Immune-modulatory Properties of the Octapeptide NAP in Campylobacter jejuni Infected Mice Suffering from Acute Enterocolitis
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Statement
2.2. Generation of Secondary Abiotic IL-10−/− Mice
2.3. C. jejuni Infection, Gastrointestinal Colonization, and Extra-Intestinal Translocation
2.4. Treatment with Synthetic NAP
2.5. Clinical Assessment
2.6. Sampling Procedures
2.7. Immunohistochemistry
2.8. Pro-Inflammatory Cytokines in Intestinal, Extra-Intestinal, and Serum Samples
2.9. Statistical Analysis
3. Results
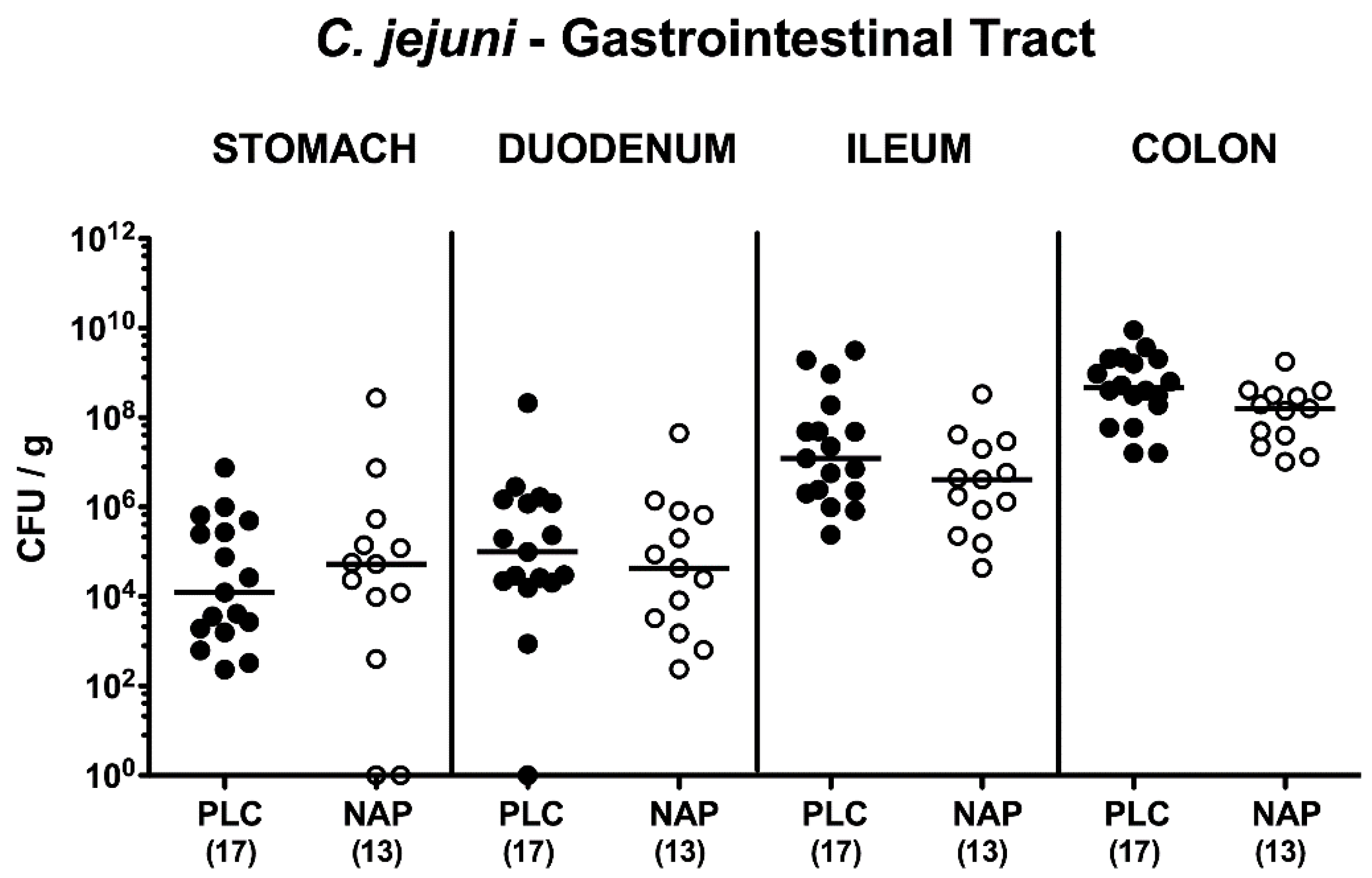
3.1. Gastrointestinal Pathogen Loads Following NAP Treatment of C. jejuni Infected Secondary Abiotic IL-10−/− Mice
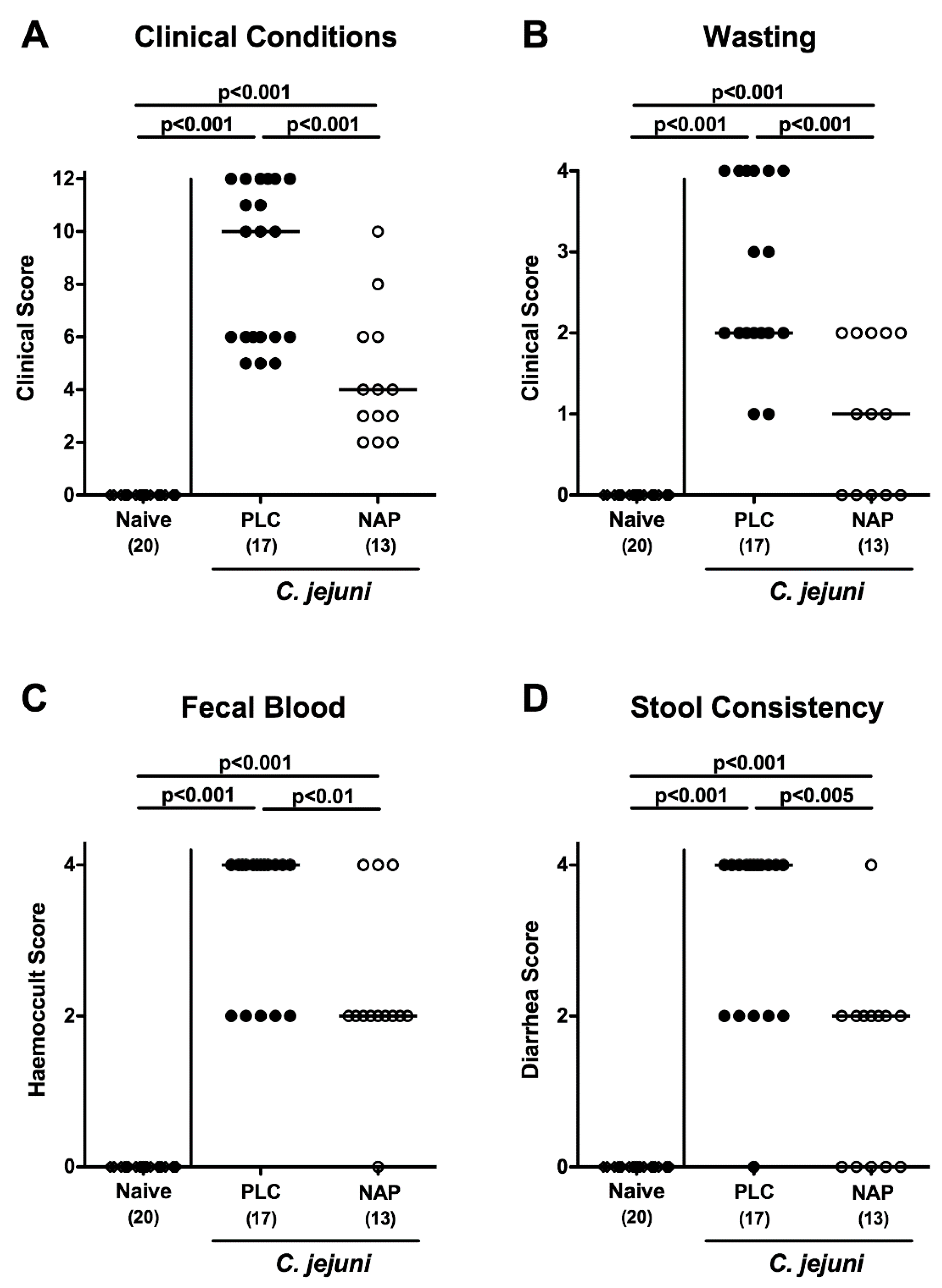
3.2. Clinical Conditions Following NAP Treatment of C. jejuni Infected Secondary Abiotic IL10−/− Mice
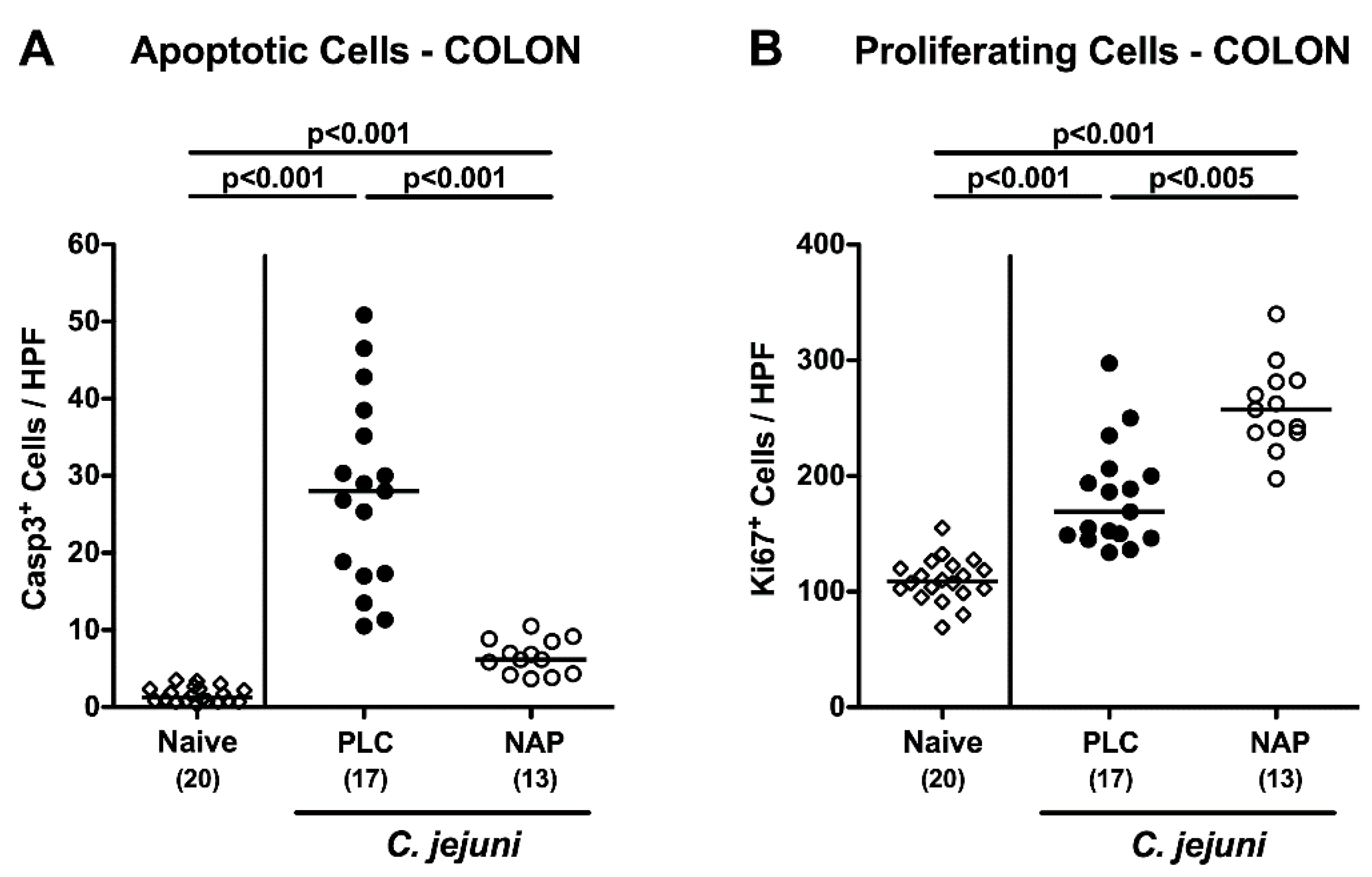
3.3. Colonic Epithelial Apoptotic and Proliferative Cell Responses following NAP Treatment of C. jejuni Infected Secondary Abiotic IL10−/− Mice
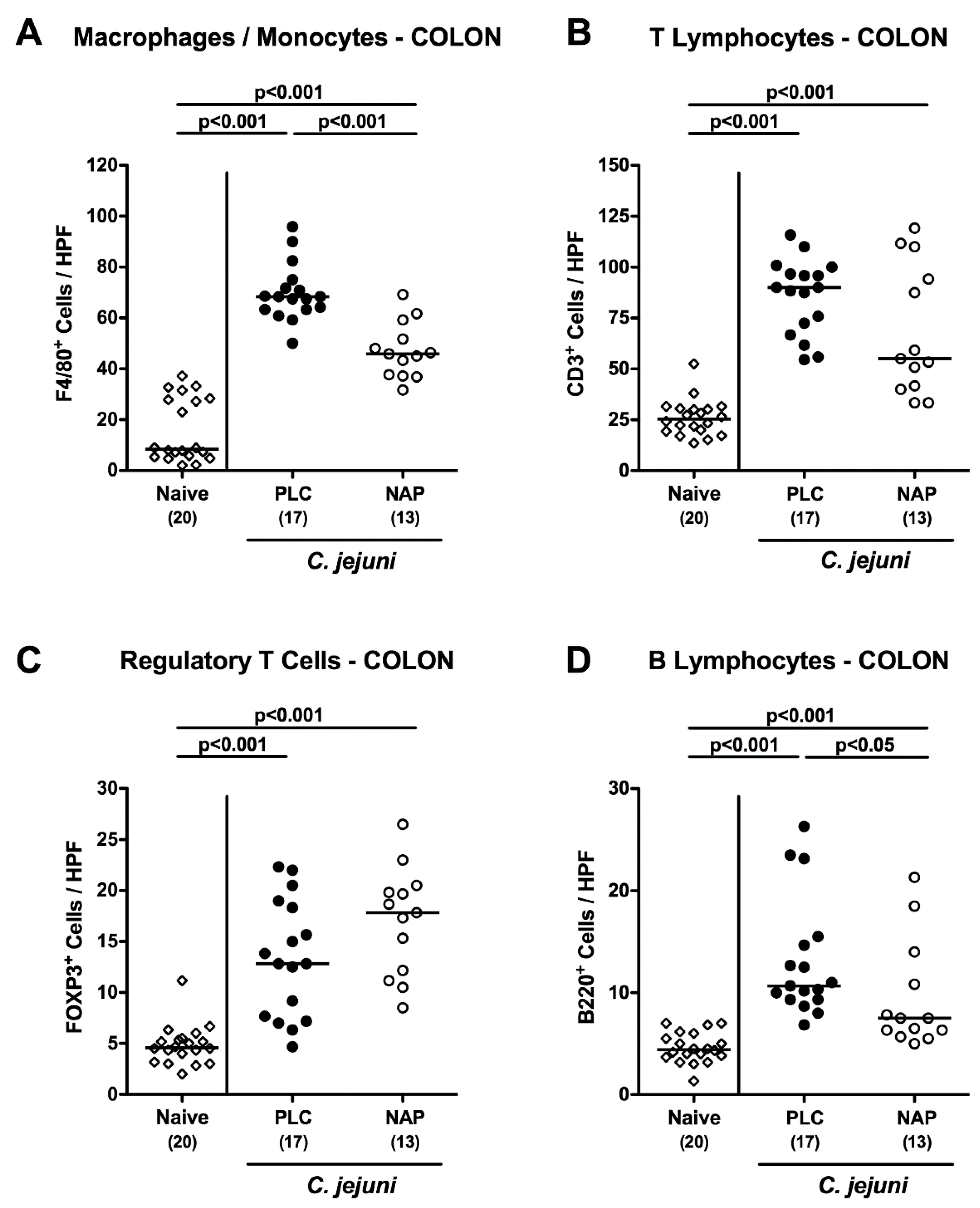
3.4. Colonic Innate and Adaptive Immune Cell Responses following NAP Treatment of C. jejuni Infected Secondary Abiotic IL10-/- Mice
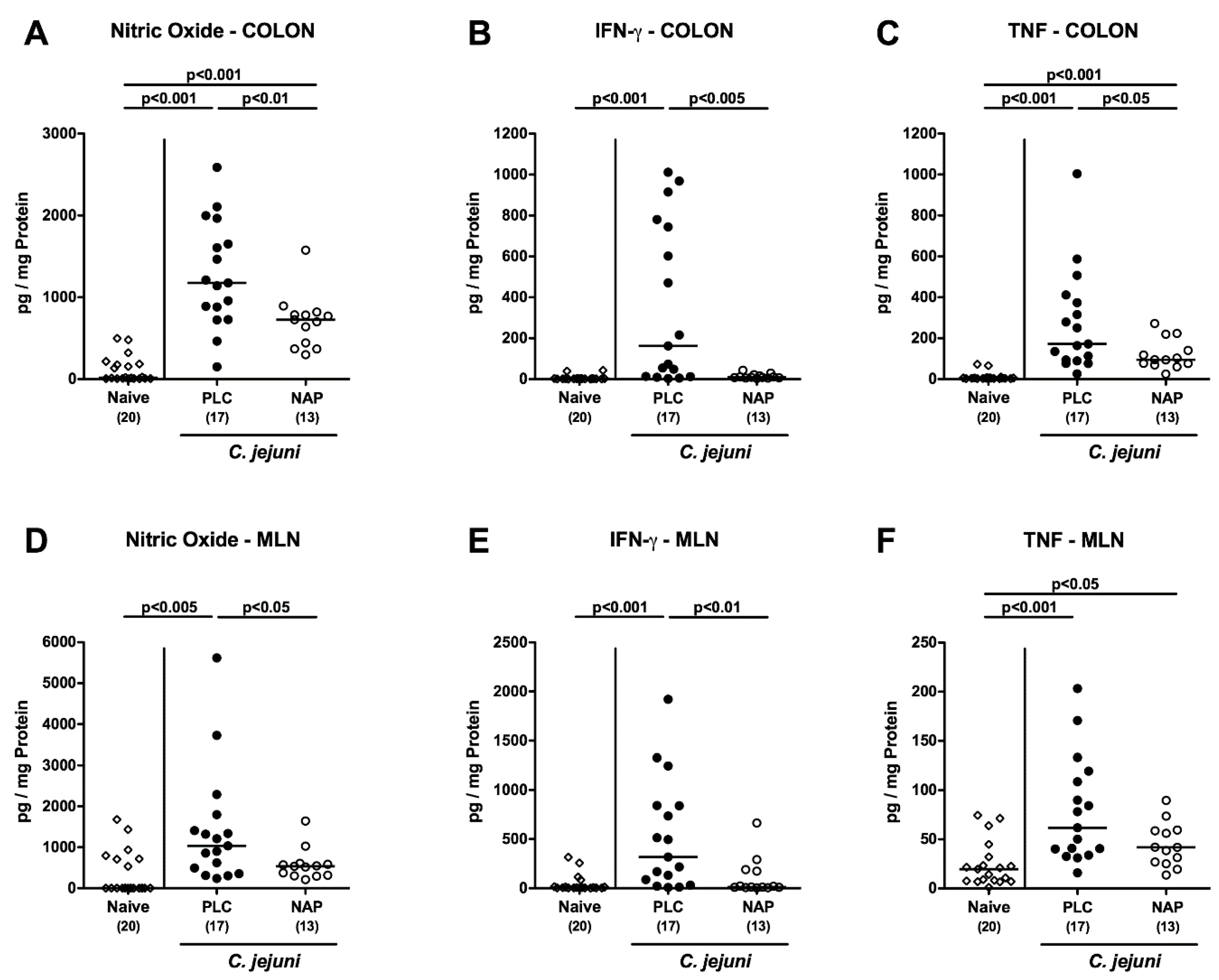
3.5. Intestinal Pro-Inflammatory Mediator Secretion following NAP Treatment of C. jejuni Infected Secondary Abiotic IL10−/− Mice
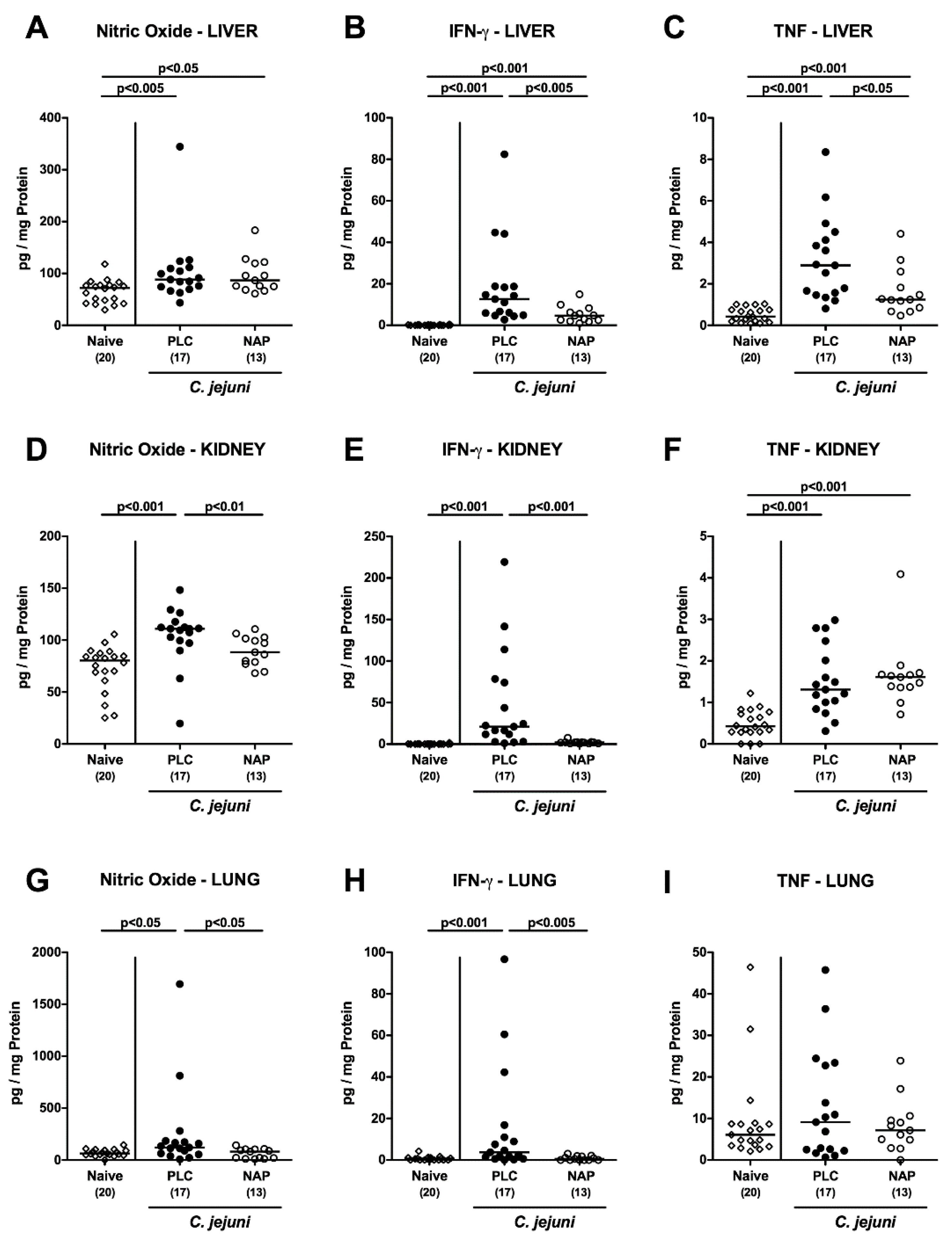
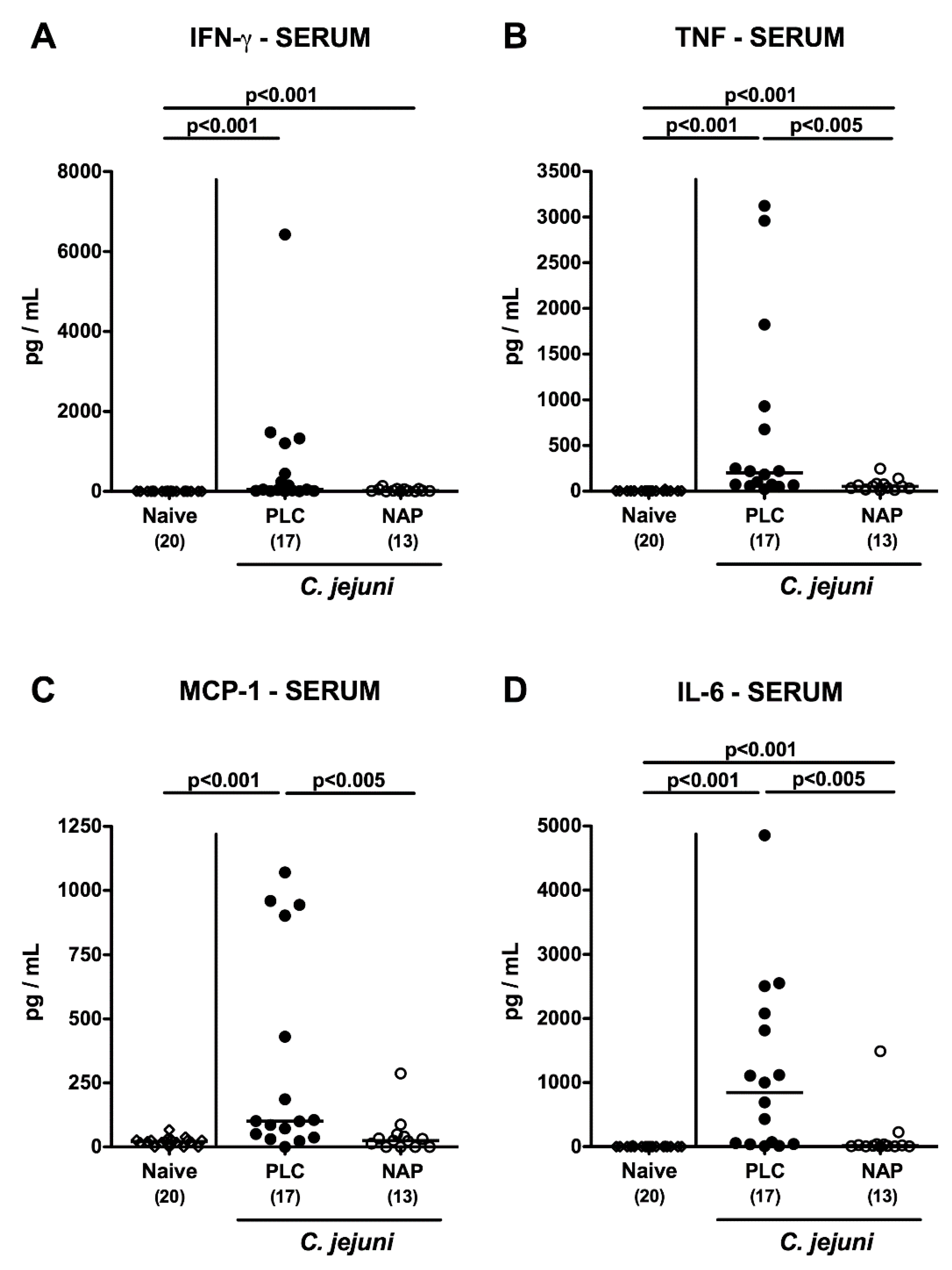
3.6. Extra-Intestinal including Systemic Pro-Inflammatory Mediator Secretion following NAP Treatment of C. jejuni Infected Secondary Abiotic IL10−/− Mice
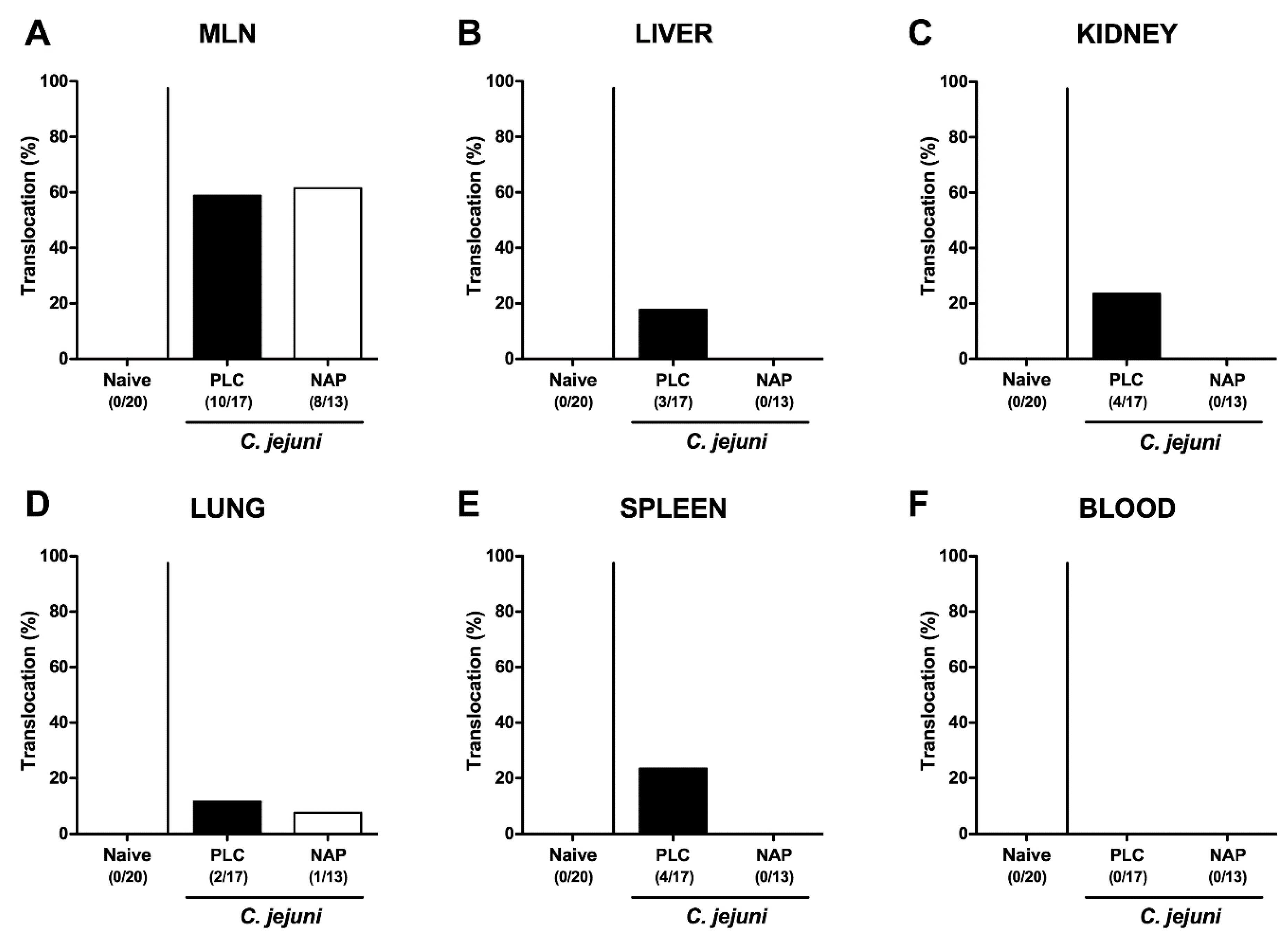
3.7. Bacterial Translocation following NAP Treatment of C. jejuni Infected Secondary Abiotic IL10-/- Mice
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ADNP | Activity-dependent neuroprotective protein |
| BCG | Bacillus Calmette–Guerin |
| Casp3 | Caspase-3 |
| CBA | Cytometric Bead Array |
| CFU | Colony forming units |
| DSS | Dextran sulfate sodium |
| ECDC | European Centre for Disease Prevention and Control |
| EU | European Union |
| GBS | Guillain–Barré syndrome |
| HPF | High power fields |
| IFN-γ | Interferon-γ |
| IL | Interleukin |
| LOS | Lipooligosaccharide |
