Phenotypic Adaptation of Pseudomonas aeruginosa in the Presence of Siderophore-Antibiotic Conjugates during Epithelial Cell Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Bacterial Strains, Plasmids, and Growth Conditions
2.3. Iron Scavenging from PVD-Fe
2.4. Growth Assays in Iron-Restricted Conditions
2.5. Iron Uptake
2.6. Infection Assay
2.7. Quantitative Real-Time PCR on Bacteria Grown in the Presence of TCVL6
2.8. Quantitative Real-Time PCR on Bacteria Infecting Epithelial Cells
2.9. Label-Free Proteomic Analysis on Bacteria Infecting Epithelial Cells
3. Results
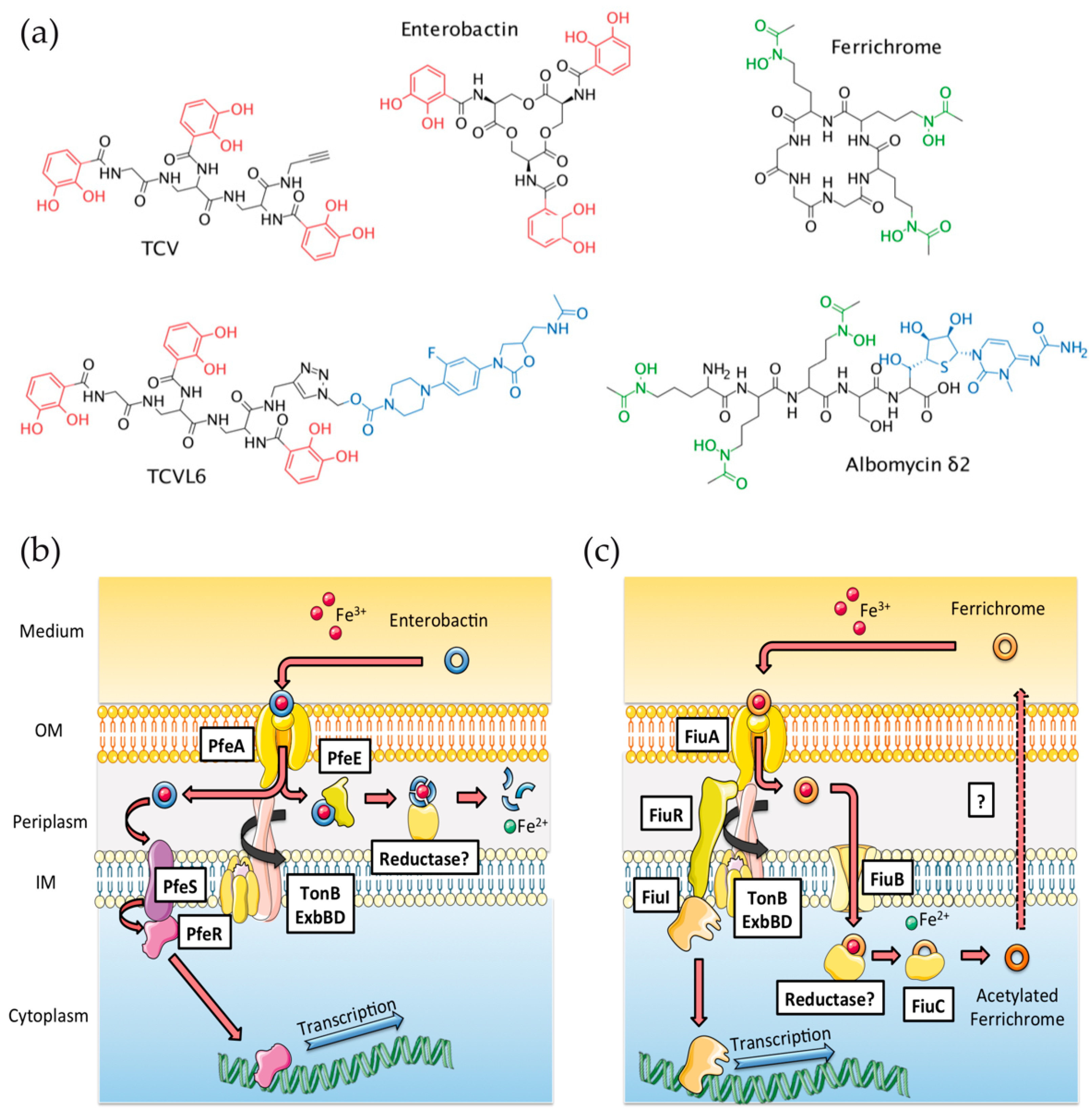
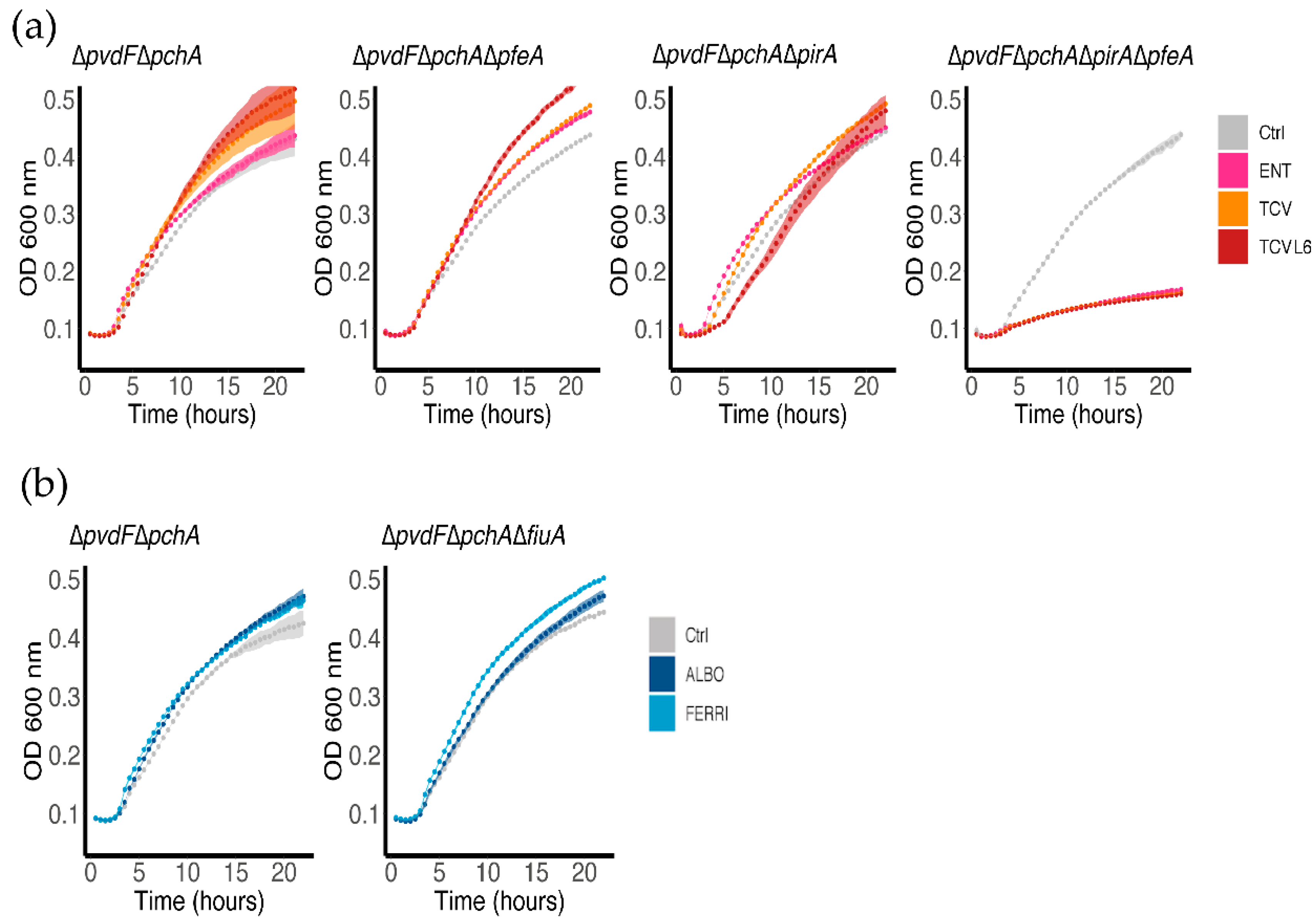
3.1. Ability of ALBO and TCVL6 to Compete for Iron with PVD
3.2. ALBO and TCVL6 Both Transport Iron into P. aeruginosa Cells
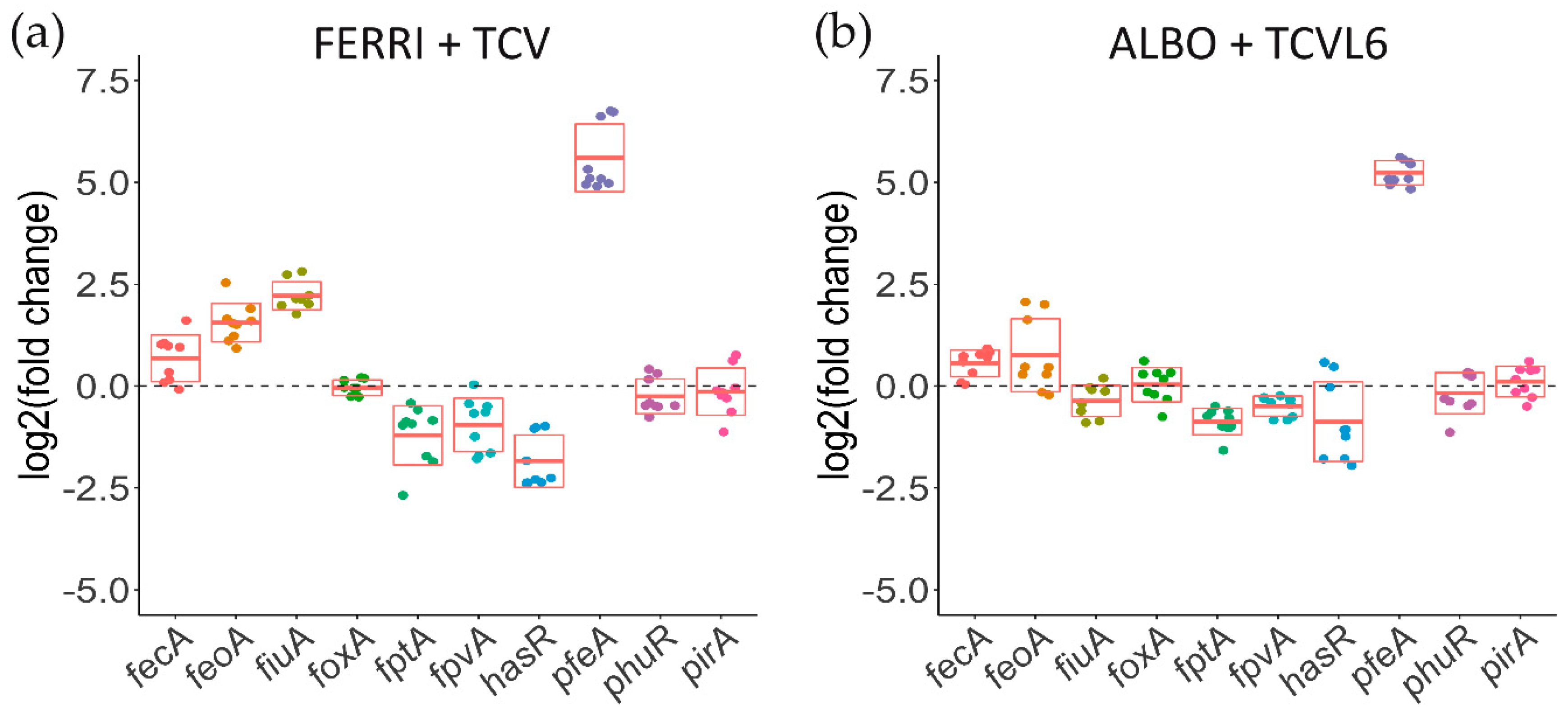
3.3. The Presence of Siderophore–Antibiotic Conjugates Induces the Transcription and Expression of their Corresponding OMTs in P. aeruginosa Cells Infecting Epithelial Cells
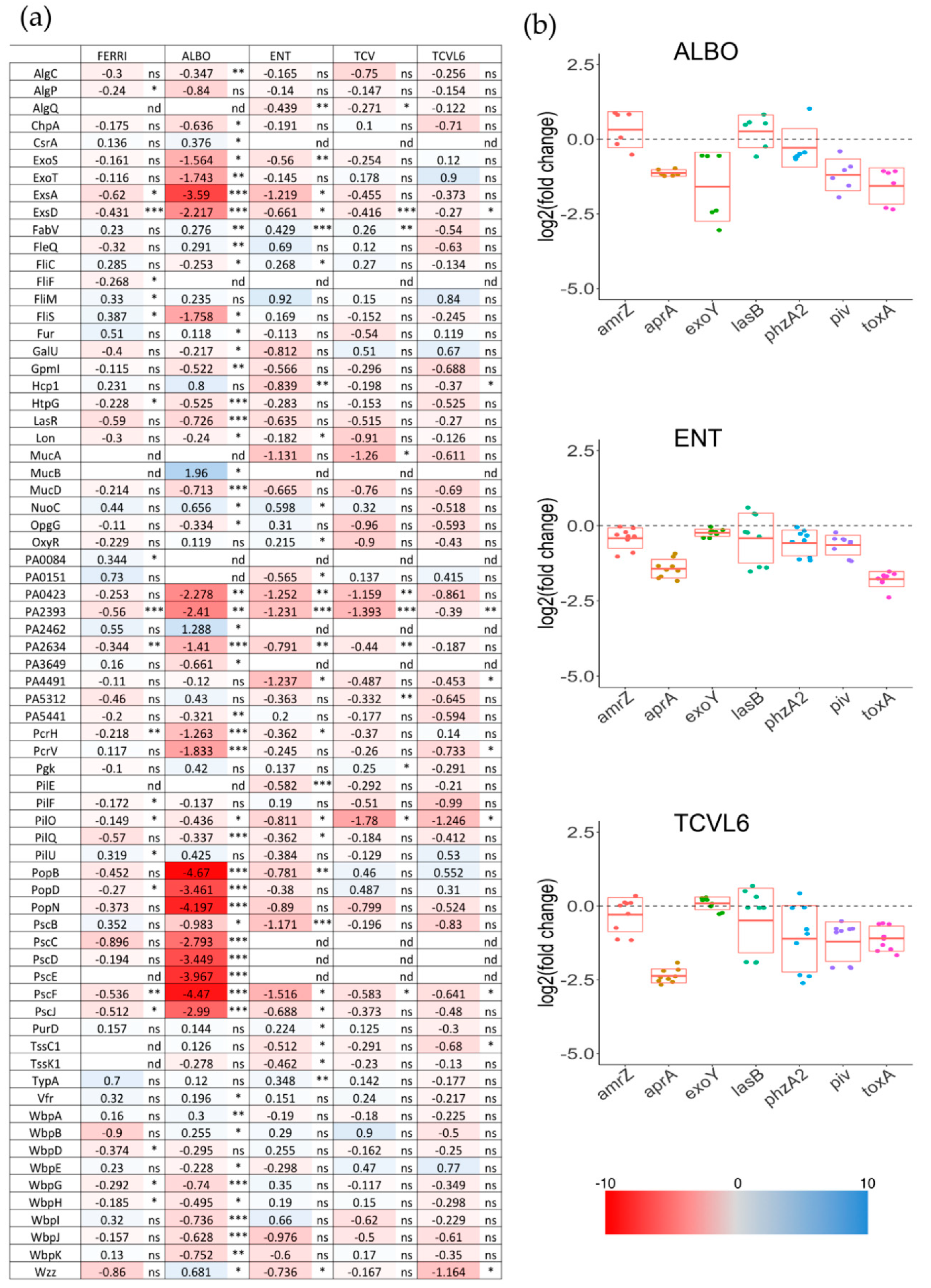
3.4. Siderophore–Antibiotic Conjugates also Affect the Expression of Virulence Factors
3.5. A Tris-Catechol Conjugate More Efficiently Induces the Transcription of its OMT than a Hydroxamate Conjugate in P. aeruginosa Cells Infecting Epithelial Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tillotson, G.S. Trojan Horse Antibiotics—A Novel Way to Circumvent Gram-Negative Bacterial Resistance? Infect. Dis. 2016, 9, 45–52. [Google Scholar] [CrossRef]
- Hider, R.C.; Kong, X. Chemistry and biology of siderophores. Nat. Prod. Rep. 2011, 27, 637–657. [Google Scholar] [CrossRef]
- Schalk, I.J.; Mislin, G.L.A.; Brillet, K. Structure, function and binding selectivity and stereoselectivity of siderophore-iron outer membrane transporters. Curr. Top. Membr. 2012, 69, 37–66. [Google Scholar] [CrossRef] [PubMed]
