; Sivanesan, S.D.; Mudliar, S.; Kaur, S.; Tripathi, A.K.; Chakrabarti, T. Bio-sequestration of carbon dioxide using carbonic anhydrase enzyme purified from citrobacter freundii. World J. Microbiol. Biotechnol. 2009, 25, 981–987. [Google Scholar] [CrossRef]
- Lee, N.M.; Meisinger, D.B.; Aubrecht, R.; Kovačik, L.; Saiz-Jimenez, C.; Baskar, S.; Baskar, R.; Liebl, W.; Porter, M.L.; Engel, A.S. Caves and Karst Environments. In Life at Extremes: Environments, Organisms and Strategies for Survival, 1st ed.; Bell, E.M., Ed.; CAB International: Oxfordshire, UK, 2012; pp. 320–344. [Google Scholar] [CrossRef]
- Eavis, A. An up to Date Report of Cave Exploration around the World. In Proceedings of the 15th International Congress of Speleology, Kerrville, TX, USA, 19–26 July 2009; pp. 21–25. [Google Scholar]
- Leadbetter, J.R. Cultivation of recalcitrant microbes: Cells are alive, well and revealing their secrets in the 21st century laboratory. Curr. Opin. Microbiol. 2003, 6, 274–281. [Google Scholar] [CrossRef]
- Probst, A.J.; Moissl-Eichinger, C. “Altiarchaeales”: Uncultivated archaea from the subsurface. Life 2015, 5, 1381–1395. [Google Scholar] [CrossRef]
- Jones, W.K.; White, W.B. Karst. In Encyclopedia of Caves, 3rd ed.; White, W.B., Culver, D.C., Pipan, T., Eds.; Elsevier: Waltham, MA, USA, 2019; pp. 609–618. [Google Scholar]
- Komac, M.; Urbanc, J. Assessment of spatial properties of karst areas on a regional scale using GIS and statistics—The case of Slovenia. J. Caves Karst. Stud. 2012, 74, 251–261. [Google Scholar] [CrossRef]
- Mulec, J. Phototrophs in Caves. In Cave Ecology; Moldovan, O.T., Kováč, Ľ., Halse, S., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 91–106. [Google Scholar] [CrossRef]
- Hill, C.A.; Forti, P. Cave Minerals of the World, 2nd ed.; National Speleological Society: Huntsvill, AL, USA, 1997. [Google Scholar]
- Mulec, J.; Kosi, G. Lampenflora algae and methods of growth control. J. Caves Karst. Stud. 2009, 71, 109–115. [Google Scholar]
- Del Rosal Padial, Y.; Jurado Lobo, V.; Hernández Mariné, M.; Roldán Molina, M.; Sáiz Jiménez, C. Biofilms en cuevas turísticas: La Cueva de Nerja y la Cueva del Tesoro. In El Karst y el Hombre: Las Cuevas Como Patrimonio Mundial; Andreo, B., Durán, J.J., Eds.; Asociación de Cuevas Turísticas Españolas: Madrid, Spain, 2016; pp. 103–114. [Google Scholar]
- Mulec, J. Chapter 75—Lampenflora. In Encyclopedia of Caves, 3rd ed.; White, W.B., Culver, D.C., Pipan, T., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 635–641. [Google Scholar]
- Romero, A. Cave Biology: Life in Darkness, 1st ed.; Cambridge University Press: Cambridge, UK, 2009; p. 182. [Google Scholar]
- Culver, D.C.; Pipan, T. Shifting Paradigms of the Evolution of Cave Life. Acta Carsol. 2015, 44, 415–425. [Google Scholar] [CrossRef]
- Reitschuler, C.; Lins, P.; Wagner, A.O.; Illmer, P. Cultivation of moonmilk-born non-extremophilic thaumand euryarchaeota in mixed culture. Anaerobe 2014, 29, 73–79. [Google Scholar] [CrossRef]
- Reitschuler, C.; Spötl, C.; Hofmann, K.; Wagner, A.O.; Illmer, P. Archaeal distribution in moonmilk deposits from Alpine caves and their ecophysiological potential. Microb. Ecol. 2016, 71, 686–699. [Google Scholar] [CrossRef]
- Offre, P.; Spang, A.; Schleper, C. Archaea in biogeochemical cycles. Annu. Rev. Microbiol. 2013, 67, 437–457. [Google Scholar] [CrossRef]
- Hamilton, T.L.; Jones, D.S.; Schaperdoth, I.; Macalady, J.L. Metagenomic insights into s(0) precipitation in a terrestrial subsurface lithoautotrophic ecosystem. Front. Microbiol. 2014, 5, 756. [Google Scholar] [CrossRef]
- Anderson, R.; Sogin, M.; Baross, J. Biogeography and ecology of the rare and abundant microbial lineages in deep-sea hydrothermal vents. FEMS Microbiol. Ecol. 2015, 91, 1–11. [Google Scholar] [CrossRef]
- Mattes, T.; Nunn, B.; Marshall, K.; Proskurowski, G.; Kelly, D.S.; Kawka, O.E.; Goodlett, D.R.; Hansell, D.A.; Morris, R.M. Sulfur oxidizers dominate carbon fixation at a biogeochemical hot spot in the dark ocean. ISME J. 2013, 7, 2349–2360. [Google Scholar] [CrossRef]
- Takai, K.; Gamo, T.; Tsunogai, U.; Nakayama, N.; Hirayama, H.; Nealson, K.H.; Horikoshi, K. Geochemical and microbiological evidence for a hydrogen-based, hyperthermophilic subsurface lithoautotrophic microbial ecosystem (HyperSLiME) beneath an active deep-sea hydrothermal field. Extremophiles 2004, 8, 269–282. [Google Scholar] [CrossRef]
- Mulec, J.; Petrič, M.; Koželj, A.; Brun, C.; Batagelj, E.; Hladnik, A.; Holko, L. A Multiparameter analysis of environmental gradients related to hydrological conditions in a binary karst system (underground course of the Pivka river, Slovenia). Acta Carsol. 2019, 48, 314–327. [Google Scholar] [CrossRef]
- Skirnisdottir, S.; Hreggvidsson, G.O.; Hjörleifsdottir, S.; Marteinsson, V.T.; Petursdottir, S.K.; Holst, O.; Kristjansson, J.K. Influence of sulfide and temperature on species composition and community structure of hot spring microbial mats. Appl. Environ. Microbiol. 2000, 66, 2835–2841. [Google Scholar] [CrossRef]
- Mulec, J.; Summers Engel, A. Karst spring microbial diversity differs across an oxygen-sulphide ecocline and reveals potential for novel taxa discovery. Acta Carsol. 2019, 48, 129–143. [Google Scholar] [CrossRef]
