Mucus: An Underestimated Gut Target for Environmental Pollutants and Food Additives
Abstract
1. Introduction
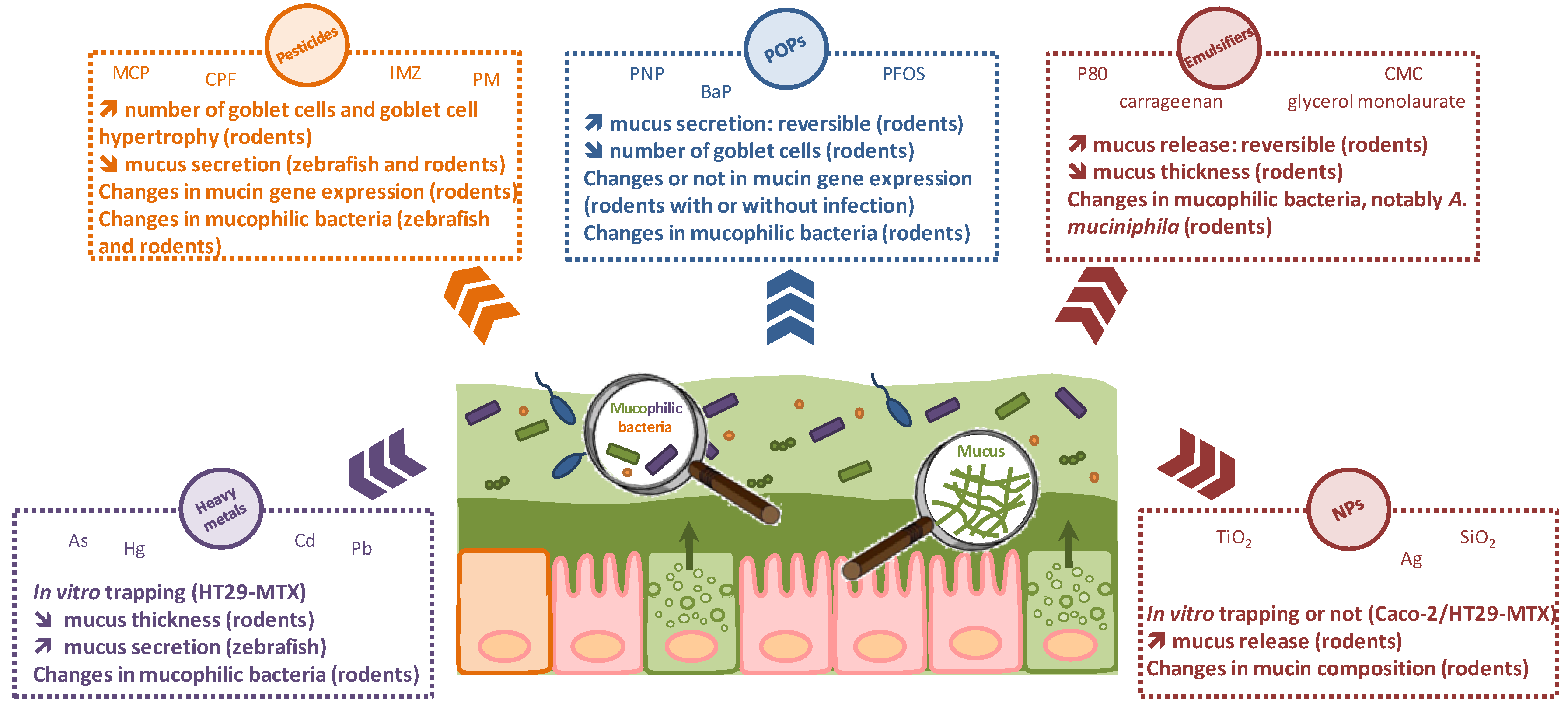
2. Effects of Environmental Pollutants and Food Additives on the Gut Mucus Barrier
2.1. Heavy Metals
2.2. Pesticides
2.3. Persistent Organic Pollutants
2.4. Food Additives
2.4.1. Emulsifiers
2.4.2. Nanomaterials
3. Conclusions
Funding
Conflicts of Interest
References
- Jurewicz, J.; Polanska, K.; Hanke, W. Chemical exposure early in life and the neurodevelopment of children–an overview of current epidemiological evidence. Ann. Agric. Environ. Med. 2013, 20, 465–486. [Google Scholar] [PubMed]
- Yuswir, N.S.; Praveena, S.M.; Aris, A.Z.; Hashim, Z. Bioavailability of heavy metals using in vitro digestion model: A state of present knowledge. Rev. Environ. Health 2013, 28, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Caravanos, J.; Dowling, R.; Tellez-Rojo, M.M.; Cantoral, A.; Kobrosly, R.; Estrada, D.; Orjuela, M.; Gualtero, S.; Ericson, B.; Rivera, A.; et al. Blood lead levels in Mexico and pediatric burden of disease implications. Ann. Glob. Health 2014, 80, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Hadrup, N.; Lam, H.R. Oral toxicity of silver ions, silver nanoparticles and colloidal silver—A review. Regul. Toxicol. Pharmacol. 2014, 68, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Theodorou, I.G.; Ryan, M.P.; Tetley, T.D.; Porter, A.E. Inhalation of silver nanomaterials—Seeing the risks. Int. J. Mol. Sci. 2014, 15, 23936–23974. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, S.; Mazumder, M.A.; Al-Attas, O.; Husain, T. Heavy metals in drinking water: Occurrences, implications, and future needs in developing countries. Sci. Total Environ. 2016, 569–570, 476–488. [Google Scholar] [CrossRef] [PubMed]
- Moller, W.; Haussinger, K.; Winkler-Heil, R.; Stahlhofen, W.; Meyer, T.; Hofmann, W.; Heyder, J. Mucociliary and long-term particle clearance in the airways of healthy nonsmoker subjects. J. Appl. Physiol. 2004, 97, 2200–2206. [Google Scholar] [CrossRef] [PubMed]
- Groh, K.J.; Geueke, B.; Muncke, J. Food contact materials and gut health: Implications for toxicity assessment and relevance of high molecular weight migrants. Food Chem. Toxicol. 2017, 109, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Kerckhoffs, A.P.; Akkermans, L.M.; de Smet, M.B.; Besselink, M.G.; Hietbrink, F.; Bartelink, I.H.; Busschers, W.B.; Samsom, M.W.R. Intestinal permeability in irritable bowel syndrome patients: Effects of NSAIDs. Dig. Dis. Sci. 2010, 55, 716–723. [Google Scholar] [CrossRef] [PubMed]
- Arrieta, M.C.; Bistritz, L.; Meddings, J.B. Alterations in intestinal permeability. Gut 2006, 55, 1512–1520. [Google Scholar] [CrossRef] [PubMed]
- Mankertz, J.; Schulzke, J.D. Altered permeability in inflammatory bowel disease: Pathophysiology and clinical implications. Curr. Opin. Gastroenterol. 2007, 23, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Liu, X.; Ma, L.; He, W.; Li, W.; Cao, Y.; Liu, Z. Food allergens affect the intestinal tight junction permeability in inducing intestinal food allergy in rats. Asian Pac. J. Allergy Immunol. 2014, 32, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Price, D.; Ackland, L.; Suphioglu, C. Nuts ‘n’ guts: Transport of food allergens across the intestinal epithelium. Asia Pac. Allergy 2013, 3, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Wang, B. Role of gut barrier function in the pathogenesis of nonalcoholic fatty liver disease. Gastroenterol. Res. Pract. 2015, 2015, 287348. [Google Scholar] [CrossRef] [PubMed]