| LPS | Lipopolysaccharide |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MLN | Mesenteric lymph nodes |
| PBS | Phosphate buffered saline |
| p.i | Post-infection |
| PLC | Placebo |
| SPF | Specific pathogen free |
| TGF-β | Transforming growth factor β |
| TLR-4 | Toll-like receptor 4 |
| TNF | Tumor necrosis factor |
| Trpm4 | Transient receptor potential cation channel subfamily M member 4. |
References
- Kaakoush, N.O.; Castano-Rodriguez, N.; Mitchell, H.M.; Man, S.M. Global Epidemiology of Campylobacter Infection. Clin. Microbiol. Rev. 2015, 28, 687–720. [Google Scholar] [CrossRef] [PubMed]
- Igwaran, A.; Okoh, A.I. Human campylobacteriosis: A public health concern of global importance. Heliyon 2019, 5, e02814. [Google Scholar] [CrossRef]
- European Food Safety Authority and European Centre for Disease Prevention and Control (EFSA and ECDC). The European Union One Health 2018 Zoonoses Report. EFSA J. 2019, 17, e05926. [Google Scholar]
- Bronowski, C.; James, C.E.; Winstanley, C. Role of environmental survival in transmission of Campylobacter jejuni. FEMS Microbiol. Lett. 2014, 356, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.A.; Mehra, R.K.; Carrington, S.D.; Hickey, R.M. The food glycome: A source of protection against pathogen colonization in the gastrointestinal tract. Int. J. Food Microbiol. 2010, 142, 1–13. [Google Scholar] [CrossRef]
- Hermans, D.; Pasmans, F.; Messens, W.; Martel, A.; Van Immerseel, F.; Rasschaert, G.; Heyndrickx, M.; Van Deun, K.; Haesebrouck, F. Poultry as a host for the zoonotic pathogen Campylobacter Jejuni. Vector Borne Zoonotic Dis. 2012, 12, 89–98. [Google Scholar] [CrossRef]
- van Spreeuwel, J.P.; Duursma, G.C.; Meijer, C.J.; Bax, R.; Rosekrans, P.C.; Lindeman, J. Campylobacter colitis: Histological immunohistochemical and ultrastructural findings. Gut 1985, 26, 945–951. [Google Scholar] [CrossRef]
- Walker, R.I.; Caldwell, M.B.; Lee, E.C.; Guerry, P.; Trust, T.J.; Ruiz-Palacios, G.M. Pathophysiology of Campylobacter enteritis. Microbiol. Rev. 1986, 50, 81–94. [Google Scholar] [CrossRef]
- Janssen, R.; Krogfelt, K.A.; Cawthraw, S.A.; van Pelt, W.; Wagenaar, J.A.; Owen, R.J. Host–pathogen interactions in Campylobacter infections: The host perspective. Clin. Microbiol. Rev. 2008, 21, 505–518. [Google Scholar] [CrossRef]