- Edgcomb, V.P.; Kysela, D.T.; Teske, A.; de Vera Gomez, A.; Sogin, M.L. Benthic eukaryotic diversity in the guaymas basin hydrothermal vent environment. Proc. Natl. Acad. Sci. USA 2002, 99, 7658–7662. [Google Scholar] [CrossRef]
- Orsi, W.; Edgcomb, V.; Jeon, S.; Leslin, C.; Bunge, J.; Taylor, G.T.; Varela, R.; Epstein, S. Protistan microbial observatory in the Cariaco Basin, Caribbean. II. Habitat Specialization. ISME J. 2011, 5, 1357–1373. [Google Scholar] [CrossRef]
- Behnke, A.; Barger, K.J.; Bunge, J.; Stoeck, T. Spatio-temporal variations in protistan communities along an O2/H2S gradient in the anoxic Framvaren Fjord (Norway). FEMS Microbiol. Ecol. 2010, 72, 89–102. [Google Scholar] [CrossRef]
- Holder, T.; Basquin, C.; Ebert, J.; Randel, N.; Jollivet, D.; Conti, E.; Jékely, G.; Bono, F. Deep transcriptome-sequencing and proteome analysis of the hydrothermal vent annelid alvinella pompejana identifies the CvP-bias as a robust measure of eukaryotic thermostability. Biol. Direct 2013, 8, 2. [Google Scholar] [CrossRef]
- López-García, P.; Rodriguez-Valera, F.; Pedrós-Alió, C.; Moreira, D. Unexpected diversity of small eukaryotes in deep-sea antarctic plankton. Nature 2001, 409, 603–607. [Google Scholar] [CrossRef]
- López-García, P.; Philippe, H.; Gail, F.; Moreira, D. Autochthonous eukaryotic diversity in hydrothermal sediment and experimental microcolonizers at the Mid-Atlantic Ridge. Proc. Natl. Acad. Sci. USA 2003, 100, 697–702. [Google Scholar] [CrossRef]
- Engel, A.S. Observations on the biodiversity of sulfidic karst habitats. J. Caves Karst Stud. 2007, 69, 187–206. [Google Scholar]
- Por, F.D.; Dimentman, C.; Frumkin, A.; Naaman, I. Animal Life in the chemoautotrophic ecosystem of the hypogenic groundwater cave of Ayyalon (Israel): A summing up. Nat. Sci. 2013, 5, 7–13. [Google Scholar] [CrossRef]
- Dimkić, I.; Stanković, S.; Kabić, J.; Stupar, M.; Nenadić, M.; Ljaljević-Grbić, M.; Žikić, V.; Vujisić, L.; Tešević, V.; Vesović, N.; et al. Bat guano-dwelling microbes and antimicrobial properties of the pygidial gland secretion of a troglophilic ground beetle against them. Appl. Microbiol. Biotechnol. 2020, 104, 4109–4126. [Google Scholar] [CrossRef] [PubMed]
- Mulec, J. Microorganisms in hypogeon: Examples from Slovenian Karst Caves. Acta Carsol. 2008, 37, 153–160. [Google Scholar] [CrossRef]
- Pipan, T. Epikarst a Promising Habitat: Copepod Fauna, Its Diversity and Ecology: A Case Study from Slovenia (Europe), 1st ed.; ZRC Publishing: Ljubljana, Slovenija, 2005. [Google Scholar]
- Gerič, B.; Pipan, T.; Mulec, J. Diversity of culturable bacteria and meiofauna in the epikarst of Škocjanske Jame Caves (Slovenia). Acta Carsol. 2016, 33, 301–309. [Google Scholar] [CrossRef]
- Poulson, T.L.; Lavoie, K.H. Trophic Basis of Subsurface Ecosystems. In Subterranean Ecosystems; Wilkens, H., Culver, D.C., Humphreys, W.F., Eds.; Elsevier Press: Amsterdam, The Netherlands, 2000; pp. 231–250. [Google Scholar]
- Banskar, S.; Bhute, S.S.; Suryavanshi, M.V.; Punekar, S.; Shouche, Y.S. Microbiome analysis reveals the abundance of bacterial pathogens in Rousettus leschenaultii Guano. Sci. Rep. 2016, 6, 36948. [Google Scholar] [CrossRef]
- Gold, T. The deep, hot biosphere. Proc. Natl. Acad. Sci. USA 1992, 89, 6045–6049. [Google Scholar] [CrossRef]
- Hanson, J.K.; Nash, G.H. Caves, 1st ed.; Infobase Publishing: New York, NY, USA, 2007. [Google Scholar]
- Porca, E.; Jurado, V.; Žgur-Bertok, D.; Saiz-Jimenez, C.; Pašić, L. Comparative analysis of yellow microbial communities growing on the walls of geographically distinct caves indicates a common core of microorganisms involved in their formation. FEMS Microbiol. Ecol. 2012, 81, 255–266. [Google Scholar] [CrossRef]
- Pedersen, K. Exploration of deep intraterrestrial microbial life: Current perspectives. FEMS Microbiol. Lett. 2000, 185, 9–16. [Google Scholar] [CrossRef]
- Mulec, J.; Zalar, P.; Hajna, N.Z.; Rupnik, M. Screening for culturable microorganisms from cave environments (Slovenia); Proučevanje Mikroorganizmov Iz Jamskih Okolij (Slovenija) z gojitvenimi tehnikami. Acta Carsol. 2002, 31, 177–187. [Google Scholar]
- Sterflinger, K.; Krumbein, W.E. Dematiaceous fungi as a major agent for biopitting on Mediterranean marbles and limestones. Geomicrobiol. J. 1997, 14, 219–230. [Google Scholar] [CrossRef]
- Viles, H.A. Micro-organisms and geomorphology. Mitt. Verb. Dtsch. Hohlen- Karstforscher 2000, 46, 116–121. [Google Scholar]
- Viles, H.A. Microbial geomorphology: A neglected link between life and landscape. Geomorphology 2012, 157–158, 6–16. [Google Scholar] [CrossRef]
- Barton, H.A.; Jurado, V. What’s up down there? Microbial diversity in caves microorganisms in caves survive under nutrient-poor conditions and are metabolically versatile and unexpectedly diverse. Microbe 2007, 2, 132–138. [Google Scholar]
- Portillo, M.C.; Gonzalez, J.M. Sulfate-reducing bacteria are common members of bacterial communities in Altamira cave (Spain). Sci. Total Environ. 2009, 407, 1114–1122. [Google Scholar] [CrossRef]
