The Neglected Microbial Components of Commercial Probiotic Formulations
Abstract
1. Introduction
2. “Overfilling” of Viable Microbial Cells during Probiotic Product Manufacturing
3. Viable Cells in Nongrowing States (the “Hidden” Viable Biomass)
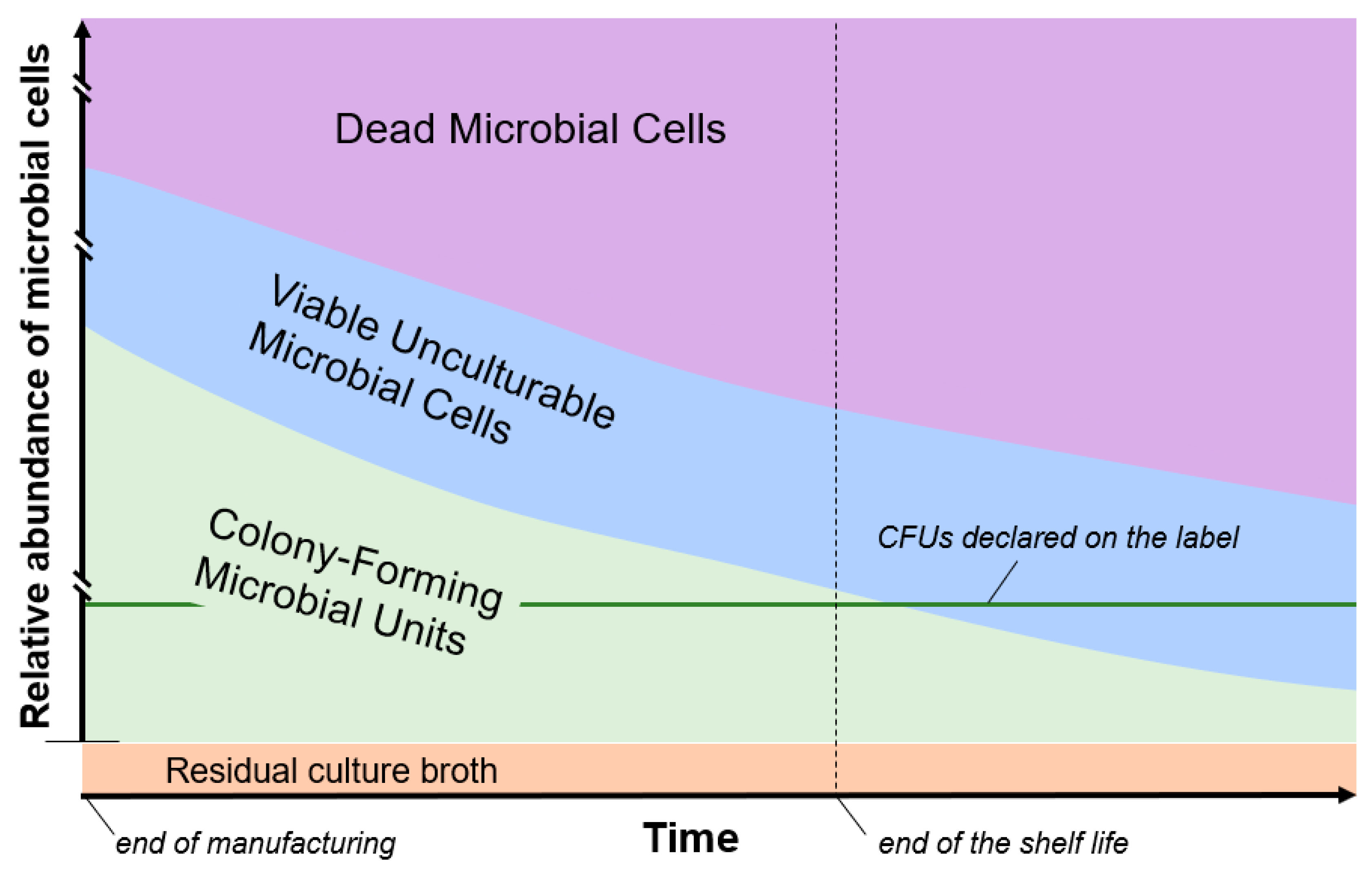
4. Non-Viable Microbial Cells in Probiotic Formulations
5. Extracellular Microbial Products in Probiotic Formulations
6. Methodologies for the Microbiological Characterization of Probiotic Products
7. Conclusions
- On the label of commercial products, in addition to the minimum number of CFUs at the end of the shelf life, the total number of microbial cells in the final product determined by FC at the end of manufacturing must be declared.