- Kist, M.; Bereswill, S. Campylobacter jejuni. Contrib. Microbiol. 2001, 8, 150–165. [Google Scholar]
- Backert, S.; Tegtmeyer, N.; Cróinín, T.Ó.; Boehm, M.; Heimesaat, M.M. Human campylobacteriosis. In Campylobacter; Klein, G., Ed.; Academic Press: Cambridge, MA, USA, 2017; pp. 1–25. [Google Scholar] [CrossRef]
- Bereswill, S.; Fischer, A.; Plickert, R.; Haag, L.M.; Otto, B.; Kuhl, A.A.; Dasti, J.I.; Zautner, A.E.; Munoz, M.; Loddenkemper, C.; et al. Novel murine infection models provide deep insights into the “menage a trois” of Campylobacter jejuni, microbiota and host innate immunity. PLoS ONE 2011, 6, e20953. [Google Scholar] [CrossRef]
- Masanta, W.O.; Heimesaat, M.M.; Bereswill, S.; Tareen, A.M.; Lugert, R.; Groß, U.; Zautner, A.E. Modification of intestinal microbiota and its consequences for innate immune response in the pathogenesis of campylobacteriosis. Clin. Dev. Immunol. 2013, 2013, 526860. [Google Scholar] [CrossRef] [PubMed]
- Fiebiger, U.; Bereswill, S.; Heimesaat, M.M. Dissecting the Interplay Between Intestinal Microbiota and Host Immunity in Health and Disease: Lessons Learned from Germfree and Gnotobiotic Animal Models. Eur. J. Microbiol. Immunol. 2016, 6, 253–271. [Google Scholar] [CrossRef] [PubMed]
- Taveira da Silva, A.M.; Kaulbach, H.C.; Chuidian, F.S.; Lambert, D.R.; Suffredini, A.F.; Danner, R.L. Brief report: Shock and multiple-organ dysfunction after self-administration of Salmonella endotoxin. N. Engl. J. Med. 1993, 328, 1457–1460. [Google Scholar] [CrossRef]
- Mousavi, S.; Bereswill, S.; Heimesaat, M.M. Novel Clinical Campylobacter jejuni Infection Models Based on Sensitization of Mice to Lipooligosaccharide, a Major Bacterial Factor Triggering Innate Immune Responses in Human Campylobacteriosis. Microorganisms 2020, 8, 482. [Google Scholar] [CrossRef]
- Haag, L.M.; Fischer, A.; Otto, B.; Plickert, R.; Kuhl, A.A.; Gobel, U.B.; Bereswill, S.; Heimesaat, M.M. Campylobacter jejuni induces acute enterocolitis in gnotobiotic IL-10-/- mice via Toll-like-receptor-2 and -4 signaling. PLoS ONE 2012, 7, e40761. [Google Scholar] [CrossRef]
- Mousavi, S.; Lobo de Sá, F.D.; Schulzke, J.-D.; Bücker, R.; Bereswill, S.; Heimesaat, M.M. Vitamin D in Acute Campylobacteriosis-Results From an Intervention Study Applying a Clinical Campylobacter jejuni Induced Enterocolitis Model. Front. Immunol. 2019, 10, 2094. [Google Scholar] [CrossRef]
