Analysis of Arbuscular Mycorrhizal Fungal Inoculant Benchmarks
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Collection
2.2. Data Analysis
3. Results
3.1. Mycorrhiza Inoculant Company by Country
3.2. Formula and Composition of Mycorrhizal Inoculants
3.3. Propagule Concentration
3.4. Industrial Claims on Effects of the AMF-Based Inoculants
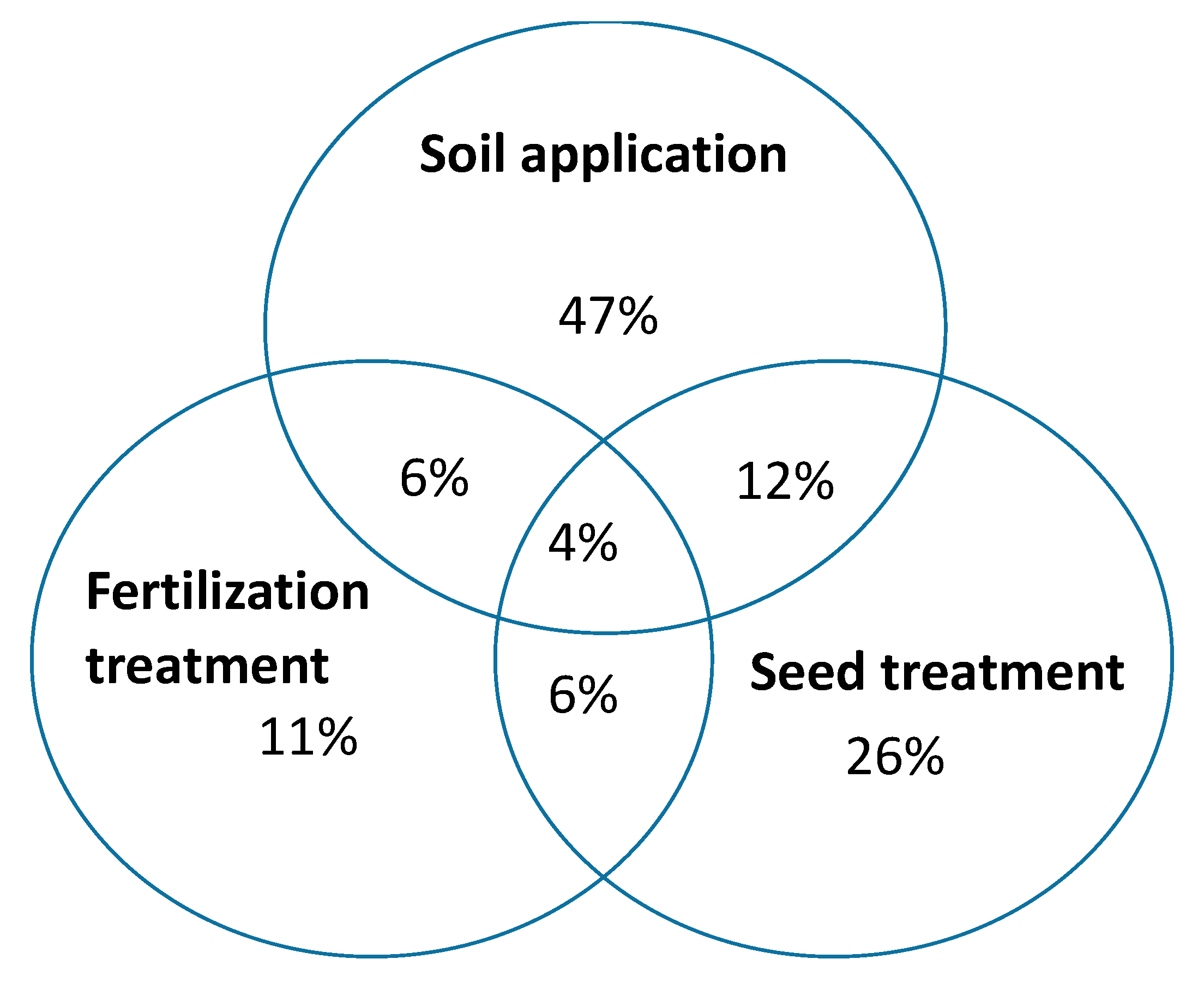
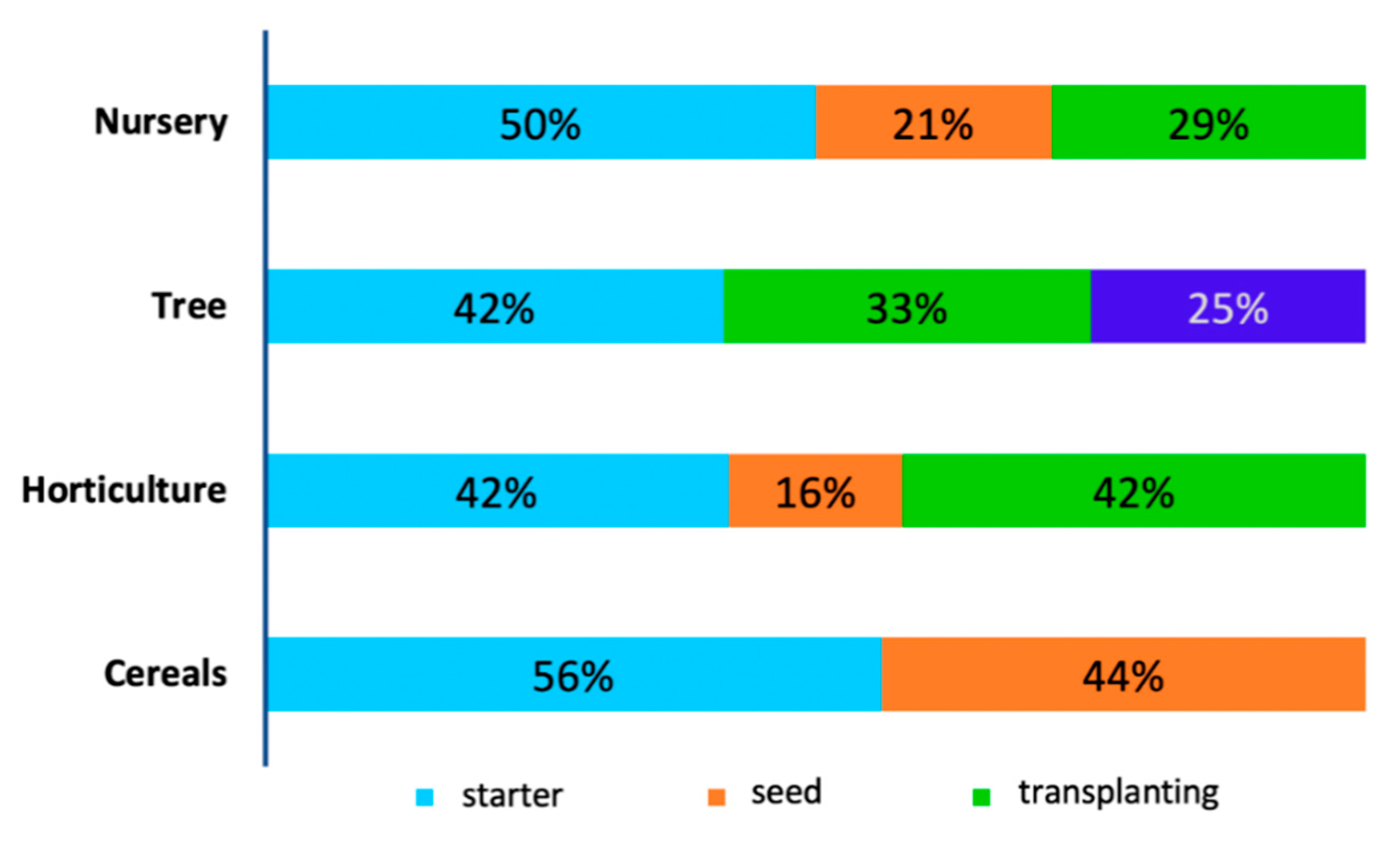
3.5. Recommended Inoculant Application Methods and Doses
4. Discussion
4.1. Product Breakdown by Country of Production
4.2. AMF-Based Inoculant Formulation
4.3. Inoculant Composition
4.4. Propagule Concentration
4.5. Industrial Claims on the Effects of AMF-Based Inoculants
4.6. Recommended Inoculant Application Methods and Doses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bio-FITProject. Biofertilizers Market Size. Available online: https://www.bio-fit.eu/hu/q5/lo12-bio-fertilizers-market-size?showall=1 (accessed on 29 December 2020).
- Fernández, M.F.; Molina, J.J.; Nicolás, E.N.; Alarcón, J.J.; Kirchmair, M.; García, F.J.; Garcia, A.J.B.; Bernal, C. Application of Arbuscular Mycorrhizae Glomus iranicum var. tenuihypharum var. nova in Intensive Agriculture: A Study Case. J. Agric. Sci. Technol. B 2017, 7. [Google Scholar] [CrossRef]
- Ortaş, İ.; Rafique, M. The Mechanisms of Nutrient Uptake by Arbuscular Mycorrhizae. In Mycorrhiza—Nutrient Uptake, Biocontrol, Ecorestoration; Varma, A., Prasad, R., Tuteja, N., Eds.; Springer: Cham, Swizerland, 2017; pp. 1–19. [Google Scholar]
- Makarov, M. The Role of Mycorrhiza in Transformation of Nitrogen Compounds in Soil and Nitrogen Nutrition of Plants: A Review. Eurasian Soil Sci. 2019, 52, 193–205. [Google Scholar] [CrossRef]
- Smith, S.; Read, D. Mycorrhizal Symbiosis; Elsevier Ltd.: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Berruti, A.; Lumini, E.; Balestrini, R.; Bianciotto, V. Arbuscular Mycorrhizal Fungi as Natural Biofertilizers: Let’s Benefit from Past Successes. Front. Microbiol. 2015, 6, 1559. [Google Scholar] [CrossRef]
- 7. UNDESA, U.N. Department of Economic and Social Affairs, Population Division. How Certain Are the United Nations Global Population Projections? World Population Prospects 2019: Ten Key Findings. 2019; Population Facts No. 2019/6; UNDESA, U.N. Department of Economic and Social Affairs, Population Division: New York, NY, USA, 2019. [Google Scholar]
- Hunter, M.C.; Smith, R.G.; Schipanski, M.E.; Atwood, L.W.; Mortensen, D.A. Agriculture in 2050: Recalibrating Targets for Sustainable Intensification. BioScience 2017, 67, 386–391. [Google Scholar] [CrossRef]
- Vosátka, M.; Albrechtová, J.; Patten, R. The international market development for mycorrhizal technology. In Mycorrhiza: State of the Art, Genetics and Molecular Biology, Eco-Function, Biotechnology, Eco-Physiology, Structure and Systematics, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 419–438. [Google Scholar]
- Subashini, H.D.; Malarvannan, S.; Kumaran, P. Effect of Biofertilizers (N-Fixers) on the Yield of Rice Varieties at Puducherry, India. Asian J. Agric. Res. 2007, 1, 146–150. [Google Scholar] [CrossRef]
- Mahdi, S.; Hassan, G.; Samoon, S.; Rather, H.; Dar, S.; Zehra, B. Bio-fertilizers in organic agriculture. J. Phytol. 2010, 2, 42–54. [Google Scholar]
