The Anti-Rotaviral Activity of Low Molecular Weight and Non-Proteinaceous Substance from Bifidobacterium longum BORI Cell Extract
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells, Viruses, and Bacteria
2.2. Preparation of Cell Extract of Tested Bacteria
2.3. Antiviral Assay
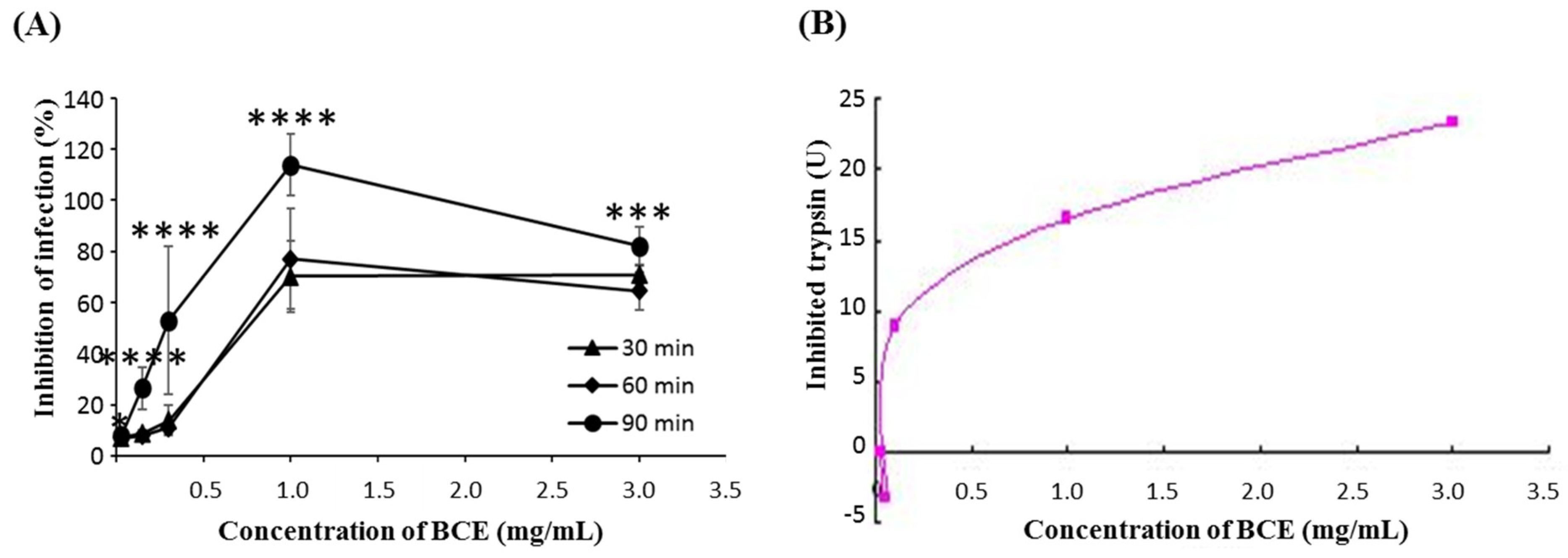
2.4. Colorimetric Trypsin Inhibitor Assay
2.5. Statistical Analysis
3. Results
3.1. Anti-Rotaviral Effect of BCE on Rotavirus
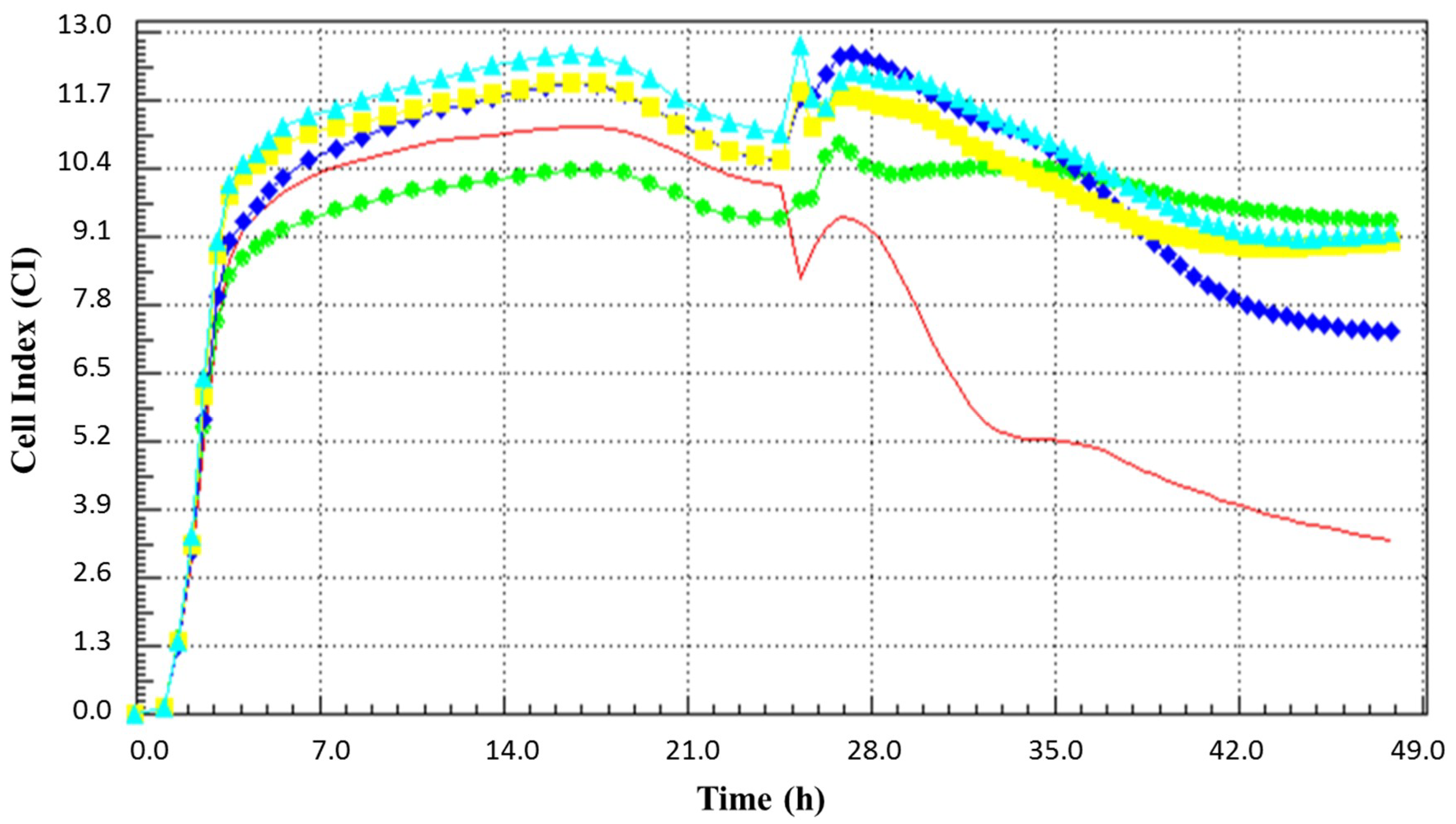
3.2. Anti-Rotaviral Effect of Heat and Proteolytic Enzyme Treated BCE
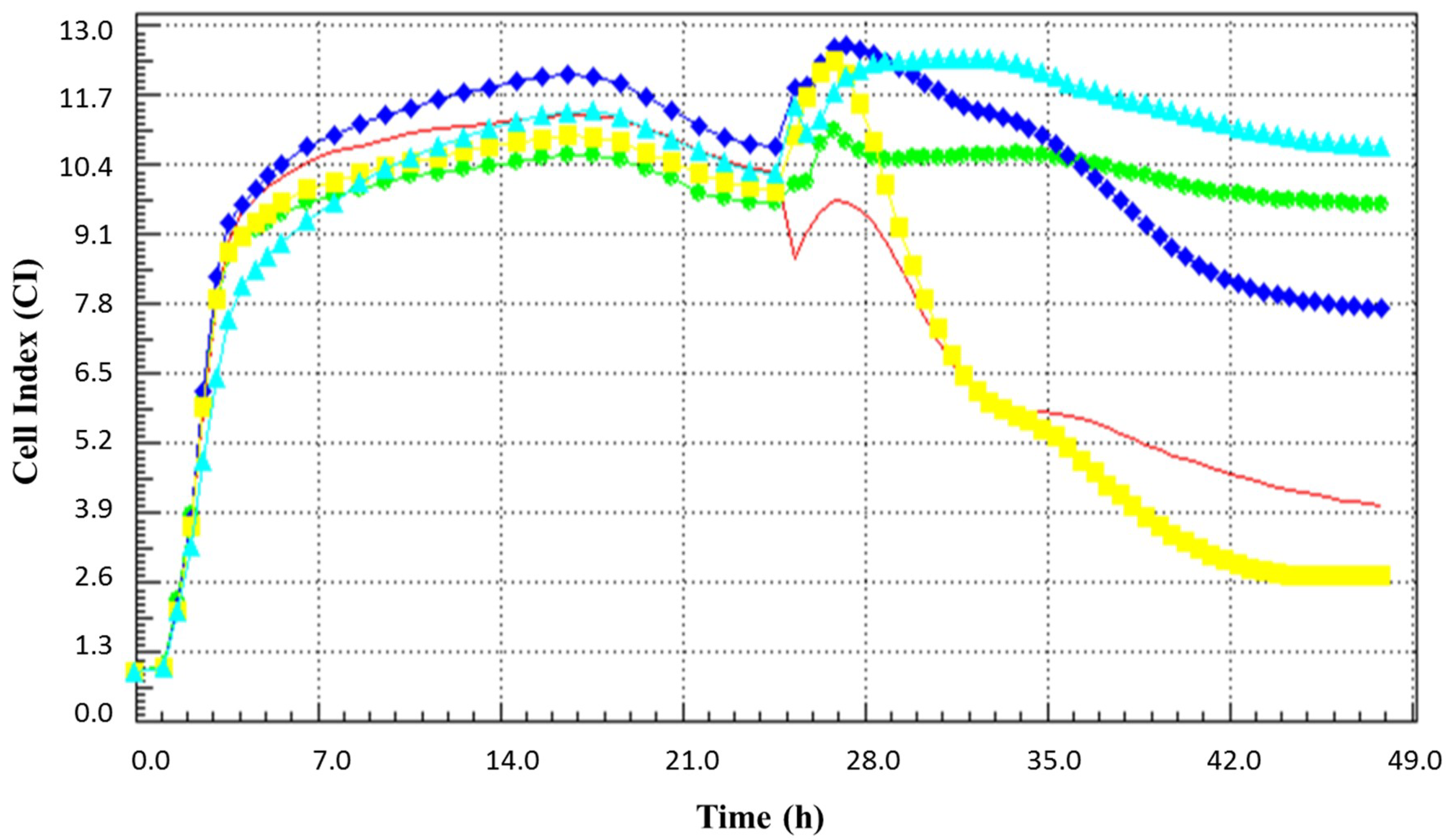
3.3. The Antiviral Effect of BCE Fractions Divided by the Molecular Weight
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Crawford, S.E.; Ramani, S.; Tate, J.E.; Parashar, U.D.; Svensson, L.; Hagbom, M.; Franco, M.A.; Greenberg, H.B.; O’Ryan, M.; Kang, G.; et al. Desselberger, Rotavirus infection. Nat. Rev. Dis. Primers 2017, 3, 17083. [Google Scholar] [CrossRef] [PubMed]
- Tate, J.E.; Burton, A.H.; Boschi-Pinto, C.; Parashar, U.D.; World Health Organization-Coordinated Global Rotavirus Surveillance Network. Global, Regional, and National Estimates of Rotavirus Mortality in Children <5 Years of Age, 2000–2013. Clin. Infect. Dis. 2016, 62, S96–S105. [Google Scholar] [PubMed]
- Guarino, A.; Dupont, C.; Gorelov, A.V.; Gottrand, F.; Lee, J.K.; Lin, Z.; Lo Vecchio, A.; Nguyen, T.D.; Salazar-Lindo, E. The management of acute diarrhea in children in developed and developing areas: From evidence base to clinical practice. Expert Opin. Pharmacother. 