- Mislin, G.L.A.; Schalk, I.J. Siderophore-dependent iron uptake systems as gates for antibiotic Trojan horse strategies against Pseudomonas aeruginosa. Metallomics 2014, 6, 408–420. [Google Scholar] [CrossRef]
- Górska, A.; Sloderbach, A.; Marszałł, M.P. Siderophore-drug complexes: Potential medicinal applications of the “Trojan horse” strategy. Trends Pharmacol. Sci. 2014, 35, 442–449. [Google Scholar] [CrossRef]
- Zhanel, G.G.; Golden, A.R.; Zelenitsky, S.; Wiebe, K.; Lawrence, C.K.; Adam, H.J.; Idowu, T.; Domalaon, R.; Schweizer, F.; Zhanel, M.A.; et al. Cefiderocol: A Siderophore Cephalosporin with Activity Against Carbapenem-Resistant and Multidrug-Resistant Gram-Negative Bacilli. Drugs 2019, 79, 271–289. [Google Scholar] [CrossRef]
- Gause, G.F. Recent studies on albomycin, a new antibiotic. Br. Med. J. 1955, 2, 1177–1179. [Google Scholar] [CrossRef]
- Benz, G.; Schroeder, T.; Kurz, J.; Wuensche, C.; Karl, W.; Steffens, G.; Pfitzner, J.; Schmidt, D. Konstitution der Desferriform der Albomycine δ1, δ2, ε. Angew. Chem. 1982, 21, 1322–1335. [Google Scholar]
- Sackmann, W.; Reusser, P.; Neipp, L.; Kradolfer, F.; Gross, F. Ferrimycin A, a new iron-containing antibiotic. Antibiot. Chemother. 1962, 12, 34–45. [Google Scholar]
- Selzer, G.B. Antibiotics: Isolation, separation, and purification. J. Pharm. Sci. 1980, 69, 754. [Google Scholar] [CrossRef]
- Vertesy, L.; Aretz, W.; Fehlhaber, H.-W.; Kogler, H. Salmycins A-D, antibiotics from Streptomyces violaceus DSM 8286 having a siderophore-aminoglycoside structure. Helv. Chim. Acta 1995, 78, 46–60. [Google Scholar]
- Cornelis, P.; Dingemans, J. Pseudomonas aeruginosa adapts its iron uptake strategies in function of the type of infections. Front. Cell. Infect. Microbiol. 2013, 3, 75. [Google Scholar] [CrossRef] [PubMed]
