Stress Response of Mesosutterella multiformis Mediated by Nitrate Reduction
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Growth Conditions
2.2. Determination of Nitrite Concentrations
2.3. Evaluation of Cell Morphology
2.4. Effect of Nitrate on the Composition of Cellular Fatty Acids
2.5. Evaluation of the Ability of Other Sutterellaceae Species to Form Filaments
2.6. Genome Analysis
2.7. Statistical Analyses
3. Results
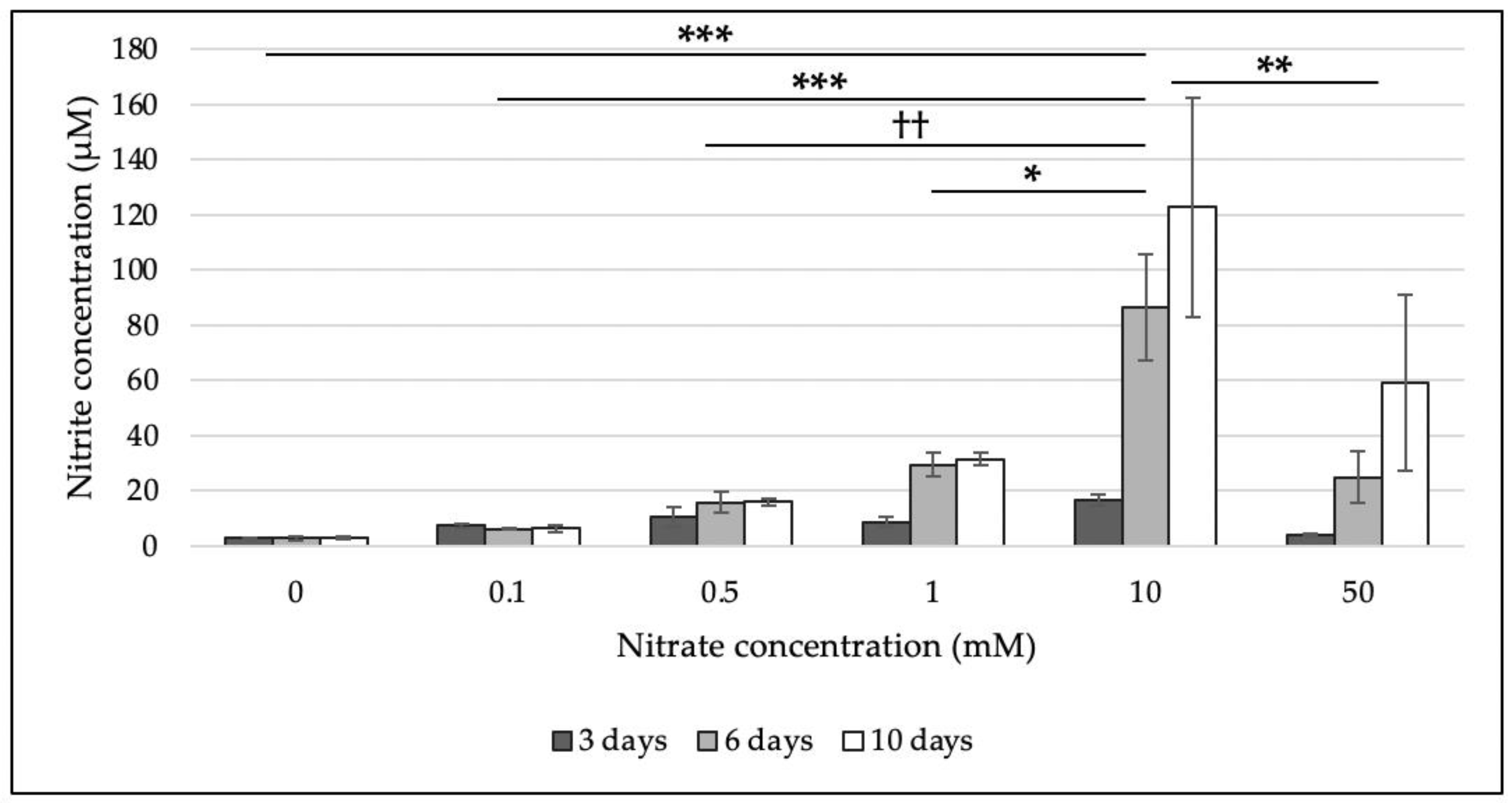
3.1. Effect of Nitrate Concentration on Ability of Nitrate Reduction
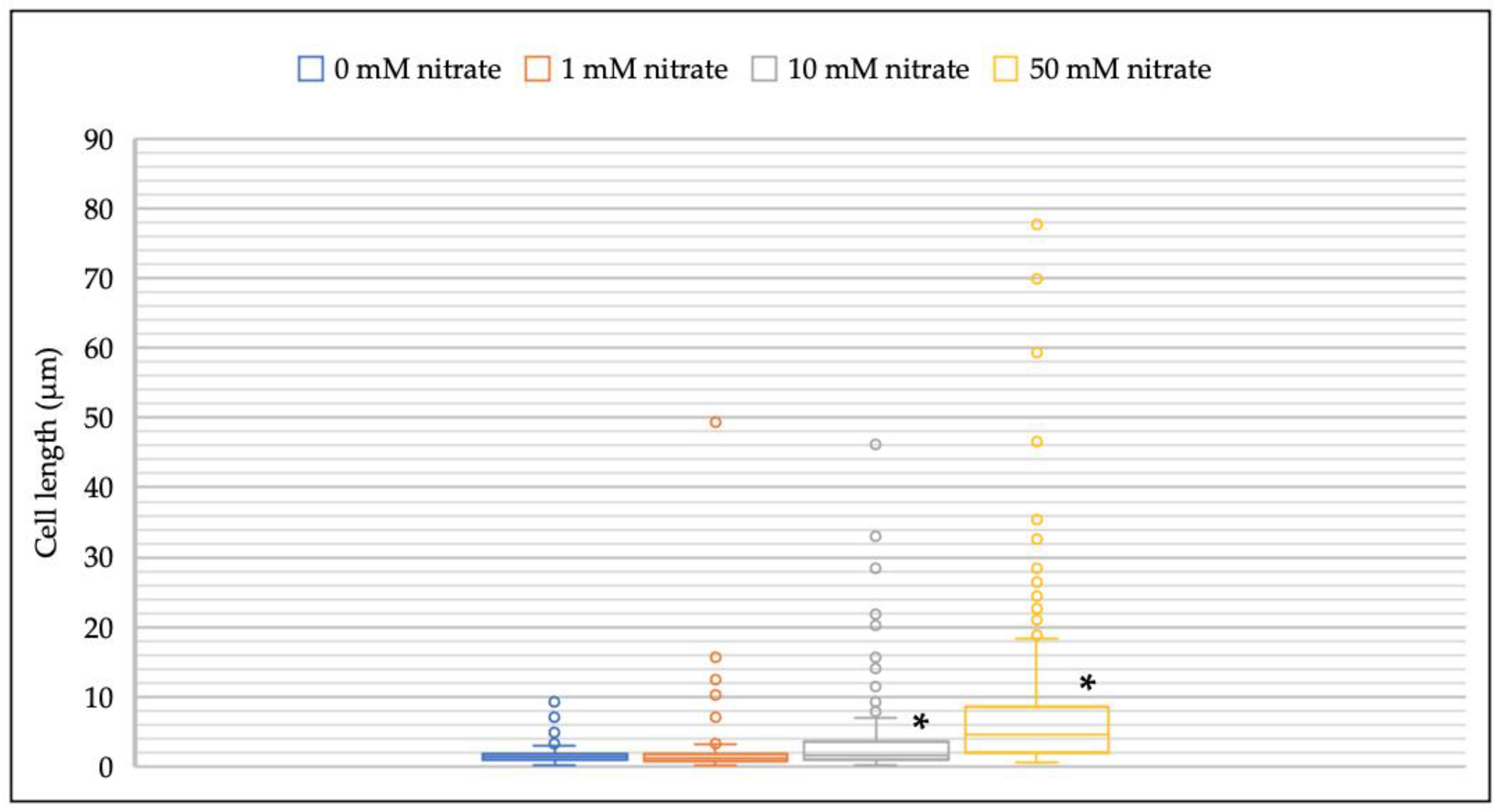
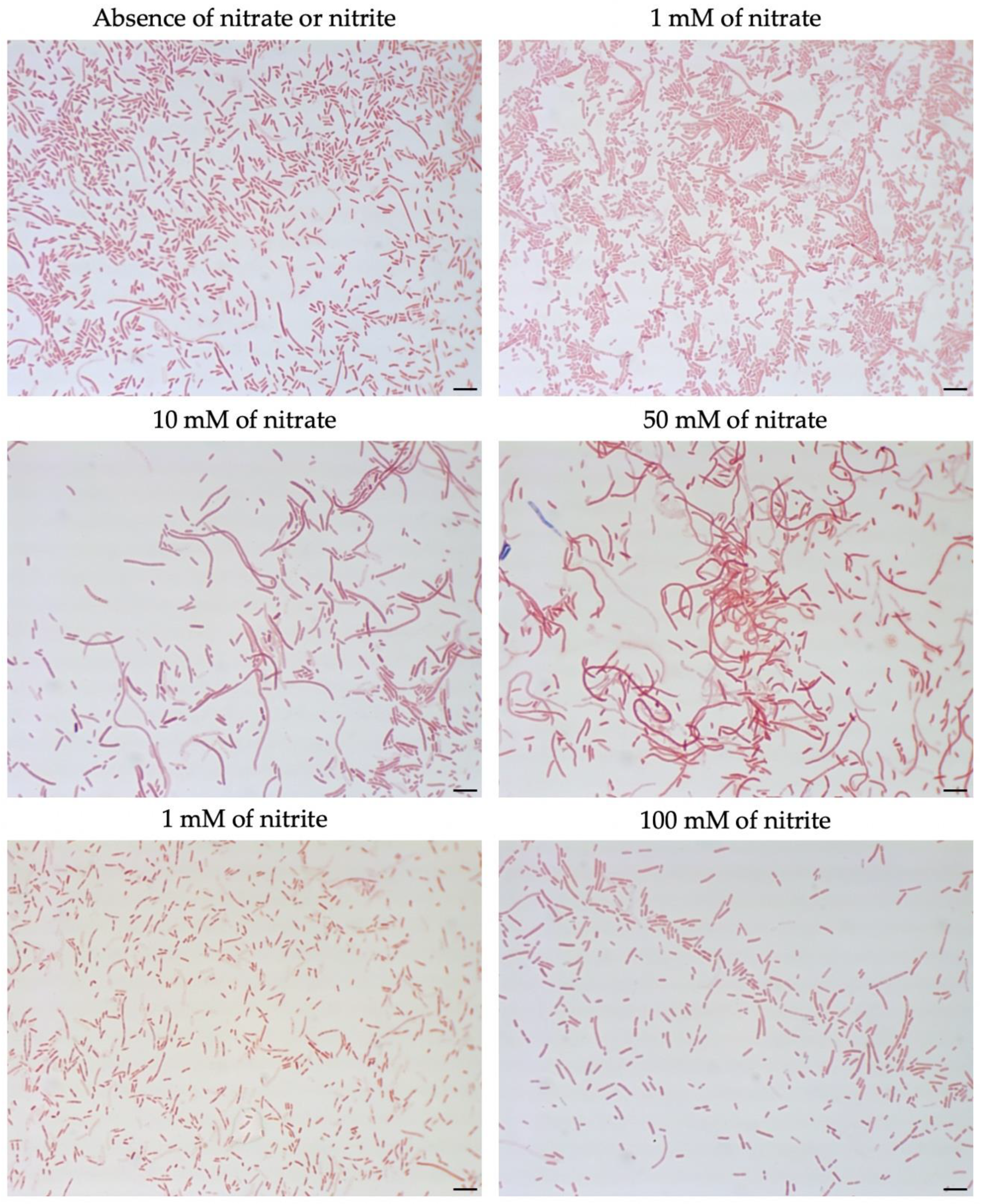
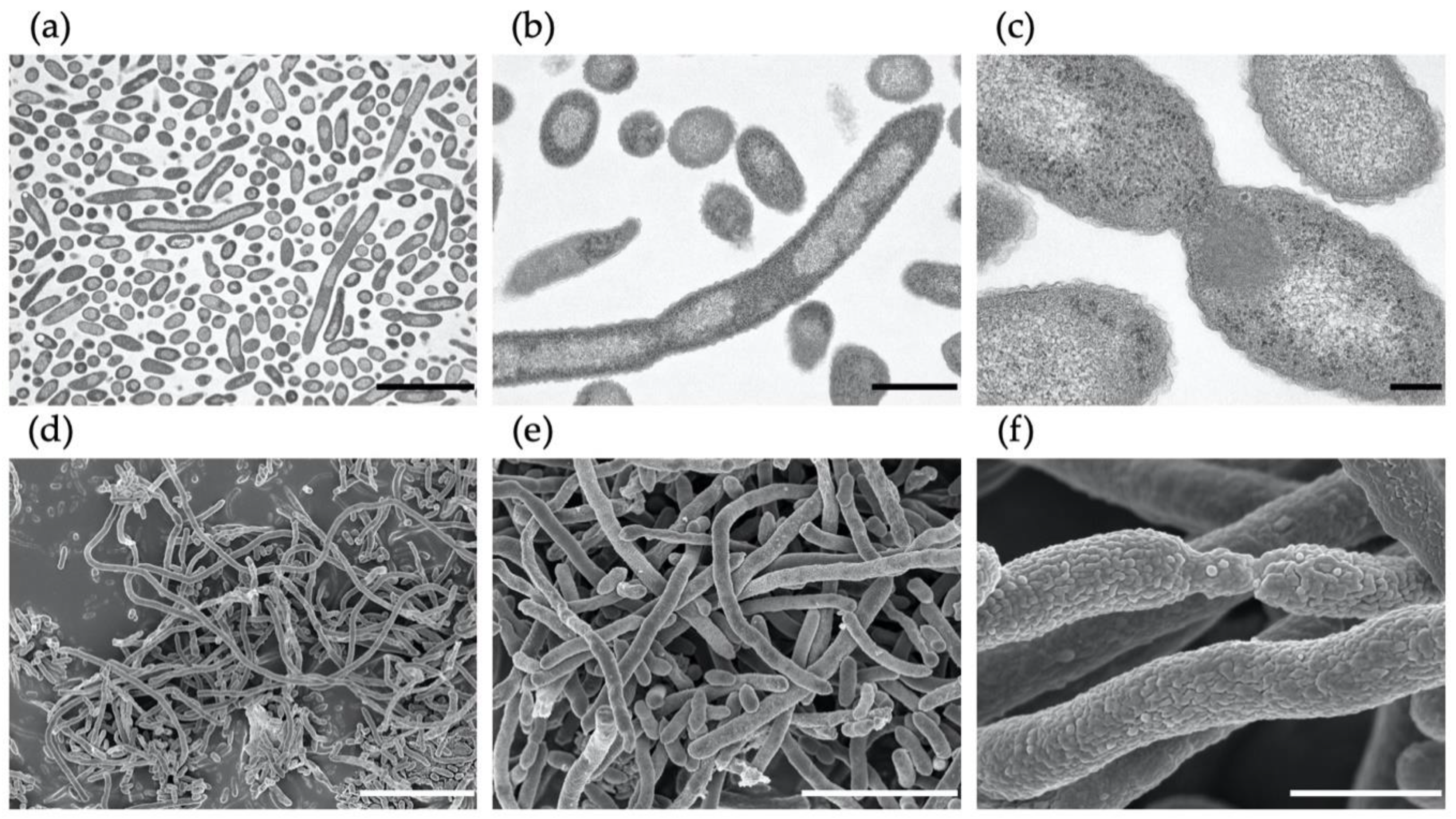
3.2. Effect of Nitrate on Cell Morphology of M. multiformis
3.3. Effect of Nitrate-Induced Filamentation on the Composition of Cellular Fatty Acids
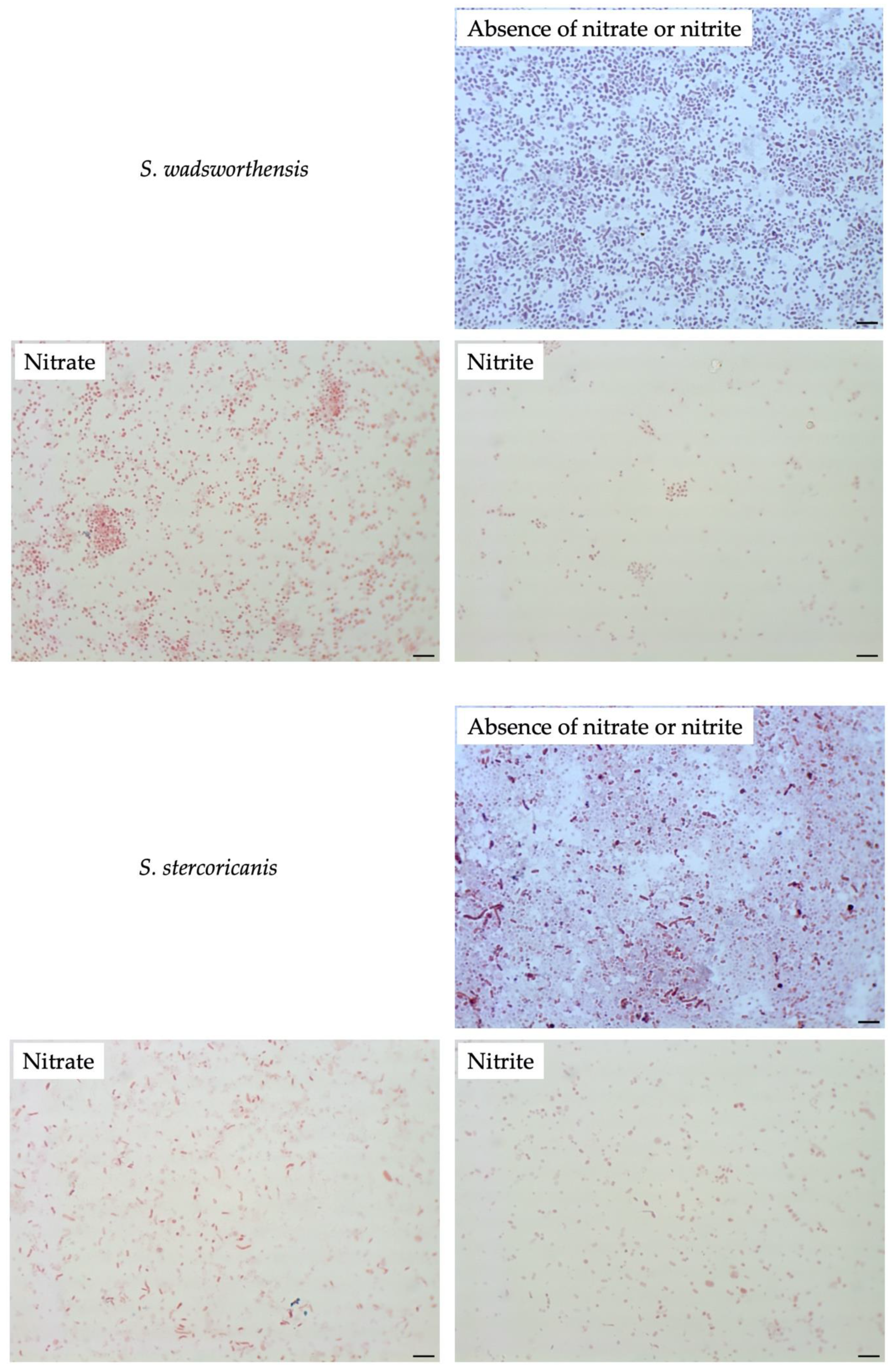
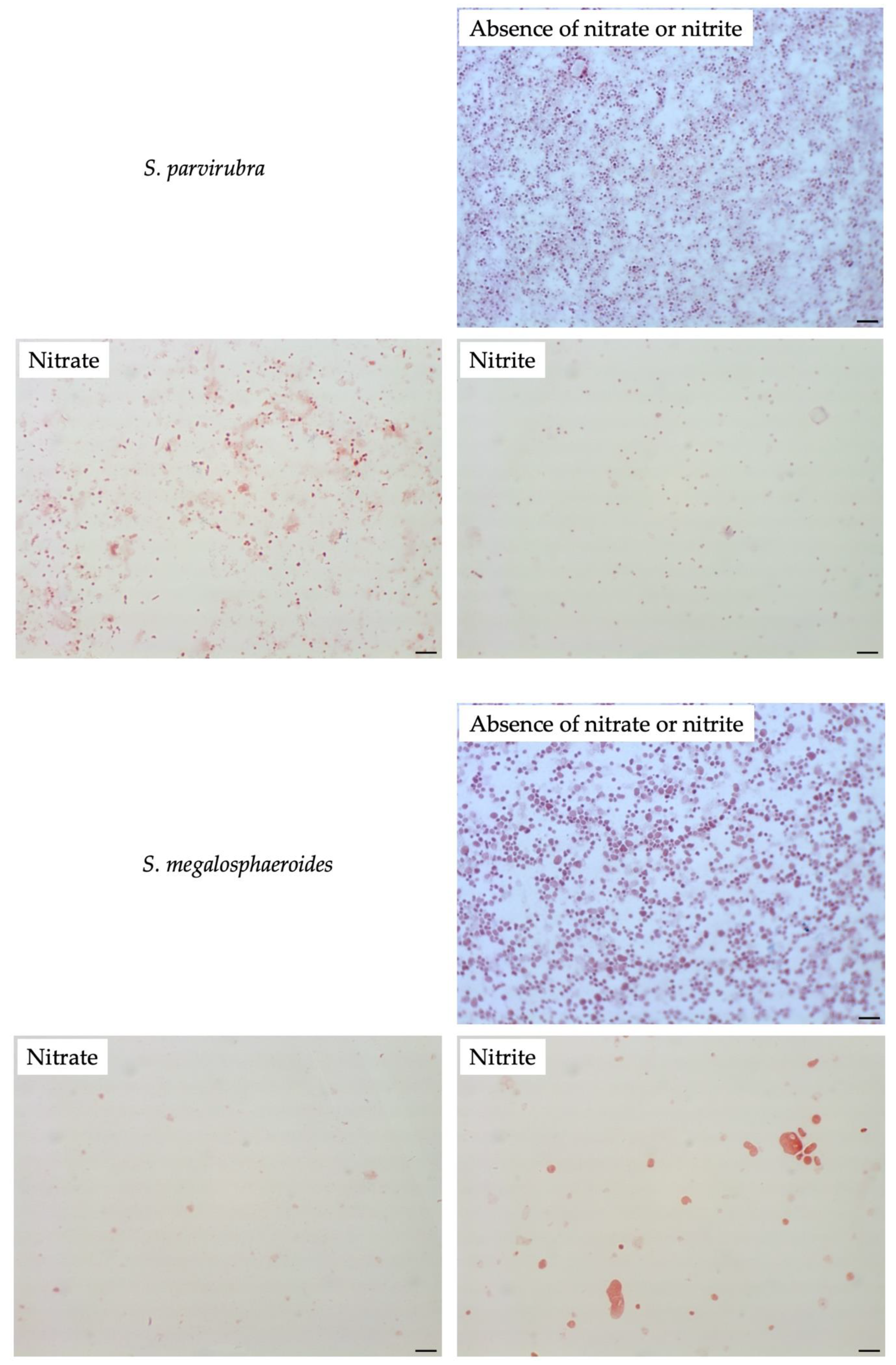
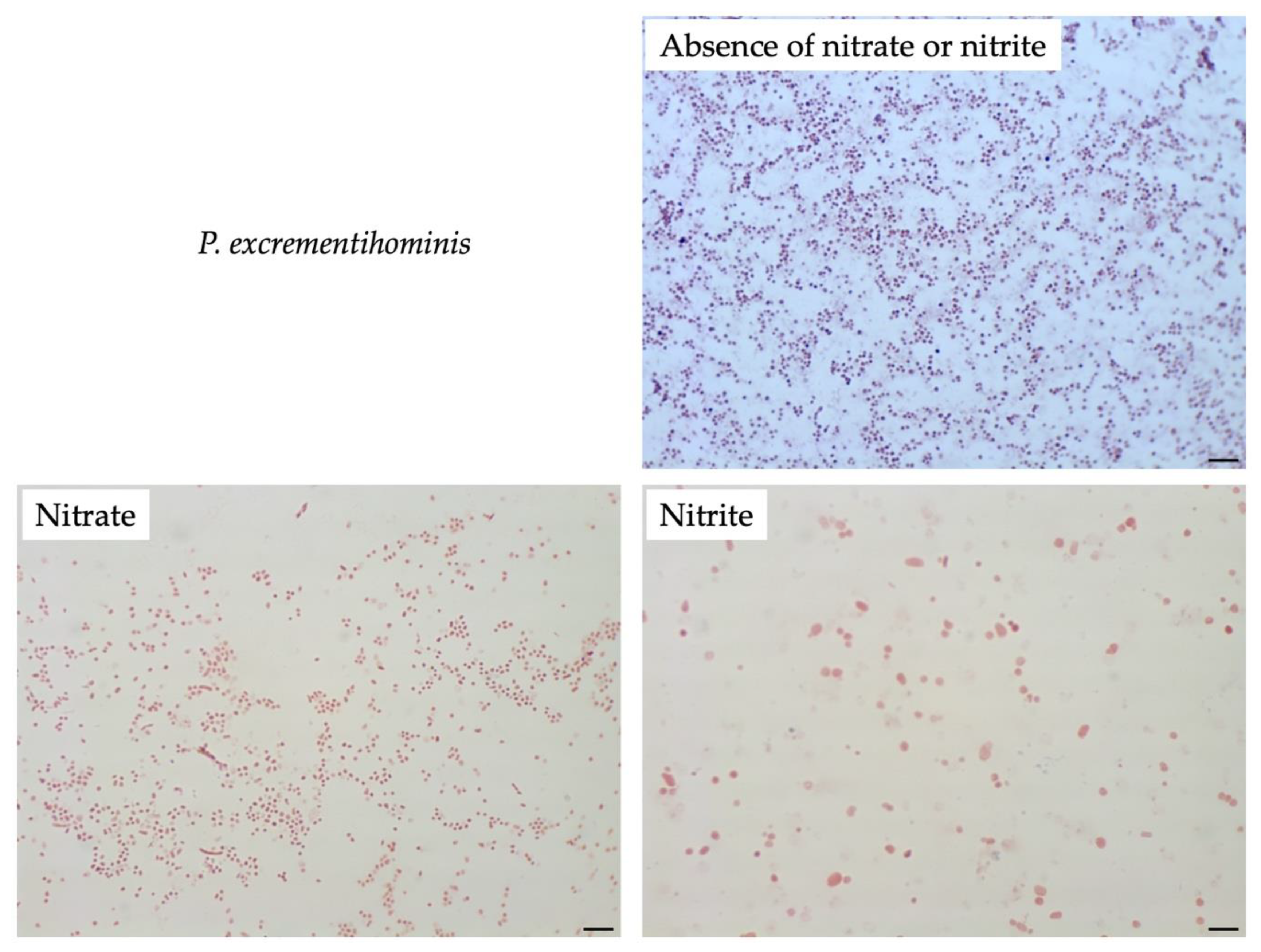
3.4. Effect of Nitrate or Nitrite on Other Sutterellaceae Species
3.5. Differences of Genes in Sutterellaceae Species
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Papadimitriou, K.; Alegría, Á.; Bron, P.A.; de Angelis, M.; Gobbetti, M.; Kleerebezem, M.; Lemos, J.A.; Linares, D.M.; Ross, P.; Stanton, C.; et al. Stress physiology of lactic acid bacteria. Microbiol. Mol. Biol. Rev. 2016, 80, 837–890. [Google Scholar] [CrossRef] [PubMed]
- Bucka-Kolendo, J.; Sokołowska, B. Lactic acid bacteria stress response to preservation processes in the beverage and juice industry. Acta Biochim. Pol. 2017, 64, 459–464. [Google Scholar] [CrossRef] [PubMed]
