Bifidobacterial Transfer from Mother to Child as Examined by an Animal Model
Abstract
1. Introduction
2. Results and Discussion
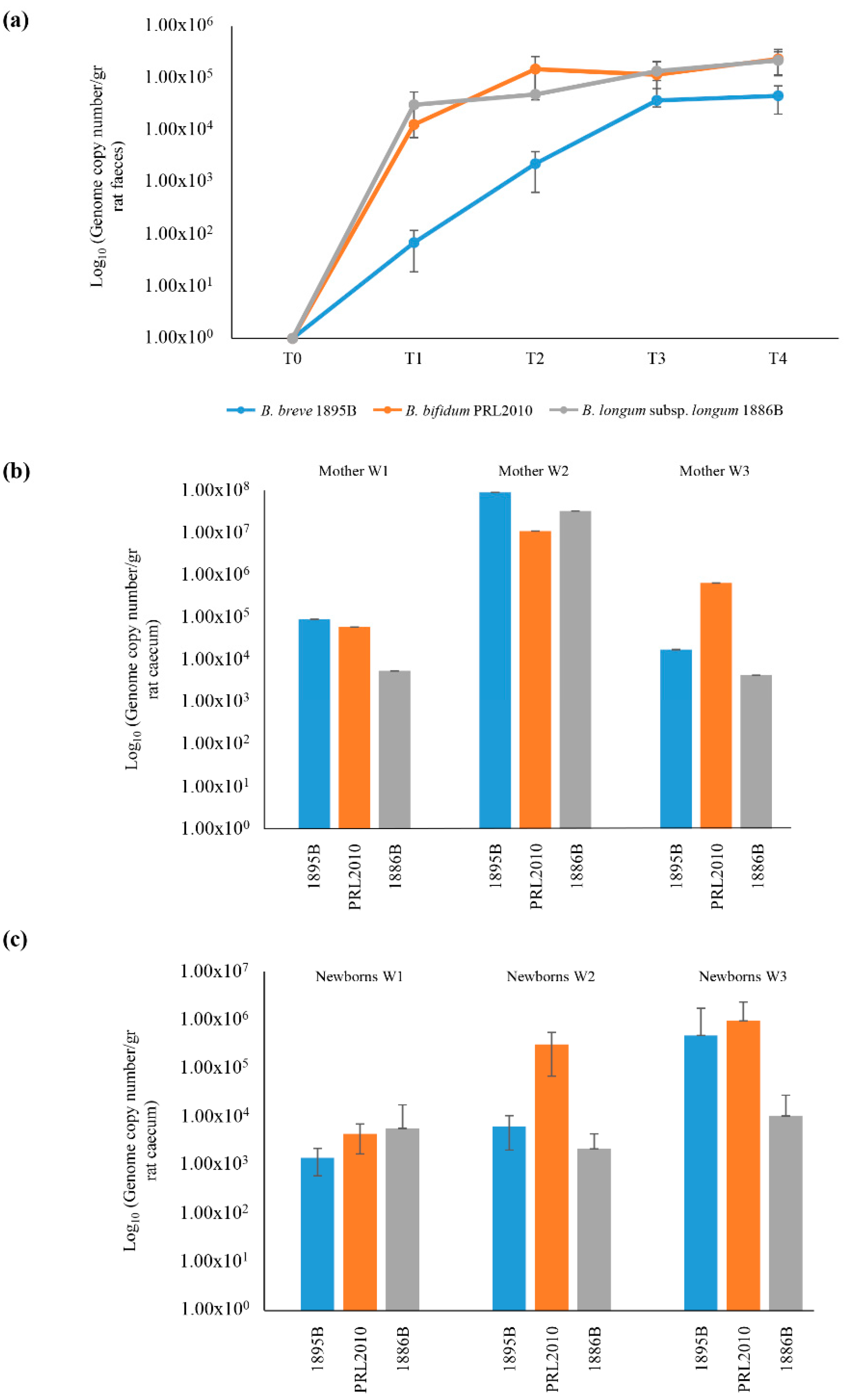
2.1. Evaluation of Vertical Transmission of Bifidobacteria under in Vivo Conditions
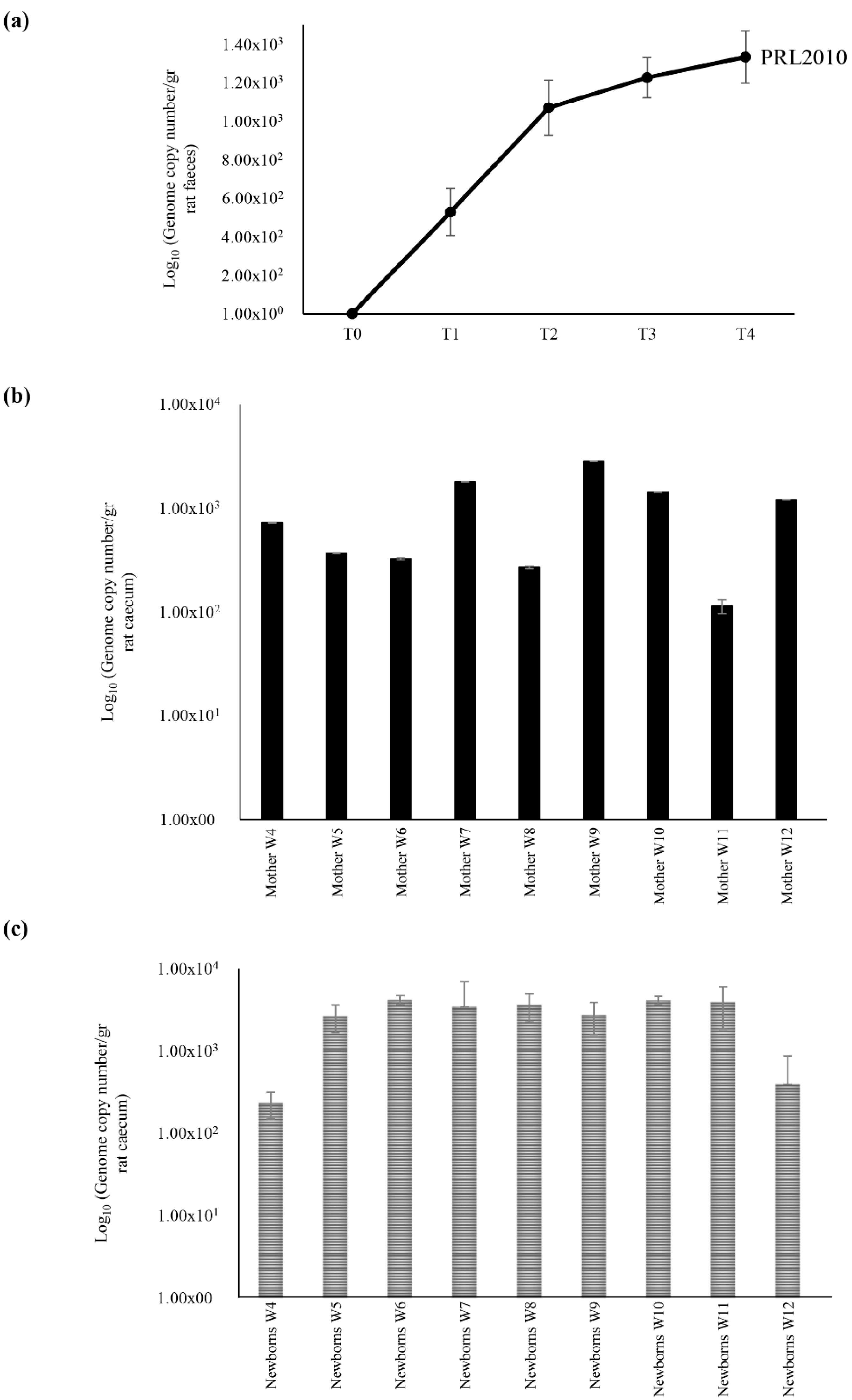
2.2. Maternal Inheritance of B. bifidum PRL2010 Strain
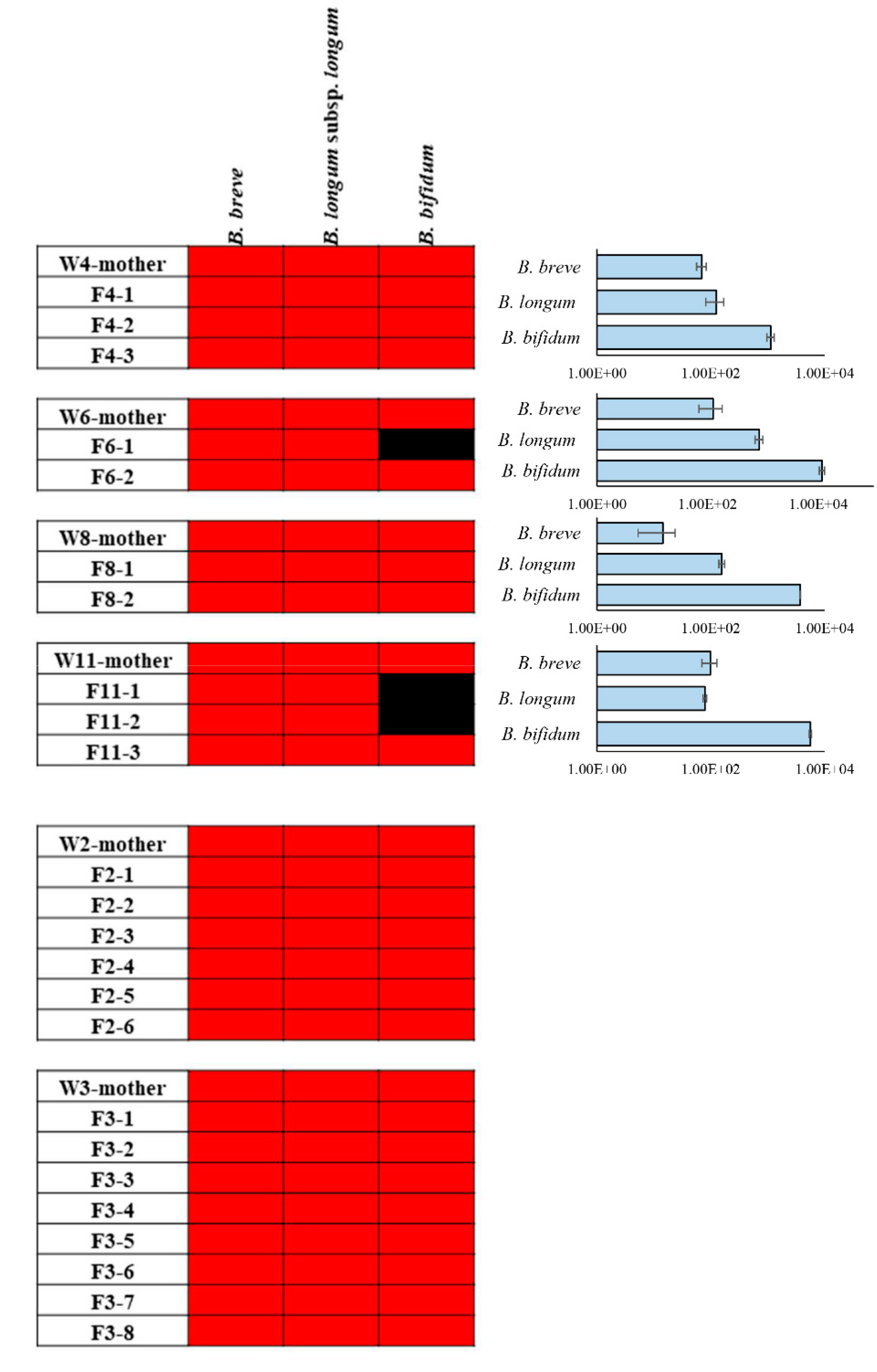
2.3. Identification of DNA Belonging to PRL2010 in Different Rat Body Sites
2.4. ITS Bifidobacterial Profiling of the Caecum of Mothers and Newborns
3. Materials and Methods
3.1. Experiment Design and Bifidobacterial Treatment of Rats
3.2. Bifidobacterium Strains Growth Conditions
3.3. DNA Extraction and qPCR
3.4. Bifidobacterium Strain Isolation From Mothers’ Caecum
3.5. Bifidobacterial ITS PCR Amplification and Sequencing
3.6. Data Deposition
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ventura, M.; Turroni, F.; Motherway, M.O.; MacSharry, J.; van Sinderen, D. Host-microbe interactions that facilitate gut colonization by commensal bifidobacteria. Trends Microbiol. 2012, 20, 467–476. [Google Scholar] [CrossRef] [PubMed]
- Turroni, F.; Milani, C.; Duranti, S.; Ferrario, C.; Lugli, G.A.; Mancabelli, L.; van Sinderen, D.; Ventura, M. Bifidobacteria and the infant gut: An example of co-evolution and natural selection. Cell. Mol. Life Sci. 2018, 75, 103–118. [Google Scholar] [CrossRef] [PubMed]
- Steed, H.; Macfarlane, G.T.; Blackett, K.L.; Macfarlane, S.; Miller, M.H.; Bahrami, B.; Dillon, J.F. Bacterial translocation in cirrhosis is not caused by an abnormal small bowel gut microbiota. FEMS Immunol. Med. Microbiol. 2011, 63, 346–354. [Google Scholar] [CrossRef] [PubMed]
- Vaishampayan, P.A.; Kuehl, J.V.; Froula, J.L.; Morgan, J.L.; Ochman, H.; Francino, M.P. Comparative metagenomics and population dynamics of the gut microbiota in mother and infant. Genome Biol. Evol. 2010, 2, 53–66. [Google Scholar] [CrossRef] [PubMed]
