Genetic Susceptibility of the Host in Virus-Induced Diabetes
Abstract
:1. Introduction
2. Host Genetic Factors in Humans
2.1. IFIH1
2.1.1. rs1990760
2.1.2. rs35667974, rs35337543, rs35744605, and rs35732034
2.1.3. rs3747517, rs2111485, and rs13422767
2.2. TYK2
2.2.1. rs2304256
2.2.2. rs12720356
2.2.3. rs2304258, rs17000728, rs17000730, rs2304259, rs891696485, and rs953883300
2.3. CXADR
2.3.1. rs6517774
2.3.2. rs2824404
2.4. BACH2
rs72928038, rs3757247, and rs11755527
2.5. PTPN22
rs2476601
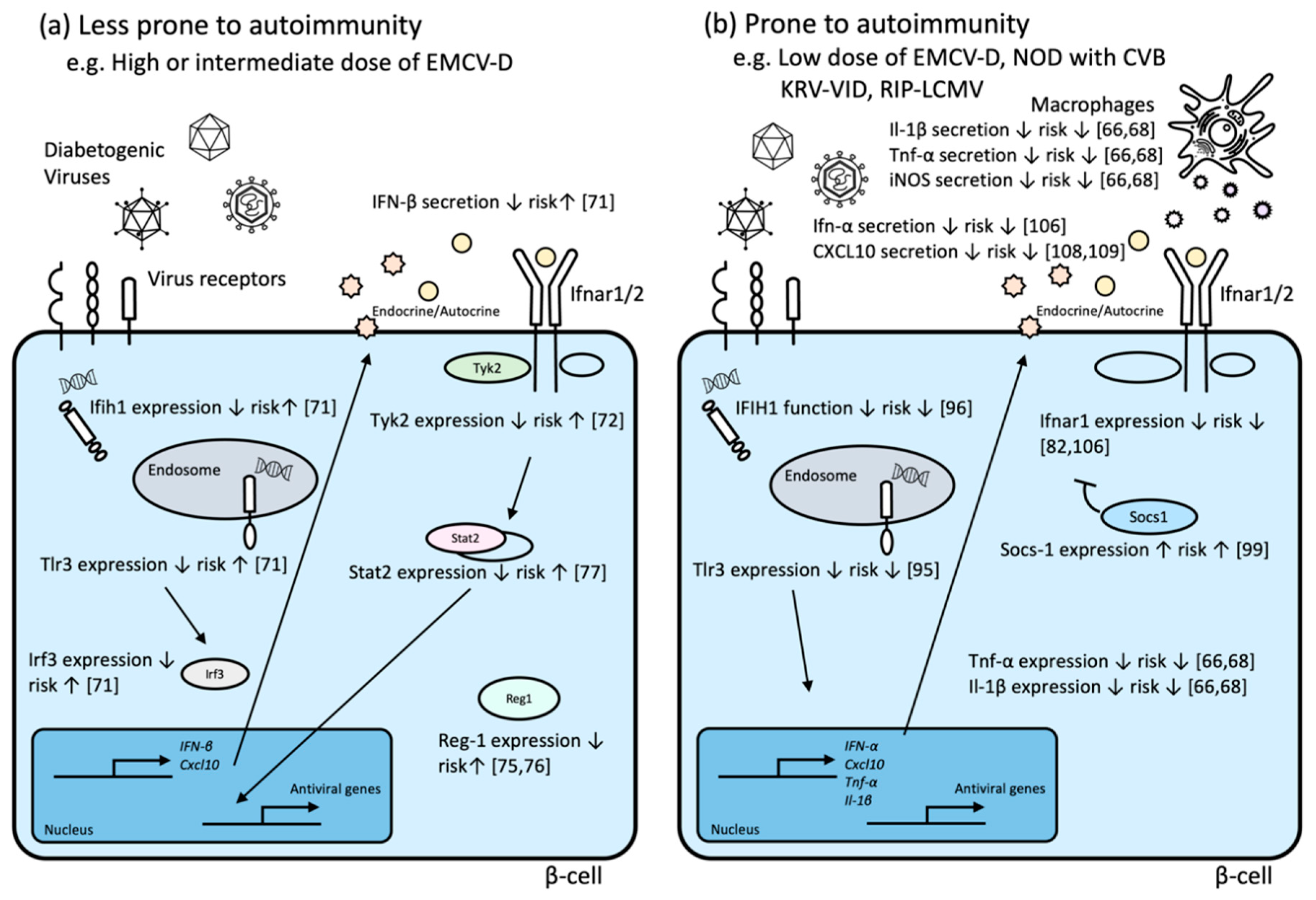
3. Host Genetic Factors in Animal Models
3.1. EMCV-Induced Diabetes Model
3.1.1. Ifih1, Tlr3, Irf3, and Ifn-β
3.1.2. Tyk2
3.1.3. Il-1β, Tnf-α, and iNOS
3.1.4. Reg1
3.1.5. Stat2
3.2. KRV-Induced Autoimmune Diabetes Model
3.2.1. MHC (RT1)
3.2.2. Ifnar1
3.3. CVB-Induced Acute Autoimmune NOD Diabetes Model
3.3.1. Tlr3
3.3.2. Ifih1
3.3.3. Socs-1
3.4. Transgenic Animal Models
3.4.1. Ifn-α
3.4.2. Cxcl10
3.5. Combined STZ and Virus Infection Model
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- International Diabetes Federation. IDF Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019; ISBN 9782930229874. [Google Scholar]
- American Diabetes Association. Classification and diagnosis of diabetes: Standards of medical care in diabetes-2019. Diabetes Care 2019, 42, S13–S28. [Google Scholar] [CrossRef] [Green Version]
- American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care 2014, 37, S81–S90. [Google Scholar] [CrossRef] [Green Version]
- Ilonen, J.; Lempainen, J.; Veijola, R. The heterogeneous pathogenesis of type 1 diabetes mellitus. Nat. Rev. Endocrinol. 2019, 15, 635–650. [Google Scholar] [CrossRef]
- Hober, D.; Sauter, P. Pathogenesis of type 1 diabetes mellitus: Interplay between enterovirus and host. Nat. Rev. Endocrinol. 2010, 6, 279–289. [Google Scholar] [CrossRef]
- Harris, H.F. A case of diabetes mellitus quickly following mumps. Bost. Med. Surg. J. 1899, 140, 465–469. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, S.; Nishida, Y.; Aida, K.; Maruyama, T.; Shimada, A.; Suzuki, M.; Shimura, H.; Takizawa, S.; Takahashi, M.; Akiyama, D.; et al. Enterovirus infection, CXC chemokine ligand 10 (CXCL10), and CXCR3 circuit a mechanism of accelerated β-cell failure in fulminant type 1 diabetes. Diabetes 2009, 58, 2285–2291. [Google Scholar] [CrossRef]
- Lönnrot, M.; Korpela, K.; Knip, M.; Ilonen, J.; Simell, O.; Korhonen, S.; Savola, K.; Muona, P.; Simell, T.; Koskela, P.; et al. Enterovirus infections as a risk factor for β-cell autoimmunity in a prospectively observed birth cohort: The Finnish Diabetes Prediction and Prevention Study. Diabetes 2000, 49, 1314–1318. [Google Scholar] [CrossRef] [Green Version]
- Rogers, M.A.M.; Basu, T.; Kim, C. Lower incidence rate of type 1 diabetes after receipt of the rotavirus vaccine in the united states, 2001–2017. Sci. Rep. 2019, 9, 7727. [Google Scholar] [CrossRef] [Green Version]
- Levet, S.; Charvet, B.; Bertin, A.; Deschaumes, A.; Perron, H.; Hober, D. Human endogenous retroviruses and type 1 diabetes. Curr. Diabetes Rep. 2019, 19, 141. [Google Scholar] [CrossRef] [Green Version]
- Levet, S.; Medina, J.; Joanou, J.; Demolder, A.; Queruel, N.; Réant, K.; Normand, M.; Seffals, M.; Dimier, J.; Germi, R.; et al. An ancestral retroviral protein identified as a therapeutic target in type-1 diabetes. JCI Insight 2017, 2. [Google Scholar] [CrossRef] [Green Version]
- Hao, W.; Serreze, D.V.; McCulloch, D.K.; Neifing, J.L.; Palmer, J.P. Insulin (auto) antibodies from human IDDM cross-react with retroviral antigen p73. J. Autoimmun. 1993, 6, 787–798. [Google Scholar] [CrossRef]
- Patterson, K.; Chandra, R.; Jenson, A.B. Congenital rubella, insulitis, and diabetes mellitus in an infent. Lancet 1981, 1, 1048–1049. [Google Scholar] [CrossRef]
- Rogers, M.A.M.; Kim, C. Congenital infections as contributors to the onset of diabetes in children: A longitudinal study in the United States, 2001–2017. Pediatr. Diabetes 2019, 21, 456–459. [Google Scholar] [CrossRef]
- Yoneda, S.; Imagawa, A.; Fukui, K.; Uno, S.; Kozawa, J.; Sakai, M.; Yumioka, T.; Iwahashi, H.; Shimomura, I. A histological study of fulminant type 1 diabetes mellitus related to human cytomegalovirus reactivation. J. Clin. Endocrinol. Metab. 2017, 102, 2394–2400. [Google Scholar] [CrossRef]
- Hiemstra, H.S.; Schloot, N.C.; Van Veelen, P.A.; Willemen, S.J.M.; Franken, K.L.M.C.; Van Rood, J.J.; De Vries, R.R.P.; Chaudhuri, A.; Behan, P.O.; Drijfhout, J.W.; et al. Cytomegalovirus in autoimmunity: T cell crossreactivity to viral antigen and autoantigen glutamic acid decarboxylase. Proc. Natl. Acad. Sci. USA 2001, 98, 3988–3991. [Google Scholar] [CrossRef] [Green Version]
- Bian, X.; Wallstrom, G.; Davis, A.; Wang, J.; Park, J.; Throop, A.; Steel, J.; Yu, X.; Wasserfall, C.; Schatz, D.; et al. Immunoproteomic profiling of antiviral antibodies in new-onset type 1 diabetes using protein arrays. Diabetes 2016, 65, 285–296. [Google Scholar] [CrossRef] [Green Version]
- Munakata, Y.; Kodera, T.; Saito, T.; Sasaki, T. Rheumatoid arthritis, type 1 diabetes, and Graves’ disease after acute parvovirus B19 infection. Lancet 2005, 366, 780. [Google Scholar] [CrossRef]