- Miele, L.; Valenza, V.; La Torre, G.; Montalto, M.; Cammarota, G.; Ricci, R.; Masciana, R.; Forgione, A.; Gabrieli, M.L.; Perotti, G.; et al. Increased intestinal permeability and tight junction alterations in nonalcoholic fatty liver disease. Hepatology 2009, 49, 1877–1887. [Google Scholar] [CrossRef] [PubMed]
- De Kort, S.; Keszthelyi, D.; Masclee, A.A. Leaky gut and diabetes mellitus: What is the link? Obes. Rev. 2011, 12, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Atkinson, M.A. The role for gut permeability in the pathogenesis of type 1 diabetes—A solid or leaky concept? Pediatr. Diabetes 2015, 16, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Vaarala, O.; Atkinson, M.A.; Neu, J. The “perfect storm” for type 1 diabetes: The complex interplay between intestinal microbiota, gut permeability, and mucosal immunity. Diabetes 2008, 57, 2555–2562. [Google Scholar] [CrossRef] [PubMed]
- Lerner, A.; Matthias, T. Changes in intestinal tight junction permeability associated with industrial food additives explain the rising incidence of autoimmune disease. Autoimmun. Rev. 2015, 14, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Robert, H.; Payros, D.; Pinton, P.; Théodorou, V.; Mercier-Bonin, M.; Oswald, I. Impact of mycotoxins on the intestine: Are mucus and microbiota new targets? J. Toxicol. Environ. Health B Crit. Rev. 2017, 21, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Joly Condette, C.; Khorsi-Cauet, H.; Morlière, P.; Zabijak, L.; Reygner, J.; Bach, V.; Gay-Quéheillard, J. Increased gut permeability and bacterial translocation after chronic chlorpyrifos exposure in rats. PLoS ONE 2014, 9, e102217. [Google Scholar] [CrossRef] [PubMed]
- Tirelli, V.; Catone, T.; Turco, L.; Di Consiglio, E.; Testai, E.; De Angelis, I. Effects of the pesticide chlorpyrifos on an in vitro model of intestinal barrier. Toxicol. In Vitro 2007, 21, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.Y.; Wang, M.; Zhang, J.; Barve, S.S.; McClain, C.J.; Joshi-Barve, S. Acrolein disrupts tight junction proteins and causes endoplasmic reticulum stress-mediated epithelial cell death leading to intestinal barrier dysfunction and permeability. Am. J. Pathol. 2017, 187, 2686–2697. [Google Scholar] [CrossRef] [PubMed]
- Kish, L.; Hotte, N.; Kaplan, G.G.; Vincent, R.; Tso, R.; Gänzle, M.; Rioux, K.P.; Thiesen, A.; Barkema, H.W.; Wine, E.; Madsen, K.L. Environmental particulate matter induces murine intestinal inflammatory responses and alters the gut microbiome. PLoS ONE 2013, 8, e62220. [Google Scholar] [CrossRef] [PubMed]
- Koeneman, B.A.; Zhang, Y.; Westerhoff, P.; Chen, Y.; Crittenden, J.C.; Capco, D.G. Toxicity and cellular responses of intestinal cells exposed to titanium dioxide. Cell Biol. Toxicol. 2010, 26, 225–238. [Google Scholar] [CrossRef] [PubMed]
- Brun, E.; Barreau, F.; Veronesi, G.; Fayard, B.; Sorieul, S.; Chanéac, C.; Carapito, C.; Rabilloud, T.; Mabondzo, A.; Herlin-Boime, N.; Carrière, M. Titanium dioxide nanoparticle impact and translocation through ex vivo, in vivo and in vitro gut epithelia. Part. Fibre Toxicol. 2014, 11, 13. [Google Scholar] [CrossRef] [PubMed]
- Williams, K.M.; Gokulan, K.; Cerniglia, C.E.; Khare, S. Size and dose dependent effects of silver nanoparticle exposure on intestinal permeability in an in vitro model of the human gut epithelium. J. Nanobiotechnol. 2016, 14, 62. [Google Scholar] [CrossRef] [PubMed]