- Hews, C.L.; Cho, T.; Rowley, G.; Raivio, T.L. Maintaining integrity under stress: Envelope stress response regulation of pathogenesis in Gram-negative bacteria. Front. Cell. Infect. Microbiol. 2019, 9, 313. [Google Scholar] [CrossRef]
- Sakamoto, M.; Ikeyama, N.; Kunihiro, T.; Iino, T.; Yuki, M.; Ohkuma, M. Mesosutterella multiformis gen. nov., sp. nov., a member of the family Sutterellaceae and Sutterella megalosphaeroides sp. nov., isolated from human faeces. Int. J. Syst. Evol. Microbiol. 2018, 68, 3942–3950. [Google Scholar] [CrossRef] [PubMed]
- Williams, B.L.; Hornig, M.; Parekh, T.; Lipkin, W.I. Application of novel PCR-based methods for detection, quantitation, and phylogenetic characterization of Sutterella species in intestinal biopsy samples from children with autism and gastrointestinal disturbances. mBio 2012, 3, e00261-11. [Google Scholar] [CrossRef] [PubMed]
- Hiippala, K.; Kainulainen, V.; Kalliomäki, M.; Arkkila, P.; Satokari, R. Mucosal prevalence and interactions with the epithelium indicate commensalism of Sutterella spp. Front. Microbiol. 2016, 7, 1706. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.J.; Wu, H.; Wu, S.D.; Lu, N.; Wang, Y.T.; Liu, H.N.; Dong, L.; Liu, T.T.; Shen, X.Z. Parasutterella, in association with irritable bowel syndrome and intestinal chronic inflammation. J. Gastroenterol. Hepatol. 2018, 33, 1844–1852. [Google Scholar] [CrossRef] [PubMed]
- Ikeyama, N.; Ohkuma, M.; Sakamoto, M. Draft genome sequence of Mesosutterella multiformis JCM 32464T, a member of the family Sutterellaceae, isolated from human feces. Microbiol. Resour. Announc. 2019, 8, e00478-19. [Google Scholar] [CrossRef]