- Sabouri, S.; Benkahla, M.A.; Kiosses, W.B.; Rodriguez-Calvo, T.; Zapardiel-Gonzalo, J.; Castillo, E.; von Herrath, M.G. Human herpesvirus-6 is present at higher levels in the pancreatic tissues of donors with type 1 diabetes. J. Autoimmun. 2020, 107, 102378. [Google Scholar] [CrossRef]
- Drescher, K.M.; Kono, K.; Bopegamage, S.; Carson, S.D.; Tracy, S. Coxsackievirus B3 infection and type 1 diabetes development in NOD mice: Insulitis determines susceptibility of pancreatic islets to virus infection. Virology 2004, 329, 381–394. [Google Scholar] [CrossRef] [Green Version]
- Yoon, J.W.; McClintock, P.R.; Onodera, T.; Notkins, A.L. Virus-induced diabetes mellitus. XVIII. Inhibition by a nondiabetogenic variant of encephalomyocarditis virus. J. Exp. Med. 1980, 152, 878–892. [Google Scholar] [CrossRef] [Green Version]
- Yoon, J.W.; Morishima, T.; McClintock, P.R.; Austin, M.; Notkins, A.L. Virus-induced diabetes mellitus: Mengovirus infects pancreatic beta cells in strains of mice resistant to the diabetogenic effect of encephalomyocarditis virus. J. Virol. 1984, 50, 684–690. [Google Scholar] [CrossRef] [Green Version]
- Pane, J.A.; Fleming, F.E.; Graham, K.L.; Thomas, H.E.; Kay, T.W.H.; Coulson, B.S. Rotavirus acceleration of type 1 diabetes in non-obese diabetic mice depends on type I interferon signalling. Sci. Rep. 2016, 6, 29697. [Google Scholar] [CrossRef] [Green Version]
- Kenji, S.; Yoon, J. Association of β-cell-specific expression of endogenous retrovirus with development of insulitis and diabetes in NOD mouse. Diabetes 1988, 37, 1722–1726. [Google Scholar] [CrossRef]
- Onodera, T.; Jenson, A.B.; Yoon, J.W.; Notkins, A.L. Virus-induced diabetes mellitus: Reovirus infection of pancreatic β cells in mice. Science 1978, 201, 529–531. [Google Scholar] [CrossRef]
- Rayfield, E.J.; Kelly, K.J.; Yoon, J.W. Rubella virus-induced diabetes in the hamster. Diabetes 1986, 35, 1278–1281. [Google Scholar] [CrossRef]
- Capua, I.; Mercalli, A.; Pizzuto, M.S.; Romero-Tejeda, A.; Kasloff, S.; De Battisti, C.; Bonfante, F.; Patrono, L.V.; Vicenzi, E.; Zappulli, V.; et al. Influenza A viruses grow in human pancreatic cells and cause pancreatitis and diabetes in an animal model. J. Virol. 2013, 87, 597–610. [Google Scholar] [CrossRef] [Green Version]
- Taniyama, H.; Ushiki, T.; Tajima, M.; Kurosawa, T.; Kitamura, N.; Takahashi, K.; Matsukawa, K.; Itakura, C. Spontaneous diabetes mellitus associated with persistent bovine viral diarrhea (BVD) virus infection in young cattle. Vet. Pathol. 1995, 32, 221–229. [Google Scholar] [CrossRef] [Green Version]
- Ohashi, P.S.; Oehen, S.; Buerki, K.; Pircher, H.; Ohashi, C.T.; Odermatt, B.; Malissen, B.; Zinkernagel, R.M.; Hengartner, H. Ablation of “tolerance” and induction of diabetes by virus infection in viral antigen transgenic mice. Cell 1991, 65, 305–317. [Google Scholar] [CrossRef]
- Guberski, D.L.; Thomas, V.A.; Shek, W.R.; Like, A.A.; Handler, E.S.; Rossini, A.A.; Wallace, J.E.; Welsh, R.M. Induction of type I diabetes by Kilham’s rat virus in diabetes-resistant BB/Wor rats. Science 1991, 254, 1010–1013. [Google Scholar] [CrossRef]
- Van Der Werf, N.; Hillebrands, J.L.; Klatter, F.A.; Bos, I.; Bruggeman, C.A.; Rozing, J. Cytomegalovirus infection modulates cellular immunity in an experimental model for autoimmune diabetes. Clin. Dev. Immunol. 2003, 10, 153–160. [Google Scholar] [CrossRef] [Green Version]
- Imagawa, A.; Hanafusa, T. Fulminant type 1 diabetes-an important subtype in East Asia. Diabetes Metab. Res. Rev. 2011, 27, 959–964. [Google Scholar] [CrossRef]
- Aida, K.; Nishida, Y.; Tanaka, S.; Maruyama, T.; Shimada, A.; Awata, T.; Suzuki, M.; Shimura, H.; Takizawa, S.; Ichijo, M.; et al. RIG-I- and MDA5-initiated innate immunity linked with adaptive immunity accelerates β-cell death in fulminant type 1 diabetes. Diabetes 2011, 60, 884–889. [Google Scholar] [CrossRef] [Green Version]
- Hosokawa, Y.; Toyoda, T.; Fukui, K.; Baden, M.Y.; Funato, M.; Kondo, Y.; Sudo, T.; Iwahashi, H.; Kishida, M.; Okada, C.; et al. Insulin-producing cells derived from ‘induced pluripotent stem cells’ of patients with fulminant type 1 diabetes: Vulnerability to cytokine insults and increased expression of apoptosis-related genes. J. Diabetes Investig. 2018, 9, 481–493. [Google Scholar] [CrossRef] [Green Version]
- De Beeck, A.O.; Eizirik, D.L. Viral infections in type 1 diabetes mellitus-why the β cells? Nat. Rev. Endocrinol. 2016, 12, 263–273. [Google Scholar] [CrossRef] [Green Version]
- Oshima, M.; Knoch, K.P.; Diedisheim, M.; Petzold, A.; Cattan, P.; Bugliani, M.; Marchetti, P.; Choudhary, P.; Huang, G.C.; Bornstein, S.R.; et al. Virus-like infection induces human β cell dedifferentiation. JCI Insight 2018, 3. [Google Scholar] [CrossRef] [Green Version]
- Lempainen, J.; Laine, A.P.; Hammais, A.; Toppari, J.; Simell, O.; Veijola, R.; Knip, M.; Ilonen, J. Non-HLA gene effects on the disease process of type 1 diabetes: From HLA susceptibility to overt disease. J. Autoimmun. 2015, 61, 45–53. [Google Scholar] [CrossRef]
- Witsø, E.; Tapia, G.; Cinek, O.; Pociot, F.M.; Stene, L.C.; Rønningen, K.S. Polymorphisms in the innate immune IFIH1 gene, frequency of enterovirus in monthly fecal samples during infancy, and islet autoimmunity. PLoS ONE 2011, 6, e27781. [Google Scholar] [CrossRef] [Green Version]