- Blatnik, M.; Culver, D.C.; Gabrovšek, F.; Knez, M.; Kogovšek, B.; Kogovšek, J.; Liu, H.; Mayaud, C.; Mihevc, A.; Mulec, J.; et al. Microbial Underground: Microorganisms and Their Habitats in Škocjanske Jame. In Karstology in the Classical Karst, 1st ed.; Knez, M., Otoničar, B., Petrič, M., Pipan, T., Slabe, T., Eds.; Springer Nature: Cham, Switzerland, 2020; pp. 169–181. [Google Scholar] [CrossRef]
- Pašić, L.; Kovče, B.; Sket, B.; Herzog-Velikonja, B. Diversity of microbial communities colonizing the walls of a Karstic Cave in Slovenia. FEMS Microbiol. Ecol. 2009, 71, 50–60. [Google Scholar] [CrossRef]
- Zhang, Z.F.; Liu, F.; Zhou, X.; Liu, X.Z.; Liu, S.J.; Cai, L. Culturable mycobiota from karst caves in China, with descriptions of 20 new species. Pers. Mol. Phylogeny Evol. Fungi 2017, 39, 1–31. [Google Scholar] [CrossRef]
- Zhang, Z.F.; Zhou, S.Y.; Eurwilaichitr, L.; Ingsriswang, S.; Raza, M.; Chen, Q.; Zhao, P.; Liu, F.; Cai, L. Culturable mycobiota from karst caves in China II, with descriptions of 33 new species. Fungal Divers. 2020, 106, 29–136. [Google Scholar] [CrossRef]
- Bosák, P.; Mihevc, A.; Pruner, P. Geomorphological evolution of the Podgorski Karst, SW Slovenia: Contribution of magnetostratigraphic research of the Črnotiče II site with Marifugia Sp. Acta Carsol. 2004, 33, 175–204. [Google Scholar] [CrossRef]
- Megušar, F.; Sket, B. On the nature of some organic covers on the cave-walls. In Proceedings of the 6th International Congress of Speleology, Academia, Olomuc, Czech Republic, 1973; pp. 159–161. [Google Scholar]
- Laiz, L.; Groth, I.; Gonzalez, I.; Saiz-Jimenez, C. Microbiological study of the dripping waters in Altamira Cave (Santillana Del Mar, Spain). J. Microbiol. Methods 1999, 36, 129–138. [Google Scholar] [CrossRef]
- Schabereiter-Gurtner, C.; Saiz-Jimenez, C.; Piñar, G.; Lubitz, W.; Rolleke, S. Altamira Cave paleolithic paintings harbor partly unknown bacterial communities. FEMS Microbiol. Lett. 2002, 211, 7–11. [Google Scholar] [CrossRef]
- Portillo, M.C.; Gonzalez, J.M.; Saiz-Jimenez, C. Metabolically active microbial communities of yellow and grey colonizations on the walls of Altamira Cave, Spain. J. Appl. Microbiol. 2008, 104, 681–691. [Google Scholar] [CrossRef]
- Schabereiter-Gurtner, C.; Saiz-Jimenez, C.; Pinar, G.; Lubitz, W.; Rolleke, S. Phylogenetic 16S rRNA analysis reveals the presence of complex and partly unknown bacterial communities in Tito Bustillo Cave, Spain, and on its palaeolithic paintings. Environ. Microbiol. 2002, 4, 392–400. [Google Scholar] [CrossRef]
- Chelius, M.K.; Moore, J.C. Molecular phylogenetic analysis of archaea and bacteria in Wind Cave, South Dakota. Geomicrobiol. J. 2004, 21, 123–134. [Google Scholar] [CrossRef]
- Angert, E.R.; Northup, D.E.; Reysenbach, A.L.; Peek, A.S.; Goebel, B.M.; Pace, N.R. Molecular phylogenetic analysis of a bacterial community in Sulphur River, Parker Cave, Kentucky. Am. Min. 1998, 83, 1583–1592. [Google Scholar] [CrossRef]
- Sarbu, S.M. Movile Cave: A chemoautotrophically based groundwater ecosystem. In Subterranean Ecosystems; Wilkens, H., Culver, D.C., Humphreys, W.F., Eds.; Elsevier Press: Amsterdam, The Netherlands, 2000; pp. 319–343. [Google Scholar]
- Engel, A.S.; Porter, M.L.; Kinkle, B.K.; Kane, T.C. Ecological assessment and geological significance of microbial communities from Cesspool Cave, Virginia. Geomicrobiol. J. 2001, 18, 259–274. [Google Scholar] [CrossRef]
- Engel, A.S.; Lee, N.; Porter, M.L.; Stern, L.A.; Bennett, P.C.; Wagner, M. Filamentous “Epsilonproteobacteria” dominate microbial mats from Sulfidic Cave Springs. Appl. Environ. Microbiol. 2003, 69, 5503–5511. [Google Scholar] [CrossRef] [PubMed]
- Macalady, J.L.; Jones, D.S.; Lyon, E.H. Extremely acidic, pendulous cave wall biofilms from the Frasassi Cave system, Italy. Environ. Microbiol. 2007, 9, 1402–1414. [Google Scholar] [CrossRef]
- Macalady, J.L.; Lyon, E.H.; Koffman, B.; Albertson, L.K.; Meyer, K.; Galdenzi, S.; Mariani, S. Dominant microbial populations in limestone-corroding stream biofilms, Frasassi Cave system, Italy. Appl. Environ. Microbiol. 2006, 72, 5596–5609. [Google Scholar] [CrossRef]
- Palleroni, N. Introduction to the Family Pseudomonadaceae. In The Prokaryotes, 2nd ed.; Balows, A., Trueper, H.G., Dworkin, M., Harder, W., Schliefer, K.H., Eds.; Springer: New York, NY, USA, 1992; Volume 3. [Google Scholar]
- Northup, D.E.; Barns, S.M.; Yu, L.E.; Spilde, M.N.; Schelble, R.T.; Dano, K.E.; Crossey, L.J.; Connolly, C.A.; Boston, P.J.; Natvig, D.O.; et al. Diverse microbial communities inhabiting ferromanganese deposits in Lechuguilla and Spider Caves. Environ. Microbiol. 2003, 5, 1071–1086. [Google Scholar] [CrossRef]
- Ikner, L.A.; Toomey, R.S.; Nolan, G.; Neilson, J.W.; Pryor, B.M.; Maier, R.M. Microbial ecology culturable microbial diversity and the impact of tourism in Kartchner Caverns, Arizona. Microb. Ecol. 2007, 53, 30–42. [Google Scholar] [CrossRef]
- Bastian, F.; Alabouvette, C.; Jurado, V.; Saiz-Jimenez, C. Impact of biocide treatments on the bacterial communities of the Lascaux Cave. Sci. Nat. 2009, 96, 863–868. [Google Scholar] [CrossRef] [PubMed]