- Mousavi, S.; Schmidt, A.-M.; Escher, U.; Kittler, S.; Kehrenberg, C.; Thunhorst, E.; Bereswill, S.; Heimesaat, M.M. Carvacrol ameliorates acute campylobacteriosis in a clinical murine infection model. Gut Pathog. 2020, 12, 2. [Google Scholar] [CrossRef]
- Mousavi, S.; Escher, U.; Thunhorst, E.; Kittler, S.; Kehrenberg, C.; Bereswill, S.; Heimesaat, M.M. Vitamin C alleviates acute enterocolitis in Campylobacter jejuni infected mice. Sci. Rep. 2020, 10, 2921. [Google Scholar] [CrossRef]
- Bassan, M.; Zamostiano, R.; Davidson, A.; Pinhasov, A.; Giladi, E.; Perl, O.; Bassan, H.; Blat, C.; Gibney, G.; Glazner, G.; et al. Complete sequence of a novel protein containing a femtomolar-activity-dependent neuroprotective peptide. J. Neurochem. 1999, 72, 1283–1293. [Google Scholar] [CrossRef]
- Zamostiano, R.; Pinhasov, A.; Gelber, E.; Steingart, R.A.; Seroussi, E.; Giladi, E.; Bassan, M.; Wollman, Y.; Eyre, H.J.; Mulley, J.C.; et al. Cloning and characterization of the human activity-dependent neuroprotective protein. J. Biol. Chem. 2001, 276, 708–714. [Google Scholar] [CrossRef] [PubMed]
- Gozes, I.; Divinski, I. The femtomolar-acting NAP interacts with microtubules: Novel aspects of astrocyte protection. J. Alzheimer’s Dis. JAD 2004, 6, S37–S41. [Google Scholar] [CrossRef] [PubMed]
- Quintana, F.J.; Zaltzman, R.; Fernandez-Montesinos, R.; Herrera, J.L.; Gozes, I.; Cohen, I.R.; Pozo, D. NAP, a peptide derived from the activity-dependent neuroprotective protein, modulates macrophage function. Ann. New York Acad. Sci. 2006, 1070, 500–506. [Google Scholar] [CrossRef] [PubMed]
- Zemlyak, I.; Furman, S.; Brenneman, D.E.; Gozes, I. A novel peptide prevents death in enriched neuronal cultures. Regul. Pept. 2000, 96, 39–43. [Google Scholar] [CrossRef]
- Ashur-Fabian, O.; Segal-Ruder, Y.; Skutelsky, E.; Brenneman, D.E.; Steingart, R.A.; Giladi, E.; Gozes, I. The neuroprotective peptide NAP inhibits the aggregation of the beta-amyloid peptide. Peptides 2003, 24, 1413–1423. [Google Scholar] [CrossRef] [PubMed]
- Beni-Adani, L.; Gozes, I.; Cohen, Y.; Assaf, Y.; Steingart, R.A.; Brenneman, D.E.; Eizenberg, O.; Trembolver, V.; Shohami, E. A peptide derived from activity-dependent neuroprotective protein (ADNP) ameliorates injury response in closed head injury in mice. J. Pharmacol. Exp. Ther. 2001, 296, 57–63. [Google Scholar]