- Domsgen, E.; Lind, K.; Kong, L.; Hühn, M.H.; Rasool, O.; Van Kuppeveld, F.; Korsgren, O.; Lahesmaa, R.; Flodström-Tullberg, M. An IFIH1 gene polymorphism associated with risk for autoimmunity regulates canonical antiviral defence pathways in Coxsackievirus infected human pancreatic islets. Sci. Rep. 2016, 6, 39378. [Google Scholar] [CrossRef]
- Onengut-Gumuscu, S.; Chen, W.M.; Burren, O.; Cooper, N.J.; Quinlan, A.R.; Mychaleckyj, J.C.; Farber, E.; Bonnie, J.K.; Szpak, M.; Schofield, E.; et al. Fine mapping of type 1 diabetes susceptibility loci and evidence for colocalization of causal variants with lymphoid gene enhancers. Nat. Genet. 2015, 47, 381–386. [Google Scholar] [CrossRef]
- Vehik, K.; Lynch, K.F.; Wong, M.C.; Tian, X.; Ross, M.C.; Gibbs, R.A.; Ajami, N.J.; Petrosino, J.F.; Rewers, M.; Toppari, J.; et al. Prospective virome analyses in young children at increased genetic risk for type 1 diabetes. Nat. Med. 2019, 25, 1865–1872. [Google Scholar] [CrossRef]
- Nagafuchi, S.; Kamada-Hibio, Y.; Hirakawa, K.; Tsutsu, N.; Minami, M.; Okada, A.; Kai, K.; Teshima, M.; Moroishi, A.; Murakami, Y.; et al. TYK2 Promoter Variant and Diabetes Mellitus in the Japanese. EBioMedicine 2015, 2, 744–749. [Google Scholar] [CrossRef] [Green Version]
- Mine, K.; Hirakawa, K.; Kondo, S.; Minami, M.; Okada, A.; Tsutsu, N.; Yokogawa, Y.; Hibio, Y.; Kojima, F.; Fujimoto, S.; et al. Subtyping of Type 1 Diabetes as Classified by Anti-GAD Antibody, IgE Levels, and Tyrosine kinase 2 (TYK2) Promoter Variant in the Japanese. EBioMedicine 2017, 23, 46–51. [Google Scholar] [CrossRef] [Green Version]
- Bradfield, J.P.; Qu, H.Q.; Wang, K.; Zhang, H.; Sleiman, P.M.; Kim, C.E.; Mentch, F.D.; Qiu, H.; Glessner, J.T.; Thomas, K.A.; et al. A genome-wide meta-analysis of six type 1 diabetes cohorts identifies multiple associated loci. PLoS Genet. 2011, 7, 1002293. [Google Scholar] [CrossRef] [Green Version]
- Kang, D.C.; Gopalkrishnan, R.V.; Wu, Q.; Jankowsky, E.; Pyle, A.M.; Fisher, P.B. mda-5: An interferon-inducible putative RNA helicase with double-stranded RNA-dependent ATPase activity and melanoma growth-suppressive properties. Proc. Natl. Acad. Sci. USA 2002, 99, 637–642. [Google Scholar] [CrossRef] [Green Version]
- Wu, B.; Peisley, A.; Richards, C.; Yao, H.; Zeng, X.; Lin, C.; Chu, F.; Walz, T.; Hur, S. Structural basis for dsRNA recognition, filament formation, and antiviral signal activation by MDA5. Cell 2013, 152, 276–289. [Google Scholar] [CrossRef] [Green Version]
- Dias Junior, A.G.; Sampaio, N.G.; Rehwinkel, J. A Balancing Act: MDA5 in Antiviral Immunity and Autoinflammation. Trends Microbiol. 2019, 27, 75–85. [Google Scholar] [CrossRef] [Green Version]
- Barrett, J.C.; Clayton, D.G.; Concannon, P.; Akolkar, B.; Cooper, J.D.; Erlich, H.A.; Julier, C.; Morahan, G.; Nerup, J.; Nierras, C.; et al. Genome-wide association study and meta-analysis find that over 40 loci affect risk of type 1 diabetes. Nat. Genet. 2009, 41, 703–707. [Google Scholar] [CrossRef] [Green Version]
- Funabiki, M.; Kato, H.; Miyachi, Y.; Toki, H.; Motegi, H.; Inoue, M.; Minowa, O.; Yoshida, A.; Deguchi, K.; Sato, H.; et al. Autoimmune disorders associated with gain of function of the intracellular sensor MDA5. Immunity 2014, 40, 199–212. [Google Scholar] [CrossRef] [Green Version]
- Shigemoto, T.; Kageyama, M.; Hirai, R.; Zheng, J.P.; Yoneyama, M.; Fujita, T. Identification of loss of function mutations in human genes encoding RIG-I and MDA5: Implications for resistance to type I diabetes. J. Biol. Chem. 2009, 284, 13348–13354. [Google Scholar] [CrossRef] [Green Version]
- Gorman, J.A.; Hundhausen, C.; Errett, J.S.; Stone, A.E.; Allenspach, E.J.; Ge, Y.; Arkatkar, T.; Clough, C.; Dai, X.; Khim, S.; et al. The A946T variant of the RNA sensor IFIH1 mediates an interferon program that limits viral infection but increases the risk for autoimmunity. Nat. Immunol. 2017, 18, 744–752. [Google Scholar] [CrossRef]
- Nejentsev, S.; Walker, N.; Riches, D.; Egholm, M.; Todd, J.A. Rare variants of IFIH1, a gene implicated in antiviral responses, protect against type 1 diabetes. Science 2009, 324, 387–389. [Google Scholar] [CrossRef] [Green Version]
- Asgari, S.; Schlapbach, L.J.; Anchisi, S.; Hammer, C.; Bartha, I.; Junier, T.; Mottet-Osman, G.; Posfay-Barbe, K.M.; Longchamp, D.; Stocker, M.; et al. Severe viral respiratory infections in children with IFIH1 loss-of-function mutations. Proc. Natl. Acad. Sci. USA 2017, 114, 8342–8347. [Google Scholar] [CrossRef] [Green Version]
- Zurawek, M.; Fichna, M.; Fichna, P.; Skowronska, B.; Dzikiewicz-Krawczyk, A.; Januszkiewicz, D.; Nowak, J. Cumulative effect of IFIH1 variants and increased gene expression associated with type 1 diabetes. Diabetes Res. Clin. Pract. 2015, 107, 259–266. [Google Scholar] [CrossRef]
- Shimoda, K.; Kato, K.; Aoki, K.; Matsuda, T.; Miyamoto, A.; Shibamori, M.; Yamashita, M.; Numata, A.; Takase, K.; Kobayashi, S.; et al. Tyk2 plays a restricted role in IFNα signaling, although it is required for IL-12-mediated T cell function. Immunity 2000, 13, 561–571. [Google Scholar] [CrossRef]
- Leitner, N.R.; Witalisz-Siepracka, A.; Strobl, B.; Müller, M. Tyrosine kinase 2 – Surveillant of tumours and bona fide oncogene. Cytokine 2017, 89, 209–218. [Google Scholar] [CrossRef] [Green Version]
