Characterization of mcr-1-Harboring Plasmids from Pan Drug-Resistant Escherichia coli Strains Isolated from Retail Raw Chicken in South Korea
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Methods
2.2. Antimicrobial Susceptibility Testing
2.3. Conjugation Assay
2.4. Whole-Genome Sequencing
3. Results and Discussion
3.1. Whole-Genome Sequencing of mcr-1-Positive E. coli Strains
3.2. Antimicrobial Susceptibility Profiles and Other Resistance Genes
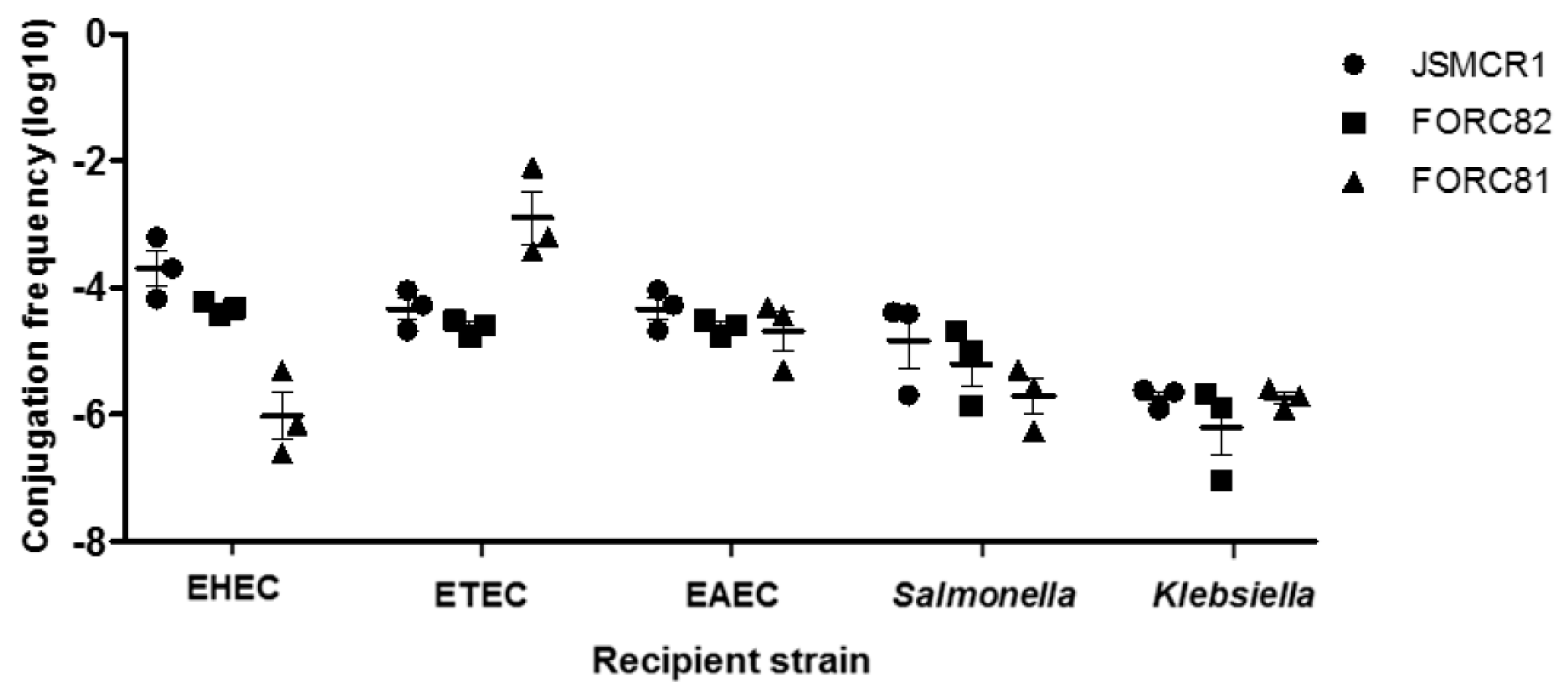
3.3. Conjugation Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gajdács, M. The concept of an ideal antibiotic: Implications for drug design. Molecules 2019, 24, 892. [Google Scholar] [CrossRef] [PubMed]
- Rhouma, M.; Beaudry, F.; Theriault, W.; Letellier, A. Colistin in pig production: Chemistry, mechanism of antibacterial action, microbial resistance emergence, and one health perspectives. Front. Microbiol. 2016, 7, 1789. [Google Scholar] [CrossRef] [PubMed]
- Gao, R.; Hu, Y.; Li, Z.; Sun, J.; Wang, Q.; Lin, J.; Ye, H.; Liu, F.; Srinivas, S.; Li, D. Dissemination and mechanism for the mcr-1 colistin resistance. PLoS Pathog. 2016, 12, e1005957. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-Y.; Wang, Y.; Walsh, T.R.; Yi, L.-X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X. Emergence of plasmid-mediated colistin resistance mechanism mcr-1 in animals and human beings in China: a microbiological and molecular biological study. Lancet Infect. Dis. 2016, 16, 161–168. [Google Scholar] [CrossRef]
- Carroll, L.M.; Gaballa, A.; Guldimann, C.; Sullivan, G.; Henderson, L.O.; Wiedmann, M. Identification of novel mobilized colistin resistance gene mcr-9 in a multidrug-resistant, colistin-susceptible Salmonella enterica serotype Typhimurium isolate. mBio 2019, 10, e00853-19. [Google Scholar] [CrossRef] [PubMed]
- El Chakhtoura, N.G.; Saade, E.; Iovleva, A.; Yasmin, M.; Wilson, B.; Perez, F.; Bonomo, R.A. Therapies for multidrug resistant and extensively drug-resistant non-fermenting Gram-negative bacteria causing nosocomial infections: a perilous journey toward ‘molecularly targeted’therapy. Expert Rev. Anti-Infect. Ther. 2018, 16, 89–110. [Google Scholar] [CrossRef] [PubMed]