- Rotstein, M.; Bassan, H.; Kariv, N.; Speiser, Z.; Harel, S.; Gozes, I. NAP enhances neurodevelopment of newborn apolipoprotein E-deficient mice subjected to hypoxia. J. Pharmacol. Exp. Ther. 2006, 319, 332–339. [Google Scholar] [CrossRef]
- Greggio, S.; de Paula, S.; de Oliveira, I.M.; Trindade, C.; Rosa, R.M.; Henriques, J.A.; DaCosta, J.C. NAP prevents acute cerebral oxidative stress and protects against long-term brain injury and cognitive impairment in a model of neonatal hypoxia-ischemia. Neurobiol. Dis. 2011, 44, 152–159. [Google Scholar] [CrossRef]
- Sokolowska, P.; Passemard, S.; Mok, A.; Schwendimann, L.; Gozes, I.; Gressens, P. Neuroprotective effects of NAP against excitotoxic brain damage in the newborn mice: Implications for cerebral palsy. Neuroscience 2011, 173, 156–168. [Google Scholar] [CrossRef]
- Divinski, I.; Mittelman, L.; Gozes, I. A femtomolar acting octapeptide interacts with tubulin and protects astrocytes against zinc intoxication. J. Biol. Chem. 2004, 279, 28531–28538. [Google Scholar] [CrossRef]
- Oz, S.; Kapitansky, O.; Ivashco-Pachima, Y.; Malishkevich, A.; Giladi, E.; Skalka, N.; Rosin-Arbesfeld, R.; Mittelman, L.; Segev, O.; Hirsch, J.A.; et al. The NAP motif of activity-dependent neuroprotective protein (ADNP) regulates dendritic spines through microtubule end binding proteins. Mol. Psychiatry 2014, 19, 1115–1124. [Google Scholar] [CrossRef] [PubMed]
- Ivashko-Pachima, Y.; Sayas, C.L.; Malishkevich, A.; Gozes, I. ADNP/NAP dramatically increase microtubule end-binding protein-Tau interaction: A novel avenue for protection against tauopathy. Mol. Psychiatry 2017, 22, 1335–1344. [Google Scholar] [CrossRef] [PubMed]
- Idan-Feldman, A.; Ostritsky, R.; Gozes, I. Tau and caspase 3 as targets for neuroprotection. Int. J. Alzheimer’s Dis. 2012, 2012, 493670. [Google Scholar] [CrossRef] [PubMed]
- Braitch, M.; Kawabe, K.; Nyirenda, M.; Gilles, L.J.; Robins, R.A.; Gran, B.; Murphy, S.; Showe, L.; Constantinescu, C.S. Expression of activity-dependent neuroprotective protein in the immune system: Possible functions and relevance to multiple sclerosis. Neuroimmunomodulation 2010, 17, 120–125. [Google Scholar] [CrossRef]