2012, 13, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Kapikian, A.Z. A rotavirus vaccine for prevention of severe diarrhoea of infants and young children: development, utilization and withdrawal. Novartis Found Symp. 2001, 238, 153–171; discussion 171–179. [Google Scholar] [PubMed]
- Patel, M.M.; Haber, P.; Baggs, J.; Zuber, P.; Bines, J.E.; Parashar, U.D. Intussusception and rotavirus vaccination: A review of the available evidence. Expert Rev. Vaccines 2009, 8, 1555–1564. [Google Scholar] [CrossRef] [PubMed]
- Burnett, E.; Tate, J.E.; Kirkwood, C.D.; Nelson, E.A.S.; Santosham, M.; Steele, A.D.; Parashar, U.D. Estimated impact of rotavirus vaccine on hospitalizations and deaths from rotavirus diarrhea among children <5 in Asia. Expert Rev. Vaccines 2018, 17, 453–460. [Google Scholar] [PubMed]
- Fernandez-Duarte, K.P.; Olaya-Galan, N.N.; Salas-Cardenas, S.P.; Lopez-Rozo, J.; Gutierrez-Fernandez, M.F. Bifidobacterium adolescentis (DSM 20083) and Lactobacillus casei (Lafti L26-DSL): Probiotics Able to Block the In Vitro Adherence of Rotavirus in MA104 Cells. Probiotics Antimicrob. Proteins 2018, 10, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Rigo-Adrover, M.D.M.; Knipping, K.; Garssen, J.; van Limpt, K.; Knol, J.; Franch, A.; Castell, M.; Rodriguez-Lagunas, M.J.; Perez-Cano, F.J. Prevention of Rotavirus Diarrhea in Suckling Rats by a Specific Fermented Milk Concentrate with Prebiotic Mixture. Nutrients 2019, 11, 189. [Google Scholar] [CrossRef] [PubMed]