- In clinical trials, viable count by plating according to standard procedures (e.g., the methods reported in the document ISTISAN 08/36 by the Italian National Institute of Health) must be determined for each lot of the probiotic formulation under investigation immediately before the beginning of the intervention to determine the actual number of viable cells administered to study participants. In addition, the probiotic formulation must be analyzed by FC according to standard methods (e.g., ISO-IDF protocols) to determine the total numbers and proportions of viable, damaged, and dead microbial cells.
Author Contributions
Funding
Conflicts of Interest
References
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Caffarelli, C.; Cardinale, F.; Povesi-Dascola, C.; Dodi, I.; Mastrorilli, V.; Ricci, G. Use of probiotics in pediatric infectious diseases. Expert Rev. Anti-Infect. Ther. 2015, 13, 1517–1535. [Google Scholar] [CrossRef] [PubMed]
- Szajewska, H.; Skorka, A.; Ruszczynski, M.; Gieruszczak-Bialek, D. Meta-analysis: Lactobacillus GG for treating acute gastroenteritis in children--updated analysis of randomised controlled trials. Aliment. Pharmacol. Ther. 2013, 38, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Khalesi, S.; Sun, J.; Buys, N.; Jayasinghe, R. Effect of probiotics on blood pressure: A systematic review and meta-analysis of randomized, controlled trials. Hypertension 2014, 64, 897–903. [Google Scholar] [CrossRef] [PubMed]
- De Simone, C. The Unregulated Probiotic Market. Clin. Gastroenterol. Hepatol. 2019, 17, 809–817. [Google Scholar] [CrossRef]
- da Cruz, A.G.; Faria, J.d.A.F.; Van Dender, A.G.F. Packaging system and probiotic dairy foods. Food Res. Int. 2007, 40, 951–956. [Google Scholar] [CrossRef]
- Savini, M.; Cecchini, C.; Verdenelli, M.C.; Silvi, S.; Orpianesi, C.; Cresci, A. Pilot-scale production and viability analysis of freeze-dried probiotic bacteria using different protective agents. Nutrients 2010, 2, 330–339. [Google Scholar] [CrossRef]
- Goderska, K. Different Methods of Probiotics Stabilization. In Probiotics; Rigobelo, E.C., Ed.; IntechOpen: London, UK, 2012. [Google Scholar] [CrossRef]
- Arioli, S.; Koirala, R.; Taverniti, V.; Fiore, W.; Guglielmetti, S. Quantitative Recovery of Viable Lactobacillus paracasei CNCM I-1572 (L. casei DG(R)) After Gastrointestinal Passage in Healthy Adults. Front. Microbiol. 2018, 9, 1720. [Google Scholar] [CrossRef]
- Mai, V.; Waugh, S.; Byrd, D.; Simpson, D.; Ukhanova, M. Novel encapsulation improves recovery of probiotic strains in fecal samples of human volunteers. Appl. Microbiol. Biotechnol. 2017, 101, 1419–1425. [Google Scholar] [CrossRef]
- Volkert, M.; Ananta, E.; Luscher, C.; Knorr, D. Effect of air freezing, spray freezing, and pressure shift freezing on membrane integrity and viability of Lactobacillus rhamnosus GG. J. Food Eng. 2008, 87, 532–540. [Google Scholar] [CrossRef]
- García-Cayuela, T.; Tabasco, R.; Peláez, C.; Requena, T. Simultaneous detection and enumeration of viable lactic acid bacteria and bifidobacteria in fermented milk by using propidium monoazide and real-time PCR. Int. Dairy J. 2009, 19, 405–409. [Google Scholar] [CrossRef]
- Davis, C. Enumeration of probiotic strains: Review of culture-dependent and alternative techniques to quantify viable bacteria. J. Microbiol. Methods 2014, 103, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Gengenbacher, M.; Kaufmann, S.H. Mycobacterium tuberculosis: Success through dormancy. FEMS Microbiol. Rev. 2012, 36, 514–532. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Mendis, N.; Trigui, H.; Oliver, J.D.; Faucher, S.P. The importance of the viable but non-culturable state in human bacterial pathogens. Front. Microbiol. 2014, 5, 258. [Google Scholar] [CrossRef] [PubMed]
