Bacillus subtilis DSM 32315 Supplementation Attenuates the Effects of Clostridium perfringens Challenge on the Growth Performance and Intestinal Microbiota of Broiler Chickens
Abstract
:1. Introduction
2. Materials and Methods
2.1. Birds, Housing and Treatments
2.2. Necrotic Enteritis Experimental Model
2.3. Data Collection, Sampling Procedures and Laboratory Assays
2.4. Intestinal Content Total DNA
2.5. PCR Amplification and Sequencing
2.6. Bioinformatics
2.7. Statistical Analysis
3. Results
3.1. Growth Performance and Necrotic Enteritis Lesion
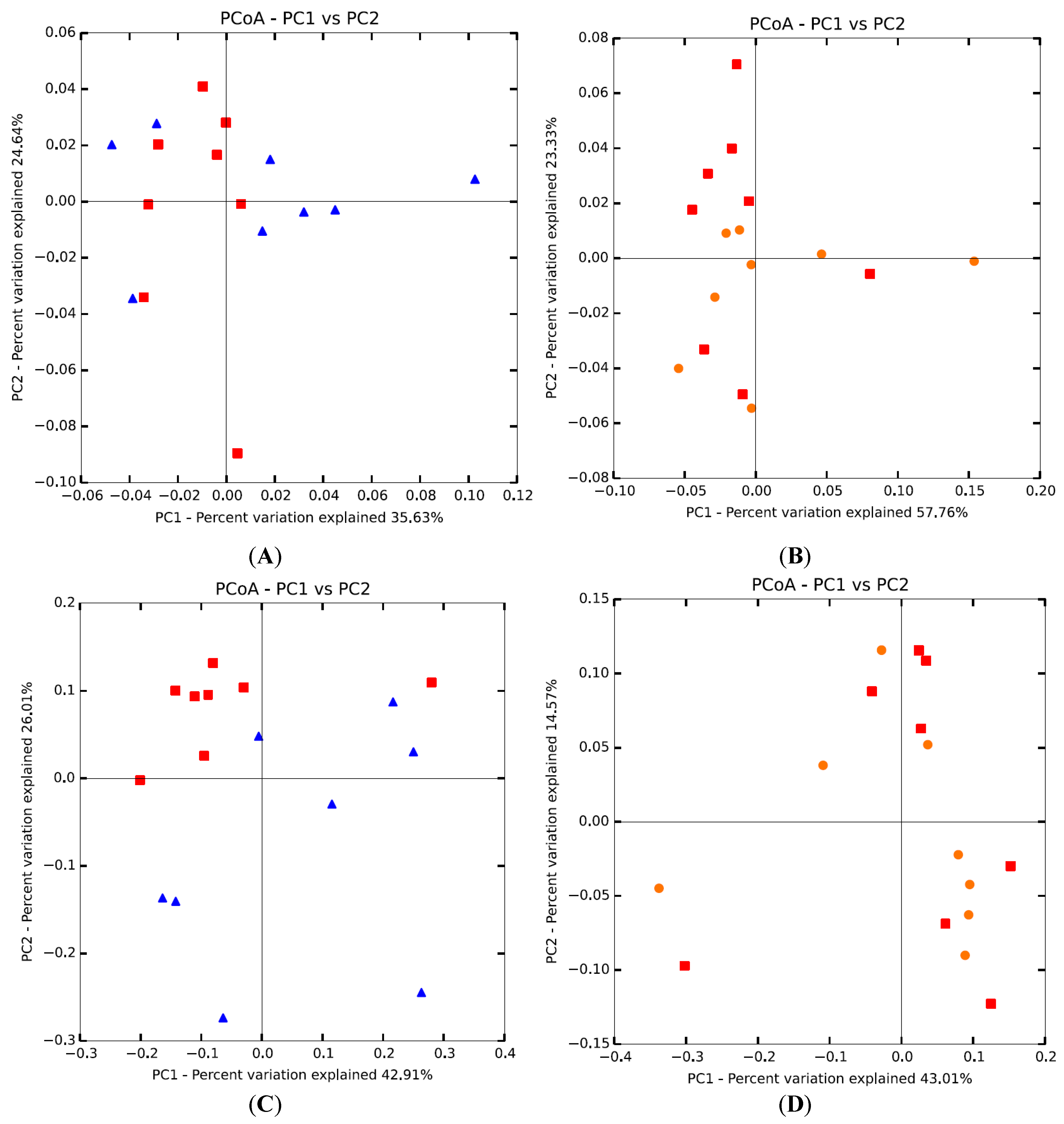
3.2. Microbial Diversity of Intestinal Microbiota
3.3. Microbial Composition of Intestinal Microbiota
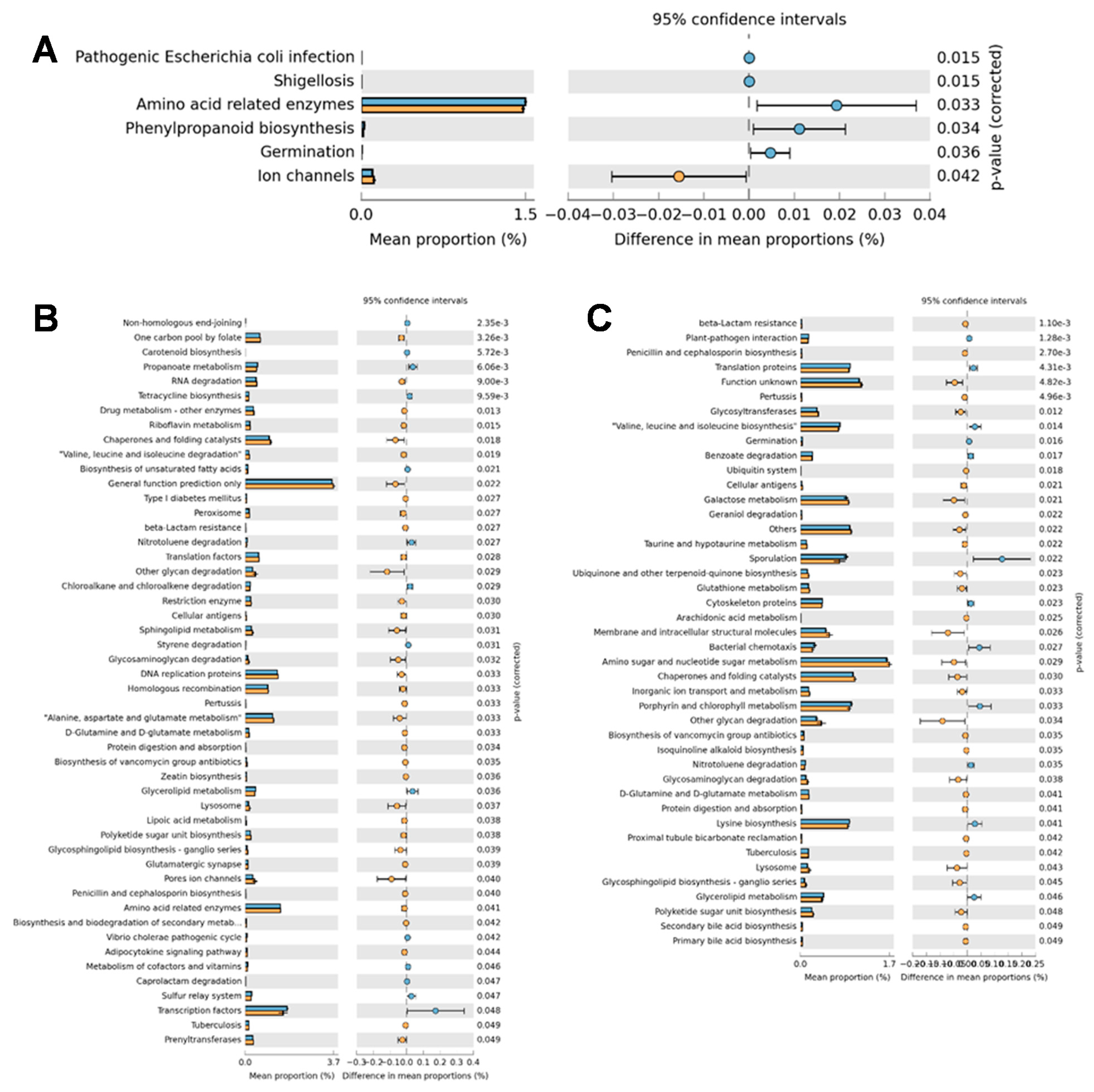
3.4. Predicted Function of Intestinal Microbiota
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Immerseel, F.V.; Buck, J.D.; Pasmans, F.; Huyghebaert, G.; Haesebrouck, F.; Ducatelle, R. Clostridium perfringens in poultry: An emerging threat for animal and public health. Avian Pathol. 2004, 33, 537–549. [Google Scholar] [CrossRef] [PubMed]
- Kiu, R.; Hall, L.J. An update on the human and animal enteric pathogen Clostridium perfringens. Emerg. Microbes Infect. 2018, 7, 141. [Google Scholar] [CrossRef] [PubMed]
- Shojadoost, B.; Vince, A.R.; Prescott, J.F. The successful experimental induction of necrotic enteritis in chickens by Clostridium perfringens: A critical review. Vet. Res. 2012, 43, 74. [Google Scholar] [CrossRef] [PubMed]
- Moore, R.J. Necrotic enteritis predisposing factors in broiler chickens. Avian Pathol. 2016, 45, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Cooper, K.K.; Songer, J.G.; Uzal, F.A. Diagnosing clostridial enteric disease in poultry. J. Vet. Diagn. Invest. 2013, 25, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Smyth, J.A. Pathology and diagnosis of necrotic enteritis: is it clear-cut? Avian Pathol. 2016, 45, 282–287. [Google Scholar] [CrossRef] [PubMed]
- Cooper, K.K.; Songer, J.G. Necrotic enteritis in chickens: A paradigm of enteric infection by Clostridium perfringens type A. Anaerobe 2009, 15, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Hofacre, C.L.; Smith, J.A.; Mathis, G.F. An optimist’s view on limiting necrotic enteritis and maintaining broiler gut health and performance in today’s marketing, food safety and regulatory climate. Poult. Sci. 2018, 97, 1929–1933. [Google Scholar] [CrossRef] [PubMed]