- Hijri, M. Analysis of a large dataset of mycorrhiza inoculation field trials on potato shows highly significant increases in yield. Mycorrhiza 2016, 26, 209–214. [Google Scholar] [CrossRef]
- Mukhongo, R.W.; Tumuhairwe, J.B.; Ebanyat, P.; AbdelGadir, A.H.; Thuita, M.; Masso, C. Production and Use of Arbuscular Mycorrhizal Fungi Inoculum in Sub-Saharan Africa: Challenges and Ways of Improving. Int. J. Soil Sci. 2016, 11, 108–122. [Google Scholar] [CrossRef]
- Vosátka, M.; Látr, A.; Gianinazzi, S.; Albrechtová, J. Development of arbuscular mycorrhizal biotechnology and industry: Current achievements and bottlenecks. Symbiosis 2013, 58, 29–37. [Google Scholar] [CrossRef]
- Adholeya, A.; Tiwari, P.; Singh, R. Large-Scale Inoculum Production of Arbuscular Mycorrhizal Fungi on Root Organs and Inoculation Strategies. In In Vitro Culture of Mycorrhizas; Declerck, S., Fortin, J.A., Strullu, D.-G., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 315–338. [Google Scholar]
- Verbruggen, E.; van der Heijden, M.G.; Rillig, M.C.; Kiers, E.T. Mycorrhizal fungal establishment in agricultural soils: Factors determining inoculation success. New Phytol. 2013, 197, 1104–1109. [Google Scholar] [CrossRef]
- Hart, M.M.; Antunes, P.M.; Chaudhary, V.B.; Abbott, L.K.; Field, K. Fungal inoculants in the field: Is the reward greater than the risk? Funct. Ecol. 2017, 32, 126–135. [Google Scholar] [CrossRef]
- Owen, D.; Williams, A.P.; Griffith, G.W.; Withers, P.J.A. Use of commercial bio-inoculants to increase agricultural production through improved phosphrous acquisition. Appl. Soil Ecol. 2015, 86, 41–54. [Google Scholar] [CrossRef]
- Berruti, A.; Borriello, R.; Lumini, E.; Scariot, V.; Bianciotto, V.; Balestrini, R. Application of laser microdissection to identify the mycorrhizal fungi that establish arbuscules inside root cells. Front. Plant Sci. 2013, 4, 135. [Google Scholar] [CrossRef]
- Renaut, S.; Daoud, R.; Masse, J.; Vialle, A.; Hijri, M. Inoculation with Rhizophagus Irregularis Does Not Alter Arbuscular Mycorrhizal Fungal Community Structure within the Roots of Corn, Wheat, and Soybean Crops. Microorganisms 2020, 8, 83. [Google Scholar] [CrossRef]
- Rosa, D.; Pogiatzis, A.; Bowen, P.; Kokkoris, V.; Richards, A.; Holland, T.; Hart, M. Performance and Establishment of a Commercial Mycorrhizal Inoculant in Viticulture. Agriculture 2020, 10, 539. [Google Scholar] [CrossRef]
- Faye, A.; Stewart, Z.P.; Ndung’u-Magiroi, K.; Diouf, M.; Ndoye, I.; Diop, T.; Dalpé, Y.; Prasad, P.V.V.; Lesueur, D. Testing of Commercial Inoculants to Enhance P Uptake and Grain Yield of Promiscuous Soybean in Kenya. Sustainability 2020, 12, 3803. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, J.; Bonini, P.; Cardarelli, M. Coating seeds with endophytic fungi enhances growth, nutrient uptake, yield and grain quality of winter wheat. Int. J. Plant Prod. 2015, 9, 171–189. [Google Scholar]
- Karaca, H.; Uygur, V.; Özkan, A.; Kaya, Z. Effects of Mycorrhizae and Fertilization on Soybean Yield and Nutrient Uptake. Commun. Soil Sci. Plant Anal. 2013, 44, 2459–2471. [Google Scholar] [CrossRef]
- Liu, N.; Chen, X.; Song, F.; Liu, F.; Liu, S.; Zhu, X. Effects of arbuscular mycorrhiza on growth and nutrition of maize plants under low temperature stress. Philipp. Agric. Sci. 2016, 99, 246–252. [Google Scholar]