- Wesche, A.M.; Gurtler, J.B.; Marks, B.P.; Ryser, E.T. Stress, sublethal injury, resuscitation, and virulence of bacterial foodborne pathogens. J. Food Prot. 2009, 72, 1121–1138. [Google Scholar] [CrossRef]
- Lahtinen, S.J.; Gueimonde, M.; Ouwehand, A.C.; Reinikainen, J.P.; Salminen, S.J. Probiotic bacteria may become dormant during storage. Appl. Environ. Microbiol. 2005, 71, 1662–1663. [Google Scholar] [CrossRef]
- Lahtinen, S.J.; Ouwehand, A.C.; Reinikainen, J.P.; Korpela, J.M.; Sandholm, J.; Salminen, S.J. Intrinsic properties of so-called dormant probiotic bacteria, determined by flow cytometric viability assays. Appl. Environ. Microbiol. 2006, 72, 5132–5134. [Google Scholar] [CrossRef]
- Lahtinen, S.J.; Ahokoski, H.; Reinikainen, J.P.; Gueimonde, M.; Nurmi, J.; Ouwehand, A.C.; Salminen, S.J. Degradation of 16S rRNA and attributes of viability of viable but nonculturable probiotic bacteria. Lett. Appl. Microbiol. 2008, 46, 693–698. [Google Scholar] [CrossRef]
- De Almada, C.N.; Almada, C.N.; Martinez, R.C.R.; Sant’Ana, A.S. Paraprobiotics: Evidences on their ability to modify biological responses, inactivation methods and perspectives on their application in foods. Trends Food Sci. Technol. 2016, 58, 96–114. [Google Scholar] [CrossRef]
- Senoh, M.; Ghosh-Banerjee, J.; Ramamurthy, T.; Colwell, R.R.; Miyoshi, S.; Nair, G.B.; Takeda, Y. Conversion of viable but nonculturable enteric bacteria to culturable by co-culture with eukaryotic cells. Microbiol. Immunol. 2012, 56, 342–345. [Google Scholar] [CrossRef]
- Bogosian, G.; Bourneuf, E.V. A matter of bacterial life and death. EMBO Rep. 2001, 2, 770–774. [Google Scholar] [CrossRef] [PubMed]
- Davey, H.M. Life, death, and in-between: Meanings and methods in microbiology. Appl. Environ. Microbiol. 2011, 77, 5571–5576. [Google Scholar] [CrossRef] [PubMed]
- Fiocco, D.; Longo, A.; Arena, M.P.; Russo, P.; Spano, G.; Capozzi, V. How probiotics face food stress: They get by with a little help. Crit. Rev. Food Sci. Nutr. 2020, 60, 1552–1580. [Google Scholar] [CrossRef] [PubMed]
- Fenster, K.; Freeburg, B.; Hollard, C.; Wong, C.; Ronhave Laursen, R.; Ouwehand, A.C. The Production and Delivery of Probiotics: A Review of a Practical Approach. Microorganisms 2019, 7, 83. [Google Scholar] [CrossRef] [PubMed]
- FAO; WHO. Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria; FAO: Rome, Italy; WHO: Córdoba, Argentina, 2001. [Google Scholar]
- Lahtinen, S.J. Probiotic viability—Does it matter? Microb. Ecol. Health Dis. 2012, 23. [Google Scholar] [CrossRef] [PubMed]
- Taverniti, V.; Guglielmetti, S. The immunomodulatory properties of probiotic microorganisms beyond their viability (ghost probiotics: Proposal of paraprobiotic concept). Genes Nutr. 2011, 6, 261–274. [Google Scholar] [CrossRef]
- Pique, N.; Berlanga, M.; Minana-Galbis, D. Health Benefits of Heat-Killed (Tyndallized) Probiotics: An Overview. Int. J. Mol. Sci. 2019, 20, 2534. [Google Scholar] [CrossRef]
- Nishida, K.; Sawada, D.; Kawai, T.; Kuwano, Y.; Fujiwara, S.; Rokutan, K. Para-psychobiotic Lactobacillus gasseri CP2305 ameliorates stress-related symptoms and sleep quality. J. Appl. Microbiol. 2017, 123, 1561–1570. [Google Scholar] [CrossRef]
- Nakata, J.; Hirota, T.; Umemura, H.; Nakagawa, T.; Kando, N.; Futamura, M.; Nakamura, Y.; Ito, K. Additive effect of Lactobacillus acidophilus L-92 on children with atopic dermatitis concomitant with food allergy. Asia Pac. Allergy 2019, 9, e18. [Google Scholar] [CrossRef]