- Wade, B.; Keyburn, A. The true cost of necrotic enteritis. World Poult. 2015, 31, 16–17. [Google Scholar]
- M’Sadeq, S.A.; Wu, S.; Swick, R.A.; Choct, M. Towards the control of necrotic enteritis in broiler chickens with in-feed antibiotics phasing-out worldwide. Anim. Nutr. 2015, 1, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Prescott, J.F.; Parreira, V.R.; Mehdizadeh Gohari, I.; Lepp, D.; Gong, J. The pathogenesis of necrotic enteritis in chickens: What we know and what we need to know: A review. Avian Pathol. 2016, 45, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Caly, D.L.; D’Inca, R.; Auclair, E.; Drider, D. Alternatives to antibiotics to prevent necrotic enteritis in broiler chickens: A microbiologist’s perspective. Front. Microbiol. 2015, 6, 1336. [Google Scholar] [CrossRef] [PubMed]
- Al-Khalaifah, H.S. Benefits of probiotics and/or prebiotics for antibiotic-reduced poultry. Poult. Sci. 2018, 97, 3807–3815. [Google Scholar] [CrossRef] [PubMed]
- Ma, T.; Suzuki, Y.; Guan, L.L. Dissect the mode of action of probiotics in affecting host-microbial interactions and immunity in food producing animals. Vet. Immunol. Immunopathol. 2018, 205, 35–48. [Google Scholar] [CrossRef] [PubMed]
- Whelan, R.A.; Doranalli, K.; Rinttil, T.; Vienola, K.; Jurgens, G.; Apajalahti, J. The impact of Bacillus subtilis DSM 32315 on the pathology, performance and intestinal microbiome of broiler chickens in a necrotic enteritis challenge. Poult. Sci. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Wang, W.; Zhang, H.; Wang, J.; Zhang, W.; Gao, J.; Wu, S.; Qi, G. Supplemental Bacillus subtilis DSM 32315 manipulates intestinal structure and microbial composition in broiler chickens. Sci. Rep. 2018, 8, 15358. [Google Scholar] [CrossRef] [PubMed]
- Kosin, B.; Rakshit, S.K. Microbial and processing criteria for production of probiotics: A review. Food Technol. Biotechnol. 2006, 44, 371–379. [Google Scholar]
- Angelakis, E. Weight gain by gut microbiota manipulation in productive animals. Microb. Pathog. 2017, 106, 162–170. [Google Scholar] [CrossRef] [PubMed]