- Meddich, A.; Mokhtar, M.A.E.; Bourzik, W.; Mitsui, T.; Baslam, M.; Hafidi, M. Optimizing Growth and Tolerance of Date Palm (Phoenix dactylifera L.) to Drought, Salinity, and Vascular Fusarium-Induced Wilt (Fusarium oxysporum) by Application of Arbuscular Mycorrhizal Fungi (AMF). In Root Biology; Giri, B., Prasad, R., Varma, A., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 239–258. [Google Scholar]
- Pal, S.; Singh, H.B.; Farooqui, A.; Rakshit, A. Commercialization of arbuscular mycorrhizal technology in agriculture and forestry. In Agriculturally Important Microorganisms: Commercialization and Regulatory Requirements in Asia; Springer: Singapore, 2016; pp. 97–105. [Google Scholar]
- Chen, M.; Arato, M.; Borghi, L.; Nouri, E.; Reinhardt, D. Beneficial Services of Arbuscular Mycorrhizal Fungi-From Ecology to Application. Front. Plant Sci. 2018, 9, 1270. [Google Scholar] [CrossRef]
- Ryan, M.H.; Kirkegaard, J.A. The agronomic relevance of arbuscular mycorrhizas in the fertility of Australian extensive cropping systems. Agric. Ecosyst. Environ. 2012, 163, 37–53. [Google Scholar] [CrossRef]
- Ayling, S.M.; Smith, S.E.; Smith, F.A. Colonisation by arbuscular mycorrhizal fungi changes the relationship between phosphorus uptake and membrane potential in leek (Allium porrum) seedlings. Aust. J. Plant Physiol. 2001, 28, 391–399. [Google Scholar] [CrossRef]
- Brundrett, M.C.; Abbott, L.K.; Jasper, D.A. Glomalean mycorrhizal fungi from tropical Australia: I. Comparison of the effectiveness and specificity of different isolation procedures. Mycorrhiza 1999, 8, 305–314. [Google Scholar] [CrossRef]
- Duponnois, R.; Plenchette, C.; Prin, Y.; Ducousso, M.; Kisa, M.; Bâ, A.M.; Galiana, A. Use of mycorrhizal inoculation to improve reafforestation process with Australian Acacia in Sahelian ecozones. Ecol. Eng. 2007, 29, 105–112. [Google Scholar] [CrossRef]
- Reiter, N.; Lawrie, A.; Walsh, N. The mycorrhizal associations of Borya mirabilis, an endangered Australian native plant. Muelleria 2013, 31, 81–88. [Google Scholar]
- Rouphael, Y.; Franken, P.; Schneider, C.; Schwarz, D.; Giovannetti, M.; Agnolucci, M.; Pascale, S.D.; Bonini, P.; Colla, G. Arbuscular mycorrhizal fungi act as biostimulants in horticultural crops. Sci. Hortic. 2015, 196, 91–108. [Google Scholar] [CrossRef]
- Tiwari, P.; Adholeya, A. In vitro co-culture of two AMF isolates Gigaspora margarita and Glomus intraradices on Ri T-DNA transformed roots. FEMS Microbiol. Lett. 2002, 206, 39–43. [Google Scholar] [CrossRef]
- Kumar, U.; Berliner, J.; Adak, T.; Rath, P.C.; Dey, A.; Pokhare, S.S.; Jambhulkar, N.N.; Panneerselvam, P.; Kumar, A.; Mohapatra, S.D. Non-target effect of continuous application of chlorpyrifos on soil microbes, nematodes and its persistence under sub-humid tropical rice-rice cropping system. Ecotoxicol. Environ. Saf 2017, 135, 225–235. [Google Scholar] [CrossRef]
- Fernández, F.; Dell’Amico, J.; Angoa, M.; de la Providencia, I. Use of a liquid inoculum of the arbuscular mycorrhizal fungi Glomus hoi in rice plants cultivated in a saline Gleysol: A new alternative to inoculate. J. Plant Breed. Crop Sci. 2011, 3, 24–33. [Google Scholar]
- Miguel, D.A.R.J.; Feliica, F.M.; Yuniesky, P.C. Liquid Mycorrhizal Inoculant; Office, E.P., Ed.; Instituto nacional de ciencias agricolas (INCA): San José de las Lajas, Cuba, 2007; pp. 1–8. [Google Scholar]