- Makino, H.; Kushiro, A.; Ishikawa, E.; Kubota, H.; Gawad, A.; Sakai, T.; Oishi, K.; Martin, R.; Ben-Amor, K.; Knol, J.; et al. Mother-to-infant transmission of intestinal bifidobacterial strains has an impact on the early development of vaginally delivered infant’s microbiota. PLoS ONE 2013, 8, e78331. [Google Scholar] [CrossRef] [PubMed]
- Makino, H.; Kushiro, A.; Ishikawa, E.; Muylaert, D.; Kubota, H.; Sakai, T.; Oishi, K.; Martin, R.; Ben Amor, K.; Oozeer, R.; et al. Transmission of intestinal bifidobacterium longum subsp. Longum strains from mother to infant, determined by multilocus sequencing typing and amplified fragment length polymorphism. Appl. Environ. Microbiol. 2011, 77, 6788–6793. [Google Scholar] [CrossRef] [PubMed]
- Milani, C.; Mancabelli, L.; Lugli, G.A.; Duranti, S.; Turroni, F.; Ferrario, C.; Mangifesta, M.; Viappiani, A.; Ferretti, P.; Gorfer, V.; et al. Exploring vertical transmission of bifidobacteria from mother to child. Appl. Environ. Microbiol. 2015, 81, 7078–7087. [Google Scholar] [CrossRef]
- Duranti, S.; Milani, C.; Lugli, G.A.; Turroni, F.; Mancabelli, L.; Sanchez, B.; Ferrario, C.; Viappiani, A.; Mangifesta, M.; Mancino, W.; et al. Insights from genomes of representatives of the human gut commensal bifidobacterium bifidum. Environ. Microbiol. 2015, 17, 2515–2531. [Google Scholar] [CrossRef]
- Duranti, S.; Lugli, G.A.; Milani, C.; James, K.; Mancabelli, L.; Turroni, F.; Alessandri, G.; Mangifesta, M.; Mancino, W.; Ossiprandi, M.C.; et al. Bifidobacterium bifidum and the infant gut microbiota: An intriguing case of microbe-host co-evolution. Environ. Microbiol. 2019. [Google Scholar] [CrossRef]
- Milani, C.; Turroni, F.; Duranti, S.; Lugli, G.A.; Mancabelli, L.; Ferrario, C.; van Sinderen, D.; Ventura, M. Genomics of the genus bifidobacterium reveals species-specific adaptation to the glycan-rich gut environment. Appl. Environ. Microbiol. 2016, 82, 980–991. [Google Scholar] [CrossRef]