- Ude, V.C.; Brown, D.M.; Viale, L.; Kanase, N.; Stone, V.; Johnston, H.J. Impact of copper oxide nanomaterials on differentiated and undifferentiated Caco-2 intestinal epithelial cells; assessment of cytotoxicity, barrier integrity, cytokine production and nanomaterial penetration. Part. Fibre Toxicol. 2017, 14, 31. [Google Scholar] [CrossRef] [PubMed]
- Coyuco, J.C.; Liu, Y.; Tan, B.J.; Ghiu, G.N. Functionalized carbon nanomaterials: Exploring the interactions with Caco-2 cells for potential oral drug delivery. Int. J. Nanomed. 2011, 6, 2253–2263. [Google Scholar]
- Chen, H.; Zhao, R.; Wang, B.; Zheng, L.; Ouyang, H.; Wang, H.; Zhou, X.; Zhang, D.; Chai, Z.; Zhao, Y.; et al. Acute oral administration of single-walled carbon nanotubes increases intestinal permeability and inflammatory responses: Association with the changes in gut microbiota in mice. Adv. Healthc. Mater. 2018. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.; Mahbub, R.; Fox, J.G. Xenobiotics: Interaction with the intestinal microflora. ILAR J. 2015, 56, 218–227. [Google Scholar] [CrossRef] [PubMed]
- Claus, S.P.; Guillou, H.; Ellero-Simatos, S. The gut microbiota: A major player in the toxicity of environmental pollutants? Biofilms Microbiomes 2016, 2, 16003. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Wu, S.; Zeng, Z.; Fu, Z. Effects of environmental pollutants on gut microbiota. Environ. Pollut. 2017, 222, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, C.S. Gut dysbiosis in animals due to environmental chemical exposures. Front. Cell. Infect. Microbiol. 2017, 7, 396. [Google Scholar] [CrossRef] [PubMed]
- Burger-van Paassen, N.; Vincent, A.; Puiman, P.J.; van der Sluis, M.; Bouma, J.; Boehm, G.; van Goudoever, J.B.; van Seuningen, I.; Renes, I.B. The regulation of intestinal mucin MUC2 expression by short-chain fatty acids: Implications for epithelial protection. Biochem. J. 2009, 420, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Burger-van Paassen, N.; van der Sluis, M.; Witte-Bouma, J.; Kerckaert, J.P.; van Goudoever, J.B.; Van Seuningen, I.; Renes, I.B. Colonic gene expression patterns of mucin Muc2 knockout mice reveal various phases in colitis development. Inflamm. Bowel Dis. 2011, 17, 2047–2057. [Google Scholar] [CrossRef] [PubMed]
- Tomas, J.; Wrzosek, L.; Bouznad, N.; Bouet, S.; Mayeur, C.; Noordine, M.L.; Honvo-Houeto, E.; Langella, P.; Thomas, M.; Cherbuy, C. Primocolonization is associated with colonic epithelial maturation during conventionalization. FASEB J. 2013, 27, 645–655. [Google Scholar] [CrossRef] [PubMed]
- Wrzosek, L.; Miquel, S.; Noordine, M.L.; Bouet, S.; Joncquel Chevalier-Curt, M.; Robert, V.; Philippe, C.; Bridonneau, C.; Cherbuy, C.; Robbe-Masselot, C.; et al. Bacteroides thetaiotaomicron and Faecalibacterium prausnitzii influence the production of mucus glycans and the development of goblet cells in the colonic epithelium of a gnotobiotic model rodent. BMC Biol. 2013, 11, 61. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, S.; Robbe-Masselot, C.; Ait-Belgnaoui, A.; Mancuso, A.; Mercade-Loubière, M.; Salvador-Cartier, C.; Gillet, M.; Ferrier, L.; Loubière, P.; Dague, E.; et al. Stress disrupts intestinal mucus barrier in rats via mucin O-glycosylation shift: Prevention by a probiotic treatment. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 307, G420–G429. [Google Scholar] [CrossRef] [PubMed]
