Evaluating the Antiparasitic Activity of Novel BPZ Derivatives Against Toxoplasma gondii
Abstract
:1. Introduction
2. Materials and Methods
2.1. Parasite Growth and Maintenance
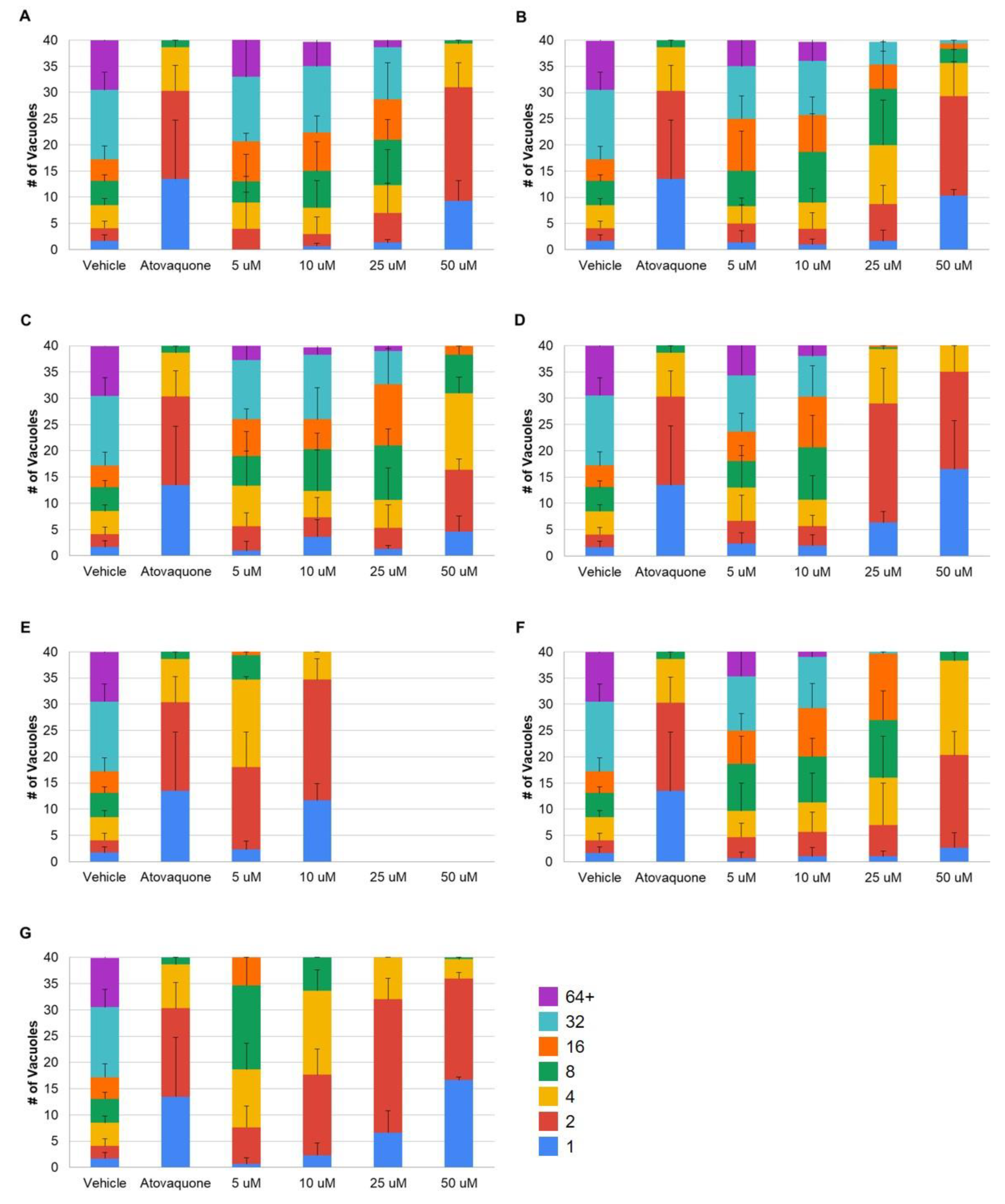
2.2. Doubling Assays
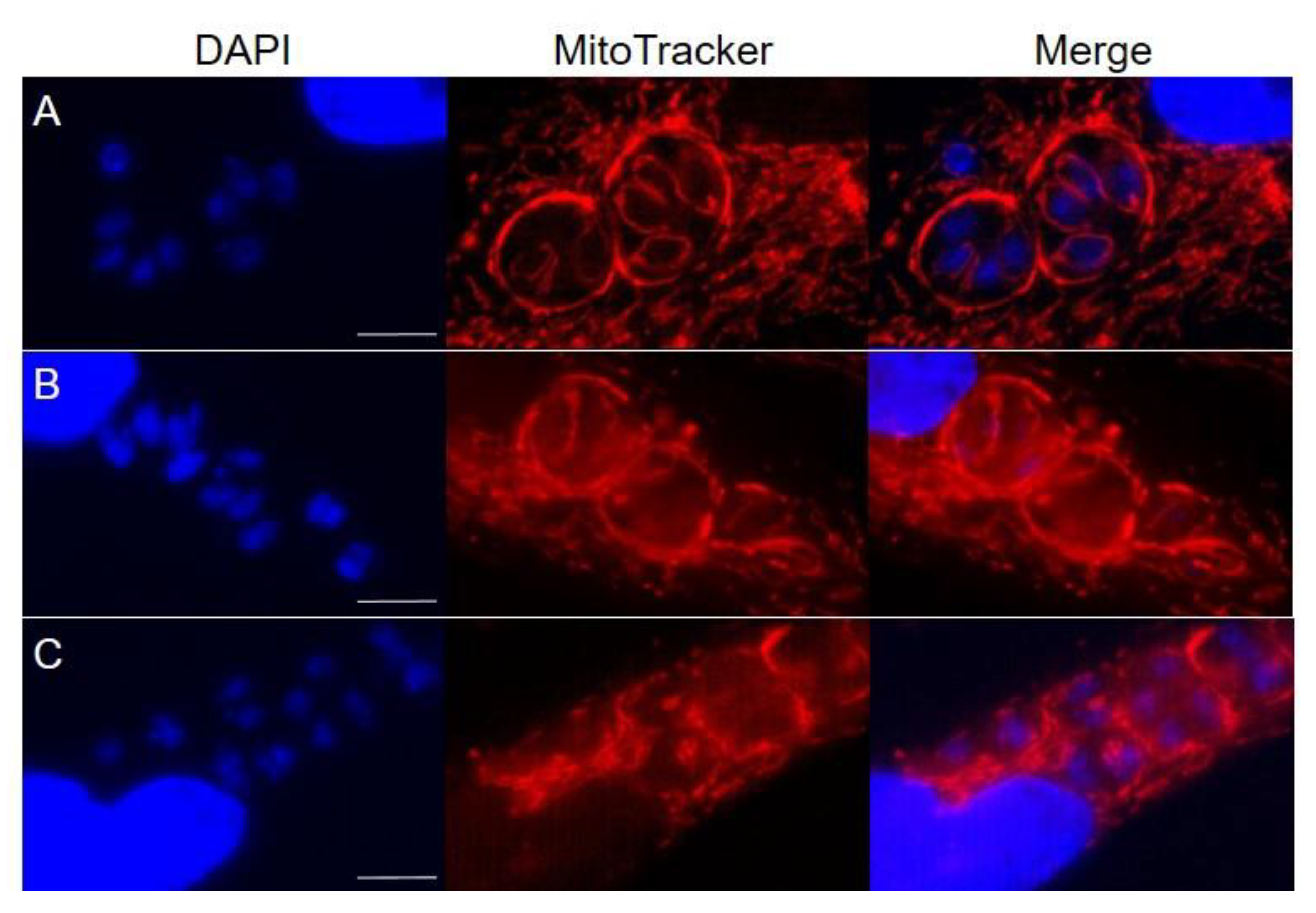
2.3. MitoTracker Staining and Fluorescence Microscopy
2.4. Statistical Analyses
3. Results
3.1. Bisphenol Z and Its Derivatives Inhibit T. gondii Replication
3.2. The Parasite Mitochondrion Is Impacted by BPZ and Its Derivatives
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Joiner, K.A.; Dubremetz, J.F. Toxoplasma gondii: A protozoan for the nineties. Infect. Immun. 1993, 61, 1169–1172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dubey, J.P. History of the discovery of the life cycle of Toxoplasma gondii. Int. J. Parasitol. 2009, 39, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Pappas, G.; Roussos, N.; Falagas, M.E. Toxoplasmosis snapshots: Global status of Toxoplasma gondii seroprevalence and implications for pregnancy and congenital toxoplasmosis. Int. J. Parasitol. 2009, 39, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Sousa, O.E.; Saenz, R.E.; Frenkel, J.K. Toxoplasmosis in Panama: A 10-year study. Am. J. Trop. Med. Hyg. 1988, 38, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.L.; Parise, M.E.; Fiore, A.E. Neglected parasitic infections in the United States: Toxoplasmosis. Am. J. Trop. Med. Hyg. 2014, 90, 794–799. [Google Scholar] [CrossRef] [Green Version]
- Guo, M.; Mishra, A.; Buchanan, R.L.; Dubey, J.P.; Hill, D.E.; Gamble, H.R.; Jones, J.L.; Pradhan, A.K. A Systematic Meta-Analysis of Toxoplasma gondii Prevalence in Food Animals in the United States. Foodborne Pathog. Dis. 2016, 13, 109–118. [Google Scholar] [CrossRef]
- Sheffield, H.G.; Melton, M.L. Toxoplasma gondii: The Oocyst, Sporozoite, and Infection of Cultured Cells. Science 1970, 167, 892–893. [Google Scholar] [CrossRef]
- Dubey, J.P.; Miller, N.L.; Frenkel, J.K. The Toxoplasma Gondii Oocyst from Cat Feces. J. Exp. Med. 1970, 132, 636–662. [Google Scholar] [CrossRef] [Green Version]
- Hutchison, W.M.; Dunachie, J.F.; Siim, J.C.; Work, K. Life cycle of Toxoplasma gondii. BMJ 1969, 4, 806. [Google Scholar] [CrossRef] [Green Version]