- Wilks, A.F.; Harpur, A.G.; Kurban, R.R.; Ralph, S.J.; Zürcher, G.; Ziemiecki, A. Two novel protein-tyrosine kinases, each with a second phosphotransferase-related catalytic domain, define a new class of protein kinase. Mol. Cell. Biol. 1991, 11, 2057–2065. [Google Scholar] [CrossRef] [Green Version]
- Marroqui, L.; Dos Santos, R.S.; Fløyel, T.; Grieco, F.A.; Santin, I.; Op De Beeck, A.; Marselli, L.; Marchetti, P.; Pociot, F.; Eizirik, D.L. TYK2, a candidate gene for type 1 diabetes, modulates apoptosis and the innate immune response in human pancreatic β-cells. Diabetes 2015, 64, 3808–3817. [Google Scholar] [CrossRef] [Green Version]
- Richter, M.F.; Duménil, G.; Uzé, G.; Fellous, M.; Pellegrini, S. Specific contribution of Tyk2 JH regions to the binding and the expression of the interferon α/β receptor component IFNAR1. J. Biol. Chem. 1998, 273, 24723–24729. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Rotival, M.; Patin, E.; Michel, F.; Pellegrini, S. Two common disease-associated TYK2 variants impact exon splicing and TYK2 dosage. PLoS ONE 2020, 15. [Google Scholar] [CrossRef] [Green Version]
- Wallace, C.; Smyth, D.J.; Maisuria-Armer, M.; Walker, N.M.; Todd, J.A.; Clayton, D.G. The imprinted DLK1-MEG3 gene region on chromosome 14q32.2 alters susceptibility to type 1 diabetes. Nat. Genet. 2010, 42, 68–71. [Google Scholar] [CrossRef] [Green Version]
- Dendrou, C.A.; Cortes, A.; Shipman, L.; Evans, H.G.; Attfield, K.E.; Jostins, L.; Barber, T.; Kaur, G.; Kuttikkatte, S.B.; Leach, O.A.; et al. Resolving TYK2 locus genotype-to-phenotype differences in autoimmunity. Sci. Transl. Med. 2016, 8, 363ra149. [Google Scholar] [CrossRef] [Green Version]
- Enerbäck, C.; Sandin, C.; Lambert, S.; Zawistowski, M.; Stuart, P.E.; Verma, D.; Tsoi, L.C.; Nair, R.P.; Johnston, A.; Elder, J.T. The psoriasis-protective TYK2 I684S variant impairs IL-12 stimulated pSTAT4 response in skin-homing CD4+ and CD8+ memory T-cells. Sci. Rep. 2018, 8, 7043. [Google Scholar] [CrossRef] [Green Version]
- Toniolo, A.; Leslie, R.D. Diabetes, the TYK2 gene and the interferon response: In search for environmental causes. EBioMedicine 2017, 24, 18–19. [Google Scholar]
- Mori, H.; Takahashi, H.; Mine, K.; Inoue, K.; Kojima, M.; Kuroki, S.; Ono, Y.; Inuzuka, S.; Nagafuchi, S.; Anzai, K. 1706-P: A promoter variant of TYK2, a putative virus-induced susceptibility gene, is associated with impaired insulin secretion and nonobese type 2 diabetes. Diabetes 2019, 68. [Google Scholar] [CrossRef]
- Tomko, R.P.; Xu, R.; Philipson, L. HCAR and MCAR: The human and mouse cellular receptors for subgroup C adenoviruses and group B coxsackieviruses. Proc. Natl. Acad. Sci. USA 1997, 94, 3352–3356. [Google Scholar] [CrossRef] [Green Version]
- Cohen, C.J.; Shieh, J.T.C.; Pickles, R.J.; Okegawa, T.; Hsieh, J.T.; Bergelson, J.M. The coxsackievirus and adenovirus receptor is a transmembrane component of the tight junction. Proc. Natl. Acad. Sci. USA 2001, 98, 15191–15196. [Google Scholar] [CrossRef] [Green Version]
- Hodik, M.; Anagandula, M.; Fuxe, J.; Krogvold, L.; Dahl-Jørgensen, K.; Hyöty, H.; Sarmiento, L.; Frisk, G. Coxsackie–adenovirus receptor expression is enhanced in pancreas from patients with type 1 diabetes. BMJ Open Diabetes Res. Care 2016, 4, e000219. [Google Scholar] [CrossRef] [Green Version]
- Bergelson, J.M.; Krithivas, A.; Celi, L.; Droguett, G.; Horwitz, M.S.; Wickham, T.; Crowell, R.L.; Finberg, R.W. The murine CAR homolog is a receptor for coxsackie B viruses and adenoviruses. J. Virol. 1998, 72, 415–419. [Google Scholar]
- Nyalwidhe, J.O.; Jurczyk, A.; Satish, B.; Redick, S.; Qaisar, N.; Trombly, M.I.; Vangala, P.; Racicot, R.; Bortell, R.; Harlan, D.M.; et al. Proteomic and transcriptional profiles of human stem cell-derived β cells following enteroviral challenge. Microorganisms 2020, 8, 295. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.; Wu, H.; Zhao, M.; Chang, C.; Lu, Q. The Bach family of transcription factors: A comprehensive review. Clin. Rev. Allergy Immunol. 2016, 50, 345–356. [Google Scholar] [CrossRef]
- Roychoudhuri, R.; Hirahara, K.; Mousavi, K.; Clever, D.; Klebanoff, C.A.; Bonelli, M.; Sciumè, G.; Zare, H.; Vahedi, G.; Dema, B.; et al. BACH2 represses effector programs to stabilize T reg-mediated immune homeostasis. Nature 2013, 498, 506–510. [Google Scholar] [CrossRef]
- Xue, A.; Wu, Y.; Zhu, Z.; Zhang, F.; Kemper, K.E.; Zheng, Z.; Yengo, L.; Lloyd-Jones, L.R.; Sidorenko, J.; Wu, Y.; et al. Genome-wide association analyses identify 143 risk variants and putative regulatory mechanisms for type 2 diabetes. Nat. Commun. 2018, 9, 2941. [Google Scholar] [CrossRef] [Green Version]
- Marroquí, L.; Santin, I.; Dos Santos, R.S.; Marselli, L.; Marchetti, P.; Eizirik, D.L. BACH2, a candidate risk gene for type 1 diabetes, regulates apoptosis in pancreatic β-cells via JNK1 modulation and crosstalk with the candidate gene PTPN2. Diabetes 2014, 63, 2516–2527. [Google Scholar] [CrossRef] [Green Version]
- Roychoudhuri, R.; Clever, D.; Li, P.; Wakabayashi, Y.; Quinn, K.M.; Klebanoff, C.A.; Ji, Y.; Sukumar, M.; Eil, R.L.; Yu, Z.; et al. BACH2 regulates CD8 + T cell differentiation by controlling access of AP-1 factors to enhancers. Nat. Immunol. 2016, 17, 851–860. [Google Scholar] [CrossRef] [Green Version]