- Birchenough, G.M.; Johansson, M.E.; Gustafsson, J.K.; Bergström, J.H.; Hansson, G.C. New developments in goblet cell mucus secretion and function. Mucosal Immunol. 2015, 8, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Birchenough, G.M.; Nystrom, E.E.; Johansson, M.E.; Hansson, G.C. A sentinel goblet cell guards the colonic crypt by triggering Nlrp6-dependent Muc2 secretion. Science 2016, 352, 1535–1542. [Google Scholar] [CrossRef] [PubMed]
- Bäckhed, F.; Ley, R.E.; Sonnenburg, J.L.; Peterson, D.A.; Gordon, J.I. Host-bacterial mutualism in the human intestine. Science 2005, 307, 1915–1920. [Google Scholar] [CrossRef] [PubMed]
- Sonnenburg, J.L.; Xu, J.; Leip, D.D.; Chen, C.H.; Westover, B.P.; Weatherford, J.; Buhler, J.D.; Gordon, J.I. Glycan foraging in vivo by an intestine-adapted bacterial symbiont. Science 2005, 307, 1955–1959. [Google Scholar] [CrossRef] [PubMed]
- El Kaoutari, A.; Armougom, F.; Gordon, J.I.; Raoult, D.; Henrissat, B. The abundance and variety of carbohydrate-active enzymes in the human gut microbiota. Nat. Rev. Microbiol. 2013, 11, 497–504. [Google Scholar] [CrossRef] [PubMed]
- Juge, N. Microbial adhesins to gastrointestinal mucus. Trends Microbiol. 2012, 20, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Tailford, L.E.; Crost, E.H.; Kavanaugh, D.; Juge, N. Mucin glycan foraging in the human gut microbiome. Front. Genet. 2015, 6, 81. [Google Scholar] [CrossRef] [PubMed]
- Derrien, M.; Vaughan, E.E.; Plugge, C.M.; de Vos, W.M. Akkermansia municiphila gen. nov., sp. nov., a human intestinal mucin-degrading bacterium. Int. J. Syst. Evol. Microbiol. 2004, 54, 1469–1476. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.Y.; Lee, S.M.; Mazmanian, S.K. The human commensal Bacteroides fragilis binds intestinal mucin. Anaerobe 2011, 17, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Crost, E.H.; Tailford, L.E.; Le Gall, G.; Fons, M.; Henrissat, B.; Juge, N. Utilisation of mucin glycans by the human gut symbiont Ruminococcus gnavus is strain-dependent. PLoS ONE 2013, 8, e76341. [Google Scholar] [CrossRef] [PubMed]
- Png, C.W.; Linden, S.K.; Gilshenan, K.S.; Zoetendal, E.G.; McSweeney, C.S.; Sly, L.I.; McGuckin, M.A.; Florin, T.H. Mucolytic bacteria with increased prevalence in IBD mucosa augment in vitro utilization of mucin by other bacteria. Am. J. Gastroenterol. 2010, 105, 2420–2428. [Google Scholar] [CrossRef] [PubMed]
- Malinen, E.; Krogius-Kurikka, L.; Lyra, A.; Nikkilä, J.; Jääskeläinen, A.; Rinttilä, T.; Vilpponen-Salmela, T.; von Wright, A.J.; Palva, A. Association of symptoms with gastrointestinal microbiota in irritable bowel syndrome. World J. Gastroenterol. 2010, 16, 4532–4540. [Google Scholar] [CrossRef] [PubMed]
- Everard, A.; Belzer, C.; Geurts, L.; Geurts, L.; Ouwerkerk, J.P.; Druart, C.; Bindels, L.B.; Guiot, Y.; Derrien, M.; Muccioli, G.G.; et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc. Natl. Acad. Sci. USA 2013, 110, 9066–9071. [Google Scholar] [CrossRef] [PubMed]
- Von Ossowski, I.; Reunanen, J.; Satokari, R.; Vesterlund, S.; Kankainen, M.; Huhtinen, H.; Tynkkynen, S.; Salminen, S.; de Vos, W.M.; Palva, A. Mucosal adhesion properties of the probiotic Lactobacillus rhamnosus GG SpaCBA and SpaFED pilin subunits. Appl. Environ. Microbiol. 2010, 76, 2049–2057. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie, D.A.; Jeffers, F.; Parker, M.L.; Vibert-Vallet, A.; Bongaerts, R.J.; Roos, S.; Walter, J.; Juge, N. Strain-specific diversity of mucus-binding proteins in the adhesion and aggregation properties of Lactobacillus reuteri. Microbiology 2010, 156, 3368–3378. [Google Scholar] [CrossRef] [PubMed]