- Calvet, C.; Camprubi, A.; Pérez-Hernández, A.; Lovato, P.E. Plant Growth Stimulation and Root Colonization Potential of In Vivo versus In Vitro Arbuscular Mycorrhizal Inocula. HortScience 2013, 48, 897. [Google Scholar] [CrossRef]
- Redecker, D.; Schüßler, A.; Stockinger, H.; Stürmer, S.L.; Morton, J.B.; Walker, C. An evidence-based consensus for the classification of arbuscular mycorrhizal fungi (Glomeromycota). Mycorrhiza 2013, 23, 515–531. [Google Scholar] [CrossRef] [PubMed]
- Stürmer, S.L.; Oliveira, L.Z.; Morton, J.B. Gigasporaceae versus Glomeraceae (phylum Glomeromycota): A biogeographic tale of dominance in maritime sand dunes. Fungal Ecol. 2018, 32, 49–56. [Google Scholar] [CrossRef]
- Helgason, T.; Merryweather, J.; Young, J.P.; Fitter, A.; Helgason, T.; Merryweather, J.W.; Young, J.P.W.; Fitter, A.H. Specificity and resilience in the arbuscular mycorrhizal fungi of a natural woodland community. J. Ecol. 2007, 95, 623–630. [Google Scholar] [CrossRef]
- Opik, M.; Vanatoa, A.; Vanatoa, E.; Moora, M.; Davison, J.; Kalwij, J.M.; Reier, U.; Zobel, M. The online database MaarjAM reveals global and ecosystemic distribution patterns in arbuscular mycorrhizal fungi (Glomeromycota). New Phytol. 2010, 188, 223–241. [Google Scholar] [CrossRef]
- Veresoglou, S.D.; Shaw, L.J.; Sen, R. Glomus intraradices and Gigaspora margarita arbuscular mycorrhizal associations differentially affect nitrogen and potassium nutrition of Plantago lanceolata in a low fertility dune soil. Plant Soil 2011, 340, 481–490. [Google Scholar] [CrossRef]
- Graham, J.H.; Abbott, L.K. Wheat responses to aggressive and non-aggressive arbuscular mycorrhizal fungi. Plant Soil 2000, 220, 207–218. [Google Scholar] [CrossRef]
- Duan, T.; Facelli, E.; Smith, S.E.; Smith, F.A.; Nan, Z. Differential effects of soil disturbance and plant residue retention on function of arbuscular mycorrhizal (AM) symbiosis are not reflected in colonization of roots or hyphal development in soil. Soil Biol. Biochem. 2011, 43, 571–578. [Google Scholar] [CrossRef]
- Rivera, R.A.; Fernaández, F. Inoculation and Management of Mycorrhizal Fungi within Tropical Agroecosystems. In Biological Approaches to Sustainable Soil Systems; Uphof, N., Ed.; Cornell University: New York, NY, USA, 2005; pp. 479–488. [Google Scholar]
- Cavagnaro, T.R.; Smith, F.A.; Smith, S.E.; Jakobsen, I. Functional diversity in arbuscular mycorrhizas: Exploitation of soil patches with different phosphate enrichment differs among fungal species. Plant Cell Environ. 2005, 28, 642–650. [Google Scholar] [CrossRef]
- Fernández, F.; Vicente-Sánchez, J.; Maestre-Valero, J.F.; Bernabé, A.J.; Nicolás, E.; Pedrero, F.; Alarcón, J.J. Physiological and growth responses of young tomato seedlings to drip-irrigation containing two low doses of the arbuscular mycorrhizal fungus Glomus iranicum var. tenuihypharum sp. nova. J. Horticult. Sci. Biotechnol. 2015, 89, 679–685. [Google Scholar] [CrossRef]
- Wagg, C.; Barendregt, C.; Jansa, J.; Van der Heijden, M. Complementarity in both plant and mycorrhizal fungal communities are not necessarily increased by diversity in the other. J. Ecol. 2015, 103. [Google Scholar] [CrossRef]