- Dubey, J.P. Advances in the life cycle of Toxoplasma gondii. Int. J. Parasitol. 1998, 28, 1019–1024. [Google Scholar] [CrossRef] [Green Version]
- Gilot-Fromont, E.; Lélu, M.; Dardé, M.-L.; Richomme, C.; Aubert, D.; Afonso, E.; Mercier, A.; Gotteland, C.; Villena, I. The Life Cycle of Toxoplasma gondii in the Natural Environment. In Toxoplasmosis - Recent Advances; Djaković, O.D., Ed.; InTechOpen: London, UK, 2012; pp. 3–36. [Google Scholar]
- Montoya, J.G.; Remington, J.S. Management of Toxoplasma gondii infection during pregnancy. Clin. Infect. Dis. 2008, 47, 554–566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krueger, W.S.; Hilborn, E.D.; Converse, R.R.; Wade, T.J. Drinking water source and human Toxoplasma gondii infection in the United States: A cross-sectional analysis of NHANES data. BMC Public Health 2014, 14, 711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilking, H.; Thamm, M.; Stark, K.; Aebischer, T.; Seeber, F. Prevalence, incidence estimations, and risk factors of Toxoplasma gondii infection in Germany: A representative, cross-sectional, serological study. Sci. Rep. 2016, 6, 22551. [Google Scholar] [CrossRef] [PubMed]
- Belluco, S.; Simonato, G.; Mancin, M.; Pietrobelli, M.; Ricci, A. Toxoplasma gondii infection and food consumption: A systematic review and meta-analysis of case-controlled studies. Crit. Rev. Food Sci. Nutr. 2018, 58, 3085–3096. [Google Scholar] [CrossRef] [PubMed]
- Kervan, U.; Ozdamar, Y.; Yurdakok, O.; Kucuker, S.A.; Pac, M. A rare ocular complication after a heart transplant: Toxoplasma retinitis. Exp. Clin. Transplant. 2014, 12, 78–80. [Google Scholar] [CrossRef] [Green Version]
- Miltgen, G.; Paillot, J.; Vanlemmens, C.; Di Martino, V.; Heyd, B.; Millon, L.; Bellanger, A.-P. Fatal acute respiratory distress by Toxoplasma gondii in a Toxoplasma seronegative liver transplant recipient. New Microbiol. 2016, 39, 160–162. [Google Scholar]
- Betancourt, E.D.; Hamid, B.; Fabian, B.T.; Klotz, C.; Hartmann, S.; Seeber, F. From Entry to Early Dissemination—Toxoplasma gondii’s Initial Encounter with Its Host. Front. Cell. Infect. Microbiol. 2019, 9, 46. [Google Scholar] [CrossRef]