- Gajdács, M.; Urbán, E. Epidemiological trends and resistance associated with Stenotrophomonas maltophilia bacteremia: a 10-year retrospective cohort study in a tertiary-care hospital in hungary. Diseases 2019, 7, 41. [Google Scholar] [CrossRef] [PubMed]
- Boucher, H.W.; Talbot, G.H.; Bradley, J.S.; Edwards, J.E.; Gilbert, D.; Rice, L.B.; Scheld, M.; Spellberg, B.; Bartlett, J. Bad bugs, no drugs: No ESKAPE! an update from the Infectious Diseases Society of America. Clin. Infect. Dis. 2009, 48, 1–12. [Google Scholar] [CrossRef] [PubMed]
- do Monte, D.F.; Fernandes, M.R.; Cerdeira, L.; Esposito, F.; Galvão, J.A.; Franco, B.D.; Lincopan, N.; Landgraf, M. Chicken meat as reservoir of colistin-resistant Escherichia coli carrying mcr-1 genes in South America. Antimicrob. Agents Chemother. 2017, 61, e02718-16. [Google Scholar] [CrossRef]
- Perrin-Guyomard, A.; Bruneau, M.; Houée, P.; Deleurme, K.; Legrandois, P.; Poirier, C.; Soumet, C.; Sanders, P. Prevalence of mcr-1 in commensal Escherichia coli from French livestock, 2007 to 2014. Eurosurveillance 2016, 21, 1–3. [Google Scholar] [CrossRef]
- Guenther, S.; Falgenhauer, L.; Semmler, T.; Imirzalioglu, C.; Chakraborty, T.; Roesler, U.; Roschanski, N. Environmental emission of multiresistant Escherichia coli carrying the colistin resistance gene mcr-1 from German swine farms. J. Antimicrob. Chemother. 2017, 72, 1289–1292. [Google Scholar] [PubMed]
- Donà, V.; Bernasconi, O.J.; Pires, J.; Collaud, A.; Overesch, G.; Ramette, A.; Perreten, V.; Endimiani, A. Heterogeneous genetic location of mcr-1 in colistin-resistant Escherichia coli isolated from humans and retail chicken meat in Switzerland: emergence of mcr-1-carrying IncK2 plasmids. Antimicrob. Agents Chemother. 2017, 61, e01245-17. [Google Scholar]
- Lim, S.-K.; Kang, H.Y.; Lee, K.; Moon, D.-C.; Lee, H.-S.; Jung, S.-C. First detection of the mcr-1 gene in Escherichia coli isolated from livestock between 2013 and 2015 in South Korea. Antimicrob. Agents Chemother. 2016, AAC, 01472-16. [Google Scholar]