- Pretzer, G.; Snel, J.; Molenaar, D.; Wiersma, A.; Bron, P.A.; Lambert, J.; de Vos, W.M.; van der Meer, R.; Smits, M.A.; Kleerebezem, M. Biodiversity-based identification and functional characterization of the mannose-specific adhesin of Lactobacillus plantarum. J. Bacteriol. 2005, 187, 6128–6136. [Google Scholar] [CrossRef] [PubMed]
- Jakobsson, H.E.; Rodríguez-Piñeiro, A.M.; Schütte, A.; Ermund, A.; Boysen, P.; Bemark, M.; Sommer, F.; Bäckhed, F.; Hansson, G.C.; Johansson, M.E. The composition of the gut microbiota shapes the colon mucus barrier. EMBO Rep. 2015, 16, 164–177. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.E.V.; Phillipson, M.; Petersson, J.; Holm, L.; Velcich, A.; Hansson, G.C. The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Proc. Natl. Acad. Sci. USA 2008, 105, 15064–15069. [Google Scholar] [CrossRef] [PubMed]
- Johansson, M.E.; Larsson, J.M.; Hansson, G.C. The two mucus layers of colon are organized by the MUC2 mucin, whereas the outer layer is a legislator of host-microbial interactions. Proc. Natl. Acad. Sci. USA 2011, 108, 4659–4665. [Google Scholar] [CrossRef] [PubMed]
- Atuma, C.; Strugala, V.; Allen, A.; Holm, L. The adherent gastrointestinal mucus gel layer: Thickness and physical state in vivo. Am. J. Physiol. Gastrointest. Liver Physiol. 2001, 280, G922–G929. [Google Scholar] [CrossRef] [PubMed]
- Gustafsson, J.K.; Ermund, A.; Johansson, M.E.; Schutte, A.; Hansson, G.C.; Sjovall, H. An ex vivo method for studying mucus formation, properties, and thickness in human colonic biopsies and mouse small and large intestinal explants. Am. J. Physiol. Gastrointest. Liver Physiol. 2012, 302, G430–G438. [Google Scholar] [CrossRef] [PubMed]
- Pullan, R.D.; Thomas, G.A.; Rhodes, M.; Newcombe, R.G.; Williams, G.T.; Allen, A.; Rhodes, J. Thickness of adherent mucus gel on colonic mucosa in humans and its relevance to colitis. Gut 1994, 35, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Kamphuis, J.B.J.; Mercier-Bonin, M.; Eutamène, H.; Théodorou, V. Mucus organisation is shaped by colonic content: A new view. Sci. Rep. 2017, 7, 8527. [Google Scholar] [CrossRef] [PubMed]
- Lukovac, S.; Belzer, C.; Pellis, L.; Keijser, B.J.; de Vos, W.M.; Montijn, R.C.; Roeselers, G. Differential modulation by Akkermansia muciniphila and Faecalibacterium prausnitzii of host peripheral lipid metabolism and histone acetylation in mouse gut organoids. mBio 2014, 5, e01438-14. [Google Scholar] [CrossRef] [PubMed]
- Kober, O.I.; Ahl, D.; Pin, C.; Holm, L.; Carding, S.R.; Juge, N. γδ T-cell-deficient mice show alterations in mucin expression, glycosylation, and goblet cells but maintain an intact mucus layer. Am. J. Physiol. Gastrointest. Liver Physiol. 2014, 306, G582–G593. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific opinion on arsenic in food. EFSA J. 2009, 7, 1351. [Google Scholar]
- Calatayud, M.; Vélez, D.; Devesa, V. Metabolism of inorganic arsenic in intestinal epithelial cell lines. Chem. Res. Toxicol. 2012, 25, 2402–2411. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific opinion on the risk for public health related to the presence of mercury and methylmercury in food. EFSA J. 2012, 10, 2985. [Google Scholar]
- Vázquez, M.; Calatayud, M.; Vélez, D.; Devesa, V. Intestinal transport of methylmercury and inorganic mercury in various models of Caco-2 and HT29-MTX cells. Toxicology 2013, 311, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Agency for Toxic Substances and Disease Registry Toxicological Profile for Cadmium. 2012. Available online: https://www.atsdr.cdc.gov/toxprofiles/tp5.pdf (accessed on 1 May 2018).