- McLeod, R.; Berry, P.F.; Marshall, W.H., Jr.; Hunt, S.A.; Ryning, F.W.; Remington, J.S. Toxoplasmosis presenting as brain abscesses. Diagnosis by computerized tomography and cytology of aspirated purulent material. Am. J. Med. 1979, 67, 711–714. [Google Scholar] [CrossRef]
- Luft, B.J.; Remington, J.S. Toxoplasmic encephalitis in AIDS. Clin. Infect. Dis. 1992, 15, 211–222. [Google Scholar] [CrossRef]
- Israelski, D.M.; Remington, J.S. Toxoplasmosis in the non-AIDS immunocompromised host. Curr. Clin. Top. Infect. Dis. 1993, 13, 322–356. [Google Scholar]
- Slavin, M.A.; Meyers, J.D.; Remington, J.S.; Hackman, R.C. Toxoplasma gondii infection in marrow transplant recipients: A 20 year experience. Bone Marrow Transplant. 1994, 13, 549–557. [Google Scholar] [PubMed]
- Gallino, A.; Maggiorini, M.; Kiowski, W.; Martin, X.; Wunderli, W.; Schneider, J.; Turina, M.; Follath, F. Toxoplasmosis in heart transplant recipients. Eur. J. Clin. Microbiol. Infect. Dis. 1996, 15, 389–393. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.Y.; Remington, J.S. Toxoplasmosis in pregnancy. Clin. Infect. Dis. 1994, 18, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Henriquez, S.A.; Brett, R.; Alexander, J.; Pratt, J.; Roberts, C.W. Neuropsychiatric disease and Toxoplasma gondii infection. Neuroimmunomodulation 2009, 16, 122–133. [Google Scholar] [CrossRef]
- Martinez, V.O.; de Mendonça Lima, F.W.; de Carvalho, C.F.; Menezes-Filho, J.A. Toxoplasma gondii infection and behavioral outcomes in humans: A systematic review. Parasitol. Res. 2018, 117, 3059–3065. [Google Scholar] [CrossRef]
- Ortiz-Guerrero, G.; Gonzalez-Reyes, R.E.; de-la-Torre, A.; Medina-Rincón, G.; Nava-Mesa, M.O. Pathophysiological Mechanisms of Cognitive Impairment and Neurodegeneration by Toxoplasma gondii Infection. Brain Sci. 2020, 10, 369. [Google Scholar] [CrossRef]
- Neville, A.J.; Zach, S.J.; Wang, X.; Larson, J.J.; Judge, A.K.; Davis, L.A.; Vennerstrom, J.L.; Davis, P.H. Clinically Available Medicines Demonstrating Anti-Toxoplasma Activity. Antimicrob. Agents Chemother. 2015, 59, 7161–7169. [Google Scholar] [CrossRef] [Green Version]
- Maldonado, Y.A.; Read, J.S.; Committee on Infectious Diseases. Diagnosis, Treatment, and Prevention of Congenital Toxoplasmosis in the United States. Pediatrics 2017, 139, e20163860. [Google Scholar] [CrossRef] [Green Version]