- Poole, K.; Young, L.; Neshat, S. Enterobactin-mediated iron transport in Pseudomonas aeruginosa. J. Bacteriol. 1990, 172, 6991–6996. [Google Scholar] [PubMed]
- Hannauer, M.; Barda, Y.; Mislin, G.L.; Shanzer, A.; Schalk, I.J. The ferrichrome uptake pathway in Pseudomonas aeruginosa involves an iron release mechanism with acylation of the siderophore and a recycling of the modified desferrichrome. J. Bacteriol. 2010, 192, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Barber, M.F.; Elde, N.C. Buried Treasure: Evolutionary Perspectives on Microbial Iron Piracy. Trends Genet. 2015, 31, 627–636. [Google Scholar] [CrossRef] [PubMed]
- Perraud, Q.; Cantero, P.; Roche, B.; Gasser, V.; Normant, V.P.; Kuhn, L.; Hammann, P.; Mislin, G.L.A.; Ehret-Sabatier, L.; Schalk, I.J. Phenotypic Adaption of Pseudomonas aeruginosa by Hacking Siderophores Produced by Other Microorganisms. Mol. Cell. Proteom. 2020, 19, 589–607. [Google Scholar] [CrossRef]
- Schalk, I.J.; Guillon, L. Fate of ferrisiderophores after import across bacterial outer membranes: Different iron release strategies are observed in the cytoplasm or periplasm depending on the siderophore pathways. Amino Acids 2013, 44, 1267–1277. [Google Scholar] [CrossRef] [PubMed]
- Dean, C.R.; Neshat, S.; Poole, K. PfeR, an enterobactin-responsive activator of ferric enterobactin receptor gene expression in Pseudomonas aeruginosa. J. Bacteriol. 1996, 178, 5361–5369. [Google Scholar]
- Visca, P.; Leoni, L.; Wilson, M.J.; Lamont, I.L. Iron transport and regulation, cell signalling and genomics: Lessons from Escherichia coli and Pseudomonas. Mol. Microbiol. 2002, 45, 1177–1190. [Google Scholar]
- Michel, L.; Gonzalez, N.; Jagdeep, S.; Nguyen-Ngoc, T.; Reimmann, C. PchR-box recognition by the AraC-type regulator PchR of Pseudomonas aeruginosa requires the siderophore pyochelin as an effector. Mol. Microbiol. 2005, 58, 495–509. [Google Scholar]