- Milani, C.; Mangifesta, M.; Mancabelli, L.; Lugli, G.A.; James, K.; Duranti, S.; Turroni, F.; Ferrario, C.; Ossiprandi, M.C.; van Sinderen, D.; et al. Unveiling bifidobacterial biogeography across the mammalian branch of the tree of life. ISME J. 2017, 11, 2834–2847. [Google Scholar] [CrossRef]
- Moossavi, S.; Sepehri, S.; Robertson, B.; Bode, L.; Goruk, S.; Field, C.J.; Lix, L.M.; de Souza, R.J.; Becker, A.B.; Mandhane, P.J.; et al. Composition and variation of the human milk microbiota are influenced by maternal and early-life factors. Cell Host Microbe 2019, 25, 324–335. [Google Scholar] [CrossRef]
- Aagaard, K.; Ma, J.; Antony, K.M.; Ganu, R.; Petrosino, J.; Versalovic, J. The placenta harbors a unique microbiome. Sci. Transl. Med. 2014, 6, 237ra65. [Google Scholar] [CrossRef]
- Perez-Munoz, M.E.; Arrieta, M.C.; Ramer-Tait, A.E.; Walter, J. A critical assessment of the “sterile womb” and “in utero colonization” hypotheses: Implications for research on the pioneer infant microbiome. Microbiome 2017, 5, 48. [Google Scholar] [CrossRef]
- Rodriguez, J.M.; Murphy, K.; Stanton, C.; Ross, R.P.; Kober, O.I.; Juge, N.; Avershina, E.; Rudi, K.; Narbad, A.; Jenmalm, M.C.; et al. The composition of the gut microbiota throughout life, with an emphasis on early life. Microb. Ecol. Health Dis. 2015, 26, 26050. [Google Scholar] [CrossRef]
- Jimenez, E.; Marin, M.L.; Martin, R.; Odriozola, J.M.; Olivares, M.; Xaus, J.; Fernandez, L.; Rodriguez, J.M. Is meconium from healthy newborns actually sterile? Res. Microbiol. 2008, 159, 187–193. [Google Scholar] [CrossRef]
- Walker, R.W.; Clemente, J.C.; Peter, I.; Loos, R.J.F. The prenatal gut microbiome: Are we colonized with bacteria in utero? Pediatric Obes. 2017, 12, 3–17. [Google Scholar] [CrossRef]