- International Agency for Research on Cancer. Arsenic, metals, fibres, and dusts. IARC Monogr. Eval. Carcinog. Risks Hum. 2012, 100, 11–465. [Google Scholar]
- Öner, G.; Izgüt-Uysal, V.N.; Sentürk, U.K. Role of lipid peroxidation in cadmium-induced impairment of the gastric mucosal barrier. Food Chem. Toxicol. 1994, 32, 799–804. [Google Scholar] [CrossRef]
- Asar, M.; Kayişli, U.A.; Izgüt-Uysal, V.N.; Oner, G.; Kaya, M.; Polat, S. Cadmium-induced changes in epithelial cells of the rat stomach. Biol. Trace Elem. Res. 2000, 77, 65–81. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Y.; Liu, K.; Shen, J. Exposing to cadmium stress cause profound toxic effect on microbiota of the mice intestinal tract. PLoS ONE 2014, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, B.O.; Birchenough, G.M.H.; Ståhlman, M.; Arike, L.; Johansson, M.E.V.; Hansson, G.C.; Bäckhed, F. Bifidobacteria or fiber protects against diet-induced microbiota-mediated colonic mucus deterioration. Cell Host Microbe 2018, 23. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.; Lu, L.; Jin, C.; Wang, S.; Zhou, J.; Ni, Y.; Fu, Z.; Jin, Y. Effects of short term lead exposure on gut microbiota and hepatic metabolism in adult zebrafish. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2018, 209, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wen, X.W.; Faulk, C.; Boehnke, K.; Zhang, H.; Dolinoy, D.C.; Xi, C. Perinatal lead exposure alters gut microbiota composition and results in sex-specific bodyweight increases in adult mice. Toxicol. Sci. 2016, 151, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Snyder, J.E.; Filipov, N.M.; Parsons, P.J.; Lawrence, D.A. The efficiency of maternal transfer of lead and its influence on plasma IgE and splenic cellularity of mice. Toxicol. Sci. 2000, 57, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Waite, D.T.; Grover, R.; Westcott, N.D.; Sommerstad, H.; Kerr, L. Pesticides in ground, water surface water and spring runoff in a small Saskatchewan watershed. Environ. Toxicol. Chem. 1992, 11, 741–748. [Google Scholar] [CrossRef]
- Kumari, B.; Madan, V.K.; Singh, J.; Singh, S.; Kathpal, T.S. Monitoring of pesticidal contamination of farmgate vegetables from Hisar. Environ. Monit. Assess. 2004, 90, 65–71. [Google Scholar] [CrossRef] [PubMed]
- Rajini, P.S. Oral exposure to the organophosphorus insecticide, Monocrotophos induces intestinal dysfunction in rats. Food Chem. Toxicol. 2014, 71, 236–243. [Google Scholar]
- Bolles, H.G.; Dixon-White, H.E.; Peterson, R.K.; Tomerlin, J.R.; Day, E.W., Jr.; Oliver, G.R. U.S. market basket study to determine residues of the insecticide chlorpyrifos. J. Agric. Food Chem. 1999, 47, 1817–1822. [Google Scholar] [CrossRef] [PubMed]
- Joly Condette, C.; Bach, V.; Mayeur, C.; Gay-Quéheillard, J.; Khorsi-Cauet, H. Chlorpyrifos exposure during perinatal period affects intestinal microbiota associated with dlay of maturation of digestive tract in rats. J. Pediatr. Gastroenterol. Nutr. 2015, 61, 30–40. [Google Scholar] [PubMed]
- Ortelli, D.; Edder, P.; Corvi, C. Pesticide residues survey in citrus fruits. Food Addit. Contam. 2005, 22, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Belenguer, V.; Martinez-Capel, F.; Masiá, A.; Picó, Y. Patterns of presence and concentration of pesticides in fish and waters of the Júcar River (Eastern Spain). J. Hazard. Mater. 2014, 265, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Masiá, A.; Vásquez, K.; Campo, J.; Picó, Y. Assessment of two extraction methods to determine pesticides in soils, sediments and sludges. Application to the Túria River Basin. J. Chromatogr. A 2015, 1378, 19–31. [Google Scholar] [CrossRef] [PubMed]
