Enteroviruses and Type 1 Diabetes Mellitus: An Overlooked Relationship in Some Regions
Abstract
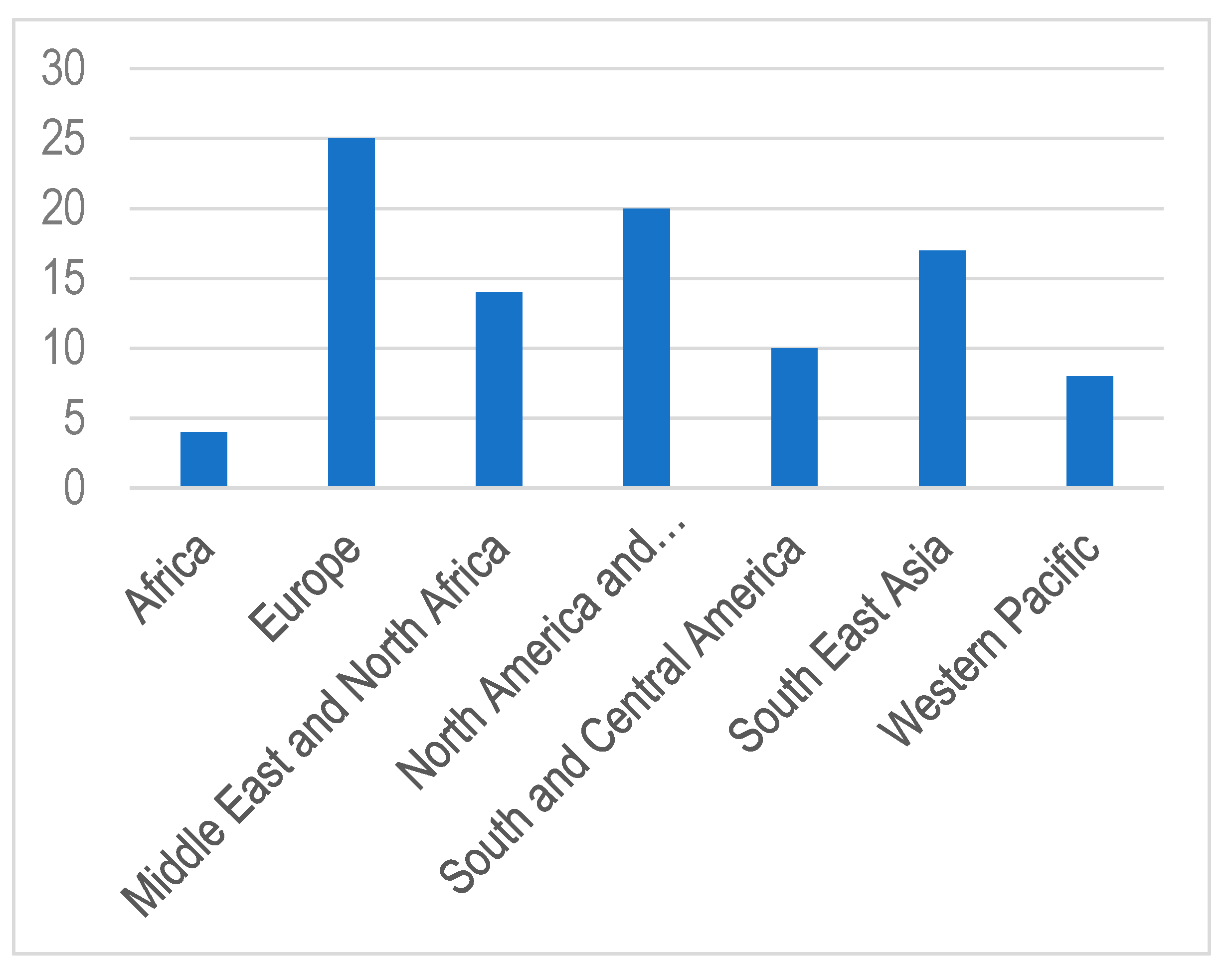
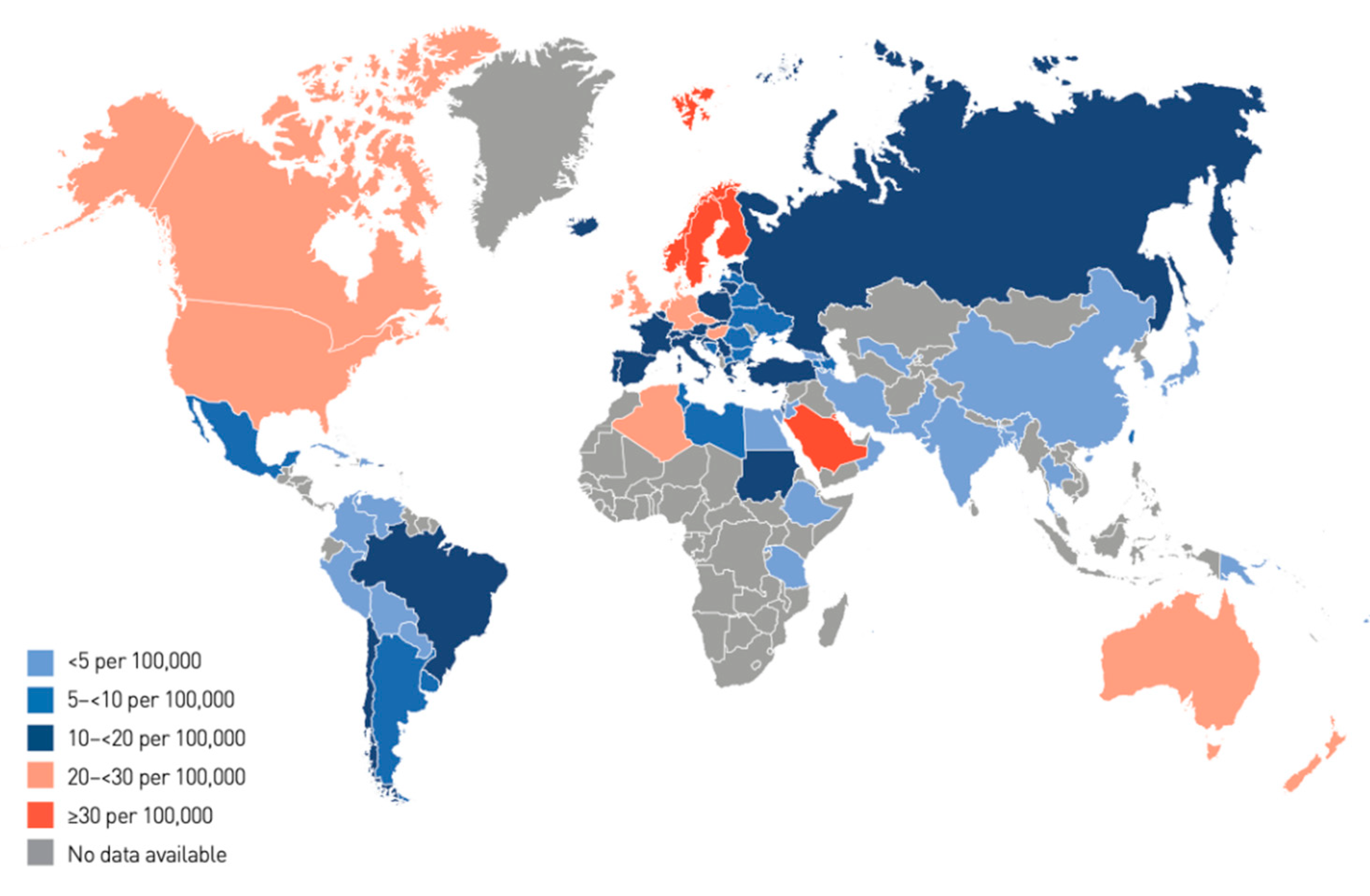
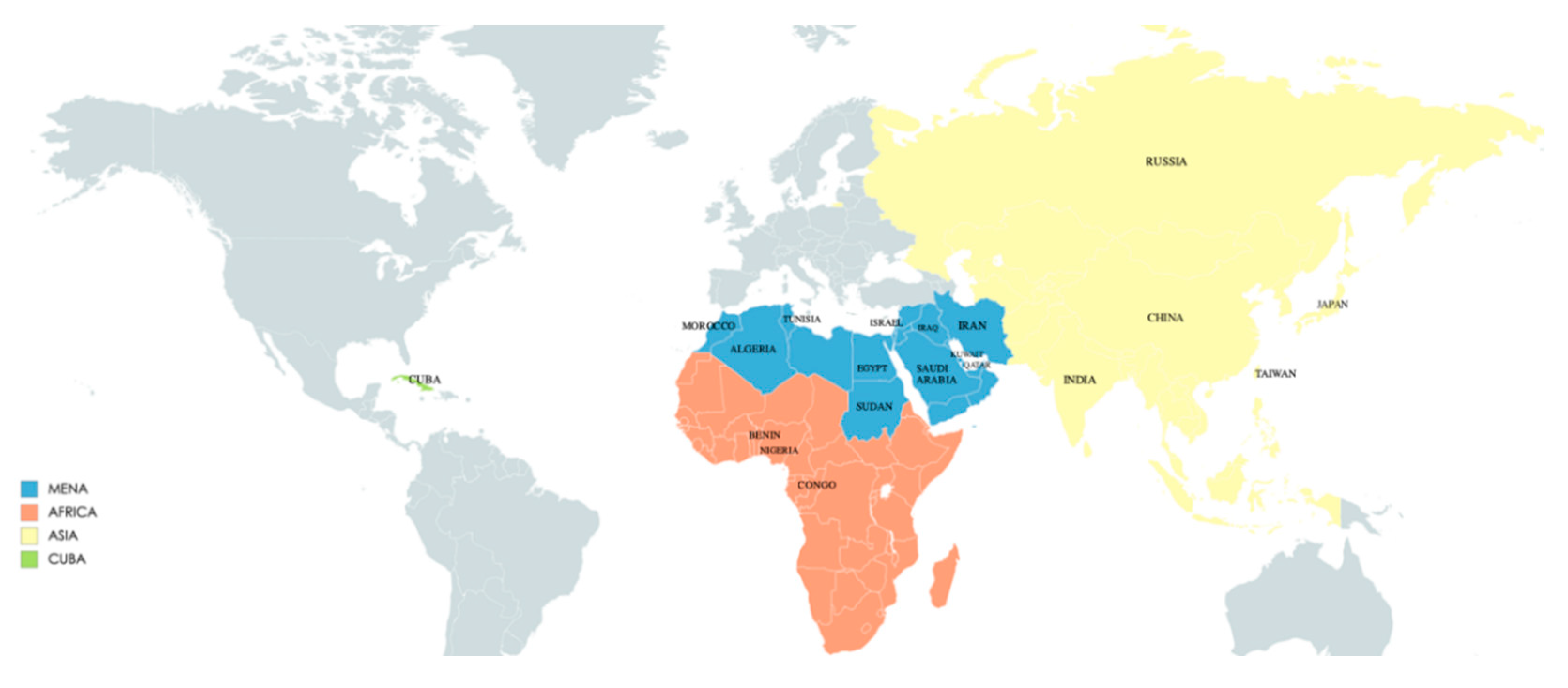
1. Introduction
2. MENA
2.1. Kuwait, Saudi Arabia, Qatar and Egypt
2.2. Iraq
2.3. Iran
2.4. Sudan
2.5. Algeria, Morocco, and Tunisia
2.6. Israel
3. Africa
3.1. Nigeria
3.2. Congo
3.3. Benin
4. Asia
4.1. India
4.2. China
4.3. Japan
4.4. Taiwan
4.5. Russia
5. Cuba
6. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lo, C.W.; Wu, K.G.; Lin, M.C.; Chen, C.J.; Ho, D.M.; Tang, R.B.; Chan, Y.J. Application of a molecular method for the classification of human enteroviruses and its correlation with clinical manifestations. J. Microbiol. Immunol. Infect. 2010, 43, 354–359. [Google Scholar] [CrossRef]
- Pons-Salort, M.; Oberste, M.S.; Pallansch, M.A.; Abedi, G.R.; Takahashi, S.; Grenfell, B.T.; Grassly, N.C. The seasonality of nonpolio enteroviruses in the United States: Patterns and drivers. Proc. Natl. Acad. Sci. USA 2018, 115, 3078–3083. [Google Scholar] [CrossRef] [PubMed]
- Alhazmi, A.; Lazrek, M.; Alidjinou, E.K.; Descombes, G.; Engelmann, I.; Hober, D. Paediatric enterovirus meningitis without cerebrospinal fluid pleocytosis. J. Infect. 2019, 79, 612–625. [Google Scholar] [CrossRef]