- Turroni, F.; Bottacini, F.; Foroni, E.; Mulder, I.; Kim, J.H.; Zomer, A.; Sanchez, B.; Bidossi, A.; Ferrarini, A.; Giubellini, V.; et al. Genome analysis of bifidobacterium bifidum prl2010 reveals metabolic pathways for host-derived glycan foraging. Proc. Natl. Acad. Sci. USA 2010, 107, 19514–19519. [Google Scholar] [CrossRef]
- Duranti, S.; Lugli, G.A.; Mancabelli, L.; Armanini, F.; Turroni, F.; James, K.; Ferretti, P.; Gorfer, V.; Ferrario, C.; Milani, C.; et al. Maternal inheritance of bifidobacterial communities and bifidophages in infants through vertical transmission. Microbiome 2017, 5, 66. [Google Scholar] [CrossRef]
- Duranti, S.; Mancabelli, L.; Mancino, W.; Anzalone, R.; Longhi, G.; Statello, R.; Carnevali, L.; Sgoifo, A.; Bernasconi, S.; Turroni, F.; et al. Exploring the effects of colostrononi on the mammalian gut microbiota composition. PLoS ONE 2019, 14, e0217609. [Google Scholar] [CrossRef]
- Duranti, S.; Gaiani, F.; Mancabelli, L.; Milani, C.; Grandi, A.; Bolchi, A.; Santoni, A.; Lugli, G.A.; Ferrario, C.; Mangifesta, M.; et al. Elucidating the gut microbiome of ulcerative colitis: Bifidobacteria as novel microbial biomarkers. FEMS Microbiol. Ecol. 2016, 92, fiw191. [Google Scholar] [CrossRef]
- Egan, M.; Motherway, M.O.; Kilcoyne, M.; Kane, M.; Joshi, L.; Ventura, M.; van Sinderen, D. Cross-feeding by bifidobacterium breve ucc2003 during co-cultivation with bifidobacterium bifidum prl2010 in a mucin-based medium. BMC Microbiol. 2014, 14, 282. [Google Scholar] [CrossRef]
- Ferrario, C.; Duranti, S.; Milani, C.; Mancabelli, L.; Lugli, G.A.; Turroni, F.; Mangifesta, M.; Viappiani, A.; Ossiprandi, M.C.; van Sinderen, D.; et al. Exploring amino acid auxotrophy in bifidobacterium bifidum prl2010. Front. Microbiol. 2015, 6, 1331. [Google Scholar] [CrossRef]
- Turroni, F.; Serafini, F.; Foroni, E.; Duranti, S.; O’Connell Motherway, M.; Taverniti, V.; Mangifesta, M.; Milani, C.; Viappiani, A.; Roversi, T.; et al. Role of sortase-dependent pili of bifidobacterium bifidum prl2010 in modulating bacterium-host interactions. Proc. Natl. Acad. Sci. USA 2013, 110, 11151–11156. [Google Scholar] [CrossRef]