- Agnolucci, M.; Avio, L.; Pepe, A.; Turrini, A.; Cristani, C.; Bonini, P.; Cirino, V.; Colosimo, F.; Ruzzi, M.; Giovannetti, M. Bacteria Associated With a Commercial Mycorrhizal Inoculum: Community Composition and Multifunctional Activity as Assessed by Illumina Sequencing and Culture-Dependent Tools. Front. Plant Sci. 2018, 9, 1956. [Google Scholar] [CrossRef] [PubMed]
- Kokkoris, V.; Hart, M. In vitro Propagation of Arbuscular Mycorrhizal Fungi May Drive Fungal Evolution. Front. Microbiol. 2019, 10, 2420. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Sharma, A.K.; Prasad, R.; Varma, A. Arbuscular mycorrhiza: A tool for enhancing crop production. In Mycorrhiza-Nutrient Uptake, Biocontrol, Ecorestoration, 4th ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 235–250. [Google Scholar]
- Cely, M.V.T.; Oliveira, A.G.; Freitas, V.F.; Luca, M.B.; Barazetti, A.R.; Santos, I.M.O.; Gionco, B.; Garcia, G.V.; Prete, C.E.C.; Andrade, G. Inoculant of Arbuscular Mycorrhizal Fungi (Rhizophagus clarus) Increase Yield of Soybean and Cotton under Field Conditions. Front Microbiol. 2016, 7, 720. [Google Scholar] [CrossRef] [PubMed]
- Castillo, C.; Oehl, F.; Sieverding, E. Arbuscular mycorrhizal fungal diversity in wheat agro-ecosystems in Southern Chile and effects of seed treatment with natural products. J. Soil Sci. Plant Nutr. 2016, 16. [Google Scholar] [CrossRef]
- Colonna, E.; Rouphael, Y.; Pascale, S.D.; Barbieri, G. Effects of mycorrhiza and plant growth promoting rhizobacteria on yield and quality of artichoke. Acta Horticult. 2016, 1147, 43–50. [Google Scholar] [CrossRef]
- Antunes, P.M.; Franken, P.; Schwarz, D.; Rillig, M.C.; Cosme, M.; Scott, M.; Hart, M.M. Linking soil biodiversity and human health: Do arbuscular mycorrhizal fungi contribute to food nutrition. In Soil Ecology and Ecosystem Services; Wall, D.H.E.A., Ed.; Oxford University Press: Oxford, UK, 2012; pp. 153–172. [Google Scholar]
- Varma, A.; Prasad, R.; Tuteja, N. Mycorrhiza-Nutrient Uptake, Biocontrol, Ecorestoration, 4th ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 1–533. [Google Scholar] [CrossRef]
- Wu, Q.-S.; Zou, Y.-N. Arbuscular Mycorrhizal Fungi and Tolerance of Drought Stress in Plants. In Arbuscular Mycorrhizas and Stress Tolerance of Plants; Springer: Singapore, 2017; pp. 25–41. [Google Scholar]
- Fernández, F.; Juárez, J.; Bernabe, A.J.; García, F.J.; Gómez, J.M. Activity of the arbuscular mycorrhizal fungus, Glomus iranicum var. tenuihypharum var. nova, and its effect on citrus development in southeastern Spain. Acta Hortic. 2019, 73–84. [Google Scholar] [CrossRef]
- Yang, H.; Schroeder-Moreno, M.; Giri, B.; Hu, S. Arbuscular Mycorrhizal Fungi and Their Responses to Nutrient Enrichment. In Root Biology; Giri, B., Prasad, R., Varma, A., Eds.; Springer International Publishing: Cham, Switzerlands, 2018; pp. 429–449. [Google Scholar]
- Lü, L.-H.; Zou, Y.-N.; Wu, Q.-S. Relationship Between Arbuscular Mycorrhizas and Plant Growth: Improvement or Depression? In Root Biology; Giri, B., Prasad, R., Varma, A., Eds.; Springer International Publishing: Cham, Switzerlands, 2018; pp. 451–464. [Google Scholar]
- Bücking, H.; Kafle, A. Role of Arbuscular Mycorrhizal Fungi in the Nitrogen Uptake of Plants: Current Knowledge and Research Gaps. Agronomy 2015, 5, 587–612. [Google Scholar] [CrossRef]