- Kim, E.S.; Chong, Y.P.; Park, S.-J.; Kim, M.-N.; Kim, S.-H.; Lee, S.-O.; Choi, S.-H.; Woo, J.H.; Jeong, J.-Y.; Kim, Y.S. Detection and genetic features of mcr-1-producing plasmid in human Escherichia coli infection in South korea. Diagn. Microbiol. Infect. Dis. 2017, 89, 158–160. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Chen, L.; Tang, Y.-W.; Kreiswirth, B.N. Emergence of the mcr-1 colistin resistance gene in carbapenem-resistant Enterobacteriaceae. Lancet Infect. Dis. 2016, 16, 287–288. [Google Scholar] [CrossRef]
- Abat, C.; Fournier, P.-E.; Jimeno, M.-T.; Rolain, J.-M.; Raoult, D. Extremely and pandrug-resistant bacteria extra-deaths: myth or reality? Eur. J. Clin. Microbiol. Infect. Dis. 2018, 37, 1687–1697. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.L.; Fratamico, P.M.; Gunther, N.W. Extraintestinal pathogenic Escherichia coli. Foodborne Pathog. Dis. 2007, 4, 134–163. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, R.; Li, J.; Wu, Z.; Yin, W.; Schwarz, S.; Tyrrell, J.M.; Zheng, Y.; Wang, S.; Shen, Z. Comprehensive resistome analysis reveals the prevalence of NDM and MCR-1 in Chinese poultry production. Nat. Microbiol. 2017, 2, 16260. [Google Scholar] [CrossRef] [PubMed]
- Schrauwen, E.J.; Huizinga, P.; van Spreuwel, N.; Verhulst, C.; Kluytmans-van den Bergh, M.F.; Kluytmans, J.A. High prevalence of the mcr-1 gene in retail chicken meat in the Netherlands in 2015. Antimicrob. Resist. Infect. Control 2017, 6, 83. [Google Scholar] [CrossRef]
- Park, H.; Kim, J.; Ryu, S.; Jeon, B. The predominance of blaCTX-M-65 and blaCTX-M-55 in extended-spectrum β-lactamase-producing Escherichia coli from retail raw chicken in South korea. J. Glob. Antimicrob. Resist. 2019, 17, 216–220. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing: M100–S27. 2017. Available online: https://clsi.org/standards-development/document-correction-notices/ (accessed on 1 January 2017).
- Bell, S.; Gatus, B.; Pham, J.; Rafferty, D. Antibiotic susceptibility testing by the CDS method. A concise laboratory manual. 1999. Available online: http://cdstest.net/manual/ (accessed on 12 July 2019).