- Rigo-Adrover, M.D.M.; van Limpt, K.; Knipping, K.; Garssen, J.; Knol, J.; Costabile, A.; Franch, A.; Castell, M.; Perez-Cano, F.J. Preventive Effect of a Synbiotic Combination of Galacto- and Fructooligosaccharides Mixture With Bifidobacterium breve M-16V in a Model of Multiple Rotavirus Infections. Front. Immunol. 2018, 9, 1318. [Google Scholar] [CrossRef] [PubMed]
- Park, M.S.; Kwon, B.; Ku, S.; Ji, G.E. The Efficacy of Bifidobacterium longum BORI and Lactobacillus acidophilus AD031 Probiotic Treatment in Infants with Rotavirus Infection. Nutrients 2017, 9, 887. [Google Scholar] [CrossRef] [PubMed]
- James, K.A.C.; Butts, C.A.; Morrison, S.C.; Koolaard, J.P.; Scott, M.F.; Scott, R.E.; Griffin, W.B.; Bang, L.M. The effects of cultivar and heat treatment on protein quality and trypsin inhibitor content of New Zealand field peas. New Zeal. J. Agr. Res. 2005, 48, 117–124. [Google Scholar] [CrossRef]
- Clark, S.M.; Roth, J.R.; Clark, M.L.; Barnett, B.B.; Spendlove, R.S. Trypsin enhancement of rotavirus infectivity: mechanism of enhancement. J. Virol. 1981, 39, 816–822. [Google Scholar] [PubMed]
- Kim, M.J.; Ku, S.; Kim, S.Y.; Lee, H.H.; Jin, H.; Kang, S.; Li, R.; Johnston, T.V.; Park, M.S.; Ji, G.E. Safety Evaluations of Bifidobacterium bifidum BGN4 and Bifidobacterium longum BORI. Int. J. Mol. Sci. 2018, 19, 1422. [Google Scholar] [CrossRef] [PubMed]
- Benureau, Y.; Huet, J.C.; Charpilienne, A.; Poncet, D.; Cohen, J. Trypsin is associated with the rotavirus capsid and is activated by solubilization of outer capsid proteins. J. Gen. Virol. 2005, 86, 3143–3151. [Google Scholar] [CrossRef] [PubMed]