- Patel, P.C.; Fisher, K.H.; Yang, E.C.; Deane, C.M.; Harrison, R.E. Proteomic analysis of microtubule-associated proteins during macrophage activation. Mol. Cell. Proteom. MCP 2009, 8, 2500–2514. [Google Scholar] [CrossRef] [PubMed]
- Escher, U.; Giladi, E.; Dunay, I.R.; Bereswill, S.; Gozes, I.; Heimesaat, M.M. Anti-inflammatory Effects of the Octapeptide NAP in Human Microbiota-Associated Mice Suffering from Subacute Ileitis. Eur. J. Microbiol. Immunol. 2018, 8, 34–40. [Google Scholar] [CrossRef]
- Heimesaat, M.M.; Fischer, A.; Kuhl, A.A.; Gobel, U.B.; Gozes, I.; Bereswill, S. Anti-Inflammatory Properties of NAP in Acute Toxoplasma Gondii-Induced Ileitis in Mice. Eur. J. Microbiol. Immunol. 2015, 5, 210–220. [Google Scholar] [CrossRef]
- Heimesaat, M.M.; Giladi, E.; Kuhl, A.A.; Bereswill, S.; Gozes, I. The octapetide NAP alleviates intestinal and extra-intestinal anti-inflammatory sequelae of acute experimental colitis. Peptides 2018, 101, 1–9. [Google Scholar] [CrossRef]
- Heimesaat, M.M.; Bereswill, S.; Fischer, A.; Fuchs, D.; Struck, D.; Niebergall, J.; Jahn, H.K.; Dunay, I.R.; Moter, A.; Gescher, D.M.; et al. Gram-negative bacteria aggravate murine small intestinal Th1-type immunopathology following oral infection with Toxoplasma gondii. J. Immunol. 2006, 177, 8785–8795. [Google Scholar] [CrossRef]
- Dangoor, D.; Biondi, B.; Gobbo, M.; Vachutinski, Y.; Fridkin, M.; Gozes, I.; Rocchi, R. Novel glycosylated VIP analogs: Synthesis, biological activity, and metabolic stability. J. Pept. Sci. 2008, 14, 321–328. [Google Scholar] [CrossRef]
- Heimesaat, M.M.; Alutis, M.; Grundmann, U.; Fischer, A.; Tegtmeyer, N.; Bohm, M.; Kuhl, A.A.; Gobel, U.B.; Backert, S.; Bereswill, S. The role of serine protease HtrA in acute ulcerative enterocolitis and extra-intestinal immune responses during Campylobacter jejuni infection of gnotobiotic IL-10 deficient mice. Front. Cell Infect. Microbiol. 2014, 4, 77. [Google Scholar] [CrossRef]
- Heimesaat, M.M.; Nogai, A.; Bereswill, S.; Plickert, R.; Fischer, A.; Loddenkemper, C.; Steinhoff, U.; Tchaptchet, S.; Thiel, E.; Freudenberg, M.A.; et al. MyD88/TLR9 mediated immunopathology and gut microbiota dynamics in a novel murine model of intestinal graft-versus-host disease. Gut 2010, 59, 1079–1087. [Google Scholar] [CrossRef]