- Reynolds, H.L.; Hartley, A.E.; Vogelsang, K.M.; Bever, J.D.; Schultz, P.A. Arbuscular mycorrhizal fungi do not enhance nitrogen acquisition and growth of old-field perennials under low nitrogen supply in glasshouse culture. New Phytol. 2005, 167, 869–880. [Google Scholar] [CrossRef]
- Wang, X.X.; Wang, X.J.; Sun, Y.; Cheng, Y.; Liu, S.; Chen, X.P.; Feng, G.; Kuyper, T.W. Arbuscular Mycorrhizal Fungi Negatively Affect Nitrogen Acquisition and Grain Yield of Maize in a N Deficient Soil. Front. Microbiol. 2018, 9, 418. [Google Scholar] [CrossRef]
- Van der Heijden, M.G.; Streitwolf-Engel, R.; Riedl, R.; Siegrist, S.; Neudecker, A.; Ineichen, K.; Boller, T.; Wiemken, A.; Sanders, I.R. The mycorrhizal contribution to plant productivity, plant nutrition and soil structure in experimental grassland. New Phytol. 2006, 172, 739–752. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.C.; Graham, J.H.; Smith, F.A. Functioning of mycorrhizal associations along the mutualism–parasitism continuum. New Phytol. 1997, 135, 575–585. [Google Scholar] [CrossRef]
- Smith, S.E.; Jakobsen, I.; Gronlund, M.; Smith, F.A. Roles of arbuscular mycorrhizas in plant phosphorus nutrition: Interactions between pathways of phosphorus uptake in arbuscular mycorrhizal roots have important implications for understanding and manipulating plant phosphorus acquisition. Plant Physiol. 2011, 156, 1050–1057. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, Y.; Zhu, J.K. Developing naturally stress-resistant crops for a sustainable agriculture. Nat. Plants 2018, 4, 989–996. [Google Scholar] [CrossRef]
- Kuswandi, P.; Sugiyarto, L. Application of mycorrhiza on planting media of tomato varieties to increase vegetable productivityin drought condition. J. Sains Dasar 2016, 4. [Google Scholar] [CrossRef]
- Wu, Q.S. Arbuscular Mycorrhizas and Stress Tolerance of Plants; Springer: Singapore, 2017; pp. 1–327. [Google Scholar]
- Adamec, S.; Andrejiová, A. Mycorrhiza and Stress Tolerance of Vegetables: A Review. Acta Hortic. Regiotect. 2018, 21, 30–35. [Google Scholar] [CrossRef]
- Wu, Q.S.; He, J.D.; Srivastava, A.K.; Zou, Y.N.; Kuca, K. Mycorrhizas enhance drought tolerance of citrus by altering root fatty acid compositions and their saturation levels. Tree Physiol. 2019, 39, 1149–1158. [Google Scholar] [CrossRef] [PubMed]
- Romero-Munar, A.; Baraza, E.; Gulías, J.; Cabot, C. Arbuscular Mycorrhizal Fungi Confer Salt Tolerance in Giant Reed (Arundo donax L.) Plants Grown Under Low Phosphorus by Reducing Leaf Na(+) Concentration and Improving Phosphorus Use Efficiency. Front. Plant Sci. 2019, 10, 843. [Google Scholar] [CrossRef]
- Van Geel, M.; De Beenhouwer, M.; Lievens, B.; Honnay, O. Crop-specific and single-species mycorrhizal inoculation is the best approach to improve crop growth in controlled environments. Agron. Sustain. Dev. 2016, 36, 37. [Google Scholar] [CrossRef]
- Rocha, I.; Ma, Y.; Souza-Alonso, P.; Vosátka, M.; Freitas, H.; Oliveira, R.S. Seed Coating: A Tool for Delivering Beneficial Microbes to Agricultural Crops. Front. Plant Sci. 2019, 10, 1357. [Google Scholar] [CrossRef]
- Michael, A.; Larry, S.; Nick, M. Inoculate with Mycorrhizae It’s as Easy as A-B-Seeds-Annotated. 2012. Available online: https://www.yumpu.com/en/document/read/15257236/inoculate-with-mycorrhizae-its-as-easy-as-a-b-seeds-mycorrhizacom (accessed on 29 December 2020).