- Porter, S.B.; Sande, M.A. Toxoplasmosis of the central nervous system in the acquired immunodeficiency syndrome. N. Engl. J. Med. 1992, 327, 1643–1648. [Google Scholar] [CrossRef]
- McLeod, R.; Khan, A.R.; Noble, G.A.; Latkany, P.; Jalbrzikowski, J.; Boyer, K. Severe sulfadiazine hypersensitivity in a child with reactivated congenital toxoplasmic chorioretinitis. Pediatric Infect. Dis. J. 2006, 25, 270–272. [Google Scholar] [CrossRef]
- Alday, P.H.; Doggett, J.S. Drugs in development for toxoplasmosis: Advances, challenges, and current status. Drug Des. Devel. Ther. 2017, 11, 273–293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Doliwa, C.; Escotte-Binet, S.; Aubert, D.; Sauvage, V.; Velard, F.; Schmid, A.; Villena, I. Sulfadiazine resistance in Toxoplasma gondii: No involvement of overexpression or polymorphisms in genes of therapeutic targets and ABC transporters. Parasite 2013, 20, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montazeri, M.; Mehrzadi, S.; Sharif, M.; Sarvi, S.; Tanzifi, A.; Aghayan, S.A.; Daryani, A. Drug Resistance in Toxoplasma gondii. Front. Microbiol. 2018, 9, 2587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stitzlein, L.M.; Stang, C.R.T.; Inbody, L.R.; Rao, P.S.S.; Schneider, R.A.; Dudley, R.W. Design, synthesis, and biological evaluation of lipophilically modified bisphenol Z derivatives. Chem. Biol. Drug Des. 2019, 94, 1574–1579. [Google Scholar] [CrossRef] [PubMed]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Salin, N.H.; Noordin, R.; Al-Najjar, B.O.; Kamarulzaman, E.E.; Yunus, M.H.; Karim, I.Z.A.; Nasim, N.N.M.; Zakaria, I.I.; Wahab, H.A. Identification of potential dual -targets anti- Toxoplasma gondii compounds through structure-based virtual screening and in-vitro studies. PLoS ONE 2020, 15, e0225232. [Google Scholar] [CrossRef]
- Pfefferkorn, E.R.; Borotz, S.E.; Nothnagel, R.F. Mutants of Toxoplasma gondii Resistant to Atovaquone (566C80) or Decoquinate. J. Parasitol. 1993, 79, 559. [Google Scholar] [CrossRef]