- Serafini, F.; Strati, F.; Ruas-Madiedo, P.; Turroni, F.; Foroni, E.; Duranti, S.; Milano, F.; Perotti, A.; Viappiani, A.; Guglielmetti, S.; et al. Evaluation of adhesion properties and antibacterial activities of the infant gut commensal bifidobacterium bifidum prl2010. Anaerobe 2013, 21, 9–17. [Google Scholar] [CrossRef]
- Serafini, F.; Turroni, F.; Ruas-Madiedo, P.; Lugli, G.A.; Milani, C.; Duranti, S.; Zamboni, N.; Bottacini, F.; van Sinderen, D.; Margolles, A.; et al. Kefir fermented milk and kefiran promote growth of bifidobacterium bifidum prl2010 and modulate its gene expression. Int. J. Food Microbiol. 2014, 178, 50–59. [Google Scholar] [CrossRef]
- Turroni, F.; Taverniti, V.; Ruas-Madiedo, P.; Duranti, S.; Guglielmetti, S.; Lugli, G.A.; Gioiosa, L.; Palanza, P.; Margolles, A.; van Sinderen, D.; et al. Bifidobacterium bifidum prl2010 modulates the host innate immune response. Appl. Environ. Microbiol. 2014, 80, 730–740. [Google Scholar] [CrossRef]
- Turroni, F.; Foroni, E.; Montanini, B.; Viappiani, A.; Strati, F.; Duranti, S.; Ferrarini, A.; Delledonne, M.; van Sinderen, D.; Ventura, M. Global genome transcription profiling of bifidobacterium bifidum prl2010 under in vitro conditions and identification of reference genes for quantitative real-time pcr. Appl. Environ. Microbiol. 2011, 77, 8578–8587. [Google Scholar] [CrossRef]
- Turroni, F.; Strati, F.; Foroni, E.; Serafini, F.; Duranti, S.; van Sinderen, D.; Ventura, M. Analysis of predicted carbohydrate transport systems encoded by bifidobacterium bifidum prl2010. Appl. Environ. Microbiol. 2012, 78, 5002–5012. [Google Scholar] [CrossRef]
- Turroni, F.; Duranti, S.; Bottacini, F.; Guglielmetti, S.; Van Sinderen, D.; Ventura, M. Bifidobacterium bifidum as an example of a specialized human gut commensal. Front. Microbiol. 2014, 5, 437. [Google Scholar] [CrossRef]