- Matamoros, S.; Hattem, J.M.; Arcilla, M.S.; Willemse, N.; Melles, D.C.; Penders, J.; Vinh, T.N.; Hoa, N.; Jong, M.D.; Schultsz, C. Global phylogenetic analysis of Escherichia coli and plasmids carrying the mcr-1 gene indicates bacterial diversity but plasmid restriction. Sci. Rep. 2017, 7, 15364. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Feng, Y.; Liu, F.; Jiang, H.; Qu, Z.; Lei, M.; Wang, J.; Zhang, B.; Hu, Y.; Ding, J. A phage-like IncY plasmid carrying the mcr-1 gene in Escherichia coli from a pig farm in China. Antimicrob. Agents Chemother. 2017, 61, e02035-16. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-Y.; Lim, S.-K.; Choi, Y.; Moon, D.-C.; Shin, J.; Ko, K.S. Whole sequences and characteristics of mcr-1-harboring plasmids of Escherichia coli strains isolated from livestock in South Korea. Microb. Drug Resist. 2018, 24, 489–492. [Google Scholar] [CrossRef] [PubMed]
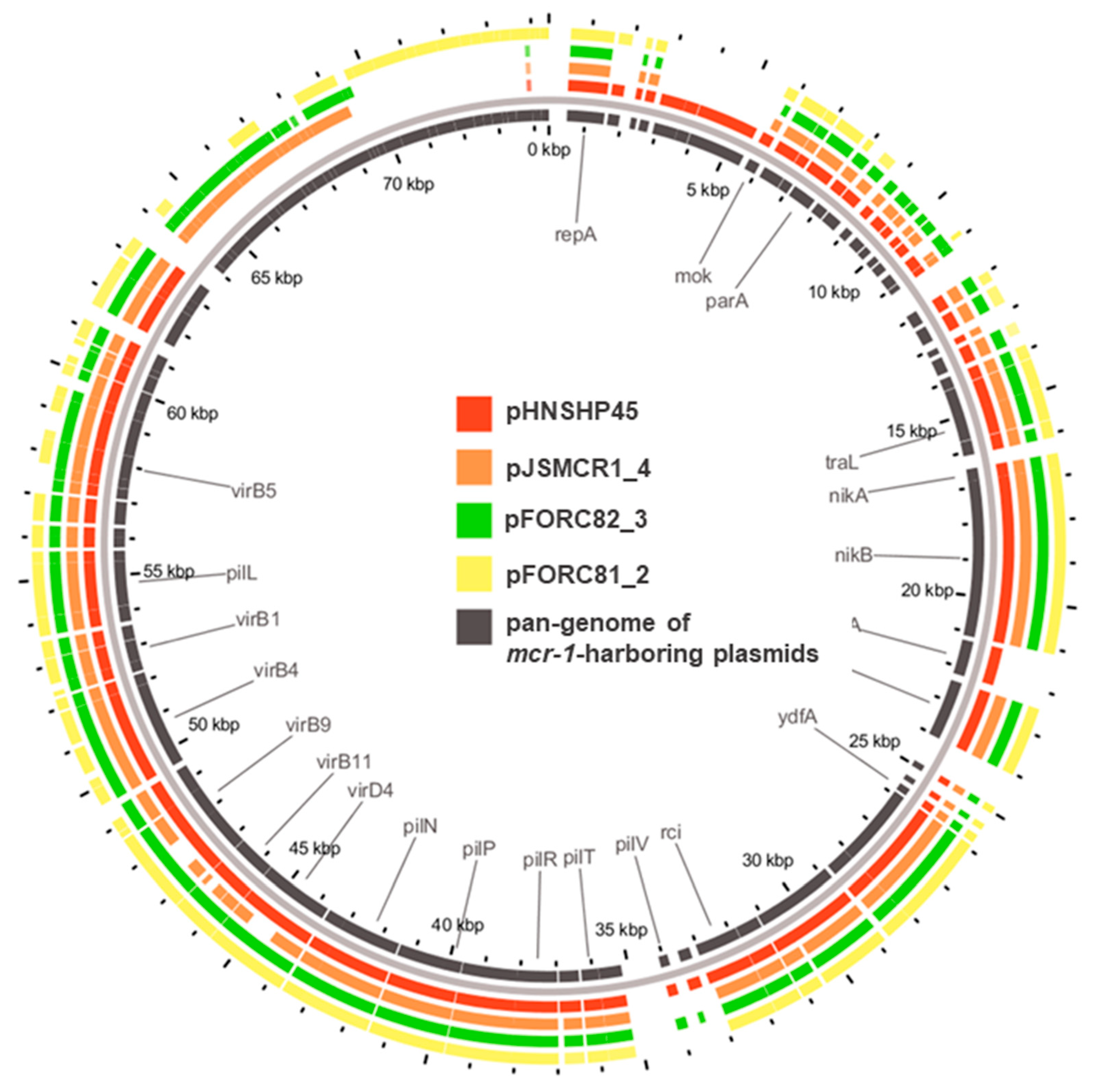
| E. coli Strain | Plasmid | Size (bp) | GenBank Accession No. | Inc Group | Resistance Genes |
|---|---|---|---|---|---|
| JSMCR1 | pJSMCR1_1 | 152,677 | CP030153 | IncFIB, IncFII | aph(3′)-Ia, aac(3)-IId, blaCTX-M-65, fosA3 |
| pJSMCR1_2 | 134,064 | CP030154 | p0111 | aadA1, blaOXA-10, qnrS1, floR, cmlA1, arr-2, tet(A), dfrA14 | |
| pJSMCR1_3 | 109,689 | CP030155 | IncI1 | - | |
| pJSMCR1_4 | 61,828 | CP030156 | IncI2 | mcr-1 | |
| pJSMCR1_5 | 29,589 | CP030157 | IncX4 | - | |
| FORC81 | pFORC81_1 | 253,947 | CP029058 | IncI1, IncFII | aadA1, aadA2, aac(3)-IId, blaTEM-1B, qnrS1, floR, cmlA1, sul3, tet(A), dfrA12 |
| pFORC81_2 | 61,917 | CP029059 | IncI2 | mcr-1 | |
| pFORC81_3 | 38,749 | CP029060 | IncX1 | - | |
| pFORC81_4 | 32,945 | CP029061 | IncI1 | blaTEM-1B, floR | |