- Bottini, N.; Peterson, E.J. Tyrosine Phosphatase PTPN22: Multifunctional Regulator of Immune Signaling, Development, and Disease. Annu. Rev. Immunol. 2014, 32, 83–119. [Google Scholar] [CrossRef]
- Brownlie, R.J.; Miosge, L.A.; Vassilakos, D.; Svensson, L.M.; Cope, A.; Zamoyska, R. Lack of the phosphatase PTPN22 increases adhesion of murine regulatory T cells to improve their immunosuppressive function. Sci. Signal. 2012, 5. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Shaked, I.; Stanford, S.M.; Zhou, W.; Curtsinger, J.M.; Mikulski, Z.; Shaheen, Z.R.; Cheng, G.; Sawatzke, K.; Campbell, A.M.; et al. The Autoimmunity-associated gene PTPN22 potentiates Toll-like receptor-driven, type 1 interferon-dependent immunity. Immunity 2013, 39, 111–122. [Google Scholar] [CrossRef] [Green Version]
- Habib, T.; Funk, A.; Rieck, M.; Brahmandam, A.; Dai, X.; Panigrahi, A.K.; Luning Prak, E.T.; Meyer-Bahlburg, A.; Sanda, S.; Greenbaum, C.; et al. Altered B cell homeostasis is associated with type I diabetes and carriers of the PTPN22 allelic variant. J. Immunol. 2012, 188, 487–496. [Google Scholar] [CrossRef] [Green Version]
- Aarnisalo, J.; Treszl, A.; Svec, P.; Marttila, J.; Öling, V.; Simell, O.; Knip, M.; Körner, A.; Madacsy, L.; Vasarhelyi, B.; et al. Reduced CD4+T cell activation in children with type 1 diabetes carrying the PTPN22/Lyp 620Trp variant. J. Autoimmun. 2008, 31, 13–21. [Google Scholar] [CrossRef]
- Krischer, J.P.; Lynch, K.F.; Lernmark, A.; Hagopian, W.A.; Rewers, M.J.; She, J.X.; Toppari, J.; Ziegler, A.G.; Akolkar, B. Genetic and environmental interactions modify the risk of diabetes-related autoimmunity by 6 years of age: The teddy study. Diabetes Care 2017, 40, 1194–1202. [Google Scholar] [CrossRef] [Green Version]
- Carocci, M.; Bakkali-Kassimi, L. The encephalomyocarditis virus. Virulence 2012, 3, 351–367. [Google Scholar] [CrossRef] [Green Version]
- Nagafuchi, S.; Kurisaki, H.; Katsuta, H. Encephalomyocarditis virus. In Diabetes and Viruses; Taylor, K.W., Hyoty, H., Toniolo, A., Zuckerman, A.J., Eds.; Springer: New York, NY, USA, 2013; pp. 37–48. ISBN 9781461440512. [Google Scholar]
- Kounoue, E.; Nagafuchi, S.; Nakamura, M.; Nakano, S.; Koga, T.; Nakayama, M.; Mituyama, M.; Niho, Y.; Takaki, R. Encephalomyocarditis (EMC) virus-induced diabetes mellitus prevented by Corynebacterium parvum in mice. Experientia 1987, 43, 430–431. [Google Scholar] [CrossRef]
- Yoon, J.W.; McClintock, P.R.; Bachurski, C.J.; Longstreth, J.D.; Notkins, A.L. Virus-induced diabetes mellitus. No evicence for immune mechanisms in the destruction of β-cells by the D-variant of encephalomyocarditis virus. Diabetes 1985, 34, 922–925. [Google Scholar] [CrossRef]
- Jun, H.S.; Yoon, J.W. A new look at viruses in type 1 diabetes. Diabetes Metab. Res. Rev. 2003, 19, 8–31. [Google Scholar] [CrossRef]
- Baek, H.S.; Yoon, J.W. Role of macrophages in the pathogenesis of encephalomyocarditis virus-induced diabetes in mice. J. Virol. 1990, 64, 5708–5715. [Google Scholar] [CrossRef] [Green Version]
- Lee, Y.S.; Li, N.; Shin, S.; Jun, H.S. Role of nitric oxide in the pathogenesis of encephalomyocarditis virus-induced diabetes in mice. J. Virol. 2009, 83, 8004–8011. [Google Scholar] [CrossRef] [Green Version]
- Baek, H.; Yoon, J. Direct involvement of macrophages in destruction of β-cells leading to development of diabetes in virus-infected mice. Diabetes 1991, 40, 1586–1597. [Google Scholar] [CrossRef]
- Hirasawa, K.; Jun, H.S.; Maeda, K.; Kawaguchi, Y.; Itagaki, S.; Mikami, T.; Baek, H.S.; Doi, K.; Yoon, J.W. Possible role of macrophage-derived soluble mediators in the pathogenesis of encephalomyocarditis virus-induced diabetes in mice. J. Virol. 1997, 71, 4024–4031. [Google Scholar] [CrossRef] [Green Version]
- Kounoue, E.; Izumi, K.I.; Ogawa, S.; Kondo, S.; Katsuta, H.; Akashi, T.; Niho, Y.; Harada, M.; Tamiya, S.; Kurisaki, H.; et al. The significance of T cells, B cells, antibodies and macrophages against encephalomyocarditis (EMC)-D virus-induced diabetes in mice. Arch. Virol. 2008, 153, 1223–1231. [Google Scholar] [CrossRef]
- Onodera, T.; Yoon, J.; Brown, K.S.; Notkins, A.L. Evidence for a single locus controlling susceptibility to virus-induced diabetes mellitus. Nature 1978, 274, 693–696. [Google Scholar] [CrossRef]
- McCartney, S.A.; Vermi, W.; Lonardi, S.; Rossini, C.; Otero, K.; Calderon, B.; Gilfillan, S.; Diamond, M.S.; Unanue, E.R.; Colonna, M. RNA sensor-induced type I IFN prevents diabetes caused by a β cell – tropic virus in mice. J. Clin. Investig. 2011, 121, 1497–1507. [Google Scholar] [CrossRef]
- Izumi, K.; Mine, K.; Inoue, Y.; Teshima, M.; Ogawa, S.; Kai, Y.; Kurafuji, T.; Hirakawa, K.; Miyakawa, D.; Ikeda, H.; et al. Reduced Tyk2 gene expression in β-cells due to natural mutation determines susceptibility to virus-induced diabetes. Nat. Commun. 2015, 6, 6748. [Google Scholar] [CrossRef]
- Chen, Z.; Downing, S.; Tzanakakis, E.S. Four decades after the discovery of regenerating islet-derived (Reg) proteins: Current understanding and challenges. Front. Cell Dev. Biol. 2019, 7, 235. [Google Scholar] [CrossRef]