- Llamas, M.A.; Sparrius, M.; Kloet, R.; Jimenez, C.R.; Vandenbroucke-Grauls, C.; Bitter, W. The heterologous siderophores ferrioxamine B and ferrichrome activate signaling pathways in Pseudomonas aeruginosa. J. Bacteriol. 2006, 188, 1882–1891. [Google Scholar] [PubMed]
- Llamas, M.A.; Imperi, F.; Visca, P.; Lamont, I.L. Cell-surface signaling in Pseudomonas: Stress responses, iron transport, and pathogenicity. FEMS Microbiol. Rev. 2014, 38, 569–597. [Google Scholar] [CrossRef] [PubMed]
- Gasser, V.; Baco, E.; Cunrath, O.; August, P.S.; Perraud, Q.; Zill, N.; Schleberger, C.; Schmidt, A.; Paulen, A.; Bumann, D.; et al. Catechol siderophores repress the pyochelin pathway and activate the enterobactin pathway in Pseudomonas aeruginosa: An opportunity for siderophore-antibiotic conjugates development. Environ. Microbiol. 2016, 18, 819–832. [Google Scholar] [CrossRef] [PubMed]
- Braun, V.; Pramanik, A.; Gwinner, T.; Koberle, M.; Bohn, E. Sideromycins: Tools and antibiotics. Biometals Int. J. Role Metal Ions Biol. Biochem. Med. 2009, 22, 3–13. [Google Scholar] [CrossRef]
- Wencewicz, T.A.; Mollmann, U.; Long, T.E.; Miller, M.J. Is drug release necessary for antimicrobial activity of siderophore-drug conjugates? Syntheses and biological studies of the naturally occurring salmycin “Trojan Horse” antibiotics and synthetic desferridanoxamine-antibiotic conjugates. Biometals Int. J. Role Metal Ions Biol. Biochem. Med. 2009, 22, 633–648. [Google Scholar] [CrossRef]
- Perraud, Q.; Moynié, L.; Gasser, V.; Munier, M.; Godet, J.; Hoegy, F.; Mély, Y.; Mislin, G.L.A.; Naismith, J.H.; Schalk, I.J. A key role for the periplasmic PfeE esterase in iron acquisition via the siderophore enterobactin in Pseudomonas aeruginosa. ACS Chem. Biol. 2018, 13, 2603–2614. [Google Scholar] [CrossRef]