| FORC82 | pFORC82_1 | 250,778 | CP026642 | IncHI2A, IncHI2, IncN | aadA1, aph(3′’)-Ib, aph(6)-Id, blaCTX-M-65, blaOXA-10, qnrS1, mph(A), floR, cmlA1, arr-2, sul2, tet(M), tet(A), dfrA14 |
| pFORC82_2 | 101,404 | CP026643 | IncFIC, IncFIB | - | |
| pFORC82_3 | 65,206 | CP026644 | IncI2 | mcr-1 |
| Strain | Origin a | mcr-1 Gene b | MIC c (μg/mL) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| AMP | CEF | TET | CHL | CIP | KAN | GEN | STR | POL | COL | |||
| E. coli JSMCR1 | WT | + | >64 | >64 | 128 | >64 | >8 | >32 | >64 | 64 | 8 | 8 |
| E. coli FORC81 | WT | + | >64 | 64 | >128 | >64 | >8 | 8 | >64 | 8 | 8 | 8 |
| E. coli FORC82 | WT | + | >64 | >64 | 128 | 64 | 2 | 4 | 2 | 2 | 8 | 8 |
| EHEC (E. coli ATCC 43889) | WT | - | ≤0.5 | ≤0.5 | ≤0.0628 | ≤0.5 | ≤0.0039 | ≤0.25 | ≤0.5 | >128 | ≤0.25 | ≤0.25 |
| pJSMCR1/EHEC | TC | + | ≤0.5 | ≤0.5 | ≤0.0628 | ≤0.5 | ≤0.0039 | ≤0.25 | ≤0.5 | >128 | 4 | 4 |
| pFORC81/EHEC | TC | + | ≤0.5 | ≤0.5 | ≤0.0628 | ≤0.5 | ≤0.0039 | ≤0.25 | ≤0.5 | >128 | 4 | 4 |
| pFORC82/EHEC | TC | + | ≤0.5 | ≤0.5 | ≤0.0628 | ≤0.5 | ≤0.0039 | ≤0.25 | ≤0.5 | >128 | 4 | 4 |
| ETEC (isolate) | WT | - | >64 | 8 | 32 | 64 | 0.0312 | >32 | >64 | >128 | 4 | 2 |
| pJSMCR1/ETEC | TC | + | >64 | 8 | 32 | 64 | 0.0312 | >32 | >64 | >128 | 8 | 8 |
| pFORC81/ETEC | TC | + | >64 | 8 | 32 | 64 | 0.0312 | >32 | >64 | >128 | 8 | 8 |
| pFORC82/ETEC | TC | + | >64 | 8 | 32 | 64 | 0.0312 | >32 | >64 | >128 | 8 | 8 |
| EAEC (E. coli NCCP 14039) | WT | - | >64 | 16 | >128 | 4 | 0.0312 | 8 | 2 | >128 | 4 | 2 |
| pJSMCR1/EAEC | TC | + | >64 | 16 | >128 | 4 | 0.0312 | 8 | 2 | >128 | 8 | 4 |
| pFORC81/EAEC | TC | + | >64 | 16 | >128 | 4 | 0.0312 | 8 | 2 | >128 | 8 | 8 |
| pFORC82/EAEC | TC | + | >64 | 16 | >128 | 4 | 0.0312 | 8 | 2 | >128 | 8 | 8 |
| S. Typhimurium SL1344 | WT | - | 2 | 2 | 0.5 | 4 | 0.0156 | 4 | 2 | >128 | 4 | 4 |
| pJSMCR1/SL1344 | TC | + | 2 | 2 | 0.5 | 4 | 0.0156 | 4 | 2 | >128 | 8 | 8 |