- Sawyer, M.H. Enterovirus infections: Diagnosis and treatment. Semin. Pediatr. Infect. Dis. 2002, 13, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Tracy, S.; Smithee, S.; Alhazmi, A.; Chapman, N. Coxsackievirus can persist in murine pancreas by deletion of 5′ terminal genomic sequences. J. Med. Virol. 2014, 87, 240–247. [Google Scholar] [CrossRef]
- Alidjinou, E.K.; Sané, F.; Engelmann, I.; Geenen, V.; Hober, D. Enterovirus persistence as a mechanism in the pathogenesis of type 1 diabetes. Discov. Med. 2014, 18, 273–282. [Google Scholar]
- Hober, D.; Sauter, P. Pathogenesis of type 1 diabetes mellitus: Interplay between enterovirus and host. Nat. Rev. Endocrinol. 2010, 6, 279–289. [Google Scholar] [CrossRef]
- Chapman, N.M.; Kim, K.S. Persistent coxsackievirus infection: Enterovirus persistence in chronic myocarditis and dilated cardiomyopathy. Curr. Top. Microbiol. Immunol. 2008, 323, 275–292. [Google Scholar]
- Tanaka, S.; Nishida, Y.; Aida, K.; Maruyama, T.; Shimada, A.; Suzuki, M.; Shimura, H.; Takizawa, S.; Takahashi, M.; Akiyama, D.; et al. Enterovirus infection, CXC chemokine ligand 10 (CXCL10), and CXCR3 circuit: A mechanism of accelerated beta-cell failure in fulminant type 1 diabetes. Diabetes 2009, 58, 2285–2291. [Google Scholar] [CrossRef]
- Robertson, C.C.; Rich, S.S. Genetics of type 1 diabetes. Curr. Opin. Genet. Dev. 2018, 50, 7–16. [Google Scholar] [CrossRef]