- Normant, V.; Josts, I.; Kuhn, L.; Perraud, Q.; Fritsch, S.; Hammann, P.; Mislin, G.L.A.; Tidow, H.; Schalk, I.J. Nocardamine-Dependent Iron Uptake in Pseudomonas aeruginosa: Exclusive Involvement of the FoxA Outer Membrane Transporter. ACS Chem. Biol. 2020. [Google Scholar] [CrossRef]
- Demange, P.; Wendenbaum, S.; Linget, C.; Mertz, C.; Cung, M.T.; Dell, A.; Abdallah, M.A. Bacterial siderophores: Structure and NMR assigment of pyoverdins PaA, siderophores of Pseudomonas aeruginosa ATCC 15692. Biol. Metals 1990, 3, 155–170. [Google Scholar]
- Baco, E.; Hoegy, F.; Schalk, I.J.; Mislin, G.L. Diphenyl-benzo[1,3]dioxole-4-carboxylic acid pentafluorophenyl ester: A convenient catechol precursor in the synthesis of siderophore vectors suitable for antibiotic Trojan horse strategies. Org. Biomol. Chem. 2014, 12, 749–757. [Google Scholar] [CrossRef]
- Clément, E.; Mesini, P.J.; Pattus, F.; Abdallah, M.A.; Schalk, I.J. The binding mechanism of pyoverdin with the outer membrane receptor FpvA in Pseudomonas aeruginosa is dependent on its iron-loaded status. Biochemistry 2004, 43, 7954–7965. [Google Scholar]
- Albrecht-Gary, A.M.; Blanc, S.; Rochel, N.; Ocacktan, A.Z.; Abdallah, M.A. Bacterial iron transport: Coordination properties of pyoverdin PaA, a peptidic siderophore of Pseudomonas aeruginosa. Inorg. Chem. 1994, 33, 6391–6402. [Google Scholar]
- Brandel, J.; Humbert, N.; Elhabiri, M.; Schalk, I.J.; Mislin, G.L.A.; Albrecht-Garry, A.-M. Pyochelin, a siderophore of Pseudomonas aeruginosa: Physicochemical characterization of the iron(III), copper(II) and zinc(II) complexes. Dalton Trans 2012, 41, 2820–2834. [Google Scholar] [PubMed]
- Folschweiller, N.; Gallay, J.; Vincent, M.; Abdallah, M.A.; Pattus, F.; Schalk, I.J. The interaction between pyoverdin and its outer membrane receptor in Pseudomonas aeruginosa leads to different conformers: A time-resolved fluorescence study. Biochemistry 2002, 41, 14591–14601. [Google Scholar] [PubMed]