- Atienza, J.M.; Yu, N.; Kirstein, S.L.; Xi, B.; Wang, X.; Xu, X.; Abassi, Y.A. Dynamic and label-free cell-based assays using the real-time cell electronic sensing system. Assay. Drug Dev. Technol. 2006, 4, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Desselberger, U. Rotaviruses. Virus Res. 2014, 190, 75–96. [Google Scholar] [CrossRef] [PubMed]
- Salas-Cárdenas, S.P.; Olaya-Galán, N.N.; Fernández, K.; Velez, F.; Guerrero, C.A.; Gutiérrez, M.F. Decreased rotavirus infection of MA104 cells via probiotic extract binding to Hsc70 and ß3 integrin receptors. Univ. Sci. 2002, 23, 219–239. [Google Scholar] [CrossRef]
- Chenoll, E.; Casinos, B.; Bataller, E.; Buesa, J.; Ramon, D.; Genoves, S.; Fabrega, J.; Rivero Urgell, M.; Moreno Munoz, J.A. Identification of a Peptide Produced by Bifidobacterium longum CECT 7210 with Antirotaviral Activity. Front. Microbiol. 2016, 7, 655. [Google Scholar] [CrossRef] [PubMed]
- Olaya Galan, N.N.; Ulloa Rubiano, J.C.; Velez Reyes, F.A.; Fernandez Duarte, K.P.; Salas Cardenas, S.P.; Gutierrez Fernandez, M.F. In vitro antiviral activity of Lactobacillus casei and Bifidobacterium adolescentis against rotavirus infection monitored by NSP4 protein production. J. Appl. Microbiol. 2016, 120, 1041–1051. [Google Scholar] [CrossRef] [PubMed]
- Bae, E.-A.; Han, M.J.; Song, M.-J.; Kim, D.-H. Purification of Rotavirus Infection—Inhibitory Protein from Bifidobacterium breve K—110. J. Microbiol. Biotechnol. 2002, 12, 553–556. [Google Scholar]

| Strain | The Number of Infected Wells (per 8 Wells) | The Inhibition of Infection (%) |
|---|---|---|
| B. longum RD43 | 2/8 | 75 |
| B. longum RD61 | 3/8 | 62.5 |
| B. longum RD65 | 6/8 | 25 |