- Möbius, K.; Arias-Cartin, R.; Breckau, D.; Hännig, A.L.; Riedmann, K.; Biedendieck, R.; Schröder, S.; Becher, D.; Magalon, A.; Moser, J.; et al. Heme biosynthesis is coupled to electron transport chains for energy generation. Proc. Natl. Acad. Sci. USA 2010, 107, 10436–10441. [Google Scholar] [CrossRef]
- Tiso, M.; Schechter, A.N. Nitrate reduction to nitrite, nitric oxide and ammonia by gut bacteria under physiological conditions. PLoS ONE 2015, 10, e0119712. [Google Scholar] [CrossRef]
- Platzen, L.; Koch-Koerfges, A.; Weil, B.; Brocker, M.; Bott, M. Role of flavohaemoprotein Hmp and nitrate reductases NarGHJI of Corynebacterium glutamicum for coping with nitrite and nitrosative stress. FEMS Microbiol. Lett. 2014, 350, 239–248. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sakamoto, M.; Iino, T.; Ohkuma, M. Faecalimonas umbilicata gen. nov., sp. nov., isolated from human faeces, and reclassification of Eubacterium contortum, Eubacterium fissicatena and Clostridium oroticum as Faecalicatena contorta gen. nov., comb. nov., Faecalicatena fissicatena comb. nov. and Faecalicatena orotica comb. nov. Int. J. Syst. Evol. Microbiol. 2017, 67, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- Kuykendall, L.D.; Roy, M.A.; O’Neill, J.J.; Devine, T.E. Fatty acids, antibiotic resistance, and deoxyribonucleic acid homology groups of Bradyrhizobium japonicum. Int. J. Syst. Bacteriol. 1988, 38, 358–361. [Google Scholar] [CrossRef]
- Miller, L.T. Single derivatization method for routine analysis of bacterial whole-cell fatty acid methyl esters, including hydroxy acids. J. Clin. Microbiol. 1982, 16, 584–586. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Furumichi, M.; Tanabe, M.; Sato, Y.; Morishima, K. KEGG: New perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. 2017, 45, D353–D361. [Google Scholar] [CrossRef] [PubMed]