- Hamel, C.; Dalpé, Y.; Furlan, V.; Parent, S.J.M. Indigenous populations of arbuscular mycorrhizal fungi and soil aggregate stability are major determinants of leek (Allium porrum L.) response to inoculation with Glomus intraradices Schenck & Smith or Glomus versiforme (Karsten) Berch. Mycorrhiza 1997, 7, 187–196. [Google Scholar]
- Acha, A.J.; Vieira, H.D. Digital image processing of coated perennial-soybean seeds and correlation with physiological atributes. J. Seed Sci. 2020, 42. [Google Scholar] [CrossRef]
- O’Callaghan, M. Microbial inoculation of seed for improved crop performance: Issues and opportunities. Appl. Microbiol. Biotechnol. 2016, 100, 5729–5746. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Hamel, C.; Hamilton, R.I.; Smith, D.L. Mycorrhizae formation and nutrient uptake of new corn (Zea mays L.) hybrids with extreme canopy and leaf architecture as influenced by soil N and P levels. Plant Soil 2000, 221, 157–166. [Google Scholar] [CrossRef]
- Grant, C.; Bittman, S.; Montreal, M.; Plenchette, C.; Morel, C.; Bittman, C.; Montreal, S. Soil and fertilizer phosphorus: Effects on plant P supply and mycorrhizal development. Can. J. Plant Sci. 2005, 85. [Google Scholar] [CrossRef]
- Rouphael, Y.; Schwarz, D.; Krumbein, A.; Colla, G. Impact of grafting on product quality of fruit vegetables. Sci. Hortic. 2010, 127, 172–179. [Google Scholar] [CrossRef]

| Company Name | Company’s Location | Product Name | Form | AMF Species |
|---|---|---|---|---|
| Ecological Resources, Inc./Oikos | USA/Chile | Oiko-Rhiza-E | powder | R. irregularis, F. mosseae, Glomus Deserticola, R. clarus |
| USA/Chile | Oiko-Rhiza-Ectosol | powder | Not specified | |
| Mycorrhizal Application, LLC/Sumitomo Chemical Chile S.A. | USA/Chile | MycoApply Endomaxx * | granular | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum |
| Helena Agri-Enterprises, LLC | USA | Myco-Sol | powder | R. aggregatum, R. irregularis, F. mosseae |
| USA | Myco-Vam Bare Root | powder | R. aggregatum, R. irregularis, F. mosseae | |
| USA | Myco-Vam Granular | granular | R. aggregatum, R. irregularis, F. mosseae | |
| USA | Myco-Vam Plus 6-3-3 | powder | R. irregularis, C. etunicatum, R. clarus | |
| Valent USA. LLC Agricultural Products | USA | MycoApply(R) Endo-Granular | granular | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum |
| USA | MycoApply(R) EndoFuse | Liquid | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum | |
| USA | MycoApply(R) EndoMAXX | powder | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum | |
| USA | MycoApply(R) EndoPrime SC(TM) | Liquid | R. aggregatum, R. irregularis, F.s mosseae, C. etunicatum | |
| USA | MycoApply(R) EndoPrime(TM) | powder | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum | |
| USA | MycoApply(R) Ultrafine Endo | powder | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum | |
| Tainio Biologicals, Inc. | USA | MycoGenesis Seed Treatment | powder | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum |
| USA | MycoGenesis Soil Amendment | powder | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum | |
| JH Biotech, Inc. | USA | MYCORMAX Biological Inoculum | powder | R. irregularis, F. mosseae |
| USA | MYCORMAX Biological Transplant Starter | powder | R.s irregularis, F. mosseae | |
| AgroScience Solutions, LLC | USA | Organic Mycorrhizal Fungi | Liquid | R. aggregatum, R. irregularis, F. mosseae |
| Tainio Biologicals, Inc. | USA | Spectrum + Myco | powder | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum |
| Sustane Natural Fertilizer, Inc. | USA | Sustane 3-7-2 with Mycorrhizae and Humates | granular | R. aggregatum, C.s etunicatum, G. Deserticola, R. clarus |
| Vegalab Inc. | USA | Vegalab MYCO BIOBOOST | powder | R. irregularis |
| Tainio Biologicals, Inc. | USA | Rhizogenesis | powder | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum |
| Shemin Garden, LLC | USA | Ecofungi | powder | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum |
| Pathway BioLogic, LLC | USA | Managefungi | powder | R. aggregatum, R. irregularis, F. mosseae, C. etunicatum |