- Hober, D.; Alidjinou, E.K. Enteroviral pathogenesis of type 1 diabetes: Queries and answers. Curr. Opin. Infect. Dis. 2013, 26, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Krogvold, L.; Edwin, B.; Buanes, T.; Frisk, G.; Skog, O.; Anagandula, M.; Korsgren, O.; Undlien, D.; Eike, M.C.; Richardson, S.J.; et al. Detection of a low-grade enteroviral infection in the islets of langerhans of living patients newly diagnosed with type 1 diabetes. Diabetes 2015, 64, 1682–1687. [Google Scholar] [CrossRef] [PubMed]
- Takita, M.; Jimbo, E.; Fukui, T.; Aida, K.; Shimada, A.; Oikawa, Y.; Yagihashi, S.; Miura, J.; Babazono, T.; Kobayashi, T. Unique Inflammatory Changes in Exocrine and Endocrine Pancreas in Enterovirus-Induced Fulminant Type 1 Diabetes. J. Clin. Endocrinol. Metab. 2019, 104, 4282–4294. [Google Scholar] [CrossRef] [PubMed]
- Oikarinen, S.; Martiskainen, M.; Tauriainen, S.; Huhtala, H.; Ilonen, J.; Veijola, R.; Simell, O.; Knip, M.; Hyöty, H. Enterovirus RNA in blood is linked to the development of type 1 diabetes. Diabetes 2011, 60, 276–279. [Google Scholar] [CrossRef]
- GLOBAL. Diabetes Data Report 9th Edition. 2019. Available online: https://diabetesatlas.org/data/en/world/ (accessed on 17 September 2020).
- Moltchanova, E.V.; Schreier, N.; Lammi, N.; Karvonen, M. Seasonal variation of diagnosis of Type 1 diabetes mellitus in children worldwide. Diabet. Med. 2009, 26, 673–678. [Google Scholar] [CrossRef]