- Aziz, R.K.; Bartels, D.; Best, A.A.; DeJongh, M.; Disz, T.; Edwards, R.A.; Formsma, K.; Gerdes, S.; Glass, E.M.; Kubal, M.; et al. The RAST Server: Rapid annotations using subsystems technology. BMC Genom. 2008, 9, 75. [Google Scholar] [CrossRef]
- Tanizawa, Y.; Fujisawa, T.; Nakamura, Y. DFAST: A flexible prokaryotic genome annotation pipeline for faster genome publication. Bioinformatics 2018, 34, 1037–1039. [Google Scholar] [CrossRef]
- Liu, Y.; Ding, X.M.; Xue, Z.L.; Hu, L.X.; Zhang, N.J.; Wang, Z.; Yang, J.W.; Cheng, Q.; Chen, M.H.; Zhang, Z.Z.; et al. The change of the state of cell membrane can enhance the synthesis of menaquinone in Escherichia coli. World J. Microbiol. Biotechnol. 2017, 33, 52. [Google Scholar] [CrossRef]
- Heinemann, I.U.; Jahn, M.; Jahn, D. The biochemistry of heme biosynthesis. Arch. Biochem. Biophys. 2008, 474, 238–251. [Google Scholar] [CrossRef]
- Wexler, H.M.; Reeves, D.; Summanen, P.H.; Molitoris, E.; McTeague, M.; Duncan, J.; Wilson, K.H.; Finegold, S.M. Sutterella wadsworthensis gen. nov., sp. nov., bile-resistant microaerophilic Campylobacter gracilis-like clinical isolates. Int. J. Syst. Bacteriol. 1996, 46, 252–258. [Google Scholar] [CrossRef]
- Greetham, H.L.; Collins, M.D.; Gibson, G.R.; Giffard, C.; Falsen, E.; Lawson, P.A. Sutterella stercoricanis sp. nov., isolated from canine faeces. Int. J. Syst. Evol. Microbiol. 2004, 54, 1581–1584. [Google Scholar] [CrossRef] [PubMed]