- Mulec, J.; Pašić, L.; Prevoršek, Z.; Šenica, P.; Šket, R.; Petrič, M.; Holko, L.; Gostinčar, C.; Stres, B. Mikroorganizmi kot prvobitni sestavni del podzemnega sistema izvira Krajcarce (Triglavski Narodni Park). Acta Triglavensia 2017, 5, 29. [Google Scholar]
- Kostanjšek, R.; Pašić, L.; Daims, H.; Sket, B. Structure and community composition of sprout-like bacterial aggregates in a Dinaric Karst subterranean stream. Microb. Ecol. 2013, 66, 5–18. [Google Scholar] [CrossRef]
- Reitschuler, C.; Lins, P.; Schwarzenauer, T.; Spötl, C.; Wagner, A.O.; Illmer, P. New undescribed lineages of non-extremophilic archaea form a homogeneous and dominant element within Alpine moonmilk microbiomes. Geomicrobiol. J. 2015, 32, 890–902. [Google Scholar] [CrossRef]
- Cañaveras, C.S.; Sergio, C.; Sanchez-Moral, J.; Leonila, L.; Miguel, L.J.; Cesareo, G.; Saiz-Jimenez, J. On the origin of fiber calcite crystals in moonmilk deposits. Sci. Nat. 2006, 93, 27–32. [Google Scholar] [CrossRef]
- Portillo, M.C.; Gonzalez, J.M. Moonmilk deposits originate from specific bacterial communities in Altamira Cave (Spain). Microb. Ecol. 2011, 61, 182–189. [Google Scholar] [CrossRef][Green Version]
- Banks, E.D.; Taylor, N.M.; Gulley, J.; Lubbers, B.R.; Giarrizzo, J.G.; Bullen, H.A.; Hoehler, T.M.; Barton, H.A. Bacterial calcium carbonate precipitation in cave environments: A function of calcium homeostasis. Geomicrobiol. J. 2010, 27, 444–454. [Google Scholar] [CrossRef]
- Dos Santos, R.S.; Albino, U.B.; Paludo, K.S.; Silva, S.Y.S.; Oliveira, M.N.; Santos, D.A.; Marinho, A.M.D.R.; Marinho, P.B.; Lima, T.C.; Dos Santos, G.D.F.N.; et al. Extract of bacterial strain isolated from cave in the eastern Amazon induces selective cytotoxicity on tumor line of murine melanoma. Sci. Plena 2021, 17, 1–9. [Google Scholar] [CrossRef]
- Barton, H.A. Introduction to cave microbiology: A review for the non-specialist. J. Caves Karst Stud. 2006, 68, 43–54. [Google Scholar]
- Gunde-Cimerman, N.; Zalar, P.; Jeram, S. Mycoflora of cave cricket Troglophilus neglectus cadavers. Mycopathologia 1998, 141, 111–114. [Google Scholar] [CrossRef]
- Zalar, P.; Hennebert, G.; Gunde-Cimerman, N.; Cimerman, A. Mucor troglophilus, a new species from cave crickets. Mycotaxon 1997, 65, 507–516. [Google Scholar]
- Glavan, G. Production of Enzymes by Mucor Fungi, Isolated from Cave Cricket Troglophilus Neglectus. Licentiate Thesis, University of Ljubljana, Biotechnical Faculty, Ljubljana, Slovenia, 1997. [Google Scholar]
- Avguštin, J.A.; Petrič, A.; Pašić, L. Screening the cultivable cave microbial mats for the production of antimicrobial compounds and antibiotic resistance. Int. J. Speleol. 2019, 48, 295–303. [Google Scholar] [CrossRef]
- Mulec, J.; Oarga-Mulec, A.; Šturm, S.; Tomazin, R.; Matos, T. Spatio-temporal distribution and tourist impact on airborne bacteria in a cave (Škocjan Caves, Slovenia). Diversity 2017, 9, 28. [Google Scholar] [CrossRef]
- Mulec, J.; Simčič, S.; Kotar, T.; Kofol, R.; Stopinšek, S. Survey of histoplasma capsulatum in bat guano and status of histoplasmosis in Slovenia, Central Europe. Int. J. Speleol. 2020, 49, 1. [Google Scholar] [CrossRef]
- Mulec, J.; Oarga-Mulec, A.; Holko, L.; Pašić, L.; Kopitar, A.N.; Eleršek, T.; Mihevc, A. Microbiota entrapped in recently-formed ice: Paradana Ice Cave, Slovenia. Sci. Rep. 2021, 11, 1993. [Google Scholar] [CrossRef]
- Procházková, L.; Remias, D.; Řezanka, T.; Nedbalová, L. Chloromonas nivalis Subsp. tatrae, Subsp. Nov. (Chlamydomonadales, Chlorophyta): Re–Examination of a Snow Alga from the High Tatra Mountains (Slovakia). Fottea 2018, 18, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Remias, D.; Pichrtová, M.; Pangratz, M.; Lütz, C.; Holzinger, A. Ecophysiology, Secondary Pigments and Ultrastructure of Chlainomonas Sp. (Chlorophyta) from the European Alps Compared with Chlamydomonas Nivalis Forming Red Snow. FEMS Microbiol. Ecol. 2016, 92, fiw030. [Google Scholar] [CrossRef] [PubMed]
- Řezanka, T.; Nedbalová, L.; Sigler, K.; Cepák, V. Identification of astaxanthin diglucoside diesters from snow alga chlamydomonas nivalis by liquid chromatography-atmospheric pressure chemical ionization mass spectrometry. Phytochemistry 2008, 69, 479–490. [Google Scholar] [CrossRef]
- Řezanka, T.; Nedbalová, L.; Kolouchová, I.; Sigler, K. LC-MS/APCI identification of glucoside esters and diesters of astaxanthin from the snow alga Chlamydomonas nivalis including their optical stereoisomers. Phytochemistry 2013, 88, 34–42. [Google Scholar] [CrossRef]
- Procházková, L.; Remias, D.; Holzinger, A.; Řezanka, T.; Nedbalová, L. Ecophysiological and morphological comparison of two populations of Chlainomonas Sp. (Chlorophyta) causing red snow on ice-covered lakes in the high Tatras and Austrian Alps. Eur. J. Phycol. 2018, 53, 230–243. [Google Scholar] [CrossRef]
- Procházková, L.; Remias, D.; Řezanka, T.; Nedbalová, L. Ecophysiology of Chloromonas hindakii Sp. Nov. (Chlorophyceae), causing orange snow blooms at different light conditions. Microorganisms 2019, 7, 434. [Google Scholar] [CrossRef]
- Lukavský, J.; Furnadzhieva, S.; Nedbalová, L. First record of cryoseston in the Vitosha Mountains (Bulgaria). Nova Hedwig. 2009, 88, 97–109. [Google Scholar] [CrossRef]