| B. longum RD69 | 2/8 | 75 |
| B. longum RD118 | 7/8 | 12.5 |
| B. longum RD138 | 4/8 | 50 |
| B. longum BORI | 1/8 | 87.5 |
| Enzyme Treatment | The Number of Infected Wells (per 8 Wells) | Inhibition Rate (%) |
|---|---|---|
| Trypsin + BCE | 2/8 | 75 |
| Proteinase K + BCE | 0/8 | 100 |
| BCE only (0.25 mg/mL) | 0/8 | 100 |
| Heat inactivated Trypsin and Proteinase K | 8/8 | 0 |
| Negative control | 8/8 | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, Y.O.; Jeong, Y.; You, H.J.; Ku, S.; Ji, G.E.; Park, M.S. The Anti-Rotaviral Activity of Low Molecular Weight and Non-Proteinaceous Substance from Bifidobacterium longum BORI Cell Extract. Microorganisms 2019, 7, 108. https://doi.org/10.3390/microorganisms7040108
Han YO, Jeong Y, You HJ, Ku S, Ji GE, Park MS. The Anti-Rotaviral Activity of Low Molecular Weight and Non-Proteinaceous Substance from Bifidobacterium longum BORI Cell Extract. Microorganisms. 2019; 7(4):108. https://doi.org/10.3390/microorganisms7040108
Chicago/Turabian StyleHan, Yeo Ok, Yunju Jeong, Hyun Ju You, Seockmo Ku, Geun Eog Ji, and Myeong Soo Park. 2019. "The Anti-Rotaviral Activity of Low Molecular Weight and Non-Proteinaceous Substance from Bifidobacterium longum BORI Cell Extract" Microorganisms 7, no. 4: 108. https://doi.org/10.3390/microorganisms7040108
APA StyleHan, Y. O., Jeong, Y., You, H. J., Ku, S., Ji, G. E., & Park, M. S. (2019). The Anti-Rotaviral Activity of Low Molecular Weight and Non-Proteinaceous Substance from Bifidobacterium longum BORI Cell Extract. Microorganisms, 7(4), 108. https://doi.org/10.3390/microorganisms7040108