- Turroni, F.; Serafini, F.; Mangifesta, M.; Arioli, S.; Mora, D.; van Sinderen, D.; Ventura, M. Expression of sortase-dependent pili of bifidobacterium bifidum prl2010 in response to environmental gut conditions. FEMS Microbiol. Lett. 2014, 357, 23–33. [Google Scholar] [CrossRef]
- Turroni, F.; Ozcan, E.; Milani, C.; Mancabelli, L.; Viappiani, A.; van Sinderen, D.; Sela, D.A.; Ventura, M. Glycan cross-feeding activities between bifidobacteria under in vitro conditions. Front. Microbiol. 2015, 6, 1030. [Google Scholar] [CrossRef]
- Turroni, F.; Milani, C.; Duranti, S.; Mancabelli, L.; Mangifesta, M.; Viappiani, A.; Lugli, G.A.; Ferrario, C.; Gioiosa, L.; Ferrarini, A.; et al. Deciphering bifidobacterial-mediated metabolic interactions and their impact on gut microbiota by a multi-omics approach. ISME J. 2016, 10, 1656–1668. [Google Scholar] [CrossRef]
- Turroni, F.; Milani, C.; van Sinderen, D.; Ventura, M. Genetic strategies for mucin metabolism in bifidobacterium bifidum prl2010: An example of possible human-microbe co-evolution. Gut Microbes 2011, 2, 183–189. [Google Scholar] [CrossRef]
- Jimenez, E.; Fernandez, L.; Marin, M.L.; Martin, R.; Odriozola, J.M.; Nueno-Palop, C.; Narbad, A.; Olivares, M.; Xaus, J.; Rodriguez, J.M. Isolation of commensal bacteria from umbilical cord blood of healthy neonates born by cesarean section. Curr. Microbiol. 2005, 51, 270–274. [Google Scholar] [CrossRef]
- Milani, C.; Lugli, G.A.; Turroni, F.; Mancabelli, L.; Duranti, S.; Viappiani, A.; Mangifesta, M.; Segata, N.; van Sinderen, D.; Ventura, M. Evaluation of bifidobacterial community composition in the human gut by means of a targeted amplicon sequencing (its) protocol. FEMS Ecol. 2014, 90, 493–503. [Google Scholar] [CrossRef]
- Ley, R.E.; Hamady, M.; Lozupone, C.; Turnbaugh, P.J.; Ramey, R.R.; Bircher, J.S.; Schlegel, M.L.; Tucker, T.A.; Schrenzel, M.D.; Knight, R.; et al. Evolution of mammals and their gut microbes. Science 2008, 320, 1647–1651. [Google Scholar] [CrossRef]