- Sakon, H.; Nagai, F.; Morotomi, M.; Tanaka, R. Sutterella parvirubra sp. nov. and Megamonas funiformis sp. nov., isolated from human faeces. Int. J. Syst. Evol. Microbiol. 2008, 58, 970–975. [Google Scholar] [CrossRef] [PubMed]
- Nagai, F.; Morotomi, M.; Sakon, H.; Tanaka, R. Parasutterella excrementihominis gen. nov., sp. nov., a member of the family Alcaligenaceae isolated from human faeces. Int. J. Syst. Evol. Microbiol. 2009, 59, 1793–1797. [Google Scholar] [CrossRef] [PubMed]
- Morotomi, M.; Nagai, F.; Watanabe, Y. Parasutterella secunda sp. nov., isolated from human faeces and proposal of Sutterellaceae fam. nov. in the order Burkholderiales. Int. J. Syst. Evol. Microbiol. 2011, 61, 637–643. [Google Scholar] [CrossRef]
- Dev Kumar, G.; Macarisin, D.; Micallef, S.A. Salmonella enterica filamentation induced by pelargonic acid is a transient morphotype. Appl. Environ. Microbiol. 2019, 85, e02191-18. [Google Scholar] [CrossRef]
- Yoon, M.Y.; Lee, K.M.; Park, Y.; Yoon, S.S. Contribution of cell elongation to the biofilm formation of Pseudomonas aeruginosa during anaerobic respiration. PLoS ONE 2011, 6, e16105. [Google Scholar] [CrossRef]
- Nishimura, T.; Teramoto, H.; Inui, M.; Yukawa, H. Gene expression profiling of Corynebacterium glutamicum during anaerobic nitrate respiration: Induction of the SOS response for cell survival. J. Bacteriol. 2011, 193, 1327–1333. [Google Scholar] [CrossRef]
- Hoffmann, T.; Troup, B.; Szabo, A.; Hungerer, C.; Jahn, D. The anaerobic life of Bacillus subtilis: Cloning of the genes encoding the respiratory nitrate reductase system. FEMS Microbiol. Lett. 1995, 131, 219–225. [Google Scholar] [CrossRef]