| Symborg Inc. | Spain | MycoUp Activ * | powder | R. iranicum |
| Spain | MycoUp Biological Inoculant * | powder | R. iranicum | |
| Symborg | Spain | ResidHC * | powder | R. iranicum |
| Spain | ResidMG * | granular | R. iranicum | |
| Atens | Spain | Bio Asir Fruit | granular | R. iranicum, F. mosseae |
| Spain | Aegis Sym irriga | powder | R. iranicum, F. mosseae | |
| Spain | Aegis Sym microgranulado | granular | R. iranicum, F. mosseae | |
| Koppert | Netherlands | Panoramix * | Liquid | Two AMF species |
| INIFAP | México | Micorriza INIFAP | powder | R. irregularis |
| Plant Health Care | Mexico | PHC VAM.PWI | powder | R. irregularis, C. etunicatum, C. etunicatum |
| Mexico | PHC ENDO-RHIZA | powder | R. irregularis | |
| Mexico | MycorTree-Injectable | powder | R.s irregularis, C. etunicatum, R. clarus | |
| Mexico | Turf Saber | powder | R. irregularis, C. etunicatum, R. clarus | |
| Mexico | Hotic Plus | powder | R. irregularis, C. etunicatum, R. clarus | |
| OBA | Mexico | HIPER-GLOM | powder | R. irregularis |
| Vergel de Occidente | Mexico | Tec-Myc 60 | powder | AMF species not specified |
| Biokrone | Mexico | Glumix | granular | R. irregularis |
| Mexico | Glumix irigation | powder | AMF species not specified | |
| Biofabrica Siglo XXI | Mexico | Micorrizafer plus | powder | R. irregularis |
| BIOMIC | Mexico | TM-73 | powder, granular | AMF species not specified |
| Italpolina SpA/ATENS- Agrotecnologías Naturales S.L./Semillas Abe Ltd.a—In Pacta SpA | Italy/Spain/Chile | Aegis Gel * | powder | R. irregularis, F. mosseae |
| Italy/Spain/Chile | Aegis Irriga * | powder | R. irregularis, F. mosseae | |
| Italy/Spain/Chile | Coveron | powder | R. irregularis, F. mosseae | |
| GroundWork BioAG | Israel | Rootella G | granular | R. irregularis |
| Israel | Rootella P | powder | R. irregularis | |
| Israel | Rootella F | powder | R. irregularis | |
| Israel | Rootella X | powder | R. irregularis, F. mosseae | |
| Israel | Rootella T | powder | R. irregularis, F. mosseae, R. clarus | |
| Israel | Rootella S | powder | R. irregularis | |
| Agronutrition | France | CONNECTIS | Liquid | R. irregularis |
| MYCOSYM TRITION SL/Biosim | España/Chile | Mycosim Tri-Ton | granular | R. irregularis |
| PremierTech | Canada | Activ pulses granular | granular | R. irregularis |
| Canada | Activ Soya granular | granular | R. irregularis | |
| Canada | Activ Soybean Powder | powder | R. irregularis | |
| Canada | Activ field crops granular | granular | R. irregularis | |
| Canada | Activ potato liquid | Liquid | R. irregularis | |
| Canada | Activ field crops liquid | Liquid | R. irregularis | |
| Canada | Activ field crops powder | powder | R. irregularis | |
| Canada | Activ specialty crops powder | powder | R. irregularis | |
| Canada | Activ specialty crops powder pea | powder | R. irregularis | |
| Symborg Business Development S.L./Symborg Chile SpA | Chile and Spain | MycoUp * | granular | R. iranicum |
| Chile and Spain | Resid HC * | granular | R. iranicum | |
| Purely Organic Products LLC | USA | Pro-Yield Purely Pro N | __ | R. irregularis |
| USA | Pro- Yield Purely Pro P | __ | R. irregularis |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basiru, S.; Mwanza, H.P.; Hijri, M. Analysis of Arbuscular Mycorrhizal Fungal Inoculant Benchmarks. Microorganisms 2021, 9, 81. https://doi.org/10.3390/microorganisms9010081
Basiru S, Mwanza HP, Hijri M. Analysis of Arbuscular Mycorrhizal Fungal Inoculant Benchmarks. Microorganisms. 2021; 9(1):81. https://doi.org/10.3390/microorganisms9010081
Chicago/Turabian StyleBasiru, Sulaimon, Hopkins Pachalo Mwanza, and Mohamed Hijri. 2021. "Analysis of Arbuscular Mycorrhizal Fungal Inoculant Benchmarks" Microorganisms 9, no. 1: 81. https://doi.org/10.3390/microorganisms9010081
APA StyleBasiru, S., Mwanza, H. P., & Hijri, M. (2021). Analysis of Arbuscular Mycorrhizal Fungal Inoculant Benchmarks. Microorganisms, 9(1), 81. https://doi.org/10.3390/microorganisms9010081