- Nedbalová, L.; Kocianova, M.; Lukavský, J. Ecology of snow algae in the Giant Mts and their relation to cryoseston in Europe. Opera Corcon. 2008, 44, 59–68. [Google Scholar]
- Procházková, L.; Leya, T.; Krížková, H.; Nedbalová, L. Sanguina Nivaloides and Sanguina Aurantia Gen. Et Spp. Nov. (Chlorophyta): The taxonomy, phylogeny, biogeography and ecology of two newly recognised algae causing red and orange snow. FEMS Microbiol. Ecol. 2019, 95, 6. [Google Scholar] [CrossRef]
- Remias, D.; Holzinger, A.; Lütz, C. Physiology, ultrastructure and habitat of the ice alga Mesotaenium Berggrenii (Zygnemaphyceae, Chlorophyta) from Glaciers in the European Alps. Phycologia 2009, 48, 302–312. [Google Scholar] [CrossRef]
- Remias, D.; Schwaiger, S.; Aigner, S.; Leya, T.; Stuppner, H.; Lütz, C. Characterization of an UV- and VIS-Absorbing, purpurogallin-derived secondary pigment new to algae and highly abundant in Mesotaenium Berggrenii (Zygnematophyceae, Chlorophyta), an extremophyte living on glaciers. FEMS Microbiol. Ecol. 2012, 79, 638–648. [Google Scholar] [CrossRef]
- Allewaert, C.C.; Vanormelingen, P.; Proschold, T.; Gomez, P.I.; Gonzalez, M.A.; Bilcke, G.; D’Hondt, S.; Vyverman, W. Species Diversity in European Haematococcus pluvialis (Chlorophyceae, Volvocales). Phycologia 2015, 54, 583–598. [Google Scholar] [CrossRef]
- Geister, I. Sečovlje Saltpans, 1st ed.; Založba Kmečki Glas: Ljubljana, Slovenija, 2004; p. 151. [Google Scholar]
- Tkavc, R.; Gostinčar, C.; Turk, M.; Visscher, P.T.; Oren, A.; Gunde-Cimerman, N. Bacterial communities in the “petola” microbial mat from the Sečovlje Salterns (Slovenia). FEMS Microbiol. Ecol. 2010, 75, 48–62. [Google Scholar] [CrossRef]
- Geisler, D. De la Mer au Sel: Les Faciès Superficiels des Marais Salants de Salin-de-Giraud (Sud de La France). Geol. Mediterr. 1982, 9, 521–549. [Google Scholar] [CrossRef]
- Pašić, L.; Bartual, S.G.; Ulrih, N.P.; Grabnar, M.; Velikonja, B.H. Diversity of halophilic archaea in the crystallizers of an Adriatic solar saltern. FEMS Microbiol. Ecol. 2005, 54, 491–498. [Google Scholar] [CrossRef]
- Pašić, L.; Ulrih, N.P.; Črnigoj, M.; Grabnar, M.; Herzog Velikonja, B. Haloarchaeal communities in the crystallizers of two Adriatic Solar Salterns. Can. J. Microbiol. 2007, 53, 8–18. [Google Scholar] [CrossRef]
- Gyula, Z.K.; Tamás, F.; Boglárka, S.; Gyöngyi, S.; Nicolae, D.; Károly, M.; Bartha, C.; Vörös, L. First record of picophytoplankton diversity in Central European hypersaline lakes. Extremophiles 2012, 16, 759–769. [Google Scholar] [CrossRef]
- Glavaš, N.; Défarge, C.; Gautret, P.; Joulian, C.; Penhoud, P.; Motelica, M.; Kovač, N. The structure and role of the “Petola” microbial mat in sea salt production of the Sečovlje (Slovenia). Sci. Total Environ. 2018, 644, 1254–1267. [Google Scholar] [CrossRef]
- Máthé, I.; Borsodi, A.K.; Tóth, E.M.; Felföldi, T.; Jurecska, L.; Krett, G.; Kelemen, Z.; Elekes, E.; Barkács, K.; Márialigeti, K. Vertical Physico-chemical gradients with distinct microbial communities in the hypersaline and heliothermal Lake Ursu (Sovata, Romania). Extremophiles 2014, 18, 501–514. [Google Scholar] [CrossRef]
- Turk, M.; Plemenitaš, A.; Gunde-Cimerman, N. Extremophilic yeasts: Plasma-membrane fluidity as determinant of stress tolerance. Fungal Biol. 2011, 115, 950–958. [Google Scholar] [CrossRef]
- Kogej, T.; Ramos, J.; Plemenitaš, A.; Gunde-Cimerman, N. The halophilic fungus Hortaea werneckii and the halotolerant fungus Aureobasidium pullulans maintain low intracellular cation concentrations in hypersaline environments. Appl. Environ. Microbiol. 2005, 71, 6600–6605. [Google Scholar] [CrossRef]
- Plemenitaš, A.; Lenassi, M.; Konte, T.; Kejžar, A.; Zajc, J.; Gostinčar, C.; Gunde-Cimerman, N. Adaptation to high salt concentrations in halotolerant/halophilic fungi: A molecular perspective. Front. Microbiol. 2014, 5, 199. [Google Scholar] [CrossRef]
- Gunde-Cimerman, N.; Zalar, P.; de Hoog, S.; Plemenitaš, A. Hypersaline waters in salterns—Natural ecological niches for halophilic black yeasts. FEMS Microbiol. Ecol. 2000, 32, 235–240. [Google Scholar] [CrossRef]
- Derekova, A.; Mandeva, R.; Kambourova, M. Phylogenetic diversity of thermophilic carbohydrate degrading bacilli from Bulgarian hot springs. World J. Microbiol. Biotechnol. 2008, 24, 1697–1702. [Google Scholar] [CrossRef]
- Valcheva, N.; Ignatov, I.; Mihaylova, S. Physiological and molecular-genetic characteristic of bacteria strains, isolated from mountain spring and mineral waters in Plovdiv Region, Bulgaria. Int. J. Pathog. Res. 2020, 4, 44–55. [Google Scholar] [CrossRef]
- Eleršek, T.; Mulec, J. The algal community at an ecocline of a cold sulphidic spring (Sovra artesian borehole, Slovenia). Environ. Earth Sci. 2014, 71, 5255–5261. [Google Scholar] [CrossRef]
- Mulec, J.; Oarga, A.; Schiller, E.K.; Perşoiu, A.; Holko, L.; Šebela, S. Assessment of the physical environment of epigean invertebrates in a unique habitat: The case of a karst sulfidic spring, Slovenia. Ecohydrology 2015, 8, 1326–1334. [Google Scholar] [CrossRef]
- Anesio, A.M.; Lutz, S.; Chrismas, N.A.M.; Benning, L.G. The microbiome of glaciers and ice sheets. NPJ Biofilms Microbiomes 2017, 3, 10. [Google Scholar] [CrossRef] [PubMed]