- Hiltunen, M.; Hyöty, H.; Knip, M.; Ilonen, J.; Reijonen, H.; Vähäsalo, P.; Roivainen, M.; Lönnrot, M.; Leinikki, P.; Hovi, T.; et al. Islet cell antibody seroconversion in children is temporally associated with enterovirus infections. Childhood Diabetes in Finland (DiMe) Study Group. J. Infect. Dis. 1997, 175, 554–560. [Google Scholar] [CrossRef]
- Laitinen, O.H.; Honkanen, H.; Pakkanen, O.; Oikarinen, S.; Hankaniemi, M.M.; Huhtala, H.; Ruokoranta, T.; Lecouturier, V.; André, P.; Harju, R.; et al. Coxsackievirus B1 is associated with induction of β-cell autoimmunity that portends type 1 diabetes. Diabetes 2014, 63, 446–455. [Google Scholar] [CrossRef]
- Salminen, K.; Sadeharju, K.; Lönnrot, M.; Vähäsalo, P.; Kupila, A.; Korhonen, S.; Ilonen, J.; Simell, O.; Knip, M.; Hyöty, H. Enterovirus infections are associated with the induction of beta-cell autoimmunity in a prospective birth cohort study. J. Med. Virol. 2003, 69, 91–98. [Google Scholar] [CrossRef]
- Oikarinen, S.; Tauriainen, S.; Hober, D.; Lucas, B.; Vazeou, A.; Sioofy-Khojine, A.; Bozas, E.; Muir, P.; Honkanen, H.; Ilonen, J.; et al. Virus antibody survey in different European populations indicates risk association between coxsackievirus B1 and type 1 diabetes. Diabetes 2014, 63, 655–662. [Google Scholar] [CrossRef]
- Middle East and North Africa Diabetes Report 9th Edition. 2019. Available online: https://diabetesatlas.org/data/en/region/4/mena.html (accessed on 17 September 2020).