- Bacon, S.; Kyithar, M.P.; Schmid, J.; Rizvi, S.R.; Bonner, C.; Graf, R.; Prehn, J.H.M.; Byrne, M.M. Serum levels of pancreatic stone protein (PSP)/reg1A as an indicator of beta-cell apoptosis suggest an increased apoptosis rate in hepatocyte nuclear factor 1 alpha (HNF1A-MODY) carriers from the third decade of life onward. BMC Endocr. Disord. 2012, 12. [Google Scholar] [CrossRef] [Green Version]
- Aida, K.; Kobayashi, T.; Takeshita, A.; Jimbo, E.; Nishida, Y.; Yagihashi, S.; Hosoi, M.; Fukui, T.; Sugawara, A.; Takasawa, S. Crucial role of Reg I from acinar-like cell cluster touching with islets (ATLANTIS) on mitogenesis of beta cells in EMC virus-induced diabetic mice. Biochem. Biophys. Res. Commun. 2018, 503, 963–969. [Google Scholar] [CrossRef]
- Aida, K.; Saitoh, S.; Nishida, Y.; Yokota, S.; Ohno, S.; Mao, X.; Akiyama, D.; Tanaka, S.; Awata, T.; Shimada, A.; et al. Distinct cell clusters touching islet cells induce islet cell replication in association with over-expression of Regenerating Gene (REG) protein in fulminant type 1 diabetes. PLoS ONE 2014, 9, e105449. [Google Scholar] [CrossRef]
- Mine, K.; Nagafuchi, S.; Hatano, S.; Tanaka, K.; Mori, H.; Takahashi, H.; Anzai, K.; Yoshikai, Y. Impaired upregulation of Stat2 gene restrictive to pancreatic β-cells is responsible for virus-induced diabetes in DBA/2 mice. Biochem. Biophys. Res. Commun. 2020, 521, 853–860. [Google Scholar] [CrossRef]
- Ellerman, K.E.; Richards, C.A.; Guberski, D.L.; Shek, W.R.; Like, A.A. Kilham rat virus triggers T-cell-dependent autoimmune diabetes in multiple strains of rat. Diabetes 1996, 45, 557–562. [Google Scholar] [CrossRef]
- Alkanani, A.K.; Hara, N.; Gianani, R.; Zipris, D. Kilham rat virus-induced type 1 diabetes involves beta cell infection and intra-islet JAK-STAT activation prior to insulitis. Virology 2014, 468, 19–27. [Google Scholar] [CrossRef] [Green Version]
- Hara, N.; Alkanani, A.K.; Dinarello, C.A.; Zipris, D. Histone deacetylase inhibitor suppresses virus-induced proinflammatory responses and type 1 diabetes. J. Mol. Med. 2014, 92, 93–102. [Google Scholar] [CrossRef]
- Qaisar, N.; Lin, S.; Ryan, G.; Yang, C.; Oikemus, S.R.; Brodsky, M.H.; Bortel, R.; Mordes, J.P.; Wang, J.P. A critical role for the type I interferon receptor in virus-induced autoimmune diabetes in rats. Diabetes 2017, 66, 145–157. [Google Scholar] [CrossRef] [Green Version]
- Reed, J.C.; Herold, K.C. Thinking bedside at the bench: The NOD mouse model of T1DM. Nat. Rev. Endocrinol. 2015, 11, 308–314. [Google Scholar] [CrossRef]
- Pearson, J.A.; Wong, F.S.; Wen, L. The importance of the Non Obese Diabetic (NOD) mouse model in autoimmune diabetes. J. Autoimmun. 2016, 66, 76–88. [Google Scholar] [CrossRef] [Green Version]
- Miyazaki, A.; Hanafusa, T.; Yamada, K.; Miyagawa, J.; Fujino-Kurihara, H.; Nakajima, H.; Nonaka, K.; Tarui, S. Predominance of T lymphocytes in pancreatic islets and spleen of pre-diabetic non-obese diabetic (NOD) mice: A longitudinal study. Clin. Exp. Immunol. 1985, 60, 622–630. [Google Scholar]
- Jansen, A.; Homo-Delarche, F.; Hooijkaas, H.; Leenen, P.J.; Dardenne, M.; Drexhage, H.A. Immunohistochemical characterization of monocytes-macrophages and dendritic cells involved in the initiation of the insulitis and β-cell destruction in NOD mice. Diabetes 1994, 43, 667–675. [Google Scholar] [CrossRef]
- Akashi, M.; Nagafuchi, S.; Anzai, K.; Kondo, S.; Kitamura, D.; Wakana, S.; Ono, J.; Kikuchi, M.; Niho, Y.; Watanabe, T. Direct evidence for the contribution of B cells to the progression of insulitis and the development of diabetes in non-obese diabetic mice. Int. Immunol. 1997, 9, 1159–1164. [Google Scholar] [CrossRef] [Green Version]
- Tracy, S.; Drescher, K.M.; Chapman, N.M.; Kim, K.S.; Carson, S.D.; Pirruccello, S.; Lane, P.H.; Romero, J.R.; Leser, J.S. Toward testing the hypothesis that group B coxsackieviruses (CVB) trigger insulin-dependent diabetes: Inoculating nonobese diabetic mice with CVB markedly lowers diabetes incidence. J. Virol. 2002, 76, 12097–12111. [Google Scholar] [CrossRef] [Green Version]
- Atkinson, M.A.; Ellis, T.M.; Serreze, D.V.; Ottendorfer, E.W.; Gauntt, C.J. Acceleration of type 1 diabetes by a coxsackievirus infection requires a preexisting critical mass of autoreactive T-cells in pancreatic islets. Diabetes 2007, 49, 708–711. [Google Scholar] [CrossRef] [Green Version]
- Serreze, D.V.; Wasserfall, C.; Ottendorfer, E.W.; Stalvey, M.; Pierce, M.A.; Gauntt, C.; O’Donnell, B.; Flanagan, J.B.; Campbell-Thompson, M.; Ellis, T.M.; et al. Diabetes acceleration or prevention by a coxsackievirus B4 infection: Critical requirements for both interleukin-4 and gamma interferon. J. Virol. 2004, 79, 1045–1052. [Google Scholar] [CrossRef] [Green Version]
- Filippi, C.M.; Estes, E.A.; Oldham, J.E.; Von Herrath, M.G. Immunoregulatory mechanisms triggered by viral infections protect from type 1 diabetes in mice. J. Clin. Investig. 2009, 119, 1515–1523. [Google Scholar] [CrossRef] [Green Version]
- Hermitte, L.; Vialettes, B.; Naquet, P.; Atlan, C.; Payan, M.J.; Vague, P. Paradoxical lessening of autoimmune processes in non-obese diabetic mice afterinfection with the diabetogenic variant of encephalomyocarditis virus. Eur. J. Immunol. 1990, 20, 1297–1303. [Google Scholar] [CrossRef]