- Bryan, N.S.; Grisham, M.B. Methods to detect nitric oxide and its metabolites in biological samples. Free Radic. Biol. Med. 2007, 43, 645–657. [Google Scholar] [CrossRef] [PubMed]
- Luangtongkum, T.; Jeon, B.; Han, J.; Plummer, P.; Logue, C.M.; Zhang, Q. Antibiotic resistance in Campylobacter: Emergence, transmission and persistence. Future Microbiol. 2009, 4, 189–200. [Google Scholar] [CrossRef] [PubMed]
- Leker, R.R.; Teichner, A.; Grigoriadis, N.; Ovadia, H.; Brenneman, D.E.; Fridkin, M.; Giladi, E.; Romano, J.; Gozes, I. NAP, a femtomolar-acting peptide, protects the brain against ischemic injury by reducing apoptotic death. Stroke 2002, 33, 1085–1092. [Google Scholar] [CrossRef] [PubMed]
- Mollinedo, P.; Kapitansky, O.; Gonzalez-Lamuno, D.; Zaslavsky, A.; Real, P.; Gozes, I.; Gandarillas, A.; Fernandez-Luna, J.L. Cellular and animal models of skin alterations in the autism-related ADNP syndrome. Sci. Rep. 2019, 9, 736. [Google Scholar] [CrossRef]
- Kapitansky, O.; Giladi, E.; Jaljuli, I.; Bereswill, S.; Heimesaat, M.M.; Gozes, I. Microbiota changes associated with ADNP deficiencies: Rapid indicators for NAP (CP201) treatment of the ADNP syndrome and beyond. J. Neural Transm. 2020, 127, 251–263. [Google Scholar] [CrossRef]
- Furman, S.; Steingart, R.A.; Mandel, S.; Hauser, J.M.; Brenneman, D.E.; Gozes, I. Subcellular localization and secretion of activity-dependent neuroprotective protein in astrocytes. Neuron Glia Biol. 2004, 1, 193–199. [Google Scholar] [CrossRef]
- Malishkevich, A.; Marshall, G.A.; Schultz, A.P.; Sperling, R.A.; Aharon-Peretz, J.; Gozes, I. Blood-Borne Activity-Dependent Neuroprotective Protein (ADNP) is Correlated with Premorbid Intelligence, Clinical Stage, and Alzheimer’s Disease Biomarkers. J. Alzheimer’s Dis. JAD 2016, 50, 249–260. [Google Scholar] [CrossRef]
- Yang, M.H.; Yang, Y.H.; Lu, C.Y.; Jong, S.B.; Chen, L.J.; Lin, Y.F.; Wu, S.J.; Chu, P.Y.; Chung, T.W.; Tyan, Y.C. Activity-dependent neuroprotector homeobox protein: A candidate protein identified in serum as diagnostic biomarker for Alzheimer’s disease. J. Proteom. 2012, 75, 3617–3629. [Google Scholar] [CrossRef]