- Association, A.D. Diagnosis and classification of diabetes mellitus. Diabetes Care 2011, 34 (Suppl. 1), S62–S69. [Google Scholar] [CrossRef]
- Genuth, S.; Alberti, K.G.; Bennett, P.; Buse, J.; Defronzo, R.; Kahn, R. Follow-up report on the diagnosis of diabetes mellitus. Diabetes Care 2003, 26, 3160–3167. [Google Scholar] [PubMed]
- Abduljabbar, M.A.; Aljubeh, J.M.; Amalraj, A.; Cherian, M.P. Incidence trends of childhood type 1 diabetes in eastern Saudi Arabia. Saudi. Med. J. 2010, 31, 413–418. [Google Scholar] [PubMed]
- Shaltout, A.A.; Wake, D.; Thanaraj, T.A.; Omar, D.M.; Al-AbdulRazzaq, D.; Channanath, A.; AlKandari, H.; Abdulrasoul, M.; Miller, S.; Conway, N.; et al. Incidence of type 1 diabetes has doubled in Kuwaiti children 0-14 years over the last 20 years. Pediatr. Diabetes 2017, 18, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Alyafei, F.; Soliman, A.; Alkhalaf, F.; Sabt, A.; De Sanctis, V.; Waseef, R.; Elsayed, N. Incidence of type 1 and type 2 diabetes, between 2012–2016, among children and adolescents in Qatar. Acta Biomed. 2018, 89, 7–10. [Google Scholar]
- Ahmad, S.; Dalwai, A.; Al-Nakib, W. Frequency of enterovirus detection in blood samples of neonates admitted to hospital with sepsis-like illness in Kuwait. J. Med. Virol. 2013, 85, 1280–1285. [Google Scholar] [CrossRef]
- Habeb, A.M.; Al-Magamsi, M.S.; Halabi, S.; Eid, I.M.; Shalaby, S.; Bakoush, O. High incidence of childhood type 1 diabetes in Al-Madinah, North West Saudi Arabia (2004–2009). Pediatr. Diabetes 2011, 12, 676–681. [Google Scholar] [CrossRef]
- Al Shaikh, A.; Farahat, F.; Saeedi, M.; Bakar, A.; Al Gahtani, A.; Al-Zahrani, N.; Jaha, L.; Aseeri, M.A.; Al-Jifree, H.M.; Al Zahrani, A. Incidence of diabetic ketoacidosis in newly diagnosed type 1 diabetes children in western Saudi Arabia: 11-year experience. J. Pediatr. Endocrinol. Metab. 2019, 32, 857–862. [Google Scholar] [CrossRef]
- Cherian, M.P.; Al-Kanani, K.A.; Al Qahtani, S.S.; Yesurathinam, H.; Mathew, A.A.; Thomas, V.S.; Mathew, A.M.; Abduljabbar, M.A. The rising incidence of type 1 diabetes mellitus and the role of environmental factors—Three decade experience in a primary care health center in Saudi Arabia. J. Pediatr. Endocrinol. Metab. 2010, 23, 685–695. [Google Scholar] [CrossRef]
- Al-Hajjar, S.; Akhter, J.; Arishi, H.; Qadri, S.M. Enteroviruses at a tertiary care center in Saudi Arabia, 1989–1995. Ann. Saudi. Med. 1997, 17, 16–19. [Google Scholar] [CrossRef]
- Abid, F.B.; Abukhattab, M.; Ghazouani, H.; Khalil, O.; Gohar, A.; Al Soub, H.; Al Maslamani, M.; Al Khal, A.; Al Masalamani, E.; Al Dhahry, S.; et al. Epidemiology and clinical outcomes of viral central nervous system infections. Int. J. Infect. Dis. 2018, 73, 85–90. [Google Scholar] [CrossRef]
- El-Ziny, M.A.; Salem, N.A.; El-Hawary, A.K.; Chalaby, N.M.; Elsharkawy, A.A. Epidemiology of childhood type 1 diabetes mellitus in Nile Delta, northern Egypt—A retrospective study. J. Clin. Res. Pediatr. Endocrinol. 2014, 6, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Afifi, S.S.; Zaki, S.A. Isolation and Identification of Non-Polio Enteroviruses from Children in Different Egyptian Governorates. Aust. J. Basic Appl. Sci. 2009, 3, 3230–3238. [Google Scholar]
- Almahfoodh, D.; Alabbood, M.; Alali, A.; Mansour, A. Epidemiology of type 1 diabetes mellitus in Basrah, Southern Iraq: A retrospective study. Diabetes Res. Clin. Pract. 2017, 133, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Bilal, R.; AL-Zobaei, M.A.; AL-Ani, Z.R. Relation of Coxsackie B3 and B4 Viral Infections for Development of type 1 Diabetes Mellitus in Children: A Case-Control Study. Egypt. Acad. J. Biol. Sci. 2019, 11, 1–12. [Google Scholar]
- Zargari Samani, O.; Jamshidian, A.; Ghatreh Samani, M.; Khaeiri, S.; Shirzad, H. Evaluation of Enterovirus infection in people with type 1 diabetes in Chaharmahal and Bakhtiari province compared to healthy subjects. J. Shahrekord. Univ. Med. Sci. 2017, 19, 24–31. [Google Scholar]