- Basto, A.P.; Müller, J.; Rubbiani, R.; Stibal, D.; Giannini, F.; Süss-Fink, G.; Balmer, V.; Hemphill, A.; Gasser, G.; Furrer, J. Characterization of the Activities of Dinuclear Thiolato-Bridged Arene Ruthenium Complexes against Toxoplasma gondii. Antimicrob. Agents Chemother. 2017, 61, e01031-17. [Google Scholar] [CrossRef] [Green Version]
- Strobl, J.S.; Seibert, C.W.; Li, Y.; Nagarkatti, R.; Mitchell, S.M.; Rosypal, A.C.; Rathore, D.; Lindsay, D.S. Inhibition of Toxoplasma gondii and Plasmodium falciparum infections in vitro by NSC3852, a redox active antiproliferative and tumor cell differentiation agent. J. Parasitol. 2009, 95, 215–223. [Google Scholar] [CrossRef]
- Chen, Q.-W.; Dong, K.; Qin, H.-X.; Yang, Y.-K.; He, J.-L.; Li, J.; Zheng, Z.-W.; Chen, D.-L.; Chen, J.-P. Direct and Indirect Inhibition Effects of Resveratrol against Toxoplasma gondii Tachyzoites In Vitro. Antimicrob. Agents Chemother. 2019, 63, e01233-18. [Google Scholar] [CrossRef] [Green Version]
- Nishi, M.; Hu, K.; Murray, J.M.; Roos, D.S. Organellar dynamics during the cell cycle of Toxoplasma gondii. J. Cell Sci. 2008, 121, 1559–1568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Garbuz, T.; Arrizabalaga, G. Lack of mitochondrial MutS homolog 1 in Toxoplasma gondii disrupts maintenance and fidelity of mitochondrial DNA and reveals metabolic plasticity. PLoS ONE 2017, 12, e0188040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melo, E.J.L.; Attias, M.; De Souza, W. The Single Mitochondrion of Tachyzoites of Toxoplasma gondii. J. Struct. Biol. 2000, 130, 27–33. [Google Scholar] [CrossRef]
- Judge, D.M.; Anderson, M.S. Ultrastructure of trypanosoma lewisi. J. Parasitol. 1964, 50, 757–762. [Google Scholar] [CrossRef]
- Rudzinska, M.A.; Trager, W. Electron-microscope studies on malaria parasites. Triangle 1959, 4, 97–104. [Google Scholar] [PubMed]
- Charvat, R.A.; Arrizabalaga, G. Oxidative stress generated during monensin treatment contributes to altered Toxoplasma gondii mitochondrial function. Sci. Rep. 2016, 6, 22997. [Google Scholar] [CrossRef] [Green Version]
- Jeffers, V.; Kamau, E.T.; Srinivasan, A.R.; Harper, J.; Sankaran, P.; Post, S.E.; Varberg, J.M.; Sullivan, W.J., Jr.; Boyle, J.P. TgPRELID, a Mitochondrial Protein Linked to Multidrug Resistance in the Parasite. mSphere 2017, 2, e00229-16. [Google Scholar] [CrossRef] [Green Version]