- Williamson, C.J.; Cameron, K.A.; Cook, J.M.; Zarsky, J.D.; Stibal, M.; Edwards, A. Glacier algae: A dark past and a darker future. Front. Microbiol. 2019, 10, 519. [Google Scholar] [CrossRef]
- Perini, L.; Gostinčar, C.; Anesio, A.M.; Williamson, C.; Tranter, M.; Gunde-Cimerman, N. Darkening of the Greenland ice sheet: Fungal abundance and diversity are associated with algal bloom. Front. Microbiol. 2019, 10, 557. [Google Scholar] [CrossRef]
- Felip, M.; Sattler, B.; Psenner, R.; Catalan, J. Highly Active microbial communities in the ice and snow cover of high mountain lakes. Appl. Environ. Microbiol. 1995, 61, 2394–2401. [Google Scholar] [CrossRef]
- Williamson, C.J.; Anesio, A.M.; Cook, J.; Tedstone, A.; Poniecka, E.; Holland, A.; Fagan, D.; Tranter, M.; Yallop, M.L. Ice algal bloom development on the surface of the greenland ice sheet. FEMS Microbiol. Ecol. 2018, 94, 25. [Google Scholar] [CrossRef]
- Perini, L.; Gostinčar, C.; Gunde-Cimerman, N. Fungal and bacterial diversity of Svalbard subglacial ice. Sci. Rep. 2019, 9, 20230. [Google Scholar] [CrossRef]
- Hoham, R.W.; Remias, D. Snow and glacial algae: A review. J. Phycol. 2020, 56, 264–282. [Google Scholar] [CrossRef]
- Remias, D.; Holzinger, A.; Aigner, S.; Lütz, C. Ecophysiology and ultrastructure of Ancylonema Nordenskiöldii (Zygnematales, Streptophyta), causing brown ice on glaciers in Svalbard (High Arctic). Polar Biol. 2012, 35, 899–908. [Google Scholar] [CrossRef]
- Lutz, S.; Anesio, A.M.; Jorge Villar, S.E.; Benning, L.G. Variations of algal communities cause darkening of a Greenland glacier. FEMS Microbiol. Ecol. 2014, 89, 402–414. [Google Scholar] [CrossRef]
- Anesio, A.M.; Laybourn-Parry, J. Glaciers and ice sheets as a biome. Trends Ecol. Evol. 2012, 27, 219–225. [Google Scholar] [CrossRef]
- Margesin, R.; Zacke, G.; Schinner, F. Characterization of heterotrophic microorganisms in Alpine glacier cryoconite. Arct. Antarct. Alp. 2002, 34, 88–93. [Google Scholar] [CrossRef]
- Lütz-Meindl, U.; Lütz, C. Analysis of element accumulation in cell wall attached and intracellular particles of snow algae by EELS and ESI. Micron 2006, 37, 452–458. [Google Scholar] [CrossRef]
- Williams, W.E.; Gorton, H.L.; Vogelmann, T.C. Surface gas-exchange processes of snow algae. Proc. Natl. Acad. Sci. USA 2003, 100, 562–566. [Google Scholar] [CrossRef]
- Kawecka, B. Biology and ecology of snow algae. I. The sexual reproduction of Chlamydomonas nivalis (Bauer) Wille (Chlorophyta, Volvocales). Acta Hidrobiol. 1978, 20, 111–116. [Google Scholar]
- Newton, A.P.W. Red-coloured snow algae in Svalbard—Some environmental factors determining the distribution of Chlamydomonas nivalis (Chlorophyta Volvocales). Polar Biol. 1982, 1, 167–172. [Google Scholar] [CrossRef]
- Gorton, H.L.; Williams, W.E.; Vogelmann, T.C. The light environment and cellular optics of the snow alga Chlamydomonas nivalis (Bauer) Wille. Photochem. Photobiol. 2007, 73, 611–620. [Google Scholar] [CrossRef]
- Hoham, R.W.; Mullet, J.E. The life history and ecology of the snow alga Chloromonas cryophila sp. nov. (Chlorophyta, Volvocales). Phycologia 1977, 16, 53–68. [Google Scholar] [CrossRef]
- Mihevc, A. Ice caves in Slovenia. In Ice Caves, 1st ed.; Persoiu, A., Lauritzen, S.-E., Eds.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 691–703. [Google Scholar]
- Carey, A.; Zorn, M.; Tičar, J.; Lipar, M.; Komac, B.; Welch, S.; Smith, D.; Lyons, W. Glaciochemistry of cave ice: Paradana and Snežna Caves, Slovenia. Geosciences 2019, 9, 94. [Google Scholar] [CrossRef]
- Kern, Z.; Széles, E.; Horvatinčić, N.H.; Fórizs, I.; Bočić, N.; Nagy, B. Glaciochemical investigations of the ice deposit of Vukušić Ice Cave, Velebit Mountain, Croatia. Cryosphere 2011, 5, 485–494. [Google Scholar] [CrossRef]
- Citterio, M.; Turri, S.; Bini, A.; Maggi, V.; Pelfini, M.; Pini, R.; Ravazzi, C.; Santilli, M.; Stenni, B.; Udisti, R. Multidisciplinary approach to the study of the Lo Lc 1650 “Abisso Sul Margine Dell’Alto Bregai” Ice Cave (Lecco, Italy). Theor. Appl. Karstol. 2004, 17, 27–44. [Google Scholar]
- Hillebrand-Voiculescu, A.; Itcus, C.; Ardelean, I.; Pascu, D.; Perşoiu, A.; Rusu, A.; Brad, T.; Popa, E.; Onac, B.P.; Purcarea, C. Searching for cold-adapted microorganisms in the underground glacier of Scarisoara ice cave, Romania. Acta Carsol. 2014, 43, 319–329. [Google Scholar] [CrossRef]
- Müller, T.; Bleiß, W.; Martin, C.D.; Rogaschewski, S.; Fuhr, G. Snow algae from Northwest Svalbard: Their identification, distribution, pigment and nutrient content. Polar Biol. 1998, 20, 14–32. [Google Scholar] [CrossRef]
- Malavasi, V.; Soru, S.; Cao, G. Extremophile microalgae: The potential for biotechnological application. J. Phycol. 2020, 3, 559–573. [Google Scholar] [CrossRef]
- Allewaert, C.C.; Vanormelingen, P.; Daveloose, I.; Verstraete, T.; Vyverman, W. Intraspecific trait variation affecting astaxanthin productivity in two Haematococcus (Chlorophyceae) species. Algal Res. 2017, 21, 191–202. [Google Scholar] [CrossRef]
- Feller, G. Psychrophilic enzymes: From folding to function and biotechnology. Scientifica 2013, 2013, 512840. [Google Scholar] [CrossRef] [PubMed]