- Emad, M.A.; Ali, Y.H.; Enan, K.A. Epidemiology of Type 1 Diabetes Mellitus Among Children in Sudan: Serological Evidence of Coxsackievirus Infection. J. Sci. Technol. 2011, 12, 64–73. [Google Scholar]
- Boussaid, I.; Boumiza, A.; Zemni, R.; Chabchoub, E.; Gueddah, L.; Slim, I.; Slama, F.B. The role of enterovirus infections in type 1 diabetes in Tunisia. J. Pediatr. Endocrinol. Metab. 2017, 30, 1245–1250. [Google Scholar] [CrossRef]
- Badia-Boungou, F.; Sane, F.; Alidjinou, E.K.; Ternois, M.; Opoko, P.A.; Haddad, J.; Stukens, C.; Lefevre, C.; Gueorguieva, I.; Hamze, M.; et al. Marker of coxsackievirus-B4 infection in saliva of patients with type 1 diabetes. Diabetes Metab. Res. Rev. 2017, 33, e2916. [Google Scholar] [CrossRef]
- Nekoua, M.P.; Yessoufou, A.; Alidjinou, E.K.; Badia-Boungou, F.; Moutairou, K.; Sane, F.; Hober, D. Salivary anti-coxsackievirus-B4 neutralizing activity and pattern of immune parameters in patients with type 1 diabetes: A pilot study. Acta Diabetol. 2018, 55, 827–834. [Google Scholar] [CrossRef]
- Lin, H.C.; Wang, C.H.; Tsai, F.J.; Hwang, K.P.; Chen, W.; Lin, C.C.; Li, T.C. Enterovirus infection is associated with an increased risk of childhood type 1 diabetes in Taiwan: A nationwide population-based cohort study. Diabetologia 2015, 58, 79–86. [Google Scholar] [CrossRef]
- Asmaa, K.; Salma, F.; Zineb, I. Epidemiological Profile of type 1 diabetes in Children: About 971 Patients. Acad. J. Ped. Neonatol. 2019, 7, 555778. [Google Scholar]
- Viskari, H.; Ludvigsson, J.; Uibo, R.; Salur, L.; Marciulionyte, D.; Hermann, R.; Soltesz, G.; Füchtenbusch, M.; Ziegler, A.G.; Kondrashova, A.; et al. Relationship between the incidence of type 1 diabetes and maternal enterovirus antibodies: Time trends and geographical variation. Diabetologia 2005, 48, 1280–1287. [Google Scholar] [CrossRef] [PubMed]
- Elfving, M.; Lindberg, B.; Lynch, K.; Ivarsson, S.A.; Lernmark, A.; Hampe, C.S. Epitope analysis of GAD65 binding in both cord blood and at the time of clinical diagnosis of childhood type 1 diabetes. Horm. Metab. Res. 2007, 39, 790–796. [Google Scholar] [CrossRef] [PubMed]
- Hermann, E.; Truyens, C.; Alonso-Vega, C.; Even, J.; Rodriguez, P.; Berthe, A.; Gonzalez-Merino, E.; Torrico, F.; Carlier, Y. Human fetuses are able to mount an adultlike CD8 T-cell response. Blood 2002, 100, 2153–2158. [Google Scholar] [CrossRef] [PubMed]
- Ould, M.A.; Luton, D.; Yadini, M.; Pedron, B.; Aujard, Y.; Jacqz-Aigrain, E.; Jacquemard, F.; Sterkers, G. Cellular immune response of fetuses to cytomegalovirus. Pediatr. Res. 2004, 55, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Shulman, L.M.; Hampe, C.S.; Ben-Haroush, A.; Perepliotchikov, Y.; Vaziri-Sani, F.; Israel, S.; Miller, K.; Bin, H.; Kaplan, B.; Laron, Z. Antibodies to islet cell autoantigens, rotaviruses and/or enteroviruses in cord blood and healthy mothers in relation to the 2010–2011 winter viral seasons in Israel: A pilot study. Diabet. Med. 2014, 31, 681–685. [Google Scholar] [CrossRef] [PubMed]
- Damiens, A.P.; Ganga–Zandzou, P.S.; Ibounde, E.T.; Baye, E.; Biloghe, P.; Kakou, C.; Ntyonga-Pono, P. Type 1 diabetes mellitus in Gabon: A study of epidemiological aspects. Int. J. Pediatr. Adolesc. Med. 2019, 6, 87–91. [Google Scholar] [CrossRef]
- Ogbera, A.O.; Ekpebegh, C. Diabetes mellitus in Nigeria: The past, present and future. World J. Diabetes 2014, 5, 905–911. [Google Scholar] [CrossRef]
- Sarfo-Kantanka, O.; Asamoah-Boaheng, M.; Arthur, J.; Agyei, M.; Barnes, N.A.; Tenkorang, E.Y.; Midodzi, W. Trends in type 1 diabetes diagnosis in Ghana. Int. Health 2020. [Google Scholar] [CrossRef]
- Ajuwon, Z.A.; Olaleye, O.D.; Omilabu, S.A.; Baba, S.S. Complement fixing antibodies against selected viruses in diabetic patients and non-diabetic control subjects in Ibadan, Nigeria. Rev. Roum. Virol. 1992, 43, 3–5. [Google Scholar]