- Kanno, T.; Kim, K.; Kono, K.; Drescher, K.M.; Chapman, N.M.; Tracy, S. Group B Coxsackievirus diabetogenic phenotype correlates with replication efficiency. J. Virol. 2006, 80, 5637–5643. [Google Scholar] [CrossRef] [Green Version]
- McCall, K.D.; Thuma, J.R.; Courreges, M.C.; Benencia, F.; James, C.B.L.; Malgor, R.; Kantake, N.; Mudd, W.; Denlinger, N.; Nolan, B.; et al. Toll-like receptor 3 is critical for coxsackievirus B4-induced type 1 diabetes in female nod mice. Endocrinology 2015, 156, 453–461. [Google Scholar] [CrossRef] [Green Version]
- Lincez, P.J.; Shanina, I.; Horwitz, M.S. Reduced expression of the MDA5 gene IFIH1 prevents autoimmune diabetes. Diabetes 2015, 64, 2184–2193. [Google Scholar] [CrossRef] [Green Version]
- Naka, T.; Narazaki, M.; Hirata, M.; Matsumoto, T.; Minamoto, S.; Aono, A.; Nishimoto, N.; Kajita, T.; Taga, T.; Yoshizaki, K.; et al. Structure and function of a new STAT-induced STAT inhibitor. Nature 1997, 387, 924–929. [Google Scholar] [CrossRef]
- Alexander, W.S.; Starr, R.; Fenner, J.E.; Scott, C.L.; Handman, E.; Sprigg, N.S.; Corbin, J.E.; Cornish, A.L.; Darwiche, R.; Owczarek, C.M.; et al. SOCS1 is a critical inhibitor of interferon γ signaling and prevents the potentially fatal neonatal actions of this cytokine. Cell 1999, 98, 597–608. [Google Scholar] [CrossRef] [Green Version]
- Flodström, M.; Maday, A.; Balakrishna, D.; Cleary, M.M.; Yoshimura, A.; Sarvetnick, N. Target cell defense prevents the development of diabetes after viral infection. Nat. Immunol. 2002, 3, 373–382. [Google Scholar] [CrossRef]
- Stone, V.M.; Hankaniemi, M.M.; Svedin, E.; Sioofy-Khojine, A.; Oikarinen, S.; Hyöty, H.; Laitinen, O.H.; Hytönen, V.P.; Flodström-Tullberg, M. A Coxsackievirus B vaccine protects against virus-induced diabetes in an experimental mouse model of type 1 diabetes. Diabetologia 2018, 61, 476–481. [Google Scholar] [CrossRef] [Green Version]
- Stone, V.M.; Hankaniemi, M.M.; Laitinen, O.H.; Sioofy-Khojine, A.B.; Lin, A.; Lozano, I.M.D.; Mazur, M.A.; Marjomäki, V.; Loré, K.; Hyöty, H.; et al. A hexavalent Coxsackievirus B vaccine is highly immunogenic and has a strong protective capacity in mice and nonhuman primates. Sci. Adv. 2020, 6, eaaz2433. [Google Scholar] [CrossRef]
- von Herrath, M.G.; Dockter, J.; Oldstone, M.B.A. How virus induces a rapid or slow onset insulin-dependent diabetes mellitus in a transgenic model. Immunity 1994, 1, 231–242. [Google Scholar] [CrossRef]
- Roman, L.M.; Simons, L.F.; Hammer, R.E.; Sambrook, J.F.; Gething, M.J.H. The expression of influenza virus hemagglutinin in the pancreatic β cells of transgenic mice results in autoimmune diabetes. Cell 1990, 61, 383–396. [Google Scholar] [CrossRef]
- Holers, V.M.; Hollis, G.F.; Schwartz, B.D.; Evans, R.J.; Strauss-Schoenberger, J.; Carel, J.C.; Barney, D.L.; Li, B.; Stafford-Hollis, J.; Lacy, P.E. Induction of peri-insulitis but not diabetes in islet transplants expressing a single foreign antigen: A multi-stage model of disease. J. Immunol. 1993, 151, 5041–5052. [Google Scholar]
- Marro, B.S.; Ware, B.C.; Zak, J.; de la Torre, J.C.; Rosen, H.; Oldstone, M.B.A. Progression of type 1 diabetes from the prediabetic stage is controlled by interferon-α signaling. Proc. Natl. Acad. Sci. USA 2017, 114, 3708–3713. [Google Scholar] [CrossRef] [Green Version]
- Jean-Baptiste, V.S.E.; Xia, C.Q.; Clare-Salzler, M.J.; Horwitz, M.S. Type 1 diabetes and type 1 interferonopathies: Localization of a type 1 common thread of virus infection in the pancreas. EBioMedicine 2017, 22, 10–17. [Google Scholar] [CrossRef] [Green Version]
- Bender, C.; Christen, S.; Scholich, K.; Bayer, M.; Pfeilschifter, J.M.; Hintermann, E.; Christen, U. Islet-expressed CXCL10 promotes autoimmune destruction of islet isografts in mice with type 1 diabetes. Diabetes 2017, 66, 113–126. [Google Scholar] [CrossRef] [Green Version]
- Rhode, A.; Pauza, M.E.; Barral, A.M.; Rodrigo, E.; Oldstone, M.B.A.; von Herrath, M.G.; Christen, U. Islet-specific expression of CXCL10 causes spontaneous islet infiltration and accelerates diabetes development. J. Immunol. 2005, 175, 3516–3524. [Google Scholar] [CrossRef]
- Yoshimatsu, G.; Kunnathodi, F.; Saravanan, P.B.; Shahbazov, R.; Chang, C.; Darden, C.M.; Zurawski, S.; Boyuk, G.; Kanak, M.A.; Levy, M.F.; et al. Pancreatic β-cell-derived IP-10/CXCL10 isletokine mediates early loss of graft function in islet cell transplantation. Diabetes 2017, 66, 2857–2867. [Google Scholar] [CrossRef] [Green Version]
- Toniolo, A.; Onodera, T.; Yoon, J.W.; Notkins, A.L. Induction of diabetes by cumulative environmental insults from viruses and chemicals. Nature 1980, 288, 383–385. [Google Scholar] [CrossRef]
- Benkahla, M.A.; Sane, F.; Bertin, A.; Vreulx, A.C.; Elmastour, F.; Jaidane, H.; Desailloud, R.; Hober, D. Impact of coxsackievirus-B4E2 combined with a single low dose of streptozotocin on pancreas of outbred mice: Investigation of viral load, pathology and inflammation. Sci. Rep. 2019, 9, 10080. [Google Scholar] [CrossRef] [Green Version]
- Capua, I.; Mercalli, A.; Romero-Tejeda, A.; Pizzuto, M.S.; Kasloff, S.; Sordi, V.; Marzinotto, I.; Lampasona, V.; Vicenzi, E.; De Battisti, C.; et al. Study of 2009 H1N1 pandemic influenza virus as a possible causative agent of diabetes. J. Clin. Endocrinol. Metab. 2018, 103, 4343–4356. [Google Scholar] [CrossRef]