- Mukhopadhya, I.; Hansen, R.; Nicholl, C.E.; Alhaidan, Y.A.; Thomson, J.M.; Berry, S.H.; Pattinson, C.; Stead, D.A.; Russell, R.K.; El-Omar, E.M.; et al. A comprehensive evaluation of colonic mucosal isolates of Sutterella wadsworthensis from inflammatory bowel disease. PLoS ONE 2011, 6, e27076. [Google Scholar] [CrossRef]
- Szalontai, B.; Nishiyama, Y.; Gombos, Z.; Murata, N. Membrane dynamics as seen by fourier transform infrared spectroscopy in a cyanobacterium, Synechocystis PCC 6803. The effects of lipid unsaturation and the protein-to-lipid ratio. Biochim. Biophys. Acta 2000, 1509, 409–419. [Google Scholar] [CrossRef]
- Sasarman, A.; Purvis, P.; Portelance, V. Role of menaquinone in nitrate respiration in Staphylococcus aureus. J. Bacteriol. 1974, 117, 911–913. [Google Scholar] [CrossRef] [PubMed]
- Haddaji, N.; Mahdhi, A.K.; Krifi, B.; Ismail, M.B.; Bakhrouf, A. Change in cell surface properties of Lactobacillus casei under heat shock treatment. FEMS Microbiol. Lett. 2015, 362, fnv047. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, A.; Shah, N.P. Effect of salt stress on morphology and membrane composition of Lactobacillus acidophilus, Lactobacillus casei, and Bifidobacterium bifidum, and their adhesion to human intestinal epithelial-like Caco-2 cells. J. Dairy Sci. 2016, 99, 2594–2605. [Google Scholar] [CrossRef] [PubMed]
- Archer, D.L. Evidence that ingested nitrate and nitrite are beneficial to health. J. Food Prot. 2002, 65, 872–875. [Google Scholar] [CrossRef]