- Maan, H.S.; Dhole, T.N.; Chowdhary, R. Identification and characterization of nonpolio enterovirus associated with nonpolio-acute flaccid paralysis in polio endemic state of Uttar Pradesh, Northern India. PLoS ONE 2019, 14, e0208902. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Shukla, D.; Kumar, R.; Idris, M.Z.; Misra, U.K.; Dhole, T.N. Molecular epidemiological study of enteroviruses associated with encephalitis in children from India. J. Clin. Microbiol. 2012, 50, 3509–3512. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Shukla, D.; Kumar, R.; Idris, M.Z.; Misra, U.K.; Dhole, T.N. An epidemic of encephalitis associated with human enterovirus B in Uttar Pradesh, India, 2008. J. Clin. Virol. 2011, 51, 142–145. [Google Scholar] [CrossRef]
- Jain, S.; Thakur, N.; Grover, N.; Vashistt, J.; Changotra, H. Prevalence of rotavirus, norovirus and enterovirus in diarrheal diseases in Himachal Pradesh, India. Virusdisease 2016, 27, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Sun, C.; Wang, C.; Li, P.; Wang, W.; Ye, J.; Gu, X.; Wang, X.; Shen, S.; Zhi, D.; et al. Rapidly rising incidence of childhood type 1 diabetes in Chinese population: Epidemiology in Shanghai during 1997–2011. Acta Diabetol. 2014, 51, 947–953. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhou, F.; Dorman, J.; Wang, H.; Zu, X.; Mazumdar, S.; LaPorte, R.E. Association between infectious diseases and type 1 diabetes: A case-crossover study. Pediatr. Diabetes 2006, 7, 146–152. [Google Scholar] [CrossRef]
- Kawasaki, E.; Matsuura, N.; Eguchi, K. Type 1 diabetes in Japan. Diabetologia 2006, 49, 828–836. [Google Scholar] [CrossRef]
- Matsuura, N.; Fukuda, K.; Okuno, A.; Harada, S.; Fukushima, N.; Koike, A.; Ito, Y.; Hotsubo, T. Descriptive epidemiology of IDDM in Hokkaido, Japan: The Childhood IDDM Hokkaido Registry. Diabetes Care 1998, 21, 1632–1636. [Google Scholar] [CrossRef]
- Kida, K.; Mimura, G.; Ito, T.; Murakami, K.; Ashkenazi, I.; Laron, Z. Incidence of Type 1 diabetes mellitus in children aged 0–14 in Japan, 1986–1990, including an analysis for seasonality of onset and month of birth: JDS study. The Data Committee for Childhood Diabetes of the Japan Diabetes Society (JDS). Diabet Med. 2000, 17, 59–63. [Google Scholar] [CrossRef]
- Nishimura, A.; Matsumura, K.; Kikuno, S.; Nagasawa, K.; Okubo, M.; Mori, Y.; Kobayashi, T. Slowly Progressive Type 1 Diabetes Mellitus: Current Knowledge And Future Perspectives. Diabetes Metab. Syndr. Obes. 2019, 12, 2461–2477. [Google Scholar] [CrossRef]
- Imagawa, A.; Hanafusa, T.; Makino, H.; Miyagawa, J.I.; Juto, P. High titres of IgA antibodies to enterovirus in fulminant type-1 diabetes. Diabetologia 2005, 48, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Akatsuka, H.; Yano, Y.; Gabazza, E.C.; Morser, J.; Sasaki, R.; Suzuki, T.; Fujiwara, R.; Katsuki, A.; Takei, Y.; Sumida, Y. A case of fulminant type 1 diabetes with coxsackie B4 virus infection diagnosed by elevated serum levels of neutralizing antibody. Diabetes Res. Clin. Pract. 2009, 84, e50–e52. [Google Scholar] [CrossRef] [PubMed]
- Viskari, H.; Ludvigsson, J.; Uibo, R.; Salur, L.; Marciulionyte, D.; Hermann, R.; Soltesz, G.; Füchtenbusch, M.; Ziegler, A.G.; Kondrashova, A.; et al. Relationship between the incidence of type 1 diabetes and enterovirus infections in different European populations: Results from the EPIVIR project. J. Med. Virol. 2004, 72, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Horta, O.; Bello, M.; Cabrera-Rode, E.; Suárez, J.; Más, P.; García, I.; Abalos, I.; Jofra, R.; Molina, G.; Díaz-Díaz, O.; et al. Echovirus 4 and type 1 diabetes mellitus. Autoimmunity 2001, 34, 275–281. [Google Scholar]
- Cabrera-Rode, E.; Sarmiento, L.; Tiberti, C.; Molina, G.; Barrios, J.; Hernandez, D.; Diaz-Horta, O.; Di Mario, U. Type 1 diabetes islet associated antibodies in subjects infected by echovirus 16. Diabetologia 2003, 46, 1348–1353. [Google Scholar] [CrossRef]