- Loomis, L.; Raymond, K.N. Solution equilibria of enterobactin complexes. Inorg. Chem. 1991, 30, 906–911. [Google Scholar]
- Anderegg, G.; L’Eplattenier, F.; Schwarzenbach, G. Hydroxamatkomplexe III. Eisen(III)-Austausch zwischen Sideraminen und Komplexonen. Diskussion der Bildungskonstanten der Hydroxamatkomplexe. Helv. Chim. Acta 1963, 46, 1409–1422. [Google Scholar] [CrossRef]
- Postle, K.; Larsen, R.A. TonB-dependent energy transduction between outer and cytoplasmic membranes. Biometals 2007, 20, 453–465. [Google Scholar] [PubMed]
- Celia, H.; Noinaj, N.; Buchanan, S.K. Structure and Stoichiometry of the Ton Molecular Motor. Int. J. Mol. Sci. 2020, 21, 375. [Google Scholar] [CrossRef]
- Cornelis, P.; Matthijs, S.; Van Oeffelen, L. Iron uptake regulation in Pseudomonas aeruginosa. Biometals 2009, 22, 15–22. [Google Scholar] [CrossRef]
- Pollack, M. The role of exotoxin A in Pseudomonas disease and immunity. Rev. Infect. Dis. 1983, 5 (Suppl. 5), S979–S984. [Google Scholar] [CrossRef]
- Chaudry, G.J.; Holmans, P.L.; Clowes, R.C.; Draper, R.K. Pseudomonas aeruginosa exotoxin A. Target Diagn. Ther. 1992, 7, 405–423. [Google Scholar]
- Thibodeaux, B.A.; Caballero, A.R.; Dajcs, J.J.; Marquart, M.E.; Engel, L.S.; O’Callaghan, R.J. Pseudomonas aeruginosa protease IV: A corneal virulence factor of low immunogenicity. Ocul. Immunol. Inflamm. 2005, 13, 169–182. [Google Scholar] [CrossRef]
- Oh, J.; Li, X.-H.; Kim, S.-K.; Lee, J.-H. Post-secretional activation of Protease IV by quorum sensing in Pseudomonas aeruginosa. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Laarman, A.J.; Bardoel, B.W.; Ruyken, M.; Fernie, J.; Milder, F.J.; van Strijp, J.A.G.; Rooijakkers, S.H.M. Pseudomonas aeruginosa alkaline protease blocks complement activation via the classical and lectin pathways. J. Immunol. 2012, 188, 386–393. [Google Scholar] [CrossRef] [PubMed]