- La Ragione, R. Competitive exclusion by Bacillus subtilis spores of Salmonella enterica serotype Enteritidis and Clostridium perfringens in young chickens. Vet. Microbiol. 2003, 94, 245–256. [Google Scholar] [CrossRef]
- Poormontaseri, M.; Hosseinzadeh, S.; Shekarforoush, S.S.; Kalantari, T. The effects of probiotic Bacillus subtilis on the cytotoxicity of Clostridium perfringens type a in Caco-2 cell culture. BMC Microbiol. 2017, 17, 150. [Google Scholar] [CrossRef] [PubMed]
- Elshaghabee, F.M.F.; Rokana, N.; Gulhane, R.D.; Sharma, C.; Panwar, H. Bacillus as potential probiotics: Status, concerns and future perspectives. Front. Microbiol. 2017, 8, 1490. [Google Scholar] [CrossRef] [PubMed]
- Burkard, M.; Entian, K.-D.; Stein, T. Development and application of a microtiter plate-based autoinduction bioassay for detection of the lantibiotic subtilin. J. Microbiol. Methods 2007, 70, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, S.W.; Jaskolla, T.W.; Bochmann, S.; Kötter, P.; Wichelhaus, T.; Karas, M.; Stein, T.; Entian, K.-D. Entianin, a novel subtilin-like lantibiotic from Bacillus subtilis subsp. spizizenii DSM 15029 T with high antimicrobial activity. Appl. Environ. Microbiol. 2011, 77, 1698–1707. [Google Scholar] [CrossRef] [PubMed]
- Grant, A.; Gay, C.G.; Lillehoj, H.S. Bacillus spp. as direct-fed microbial antibiotic alternatives to enhance growth, immunity and gut health in poultry. Avian Pathol. 2018, 47, 339–351. [Google Scholar] [CrossRef] [PubMed]
- McGlone, J. Guide for the Care and Use of Agricultural Animals in Research and Teaching, 3rd ed. Available online: https://www.aaalac.org/about/ag_guide_3rd_ed.pdf (accessed on 4 March 2019).
- Recommendations for Broilers, Amino Acids and More. Available online: https://animal-nutrition.evonik.com/product/feed-additives/downloads/recommendations_broiler_03_16.pdf (accessed on 4 March 2019).
- Hofacre, C.L.; Froyman, R.; Gautrias, B.; George, B.; Goodwin, M.A.; Brown, J. Use of Aviguard and other intestinal bioproducts in experimental Clostridium perfringens-associated necrotizing enteritis in broiler chickens. Avian Dis. 1998, 42, 579–584. [Google Scholar] [CrossRef] [PubMed]