- Butinar, L.; Sonjak, S.; Zalar, P.; Plemenitaš, A.; Gunde-Cimerman, N. Melanized halophilic fungi are eukaryotic members of microbial communities in hypersaline waters of solar salterns. Bot. Mar. 2005, 48, 73–79. [Google Scholar] [CrossRef]
- Gunde-Cimerman, N.; Zalar, P.; Petrovič, U.; Turk, M.; Kogej, T.; de Hoog, G.S.; Plemenitaš, A. Fungi in Salterns. In Halophilic Microorganisms; Ventosa, A., Ed.; Springer: Berlin/Heidelberg, Germany, 2004; pp. 103–113. [Google Scholar]
- Chung, D.; Kim, H.; Choi, H.S. Fungi in salterns. J. Microbiol. 2019, 57, 717–724. [Google Scholar] [CrossRef]
- Oren, A. A hundred years of Dunaliella research: 1905–2005. Saline Syst. 2005, 1, 2. [Google Scholar] [CrossRef] [PubMed]
- Ventosa, A.; Arahal, D.R. Physico-chemical characteristics of hypersaline environments and their biodiversity. Extremophiles 2009, 2, 247–262. [Google Scholar]
- Ma, Y.; Galinski, E.A.; Grant, W.D.; Oren, A.; Ventosa, A. Halophiles 2010: Life in saline environments. Appl. Environ. Microbiol. 2010, 76, 6971–6981. [Google Scholar] [CrossRef]
- Oren, A. Microscopic Examination of microbial communities along a salinity gradient in saltern evaporation ponds: A ‘Halophilic Safari’. In Adaptation to Life at High Salt Concentrations in Archaea, Bacteria, and Eukarya; Gunde-Cimerman, N., Oren, A., Plemenitaš, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 41–57. [Google Scholar]
- Oren, A. The halophilic world of Lourens Baas Becking. In Halophiles and Hypersaline Environments; Ventosa, A., Oren, A., Ma, Y., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; pp. 215–232. [Google Scholar]
- Oren, A.; Meng, F.W. “Red—The magic color for solar salt production”—The since when? FEMS Microbiol. Lett. 2019, 366, 50. [Google Scholar] [CrossRef]
- Pedrós-Alió, C. Diversity of Microbial Communities: The Case of Solar Salterns. In Adaptation to Life at High Salt Concentrations in Archaea, Bacteria, and Eukarya; Gunde-Cimerman, N., Oren, A., Plemenitaš, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 71–90. [Google Scholar]
- Oren, A. Microbial life at high salt concentrations: Phylogenetic and metabolic diversity. Saline Syst. 2008, 4, 2. [Google Scholar] [CrossRef]
- Pérez-Llano, Y.; Rodríguez-Pupo, E.C.; Druzhinina, I.S.; Chenthamara, K.; Cai, F.; Gunde-Cimerman, N.; Zalar, P.; Gostinčar, C.; Kostanjšek, R.; Folch-Mallol, J.L.; et al. Stress reshapes the physiological response of halophile fungi to salinity. Cells 2020, 9, 525. [Google Scholar] [CrossRef]
- Gunde-Cimerman, N.; Butinar, L.; Sonjak, S.; Turk, M.; Uršič, V.; Zalar, P. Cellular Responses in the Halophilic Black Yeast Hortaea Werneckii to High Environmental Salinity. In Adaptation to Life at High Salt Concentrations in Archaea, Bacteria, and Eukarya; Gunde-Cimerman, N., Oren, A., Plemenitaš, A., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 453–470. [Google Scholar]
- Antón, J.; Rosselló-Mora, R.; Rodríguez-Valera, F.; Amann, R. Extremely halophilic bacteria in crystallizer ponds from solar salterns. Appl. Environ. Microbiol. 2000, 66, 3052–3057. [Google Scholar] [CrossRef]
- Oren, A. The microbiology of red brines. In Advances in Applied Microbiology, 1st ed.; Jackman, A.M., Johnson, H., Eds.; Elsevier: London, UK, 2020; Volume 113, pp. 57–110. [Google Scholar]
- Oren, A. Molecular ecology of extremely halophilic archaea and bacteria. FEMS Microbiol. Ecol. 2002, 39, 1–7. [Google Scholar] [CrossRef]
- Bardavid, R.E.; Polina, K.; Oren, A. Interrelationships between Dunaliella and halophilic prokaryotes in saltern crystallizer ponds. Extremophiles 2008, 12, 5–14. [Google Scholar] [CrossRef]
- Gunde-Cimerman, N.; Ramos, J.; Plemenitaš, A. Halotolerant and halophilic fungi. Mycol. Res. 2009, 113, 1231–1241. [Google Scholar] [CrossRef]
- Zalar, P.; Kocuvan, M.A.; Plemenitaš, A.; Gunde-Cimerman, N. Halophilic black yeasts colonize wood immersed in hypersaline water. Bot. Mar. 2005, 48, 323–326. [Google Scholar] [CrossRef]
- Oh, D.; Porter, K.; Russ, B.; Burns, D.; Dyall-Smith, M. Diversity of Haloquadratum and other haloarchaea in three, geographically distant, Australian saltern crystallizer ponds. Extremophiles 2010, 14, 161–169. [Google Scholar] [CrossRef]
- Ghai, R.; Pašic, L.; Fernández, A.B.; Martin-Cuadrado, A.-B.B.; Mizuno, C.M.; Mcmahon, K.D.; Papke, R.T.; Stepanauskas, R.; Rodriguez-Brito, B.; Rohwer, F.; et al. New abundant microbial groups in aquatic hypersaline environments. Sci. Rep. 2011, 1, 135. [Google Scholar] [CrossRef]
- Zafrilla, B.; Martínez-Espinosa, R.M.; Alonso, M.A.; Bonete, M.J. Biodiversity of archaea and floral of two inland saltern ecosystems in the Alto Vinalopó Valley, Spain. Saline Syst. 2010, 6, 10. [Google Scholar] [CrossRef]
- Pahor, M.; Poberaj, T. Stare Piranske Soline, 1st ed.; Mladinska Knjiga: Ljubljana, Slovenija, 1963; p. 45. [Google Scholar]
- Škrinjar, P.; Faganeli, J.; Ogrinc, N. The Role of stromatolites in explaining patterns of carbon, nitrogen, phosphorus, and silicon in the Sečovlje saltern evaporation ponds (Northern Adriatic Sea). J. Soils Sediments 2012, 12, 1641–1648. [Google Scholar] [CrossRef]