- Mine, K.; Takahashi, H.; Nagafuchi, S. Model animal mimicking human virus-induced diabetes. EBioMedicine 2018, 32, 8. [Google Scholar] [CrossRef] [Green Version]
- Nagafuchi, S.; Mine, K.; Takahashi, H.; Anzai, K.; Yoshikai, Y. Viruses with masked pathogenicity and genetically susceptible hosts—How to discover potentially pathogenic viruses. J. Med. Virol. 2019, 91, 1365–1367. [Google Scholar] [CrossRef]

| Humans | ||
| Virus | Ref. | |
| RNA virus | Enterovirus | [7,8] |
| Rotavirus | [9] | |
| Retrovirus | [10,11,12] | |
| Rubella virus | [13,14] | |
| Mumps virus | [6] | |
| DNA virus | Cytomegalovirus | [15,16] |
| Epstein-Barr virus | [17] | |
| Parvovirus | [18] | |
| Herpesvirus | [19] | |
| Animals | ||
| Virus | Ref. | |
| RNA virus | Enterovirus | [20] |
| Encephalomyocarditis virus | [21] | |
| Mengovirus | [22] | |
| Rotavirus | [23] | |
| Retrovirus | [24] | |
| Reovirus | [25] | |
| Rubella virus | [26] | |
| Influenza A virus | [27] | |
| Bovine viral diarrhea-mucosal disease virus | [28] | |
| Lymphocytic choriomeningitis virus | [29] | |
| DNA virus | Kilham rat virus (Rat Parvovirus) | [30] |
| Cytomegalovirus | [31] |
| Candidate Genes | SNPs | Nucleoside Change (Amino Acid Change) | The Effect of the SNPs (OR, Odds Ratio; HR, Hazard Ratio) | Potential Association with VID |
|---|---|---|---|---|
| IFIH1 | rs1990760 | NC_000002.12:g.162267541C>T (NP_071451.2:p.Ala946Thr) | T1D, HR 1.21 [37]; EV prevalence in stools, OR 1.22 [38]; The association with islet autoimmunity, OR 1.2 [38] | Linked with the high expression of type III IFNs but not with IFN-β in pancreatic islets from human donors following CVB3 infection [39]; Associated with the production of β-cell autoantibodies (IAA, GADA, IA-2A) but not the development of T1D in Finnish population (Finnish DIPP study) [37]. |
| rs35667974 | NC_000002.12:g.162268127T>C (NP_071451.2:p.Ile923Val) | T1D OR 0.59 [40]; EV prevalence in stools, OR 0.86 [38] | Association was not observed with the frequency of EV RNA in stools from Norwegian newborns [38]. | |
| rs35337543 | NC_000002.12:g.162279995C>G; NC_000002.12:g.162279995C>T | EV prevalence in stools, OR 0.78 [38] | Association was not observed with the frequency of EV RNA in stools from Norwegian newborns [38]. | |
| rs35744605 | NC_000002.12:g.162277580C>A, C>G, C>T; (NP_071451.2:p.Glu627Ter, Gln, Lys) | EV prevalence in stools, OR 0.90 [38] | Association was not observed with the frequency of EV RNA in stools from Norwegian newborns [38]. | |
| rs35732034 | NC_000002.12:g.162268086C>T | EV prevalence in stools, OR 2.47 [38] | Marginally increased prevalence of EV RNA was observed in stools from Norwegian newborns [38]. | |
| TYK2 | rs2304256 | NC_000019.10:g.10364976C>A (NP_003322.3:p.Val362Phe) | - | Associated with EVB presence in stools, but not with the presence of islet autoantibodies in the United States and European population [41]; Association was not observed with the presence of flu-like syndrome at T1D onset in Japanese population [42]. |
| rs2304258, rs17000728, rs17000730, rs2304259, rs891696485, rs953883300 | NC_000019.10:g.10380510C>T; NC_000019.10:g.10380511C>T; NC_000019.10:g.10380572T>C; NC_000019.10:g.10380676T>G; NC_000019.10:g.10381501A>C, A>T; NC_000019.10:g.10381502C>T | T1D, OR 2.4 [42]; T1D with flu-like syndrome, OR 3.6 [42]; T1D without GADA, OR 3.3 [42]; T2D, OR 2.1 [42]; T1D, OR 1.89; T1D, HR 1.21 [37] | These SNPs, named TYK2 promoter variant (ClinVar, 440728), showed the risk of T1D, especially high risk in T1D associated with flu-like syndrome at the onset and those with GADA negative in Japanese population [42,43]. | |
| CXADR | rs6517774 | NC_000021.9:g.17449955A>G | The association with islet autoimmunity, OR 1.47 [41] | The lower detection of EVB in stools in the United States and European population [41]. |
| PTPN22 | rs2476601 | NC_000001.11:g.113834946A>G (NP_057051.3:p.Arg620Trp) | T1D, OR 1.89 [40]; T1D, OR 1.96 [44]; T1D, HR 1.97 [37]; The association with islet autoimmunity, OR 1.99 [41] | The prolonged shedding or consecutive positive for EVB, HAdV-C, HAdV-F in stools in the United States and European population [41]. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mine, K.; Yoshikai, Y.; Takahashi, H.; Mori, H.; Anzai, K.; Nagafuchi, S. Genetic Susceptibility of the Host in Virus-Induced Diabetes. Microorganisms 2020, 8, 1133. https://doi.org/10.3390/microorganisms8081133
Mine K, Yoshikai Y, Takahashi H, Mori H, Anzai K, Nagafuchi S. Genetic Susceptibility of the Host in Virus-Induced Diabetes. Microorganisms. 2020; 8(8):1133. https://doi.org/10.3390/microorganisms8081133
Chicago/Turabian StyleMine, Keiichiro, Yasunobu Yoshikai, Hirokazu Takahashi, Hitoe Mori, Keizo Anzai, and Seiho Nagafuchi. 2020. "Genetic Susceptibility of the Host in Virus-Induced Diabetes" Microorganisms 8, no. 8: 1133. https://doi.org/10.3390/microorganisms8081133
APA StyleMine, K., Yoshikai, Y., Takahashi, H., Mori, H., Anzai, K., & Nagafuchi, S. (2020). Genetic Susceptibility of the Host in Virus-Induced Diabetes. Microorganisms, 8(8), 1133. https://doi.org/10.3390/microorganisms8081133