- Qihui, L.; Pei, Z.; Shanshan, L.; Mengyan, T.; Yuanxin, W.; Yuan, T.; Zheng, J.; Dong, L.; Dongmei, Y. NMAAP1 Maintains M1 Phenotype in Macrophages Through Binding to IP3R and Activating Calcium-related Signaling Pathways. Protein Pept. Lett. 2019, 26, 751–757. [Google Scholar] [CrossRef]
- Yan, B.; Xie, S.; Liu, Z.; Ran, J.; Li, Y.; Wang, J.; Yang, Y.; Zhou, J.; Li, D.; Liu, M. HDAC6 deacetylase activity is critical for lipopolysaccharide-induced activation of macrophages. PLoS ONE 2014, 9, e110718. [Google Scholar] [CrossRef] [PubMed]
- Blanco, C.; Morales, D.; Mogollones, I.; Vergara-Jaque, A.; Vargas, C.; Álvarez, A.; Riquelme, D.; Leiva-Salcedo, E.; González, W.; Morales, D. EB1-and EB2-dependent anterograde trafficking of TRPM4 regulates focal adhesion turnover and cell invasion. FASEB J. 2019, 33, 9434–9452. [Google Scholar] [CrossRef] [PubMed]
- Oz, S.; Ivashko-Pachima, Y.; Gozes, I. The ADNP derived peptide, NAP modulates the tubulin pool: Implication for neurotrophic and neuroprotective activities. PLoS ONE 2012, 7, e51458. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Heimesaat, M.M.; Mousavi, S.; Kløve, S.; Genger, C.; Weschka, D.; Giladi, E.; Bereswill, S.; Gozes, I. Immune-modulatory Properties of the Octapeptide NAP in Campylobacter jejuni Infected Mice Suffering from Acute Enterocolitis. Microorganisms 2020, 8, 802. https://doi.org/10.3390/microorganisms8060802
Heimesaat MM, Mousavi S, Kløve S, Genger C, Weschka D, Giladi E, Bereswill S, Gozes I. Immune-modulatory Properties of the Octapeptide NAP in Campylobacter jejuni Infected Mice Suffering from Acute Enterocolitis. Microorganisms. 2020; 8(6):802. https://doi.org/10.3390/microorganisms8060802
Chicago/Turabian StyleHeimesaat, Markus M., Soraya Mousavi, Sigri Kløve, Claudia Genger, Dennis Weschka, Eliezer Giladi, Stefan Bereswill, and Illana Gozes. 2020. "Immune-modulatory Properties of the Octapeptide NAP in Campylobacter jejuni Infected Mice Suffering from Acute Enterocolitis" Microorganisms 8, no. 6: 802. https://doi.org/10.3390/microorganisms8060802
APA StyleHeimesaat, M. M., Mousavi, S., Kløve, S., Genger, C., Weschka, D., Giladi, E., Bereswill, S., & Gozes, I. (2020). Immune-modulatory Properties of the Octapeptide NAP in Campylobacter jejuni Infected Mice Suffering from Acute Enterocolitis. Microorganisms, 8(6), 802. https://doi.org/10.3390/microorganisms8060802