- Muriel, C.; Arrebola, E.; Redondo-Nieto, M.; Martínez-Granero, F.; Jalvo, B.; Pfeilmeier, S.; Blanco-Romero, E.; Baena, I.; Malone, J.G.; Rivilla, R.; et al. AmrZ is a major determinant of c-di-GMP levels in Pseudomonas fluorescens F113. Sci. Rep. 2018, 8, 1979. [Google Scholar] [CrossRef] [PubMed]
- Mavrodi, D.V.; Bonsall, R.F.; Delaney, S.M.; Soule, M.J.; Phillips, G.; Thomashow, L.S. Functional analysis of genes for biosynthesis of pyocyanin and phenazine-1-carboxamide from Pseudomonas aeruginosa PAO1. J. Bacteriol. 2001, 183, 6454–6465. [Google Scholar] [CrossRef]
- Recinos, D.A.; Sekedat, M.D.; Hernandez, A.; Cohen, T.S.; Sakhtah, H.; Prince, A.S.; Price-Whelan, A.; Dietrich, L.E.P. Redundant phenazine operons in Pseudomonas aeruginosa exhibit environment-dependent expression and differential roles in pathogenicity. Proc. Natl. Acad. Sci. USA 2012, 109, 19420–19425. [Google Scholar] [CrossRef]
- Duong, F.; Bonnet, E.; Géli, V.; Lazdunski, A.; Murgier, M.; Filloux, A. The AprX protein of Pseudomonas aeruginosa: A new substrate for the Apr type I secretion system. Gene 2001, 262, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Wilderman, P.J.; Vasil, A.I.; Johnson, Z.; Wilson, M.J.; Cunliffe, H.E.; Lamont, I.L.; Vasil, M.L. Characterization of an endoprotease (PrpL) encoded by a PvdS-regulated gene in Pseudomonas aeruginosa. Infect. Immun. 2001, 69, 5385–5394. [Google Scholar] [CrossRef]
- Casilag, F.; Lorenz, A.; Krueger, J.; Klawonn, F.; Weiss, S.; Häussler, S. The LasB Elastase of Pseudomonas aeruginosa Acts in Concert with Alkaline Protease AprA To Prevent Flagellin-Mediated Immune Recognition. Infect. Immun. 2016, 84, 162–171. [Google Scholar] [CrossRef]