- Adams, C.A. The probiotic paradox: Live and dead cells are biological response modifiers. Nutr. Res. Rev. 2010, 23, 37–46. [Google Scholar] [CrossRef]
- Ananta, E.; Knorr, D. Comparison of inactivation pathways of thermal or high pressure inactivated Lactobacillus rhamnosus ATCC 53103 by flow cytometry analysis. Food Microbiol. 2009, 26, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Barros, C.P.; Guimarães, J.T.; Esmerino, E.A.; Duarte, M.C.K.H.; Silva, M.C.; Silva, R.; Ferreira, B.M.; Sant’Ana, A.S.; Freitas, M.Q.; Cruz, A.G. Paraprobiotics and postbiotics: Concepts and potential applications in dairy products. Curr. Opin. Food Sci. 2020, 32, 1–8. [Google Scholar] [CrossRef]
- Shigwedha, N.; Zhang, L.; Sichel, L.; Jia, L.; Gong, P.; Liu, W.; Wang, S.; Zhang, S.; Han, X.; Gao, W. More than a Few LAB Alleviate Common Allergies: Impact of Paraprobiotics in Comparison to Probiotical Live Cells. J. Biosci. Med. 2014, 2, 9. [Google Scholar] [CrossRef][Green Version]
- Derrien, M.; van Hylckama Vlieg, J.E. Fate, activity, and impact of ingested bacteria within the human gut microbiota. Trends Microbiol. 2015, 23, 354–366. [Google Scholar] [CrossRef] [PubMed]
- Smelt, M.J.; de Haan, B.J.; Bron, P.A.; van Swam, I.; Meijerink, M.; Wells, J.M.; Kleerebezem, M.; Faas, M.M.; de Vos, P. The impact of Lactobacillus plantarum WCFS1 teichoic acid D-alanylation on the generation of effector and regulatory T-cells in healthy mice. PLoS ONE 2013, 8, e63099. [Google Scholar] [CrossRef] [PubMed]
- Taverniti, V.; Stuknyte, M.; Minuzzo, M.; Arioli, S.; De Noni, I.; Scabiosi, C.; Cordova, Z.M.; Junttila, I.; Hamalainen, S.; Turpeinen, H.; et al. S-layer protein mediates the stimulatory effect of Lactobacillus helveticus MIMLh5 on innate immunity. Appl. Environ. Microbiol. 2013, 79, 1221–1231. [Google Scholar] [CrossRef]
- Balzaretti, S.; Taverniti, V.; Guglielmetti, S.; Fiore, W.; Minuzzo, M.; Ngo, H.N.; Ngere, J.B.; Sadiq, S.; Humphreys, P.N.; Laws, A.P. A Novel Rhamnose-Rich Hetero-exopolysaccharide Isolated from Lactobacillus paracasei DG Activates THP-1 Human Monocytic Cells. Appl. Environ. Microbiol. 2017, 83. [Google Scholar] [CrossRef]
- Guglielmetti, S.; Zanoni, I.; Balzaretti, S.; Miriani, M.; Taverniti, V.; De Noni, I.; Presti, I.; Stuknyte, M.; Scarafoni, A.; Arioli, S.; et al. Murein lytic enzyme TgaA of Bifidobacterium bifidum MIMBb75 modulates dendritic cell maturation through its cysteine- and histidine-dependent amidohydrolase/peptidase (CHAP) amidase domain. Appl. Environ. Microbiol. 2014, 80, 5170–5177. [Google Scholar] [CrossRef]
- Guglielmetti, S.; Tamagnini, I.; Mora, D.; Minuzzo, M.; Scarafoni, A.; Arioli, S.; Hellman, J.; Karp, M.; Parini, C. Implication of an outer surface lipoprotein in adhesion of Bifidobacterium bifidum to Caco-2 cells. Appl. Environ. Microbiol. 2008, 74, 4695–4702. [Google Scholar] [CrossRef]
- Qi, S.R.; Cui, Y.J.; Liu, J.X.; Luo, X.; Wang, H.F. Lactobacillus rhamnosus GG components, SLP, gDNA and CpG, exert protective effects on mouse macrophages upon lipopolysaccharide challenge. Lett. Appl. Microbiol. 2020, 70, 118–127. [Google Scholar] [CrossRef]
- Bauerl, C.; Abitayeva, G.; Sosa-Carrillo, S.; Mencher-Beltran, A.; Navarro-Lleo, N.; Coll-Marques, J.M.; Zuniga-Cabrera, M.; Shaikhin, S.; Perez-Martinez, G. P40 and P75 Are Singular Functional Muramidases Present in the Lactobacillus casei/paracasei/rhamnosus Taxon. Front. Microbiol. 2019, 10, 1420. [Google Scholar] [CrossRef] [PubMed]