- Bortoluzzi, C.; Pedroso, A.A.; Mallo, J.J.; Puyalto, M.; Kim, W.K.; Applegate, T.J. Sodium butyrate improved performance while modulating the cecal microbiota and regulating the expression of intestinal immune-related genes of broiler chickens. Poult. Sci. 2017, 96, 3981–3993. [Google Scholar] [CrossRef] [PubMed]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acids Res. 2013, 41, e1. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Pruesse, E.; Quast, C.; Knittel, K.; Fuchs, B.M.; Ludwig, W.; Peplies, J.; Glockner, F.O. SILVA: A comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res. 2007, 35, 7188–7196. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [PubMed]
- Gihring, T.M.; Green, S.J.; Schadt, C.W. Massively parallel rRNA gene sequencing exacerbates the potential for biased community diversity comparisons due to variable library sizes: Pyrosequencing exacerbates sample size bias. Environ. Microbiol. 2012, 14, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Langille, M.G.I.; Zaneveld, J.; Caporaso, J.G.; McDonald, D.; Knights, D.; Reyes, J.A.; Clemente, J.C.; Burkepile, D.E.; Vega Thurber, R.L.; Knight, R.; et al. Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat. Biotechnol. 2013, 31, 814–821. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Goto, S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef] [PubMed]
- Parks, D.H.; Tyson, G.W.; Hugenholtz, P.; Beiko, R.G. STAMP: Statistical analysis of taxonomic and functional profiles. Bioinformatics 2014, 30, 3123–3124. [Google Scholar] [CrossRef] [PubMed]
- Shang, Y.; Kumar, S.; Oakley, B.; Kim, W.K. Chicken gut microbiota: Importance and detection technology. Front. Vet. Sci. 2018, 5, 254. [Google Scholar] [CrossRef] [PubMed]
- Mosca, A.; Leclerc, M.; Hugot, J.P. Gut microbiota diversity and human diseases: Should we reintroduce key predators in our ecosystem? Front. Microbiol. 2016, 7, 455. [Google Scholar] [CrossRef] [PubMed]
- Borda-Molina, D.; Seifert, J.; Camarinha-Silva, A. Current perspectives of the chicken gastrointestinal tract and its microbiome. Comput. Struct. Biotechnol. J. 2018, 16, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Chang, Q.; Luan, Y.; Sun, F. Variance adjusted weighted UniFrac: A powerful beta diversity measure for comparing communities based on phylogeny. BMC Bioinforma. 2011, 12, 118. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Idris, U.; Harmon, B.; Hofacre, C.; Maurer, J.J.; Lee, M.D. Diversity and succession of the intestinal bacterial community of the maturing broiler chicken. Appl. Environ. Microbiol. 2003, 69, 6816–6824. [Google Scholar] [CrossRef] [PubMed]