- Singh, R.P.; Manchanda, G.; Maurya, I.K.; Wei, Y. Microbial Versatility in Varied Environments: Microbes in Sensitive Environments; Springer Nature: Singapore, 2020. [Google Scholar] [CrossRef]
- Mishra, S.S.; Ray, R.C.; Rosell, C.M.; Panda, D. Microbial Enzymes in Food Applications: History of Progress. In Microbal Enzyme Technology in Food Applications; Ray, R.C., Rosell, C.M., Eds.; Taylor & Francis Group: New York, NY, USA, 2017; pp. 3–18. [Google Scholar] [CrossRef]
- Oren, A. Industrial and environmental applications of halophilic microorganisms. Environ. Technol. 2010, 31, 825–834. [Google Scholar] [CrossRef]
- Graf, R.; Anzali, S.; Buenger, J.; Pfluecker, F.; Driller, H. The Multifunctional role of ectoine as a natural cell protectant. Clin. Dermatol. 2008, 26, 326–333. [Google Scholar] [CrossRef] [PubMed]
- Pašić, L.; Velikonja, B.H.; Ulrih, N.P. Optimization of the culture conditions for the production of a bacteriocin from halophilic archaeon Sech7a. Prep. Biochem. Biotechnol. 2008, 38, 229–245. [Google Scholar] [CrossRef] [PubMed]
- DasSarma, S.; DasSarma, P. Halophiles and their enzymes: Negativity put to good use. Curr. Opin. Microbiol. 2015, 25, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Castenholz, R.W. Thermophilic blue-green algae and the thermal environment. Bacteriol. Rev. 1969, 33, 476–504. [Google Scholar] [CrossRef]
- Horvatinčić, N.; Srdoč, D.; Krajcar, B.I.; Pezdic, J.; Kapelj, S.; Sliepčević, A. A Study of geothermal waters in Northwest Croatia and East Slovenia. Isotopes in water resources management. In Proceedings of the Symposium, Vienna, Austria, 20–24 March 1995; International Atomic Energy Agency (IAEA): Vienna, Austria, 1996; Volume 2. [Google Scholar]
- Zmazek, B.; Italiano, F.; Ivči, M.; Vaupotič, J.; Kobal, I.; Martinelli, G. Geochemical monitoring of thermal waters in Slovenia: Relationships to seismic activity. Appl. Radiat. Isot. 2002, 57, 919–930. [Google Scholar] [CrossRef]
- Kralj, P. Chemical composition of low temperature (<20–40 °C) thermal waters in Slovenia. Environ. Geol. 2004, 46, 635–642. [Google Scholar] [CrossRef]
- Šajna, N.; Haler, M.; Škornik, S.; Kaligarič, M. Survival and expansion of Pistia Stratiotes L. in a thermal stream in Slovenia. Aquat. Bot. 2007, 87, 75–79. [Google Scholar] [CrossRef]
- De Mattia, W. A new subterranean hydrobiid from a thermal spring in Eastern Slovenia: “Iglica” Velkovrhi n. Sp. (Gastropoda: Prosobranchia: Hydrobiidae). Mollusca 2007, 25, 27–31. [Google Scholar]
- Tumbarski, Y.; Valcheva-Zhekova, N.; Koleva, I.; Denkova, Z. Antimicrobial activity against some saprophytic and pathogenic microorganisms of bacillus species strains isolated from natural spring waters in Bulgaria. Br. Microbiol. Res. J. 2014, 4, 1353–1369. [Google Scholar] [CrossRef]
- Kosseva, M.R.; Fatmawati, A.; Palatova, M.; Kent, C.A. Modelling thermophilic cheese whey bioremediation in a one-stage process. Biochem. Eng. J. 2007, 35, 281–288. [Google Scholar] [CrossRef]
- Zottola, T.; Briganti, P.; Cuoco, E.; D’Amici, L.; De Gregorio, A.; Guzzon, L.; Manocchio, A.; Mancuso, M.; Condoleo, R.U. Study of the microbiological hazards in the prodution of the mozzarella buffalo cheese. Ital. J. Food Saf. 2009, 1, 39–43. [Google Scholar] [CrossRef][Green Version]
- André, S.; Zuber, F.; Remize, F. Thermophilic spore-forming bacteria isolated from spoiled canned food and their heat resistance. Results of a French ten-year survey. Int. J. Food Microbiol. 2013, 165, 134–143. [Google Scholar] [CrossRef]
- Lee, J.; Shin, S.G.; Han, G.; Koo, T.; Hwang, S. Bacteria and archaea communities in full-scale thermophilic and mesophilic anaerobic digesters treating food wastewater: Key process parameters and microbial indicators of process instability. Bioresour. Technol. 2017, 245, 689–697. [Google Scholar] [CrossRef]
- Schlesinger, W.H.; Bernhardt, E.S. Origins. In Biogeochemistry: An Analysis of Global Change, 3rd ed.; Schlesinger, W.H., Bernhardt, E.S., Eds.; Academic Press: Waltham, MA, USA, 2013; pp. 15–48. [Google Scholar]
- Overmann, J.; van Gemerden, H. Microbial interactions involving sulfur bacteria: Implications for the ecology and evolution of bacterial communities. FEMS Microbiol. Rev. 2000, 24, 591–599. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zgonik, V.; Mulec, J.; Eleršek, T.; Ogrinc, N.; Jamnik, P.; Ulrih, N.P. Extremophilic Microorganisms in Central Europe. Microorganisms 2021, 9, 2326. https://doi.org/10.3390/microorganisms9112326
Zgonik V, Mulec J, Eleršek T, Ogrinc N, Jamnik P, Ulrih NP. Extremophilic Microorganisms in Central Europe. Microorganisms. 2021; 9(11):2326. https://doi.org/10.3390/microorganisms9112326
Chicago/Turabian StyleZgonik, Vera, Janez Mulec, Tina Eleršek, Nives Ogrinc, Polona Jamnik, and Nataša Poklar Ulrih. 2021. "Extremophilic Microorganisms in Central Europe" Microorganisms 9, no. 11: 2326. https://doi.org/10.3390/microorganisms9112326
APA StyleZgonik, V., Mulec, J., Eleršek, T., Ogrinc, N., Jamnik, P., & Ulrih, N. P. (2021). Extremophilic Microorganisms in Central Europe. Microorganisms, 9(11), 2326. https://doi.org/10.3390/microorganisms9112326