- Lavine, M.D.; Arrizabalaga, G. Analysis of monensin sensitivity in Toxoplasma gondii reveals autophagy as a mechanism for drug induced death. PLoS ONE 2012, 7, e42107. [Google Scholar] [CrossRef] [Green Version]
- Desoti, V.C.; Lazarin-Bidóia, D.; Sudatti, D.B.; Pereira, R.C.; Alonso, A.; Ueda-Nakamura, T.; Dias Filho, B.P.; Nakamura, C.V.; de Oliveira Silva, S. Trypanocidal action of (-)-elatol involves an oxidative stress triggered by mitochondria dysfunction. Mar. Drugs 2012, 10, 1631–1646. [Google Scholar] [CrossRef] [Green Version]
- Garrison, E.M.; Arrizabalaga, G. Disruption of a mitochondrial MutS DNA repair enzyme homologue confers drug resistance in the parasite Toxoplasma gondii. Mol. Microbiol. 2009, 72, 425–441. [Google Scholar] [CrossRef] [Green Version]
- McFadden, D.C.; Tomavo, S.; Berry, E.A.; Boothroyd, J.C. Characterization of cytochrome b from Toxoplasma gondii and Q(o) domain mutations as a mechanism of atovaquone-resistance. Mol. Biochem. Parasitol. 2000, 108, 1–12. [Google Scholar] [CrossRef]
- Srivastava, I.K.; Rottenberg, H.; Vaidya, A.B. Atovaquone, a Broad Spectrum Antiparasitic Drug, Collapses Mitochondrial Membrane Potential in a Malarial Parasite. J. Biol. Chem. 1997, 272, 3961–3966. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, J.; Lendahl, U.; Nistér, M. Regulation of mitochondrial dynamics: Convergences and divergences between yeast and vertebrates. Cell. Mol. Life Sci. 2013, 70, 951–976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jacobs, K.; Charvat, R.; Arrizabalaga, G. Identification of Fis1 interactors in Toxoplasma gondii reveals a novel protein required for peripheral distribution of the mitochondrion. Mbio 2020, 11, e02732-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heredero-Bermejo, I.; Varberg, J.M.; Charvat, R.; Jacobs, K.; Garbuz, T.; Sullivan, W.J., Jr.; Arrizabalaga, G. TgDrpC, an atypical dynamin-related protein in Toxoplasma gondii, is associated with vesicular transport factors and parasite division. Mol. Microbiol. 2019, 111, 46–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melatti, C.; Pieperhoff, M.; Lemgruber, L.; Pohl, E.; Sheiner, L.; Meissner, M. A unique dynamin-related protein is essential for mitochondrial fission in Toxoplasma gondii. PLoS Pathog. 2019, 15, e1007512. [Google Scholar] [CrossRef]