- Ccanccapa, A.; Masiá, A.; Navarroortega, A.; Picó, Y.; Barceló, D. Pesticides in the Ebro river basin: Occurrence and risk assessment. Environ. Pollut. 2016, 211, 414–424. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Luo, T.; Zhu, Z.; Pan, Z.; Yang, J.; Wang, W.; Fu, Z.; Jin, Y. Imazalil exposure induces gut microbiota dysbiosis and hepatic metabolism disorder in zebrafish. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2017, 202, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Zeng, Z.; Fu, Z.; Jin, Y. Oral imazalil exposure induces gut microbiota dysbiosis and colonic inflammation in mice. Chemosphere 2016, 160, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Xia, J.; Wu, S.; Tu, W.; Pan, Z.; Fu, Z.; Wang, Y.; Jin, Y. Insights into a possible influence on gut microbiota and intestinal barrier function during chronic exposure of mice to imazalil. Toxicol. Sci. 2018, 162, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Katz, J.P.; Perreault, N.; Goldstein, B.G.; Lee, C.S.; Labosky, P.A.; Yang, V.W.; Kaestner, K.H. The zinc-finger transcription factor Klf4 is required for terminal differentiation of goblet cells in the colon. Development 2002, 129, 2619–2628. [Google Scholar] [PubMed]
- Schütte, A.; Ermund, A.; Becker-Pauly, C.; Johansson, M.E.; Rodriguez-Pineiro, A.M.; Bäckhed, F.; Müller, S.; Lottaz, D.; Bond, J.S.; Hansson, G.C. Microbial-induced meprin β cleavage in MUC2 mucin and a functional CFTR channel are required to release anchored small intestinal mucus. Proc. Natl. Acad. Sci. USA 2014, 111, 12396–12401. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Jin, C.; Wang, Y.; Fu, Z.; Jin, Y. Exposure to the fungicide propamocarb causes gut microbiota dysbiosis and metabolic disorder in mice. Environ. Pollut. 2018, 237, 775–783. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Conclusion on the peer review of the pesticide risk assessment of the active substance propamocarb. EFSA Sci. Rep. 2006, 78, 1–80. [Google Scholar]
- Noya, Y.; Mikami, Y.; Taneda, S.; Mori, Y.; Suzuki, A.K.; Ohkura, K.; Yamaki, K.; Yoshino, S.; Seki, K.I. Improvement of an efficient separation method for chemicals in diesel exhaust particles: Analysis for nitrophenols. Environ. Sci. Pollut. Res. Int. 2008, 15, 318–321. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Song, M.; Watanabe, G.; Nagaoka, K.; Rui, X.; Li, C. Effects of 4-nitrophenol on expression of the ER-α and AhR signaling pathway-associated genes in the small intestine of rats. Environ. Pollut. 2016, 216, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Phillips, D.H. Polycyclic aromatic hydrocarbons in the diet. Mutat. Res. 1999, 443, 139–147. [Google Scholar] [CrossRef]
- Lewtas, J. Air pollution combustion emissions: Characterization of causative agents and mechanisms associated with cancer, reproductive, and cardiovascular effects. Mutat. Res. 2007, 636, 95–133. [Google Scholar] [CrossRef] [PubMed]
- Ribière, C.; Peyret, P.; Pariso, T.N.; Darcha, C.; Déchelotte, P.J.; Barnich, N.; Peyretaillade, E.; Boucher, D. Oral exposure to environmental pollutant benzo[a]pyrene impacts the intestinal epithelium and induces gut microbial shifts in murine model. Sci. Rep. 2016, 6, 31027. [Google Scholar] [CrossRef] [PubMed]
- United Nations Environment Programme. Secretariat of the Stockholm Convention: The New POPs under the Stockholm Convention, 2011. Available online: http://www.pops-gmp.org/res/file/UNEP-POPS-COP_5-INF-27.pdf (accessed on 1 May 2018).