| pFORC81/SL1344 | TC | + | 2 | 2 | 0.5 | 4 | 0.0156 | 4 | 2 | >128 | 8 | 8 |
| pFORC82/SL1344 | TC | + | 2 | 2 | 0.5 | 4 | 0.0156 | 4 | 2 | >128 | 8 | 8 |
| Klebsiella (isolate) | WT | - | >64 | 16 | >128 | >64 | >16 | >32 | 1 | >128 | 4 | 4 |
| pJSMCR1/Klebsiella | TC | + | >64 | 16 | >128 | >64 | >16 | >32 | 1 | >128 | 8 | 16 |
| pFORC81/Klebsiella | TC | + | >64 | 16 | >128 | >64 | >16 | >32 | 1 | >128 | 8 | 16 |
| pFORC82/Klebsiella | TC | + | >64 | 16 | >128 | >64 | >16 | >32 | 1 | >128 | 32 | 16 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.; Hwang, B.K.; Choi, H.; Wang, Y.; Choi, S.H.; Ryu, S.; Jeon, B. Characterization of mcr-1-Harboring Plasmids from Pan Drug-Resistant Escherichia coli Strains Isolated from Retail Raw Chicken in South Korea. Microorganisms 2019, 7, 344. https://doi.org/10.3390/microorganisms7090344
Kim J, Hwang BK, Choi H, Wang Y, Choi SH, Ryu S, Jeon B. Characterization of mcr-1-Harboring Plasmids from Pan Drug-Resistant Escherichia coli Strains Isolated from Retail Raw Chicken in South Korea. Microorganisms. 2019; 7(9):344. https://doi.org/10.3390/microorganisms7090344
Chicago/Turabian StyleKim, Jinshil, Bo Kyoung Hwang, HyeLim Choi, Yang Wang, Sang Ho Choi, Sangryeol Ryu, and Byeonghwa Jeon. 2019. "Characterization of mcr-1-Harboring Plasmids from Pan Drug-Resistant Escherichia coli Strains Isolated from Retail Raw Chicken in South Korea" Microorganisms 7, no. 9: 344. https://doi.org/10.3390/microorganisms7090344
APA StyleKim, J., Hwang, B. K., Choi, H., Wang, Y., Choi, S. H., Ryu, S., & Jeon, B. (2019). Characterization of mcr-1-Harboring Plasmids from Pan Drug-Resistant Escherichia coli Strains Isolated from Retail Raw Chicken in South Korea. Microorganisms, 7(9), 344. https://doi.org/10.3390/microorganisms7090344