- Biddle, A.; Stewart, L.; Blanchard, J.; Leschine, S. Untangling the genetic basis of fibrolytic specialization by Lachnospiraceae and Ruminococcaceae in diverse gut communities. Diversity 2013, 5, 627–640. [Google Scholar] [CrossRef]
- Qaisrani, S.N.; van Krimpen, M.M.; Kwakkel, R.P.; Verstegen, M.W.A.; Hendriks, W.H. Dietary factors affecting hindgut protein fermentation in broilers: A review. Worlds Poult. Sci. J. 2015, 71, 139–160. [Google Scholar] [CrossRef]

| Ingredients (%) | Starter (d 1–14) | Grower (d 15–28) | Finisher (d 29–42) |
|---|---|---|---|
| Corn | 50.62 | 58.78 | 60.85 |
| Soybean meal, 48% CP | 32.30 | 23.86 | 20.99 |
| DDGS | 8.00 | 8.00 | 8.00 |
| Meat and Bone meal | 4.00 | 4.00 | 4.00 |
| Soybean oil | 2.67 | 2.87 | 3.97 |
| Dicalcium phosphate | 1.12 | 1.14 | 1.16 |
| Limestone (CaCO3) | 0.35 | 0.40 | 0.15 |
| NaCl | 0.252 | 0.257 | 0.258 |
| dl-Met | 0.278 | 0.219 | 0.196 |
| l-Lys HCl | 0.197 | 0.202 | 0.177 |
| l-Thr | 0.054 | 0.047 | 0.037 |
| Choline chrolide, 60% | 0.100 | 0.100 | 0.100 |
| Vitamin premix 1 | 0.050 | 0.050 | 0.050 |
| Trace mineral premix 2 | 0.075 | 0.075 | 0.075 |
| Nutrients (%, unless otherwise indicated) | |||
| AMEn (kcal/kg) | 2950 | 3050 | 3150 |
| Crude protein | 23.85 | 20.44 | 19.19 |
| SID Lys | 1.22 | 1.02 | 0.93 |
| SID Met+Cys | 0.89 | 0.76 | 0.71 |
| SID Thr | 0.78 | 0.66 | 0.61 |
| SID Trp | 0.23 | 0.18 | 0.17 |
| SID Arg | 1.39 | 1.15 | 1.06 |
| SID Ile | 0.85 | 0.71 | 0.66 |
| SID Leu | 1.79 | 1.59 | 1.52 |
| SID Val | 0.95 | 0.81 | 0.76 |
| Treatment | NE Lesion Score | BW Gain (g) | FI (g) | FCR (g/g) | BW Gain (g) | FI (g) | FCR (g/g) | Overall Mort, % | |
|---|---|---|---|---|---|---|---|---|---|
| 21 d | 28 d | 1–28 d | 1–42 d | ||||||
| Uninfected | 0.00 c | 0.30 | 1107 a | 1717 | 1.552 c | 2464 a | 4357 | 1.769 c | 8.0 b |
| Infected | 1.20 a | 0.30 | 975 b | 1645 | 1.689 a | 2187 c | 4351 | 1.990 a | 16.9 a |
| Infected+Prob | 0.50 b | 0.30 | 1032 b | 1645 | 1.594 b | 2298 b | 4408 | 1.918 b | 11.1 b |