- Cabrera-Rode, E.; Sarmiento, L.; Molina, G.; Pérez, C.; Arranz, C.; Galvan, J.A.; Prieto, M.; Barrios, J.; Palomera, R.; Fonseca, M.; et al. Islet cell related antibodies and type 1 diabetes associated with echovirus 30 epidemic: A case report. J. Med. Virol. 2005, 76, 373–377. [Google Scholar] [CrossRef]
- Sarmiento, L.; Cabrera-Rode, E.; Lekuleni, L.; Cuba, I.; Molina, G.; Fonseca, M.; Heng-Hung, L.; Borroto, A.D.; Gonzalez, P.; Mas-Lago, P.; et al. Occurrence of enterovirus RNA in serum of children with newly diagnosed type 1 diabetes and islet cell autoantibody-positive subjects in a population with a low incidence of type 1 diabetes. Autoimmunity 2007, 40, 540–545. [Google Scholar] [CrossRef]
| Rank | Country | Incidence Rate of T1DM for Patients Aged 0–14 Years (Per 100,000 Per Year) |
|---|---|---|
| 1 | Finland | 62.3 |
| 2 | Sweden | 43.2 |
| 3 | Kuwait | 41.7 |
| 4 | Norway | 33.6 |
| 5 | Saudi Arabia | 31.4 |
| 6 | Canada | 29.9 |
| 7 | United Kingdom | 29.4 |
| 8 | Qatar | 28.4 |
| 9 | Ireland | 27.5 |
| 10 | Denmark | 27 |
| Study | Year of Publication | Type of Study | Population | Country | Method of Detection | Serotype Detected |
|---|---|---|---|---|---|---|
| Bilal et al. [36] | 2019 | Case-control | Aged < 17 years (60 T1DM cases and 120 controls) | Iraq | - ELISA (IgG and IgM) - RT-PCR | CVB 3 CVB 4 |
| Samani et al. [37] | 2017 | Case-control | Aged < 30 years (35 T1DM cases and 35 controls) | Iran | - ELISA (IgG and IgM) - RT-PCR | - |
| Emad et al. [38] | 2011 | Questionnaire-based | 101 children (age not specified) | Sudan | - ELISA (IgG and IgM) | - |
| Boussaid et al. [39] | 2017 | Case-control | - Children (2–15 years) and adults (16–48 years) - 95 T1DM cases and 141 controls | Tunisia | RT-PCR | - |
| Badia-Boungou et al. [40] | 2017 | Case-control | Mean age, 14 years (181 T1DM cases and 135 controls) | Congo Lebanon France | Neutralization assay | CVB |
| Nekoua et al. [41] | 2018 | Case-control | Mean age, 23 years (15 T1DM cases and 8 controls) | Benin | Neutralization assay | CVB 4 |
| Lin et al. [42] | 2015 | Retrospective | Aged < 18 years | Taiwan | NA | - |
| Imagawa et al. (64) | 2005 | Case-control | 19 patients with fulminant T1DM, 18 patients with typical T1DM, and 19 healthy control | Japan | ELISA (IgM, IgA, and IgG) | - |
| Diaz-Horta et al. (67) | 2001 | Case-control | 33 T1DM patients at diagnosis, 43 healthy children of parents with T1DM without ICA, and 57 healthy subjects | Cuba | Neutralization assay | Echovirus 4 |
| Cabrera-Rode et al. (68) | 2003 | Case-control | 118 children and adolescents (38 T1DM cases and 80 controls) | Cuba | Neutralization assay | Echovirus 16 |
| Sarmiento et al. (70) | 2007 | Case-control | Aged 1–15 years (34 newly diagnosed T1DM, 31 positive ICA, and 32 negative ICA cases and 194 controls) | Cuba | RT-PCR | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alhazmi, A.; Sane, F.; Lazrek, M.; Nekoua, M.P.; Badia-Boungou, F.; Engelmann, I.; Alidjinou, E.K.; Hober, D. Enteroviruses and Type 1 Diabetes Mellitus: An Overlooked Relationship in Some Regions. Microorganisms 2020, 8, 1458. https://doi.org/10.3390/microorganisms8101458
Alhazmi A, Sane F, Lazrek M, Nekoua MP, Badia-Boungou F, Engelmann I, Alidjinou EK, Hober D. Enteroviruses and Type 1 Diabetes Mellitus: An Overlooked Relationship in Some Regions. Microorganisms. 2020; 8(10):1458. https://doi.org/10.3390/microorganisms8101458
Chicago/Turabian StyleAlhazmi, Abdulaziz, Famara Sane, Mouna Lazrek, Magloire Pandoua Nekoua, Francis Badia-Boungou, Ilka Engelmann, Enagnon Kazali Alidjinou, and Didier Hober. 2020. "Enteroviruses and Type 1 Diabetes Mellitus: An Overlooked Relationship in Some Regions" Microorganisms 8, no. 10: 1458. https://doi.org/10.3390/microorganisms8101458
APA StyleAlhazmi, A., Sane, F., Lazrek, M., Nekoua, M. P., Badia-Boungou, F., Engelmann, I., Alidjinou, E. K., & Hober, D. (2020). Enteroviruses and Type 1 Diabetes Mellitus: An Overlooked Relationship in Some Regions. Microorganisms, 8(10), 1458. https://doi.org/10.3390/microorganisms8101458