- Yahr, T.L.; Vallis, A.J.; Hancock, M.K.; Barbieri, J.T.; Frank, D.W. ExoY, an adenylate cyclase secreted by the Pseudomonas aeruginosa type III system. Proc. Natl. Acad. Sci. USA 1998, 95, 13899–13904. [Google Scholar] [CrossRef]
- Wolz, C.; Hellstern, E.; Haug, M.; Galloway, D.R.; Vasil, M.L.; Döring, G. Pseudomonas aeruginosa LasB mutant constructed by insertional mutagenesis reveals elastolytic activity due to alkaline proteinase and the LasA fragment. Mol. Microbiol. 1991, 5, 2125–2131. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perraud, Q.; Cantero, P.; Munier, M.; Hoegy, F.; Zill, N.; Gasser, V.; Mislin, G.L.A.; Ehret-Sabatier, L.; Schalk, I.J. Phenotypic Adaptation of Pseudomonas aeruginosa in the Presence of Siderophore-Antibiotic Conjugates during Epithelial Cell Infection. Microorganisms 2020, 8, 1820. https://doi.org/10.3390/microorganisms8111820
Perraud Q, Cantero P, Munier M, Hoegy F, Zill N, Gasser V, Mislin GLA, Ehret-Sabatier L, Schalk IJ. Phenotypic Adaptation of Pseudomonas aeruginosa in the Presence of Siderophore-Antibiotic Conjugates during Epithelial Cell Infection. Microorganisms. 2020; 8(11):1820. https://doi.org/10.3390/microorganisms8111820
Chicago/Turabian StylePerraud, Quentin, Paola Cantero, Mathilde Munier, Françoise Hoegy, Nicolas Zill, Véronique Gasser, Gaëtan L. A. Mislin, Laurence Ehret-Sabatier, and Isabelle J. Schalk. 2020. "Phenotypic Adaptation of Pseudomonas aeruginosa in the Presence of Siderophore-Antibiotic Conjugates during Epithelial Cell Infection" Microorganisms 8, no. 11: 1820. https://doi.org/10.3390/microorganisms8111820
APA StylePerraud, Q., Cantero, P., Munier, M., Hoegy, F., Zill, N., Gasser, V., Mislin, G. L. A., Ehret-Sabatier, L., & Schalk, I. J. (2020). Phenotypic Adaptation of Pseudomonas aeruginosa in the Presence of Siderophore-Antibiotic Conjugates during Epithelial Cell Infection. Microorganisms, 8(11), 1820. https://doi.org/10.3390/microorganisms8111820