- Ovciarikova, J.; Lemgruber, L.; Stilger, K.L.; Sullivan, W.J.; Sheiner, L. Mitochondrial behaviour throughout the lytic cycle of Toxoplasma gondii. Sci. Rep. 2017, 7, 42746. [Google Scholar] [CrossRef] [Green Version]
- Wildführ, W. Electron microscopic studies on the morphology and reproduction of Toxoplasma gondii. II. Observations on the reproduction of Toxoplasma gondii (endodyogeny). Zentralbl. Bakteriol. Orig. 1966, 201, 110–130. [Google Scholar]
- Black, M.W.; Boothroyd, J.C. Lytic cycle of Toxoplasma gondii. Microbiol. Mol. Biol. Rev. 2000, 64, 607–623. [Google Scholar] [CrossRef] [Green Version]
| Treatment | 5 µM | 10 µM | 25 µM | 50 µM |
|---|---|---|---|---|
| DMSO 1 | 28.84 | |||
| atovaquone 2 | 2.28 | |||
| BPZ | 26.17 | 22.57 | 15.78 | 2.28 |
| amino-BPZ | 22.95 | 24.21 | 22.28 | 16.42 |
| methyl-BPZ | 21.88 | 19.54 | 8.99 | 3.31 |
| palmitoyl-BPZ | 18.29 | 16.64 | 14.17 | 4.30 |
| ethyl-BPZ | 21.78 | 15.93 | 2.53 | 1.84 |
| isopropyl-BPZ | 3.71 | 1.98 | - 3 | - |
| hexanoyl-BPZ | 20.78 | 15.63 | 8.76 | 3.08 |
| hydroxyl-BPZ | 25.58 | 20.34 | 22.50 | 20.87 |
| dansyl-BPZ | 6.80 | 3.69 | 2.23 | 1.82 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hatton, O.; Stitzlein, L.; Dudley, R.W.; Charvat, R.A. Evaluating the Antiparasitic Activity of Novel BPZ Derivatives Against Toxoplasma gondii. Microorganisms 2020, 8, 1159. https://doi.org/10.3390/microorganisms8081159
Hatton O, Stitzlein L, Dudley RW, Charvat RA. Evaluating the Antiparasitic Activity of Novel BPZ Derivatives Against Toxoplasma gondii. Microorganisms. 2020; 8(8):1159. https://doi.org/10.3390/microorganisms8081159
Chicago/Turabian StyleHatton, Olivia, Lea Stitzlein, Richard W. Dudley, and Robert A. Charvat. 2020. "Evaluating the Antiparasitic Activity of Novel BPZ Derivatives Against Toxoplasma gondii" Microorganisms 8, no. 8: 1159. https://doi.org/10.3390/microorganisms8081159
APA StyleHatton, O., Stitzlein, L., Dudley, R. W., & Charvat, R. A. (2020). Evaluating the Antiparasitic Activity of Novel BPZ Derivatives Against Toxoplasma gondii. Microorganisms, 8(8), 1159. https://doi.org/10.3390/microorganisms8081159