- Tsilingiri, K.; Rescigno, M. Postbiotics: What else? Benef. Microbes 2013, 4, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Li, Y.; Wan, Y.; Hu, T.; Liu, L.; Yang, S.; Gong, Z.; Zeng, Q.; Wei, Y.; Yang, W.; et al. A Novel Postbiotic From Lactobacillus rhamnosus GG With a Beneficial Effect on Intestinal Barrier Function. Front. Microbiol. 2019, 10, 477. [Google Scholar] [CrossRef] [PubMed]
- Aguilar-Toala, J.E.; Hall, F.G.; Urbizo-Reyes, U.C.; Garcia, H.S.; Vallejo-Cordoba, B.; Gonzalez-Cordova, A.F.; Hernandez-Mendoza, A.; Liceaga, A.M. In Silico Prediction and In Vitro Assessment of Multifunctional Properties of Postbiotics Obtained From Two Probiotic Bacteria. Probiotics Antimicrob. Proteins 2019. [Google Scholar] [CrossRef]
- Tsilingiri, K.; Barbosa, T.; Penna, G.; Caprioli, F.; Sonzogni, A.; Viale, G.; Rescigno, M. Probiotic and postbiotic activity in health and disease: Comparison on a novel polarised ex-vivo organ culture model. Gut 2012, 61, 1007–1015. [Google Scholar] [CrossRef]
- Chiron, C.; Tompkins, T.A.; Burguiere, P. Flow cytometry: A versatile technology for specific quantification and viability assessment of micro-organisms in multistrain probiotic products. J. Appl. Microbiol. 2018, 124, 572–584. [Google Scholar] [CrossRef]
- Jackson, S.A.; Schoeni, J.L.; Vegge, C.; Pane, M.; Stahl, B.; Bradley, M.; Goldman, V.S.; Burguiere, P.; Atwater, J.B.; Sanders, M.E. Improving End-User Trust in the Quality of Commercial Probiotic Products. Front. Microbiol. 2019, 10, 739. [Google Scholar] [CrossRef]
- Van Nevel, S.; Koetzsch, S.; Proctor, C.R.; Besmer, M.D.; Prest, E.I.; Vrouwenvelder, J.S.; Knezev, A.; Boon, N.; Hammes, F. Flow cytometric bacterial cell counts challenge conventional heterotrophic plate counts for routine microbiological drinking water monitoring. Water Res. 2017, 113, 191–206. [Google Scholar] [CrossRef]
- Kramer, M.; Obermajer, N.; Bogovic Matijasic, B.; Rogelj, I.; Kmetec, V. Quantification of live and dead probiotic bacteria in lyophilised product by real-time PCR and by flow cytometry. Appl. Microbiol. Biotechnol. 2009, 84, 1137–1147. [Google Scholar] [CrossRef]
- Mora, D.; Filardi, R.; Arioli, S.; Boeren, S.; Aalvink, S.; de Vos, W.M. Development of omics-based protocols for the microbiological characterization of multi-strain formulations marketed as probiotics: The case of VSL#3. Microb. Biotechnol. 2019, 12, 1371–1386. [Google Scholar] [CrossRef]
- Majeed, M.; Majeed, S.; Nagabhushanam, K.; Punnapuzha, A.; Philip, S.; Mundkur, L. Rapid assessment of viable but non-culturable Bacillus coagulans MTCC 5856 in commercial formulations using Flow cytometry. PLoS ONE 2018, 13, e0192836. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fiore, W.; Arioli, S.; Guglielmetti, S. The Neglected Microbial Components of Commercial Probiotic Formulations. Microorganisms 2020, 8, 1177. https://doi.org/10.3390/microorganisms8081177
Fiore W, Arioli S, Guglielmetti S. The Neglected Microbial Components of Commercial Probiotic Formulations. Microorganisms. 2020; 8(8):1177. https://doi.org/10.3390/microorganisms8081177
Chicago/Turabian StyleFiore, Walter, Stefania Arioli, and Simone Guglielmetti. 2020. "The Neglected Microbial Components of Commercial Probiotic Formulations" Microorganisms 8, no. 8: 1177. https://doi.org/10.3390/microorganisms8081177
APA StyleFiore, W., Arioli, S., & Guglielmetti, S. (2020). The Neglected Microbial Components of Commercial Probiotic Formulations. Microorganisms, 8(8), 1177. https://doi.org/10.3390/microorganisms8081177