- Wolfensohn, S.; Lloyd, M. Handbook of Laboratory Animal Management and Welfare, 3rd ed.; Oxford University Press: London, UK, 2003; Volume 1, p. 416. [Google Scholar]
- Carnevali, L.; Montano, N.; Statello, R.; Coude, G.; Vacondio, F.; Rivara, S.; Ferrari, P.F.; Sgoifo, A. Social stress contagion in rats: Behavioural, autonomic and neuroendocrine correlates. Psychoneuroendocrinology 2017, 82, 155–163. [Google Scholar] [CrossRef]
- Alessandri, G.; Milani, C.; Duranti, S.; Mancabelli, L.; Ranjanoro, T.; Modica, S.; Carnevali, L.; Statello, R.; Bottacini, F.; Turroni, F.; et al. Ability of bifidobacteria to metabolize chitin-glucan and its impact on the gut microbiota. Sci. Rep. 2019, 9, 5755. [Google Scholar] [CrossRef]
- Ventura, M.; Elli, M.; Reniero, R.; Zink, R. Molecular microbial analysis of bifidobacterium isolates from different environments by the species-specific amplified ribosomal DNA restriction analysis (ardra). FEMS Microbiol. Ecol. 2001, 36, 113–121. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. Qiime allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Edgar, R.C. Search and clustering orders of magnitude faster than blast. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Bokulich, N.A.; Kaehler, B.D.; Rideout, J.R.; Dillon, M.; Bolyen, E.; Knight, R.; Huttley, G.A.; Gregory Caporaso, J. Optimizing taxonomic classification of marker-gene amplicon sequences with qiime 2’s q2-feature-classifier plugin. Microbiome 2018, 6, 90. [Google Scholar] [CrossRef]

| Experiment Groups | Samples Name | Input Reads | Final Reads |
|---|---|---|---|
| PG | W4-mother | 3423 | 3126 |
| PG | F4-1 | 2589 | 2386 |
| PG | F4-2 | 8551 | 8012 |
| PG | F4-3 | 4126 | 3776 |
| PG | W6-mother | 2078 | 1970 |
| PG | F6-1 | 2164 | 2036 |
| PG | F6-2 | 5050 | 4852 |
| PG | W8-mother | 1239 | 1201 |
| PG | F8-1 | 6365 | 5797 |
| PG | F8-2 | 4527 | 4050 |
| PG | W11-mother | 1009 | 942 |