| SEM | 0.30 | 0.30 | 0.061 | 0.096 | 0.034 | 0.074 | 0.180 | 0.049 | 4.10 |
| p value | <0.001 | 0.98 | 0.004 | 0.26 | <0.001 | <0.001 | 0.836 | <0.001 | 0.005 |
| Treatment | Ileal Digesta | Cecal Content | ||||||
|---|---|---|---|---|---|---|---|---|
| Chao | OS | PD | Shannon | Chao | OS | PD | Shannon | |
| Uninfected (A) | 198.5 | 156.6 | 9.60 | 2.92 | 380.9 | 315.4 | 23.0 | 5.90 |
| Infected (B) | 150.5 | 118.4 | 6.40 | 2.45 | 328.9 | 270.5 | 19.4 | 5.30 |
| Infected+Prob (C) | 215.9 | 169.5 | 10.4 | 2.88 | 395.2 | 326.7 | 22.9 | 5.80 |
| p value | ||||||||
| A versus B | 0.02 | 0.007 | 0.03 | 0.06 | 0.09 | 0.10 | 0.07 | 0.009 |
| A versus C | 0.59 | 0.64 | 0.75 | 0.89 | 0.33 | 0.46 | 0.89 | 0.76 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bortoluzzi, C.; Serpa Vieira, B.; de Paula Dorigam, J.C.; Menconi, A.; Sokale, A.; Doranalli, K.; Applegate, T.J. Bacillus subtilis DSM 32315 Supplementation Attenuates the Effects of Clostridium perfringens Challenge on the Growth Performance and Intestinal Microbiota of Broiler Chickens. Microorganisms 2019, 7, 71. https://doi.org/10.3390/microorganisms7030071
Bortoluzzi C, Serpa Vieira B, de Paula Dorigam JC, Menconi A, Sokale A, Doranalli K, Applegate TJ. Bacillus subtilis DSM 32315 Supplementation Attenuates the Effects of Clostridium perfringens Challenge on the Growth Performance and Intestinal Microbiota of Broiler Chickens. Microorganisms. 2019; 7(3):71. https://doi.org/10.3390/microorganisms7030071
Chicago/Turabian StyleBortoluzzi, Cristiano, Bruno Serpa Vieira, Juliano Cesar de Paula Dorigam, Anita Menconi, Adebayo Sokale, Kiran Doranalli, and Todd Jay Applegate. 2019. "Bacillus subtilis DSM 32315 Supplementation Attenuates the Effects of Clostridium perfringens Challenge on the Growth Performance and Intestinal Microbiota of Broiler Chickens" Microorganisms 7, no. 3: 71. https://doi.org/10.3390/microorganisms7030071
APA StyleBortoluzzi, C., Serpa Vieira, B., de Paula Dorigam, J. C., Menconi, A., Sokale, A., Doranalli, K., & Applegate, T. J. (2019). Bacillus subtilis DSM 32315 Supplementation Attenuates the Effects of Clostridium perfringens Challenge on the Growth Performance and Intestinal Microbiota of Broiler Chickens. Microorganisms, 7(3), 71. https://doi.org/10.3390/microorganisms7030071