- Shaw, M.K. Formation of filaments and synthesis of macromolecules at temperatures below the minimum for growth of Escherichia coli. J. Bacteriol. 1968, 95, 221–230. [Google Scholar] [CrossRef]
- Sajbidor, J. Effect of some environmental factors on the content and composition of microbial membrane lipids. Crit. Rev. Biotechnol. 1997, 17, 87–103. [Google Scholar] [CrossRef]
- Crompton, M.J.; Dunstan, R.H.; Macdonald, M.M.; Gottfries, J.; von Eiff, C.; Roberts, T.K. Small changes in environmental parameters lead to alterations in antibiotic resistance, cell morphology and membrane fatty acid composition in Staphylococcus lugdunensis. PLoS ONE 2014, 9, e92296. [Google Scholar] [CrossRef]
- Wang, L.; Christophersen, C.T.; Sorich, M.J.; Gerber, J.P.; Angley, M.T.; Conlon, M.A. Increased abundance of Sutterella spp. and Ruminococcus torques in feces of children with autism spectrum disorder. Mol. Autism 2013, 4, 42. [Google Scholar] [CrossRef]

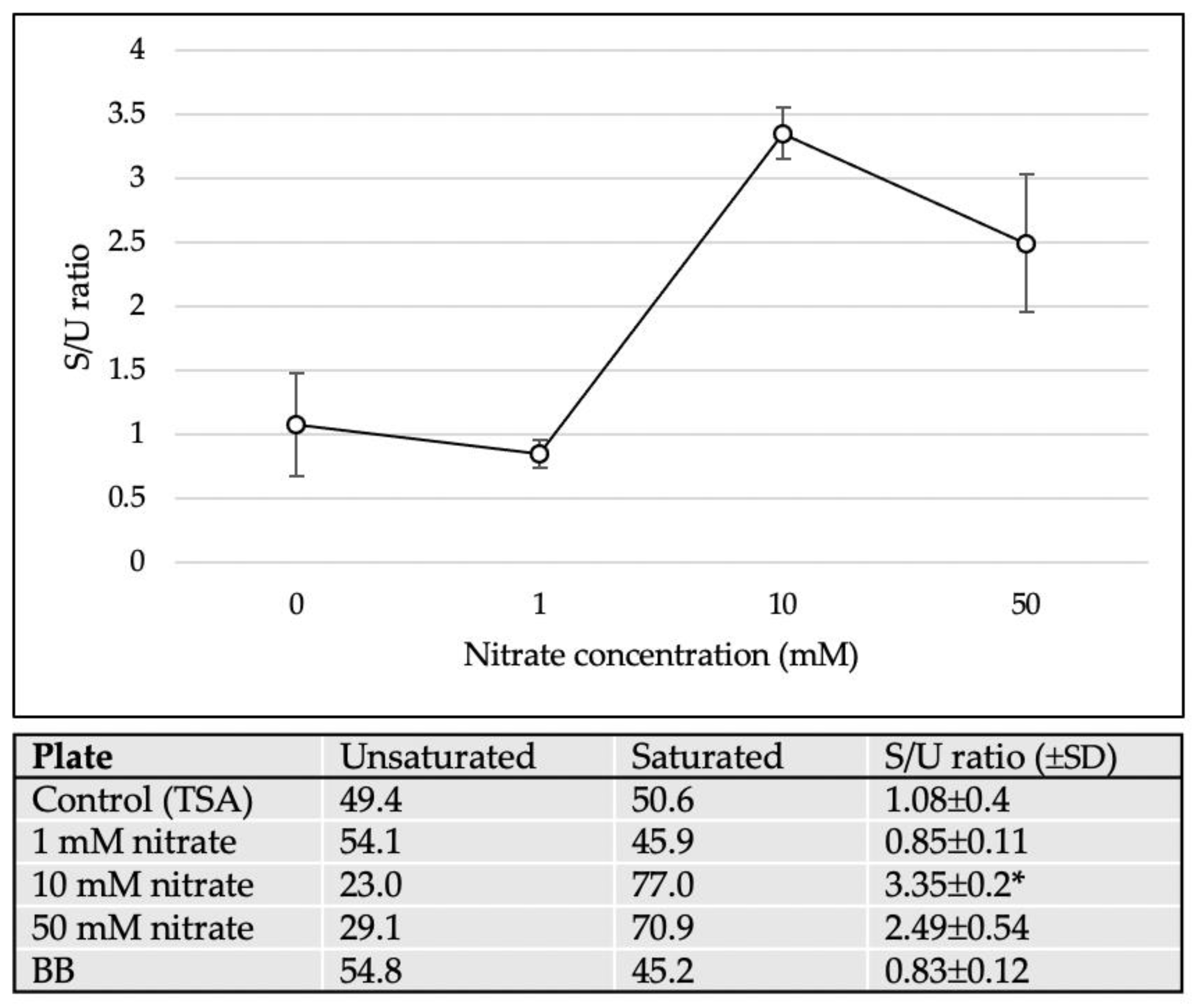
| Fatty Acids | 0 mM Nitrate | 1 mM Nitrate | 10 mM Nitrate | 50 mM Nitrate |
|---|---|---|---|---|
| Saturated: | ||||
| C9:0 | 1.4 | - | - | 3.2 |
| C12:0 | 6.0 | 3.1 | 9.8 ± 8.1 | 3.9 ± 1.7 |
| C14:0 | 2.3 | 3.1 | 23.0 ± 2.3 * | 10.7 ± 2.5 * |
| C16:0 | 32.8 ± 5.1 | 39.8 ± 7.2 | 50.8 ± 4.2 * | 48.5 ± 1.3 * |
| C18:0 | 9.4 ± 0.5 | 6.1 | -* | 5.8 ± 2.1 |
| C16:0 ALDE | 2.1 | - | - | - |
| C16:0 DMA | 3.8 | - | - | - |
| Summed Feature 5 * (C15:0 DMA and/or unknown 17.834) | - | - | - | 2.9 |
| Unsaturated: | ||||
| C16:1ω7c | 5.3 ± 0.6 | 6.6 | 11.6 ± 0.8 | 11.0 ± 0.4 |
| Summed Feature 10 * (C18:1c11/t9/t6 and/or unknown 17.834) | 44.1 ± 9.0 | 49.7 ± 2.8 | 11.5 ± 0.7 * | 18.1 ± 3.9 * |