| PG | F11-1 | 4106 | 3908 |
| PG | F11-2 | 2133 | 2036 |
| PG | F11-3 | 1692 | 1642 |
| MCG | W2-mother | 22,528 | 22,026 |
| MCG | F2-1 | 12,703 | 11,906 |
| MCG | F2-2 | 32,065 | 29,485 |
| MCG | F2-3 | 11,715 | 10,850 |
| MCG | F2-4 | 17,106 | 15,929 |
| MCG | F2-5 | 13,992 | 12,724 |
| MCG | F2-6 | 10,339 | 9739 |
| MCG | W3-mother | 2686 | 2640 |
| MCG | F3-1 | 4818 | 4620 |
| MCG | F3-2 | 8970 | 8489 |
| MCG | F3-3 | 5346 | 4987 |
| MCG | F3-4 | 5664 | 5373 |
| MCG | F3-5 | 2765 | 2584 |
| MCG | F3-6 | 12,523 | 11,182 |
| MCG | F3-7 | 6129 | 5701 |
| MCG | F3-8 | 8524 | 8065 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mancino, W.; Duranti, S.; Mancabelli, L.; Longhi, G.; Anzalone, R.; Milani, C.; Lugli, G.A.; Carnevali, L.; Statello, R.; Sgoifo, A.; et al. Bifidobacterial Transfer from Mother to Child as Examined by an Animal Model. Microorganisms 2019, 7, 293. https://doi.org/10.3390/microorganisms7090293
Mancino W, Duranti S, Mancabelli L, Longhi G, Anzalone R, Milani C, Lugli GA, Carnevali L, Statello R, Sgoifo A, et al. Bifidobacterial Transfer from Mother to Child as Examined by an Animal Model. Microorganisms. 2019; 7(9):293. https://doi.org/10.3390/microorganisms7090293
Chicago/Turabian StyleMancino, Walter, Sabrina Duranti, Leonardo Mancabelli, Giulia Longhi, Rosaria Anzalone, Christian Milani, Gabriele Andrea Lugli, Luca Carnevali, Rosario Statello, Andrea Sgoifo, and et al. 2019. "Bifidobacterial Transfer from Mother to Child as Examined by an Animal Model" Microorganisms 7, no. 9: 293. https://doi.org/10.3390/microorganisms7090293
APA StyleMancino, W., Duranti, S., Mancabelli, L., Longhi, G., Anzalone, R., Milani, C., Lugli, G. A., Carnevali, L., Statello, R., Sgoifo, A., van Sinderen, D., Ventura, M., & Turroni, F. (2019). Bifidobacterial Transfer from Mother to Child as Examined by an Animal Model. Microorganisms, 7(9), 293. https://doi.org/10.3390/microorganisms7090293