| Isolation Source | Nitrate Reductase | Nitrite Reductase | hemG | Major Quinones | Cell Mutation | References | ||
|---|---|---|---|---|---|---|---|---|
| Nitrate | Nitrite | |||||||
| Mesosutterella multiformis | Human feces | + | + | + | MK-6, MMK-6 | + | – | [4] |
| Sutterella megalosphaeroides | Human feces | – | – | + | MK-5, MMK-5 | – | + | |
| Sutterella wadsworthensis | Gastrointestinal tract infection | – | + | + | MK-5, MMK-5 | – | – | [20] |
| Sutterella stercoricanis | Dog feces | NA | NA | NA | MK-5, MMK-5 | – | – | [21] |
| Sutterella parvirubra | Human feces | – | – | + | MMK-5 | – | – | [22] |
| Parasutterella excrementihominis | Human feces | + | – | + | MK-6, MMK-6 | – | + | [23] |
| Parasutterella secunda | Human feces | NA | NA | NA | MK-5, MMK-5 | – | NA | [24] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ikeyama, N.; Ohkuma, M.; Sakamoto, M. Stress Response of Mesosutterella multiformis Mediated by Nitrate Reduction. Microorganisms 2020, 8, 2003. https://doi.org/10.3390/microorganisms8122003
Ikeyama N, Ohkuma M, Sakamoto M. Stress Response of Mesosutterella multiformis Mediated by Nitrate Reduction. Microorganisms. 2020; 8(12):2003. https://doi.org/10.3390/microorganisms8122003
Chicago/Turabian StyleIkeyama, Nao, Moriya Ohkuma, and Mitsuo Sakamoto. 2020. "Stress Response of Mesosutterella multiformis Mediated by Nitrate Reduction" Microorganisms 8, no. 12: 2003. https://doi.org/10.3390/microorganisms8122003
APA StyleIkeyama, N., Ohkuma, M., & Sakamoto, M. (2020). Stress Response of Mesosutterella multiformis Mediated by Nitrate Reduction. Microorganisms, 8(12), 2003. https://doi.org/10.3390/microorganisms8122003