- Eriksen, K.T.; Raaschou-Nielsen, O.; McLaughlin, J.K.; Lipworth, L.; Tjønneland, A.; Overvad, K.; Sørensen, M. Association between plasma PFOA and PFOS levels and total cholesterol in a middle-aged Danish population. PLoS ONE 2013, 8, e56969. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Wang, T.; Liu, S.; Jones, K.C.; Sweetman, A.J.; Lu, Y. Industrial source identification and emission estimation of perfluorooctane sulfonate in China. Environ. Int. 2013, 52, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Olsen, G.W.; Burris, J.M.; Ehresman, D.J.; Froehlich, J.W.; Seacat, A.M.; Butenhoff, J.L.; Zobel, L.R. Half-life of serum elimination of perfluorooctanesulfonate, perfluorohexanesulfonate, and perfluorooctanoate in retired fluorochemical production workers. Environ. Health Perspect. 2007, 115, 1298–1305. [Google Scholar] [CrossRef] [PubMed]
- Suo, C.; Fan, Z.; Zhou, L.; Qiu, J. Perfluorooctane sulfonate affects intestinal immunity against bacterial infection. Sci. Rep. 2017, 7, 5166. [Google Scholar] [CrossRef] [PubMed]
- Engelen, L.; Van Der Bilt, A. Oral physiology and texture perception of semisolids. J. Texture Stud. 2008, 39, 83–113. [Google Scholar] [CrossRef]
- Oberle, R.L.; Moore, T.J.; Krummel, D.A.P. Evaluation of mucosal damage of surfactants in rat jejunum and colon. J. Pharmacol. Toxicol. Methods 1995, 33, 75–81. [Google Scholar] [CrossRef]
- Chassaing, B.; Koren, O.; Goodrich, J.K.; Poole, A.C.; Srinivasan, S.; Ley, R.E.; Gewirtz, A.T. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature 2015, 519, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Zhao, M.; Zhang, H.; Li, Y.; Liu, M.; Feng, F. Antimicrobial emulsifier-glycerol monolaurate induces metabolic syndrome, gut microbiota dysbiosis, and systemic low-grade inflammation in low-fat diet fed mice. Mol. Nutr. Food Res. 2018, 62. [Google Scholar] [CrossRef] [PubMed]
- Shang, Q.; Sun, W.; Shan, X.; Jiang, H.; Cai, C.; Hao, J.; Li, G.; Yu, G. Carrageenan-induced colitis is associated with decreased population of anti-inflammatory bacterium, Akkermansia muciniphila, in the gut microbiota of C57BL/6J mice. Toxicol. Lett. 2017, 279, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Necas, J.; Bartosikova, L. Carrageenan: A review. Vet. Med. 2013, 58, 187–205. [Google Scholar] [CrossRef]
- Tobacman, J.K. Review of harmful gastrointestinal effects of carrageenan in animal experiments. Environ. Health Perspect. 2001, 109, 983–994. [Google Scholar] [CrossRef] [PubMed]
- Watson, D.B. Public health and carrageenan regulation: A review and analysis. J. Appl. Phycol. 2008, 20, 505–513. [Google Scholar] [CrossRef]
- Ensign, L.M.; Cone, R.; Hanes, J. Oral drug delivery with polymeric nanoparticles: The gastrointestinal mucus barriers. Adv. Drug Deliv. Rev. 2012, 64, 557–570. [Google Scholar] [CrossRef] [PubMed]
- Behrens, I.; Pena, A.I.; Alonso, M.J.; Kissel, T. Comparative uptake studies of bioadhesive and non-bioadhesive nanoparticles in human intestinal cell lines and rats: The effect of mucus on particle adsorption and transport. Pharm. Res. 2002, 19, 1185–1193. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, S.; Mahon, E.; Harrison, S.M.; McGetrick, J.; Muniyappa, M.; Carrington, S.D.; Brayden, D.J. Nanoparticle passage through porcine jejunal mucus: Microfluidics and rheology. Nanomedicine 2017, 13, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.J.B.; Bouwmeester, H.; Gottardo, S.; Amenta, V.; Arena, M.; Brandhoff, P.; Marvin, H.J.P.; Mech, A.; Moniz, F.B.; Pesudo, L.Q.; et al. Nanomaterials for products and application in agriculture, feed and food. Trends Food Sci. Technol. 2016, 54, 155–164. [Google Scholar] [CrossRef]
- Piperigkou, Z.; Karamanou, K.; Engin, A.B.; Gialeli, C.; Docea, A.O.; Vynios, D.H.; Pavao, M.S.; Golokhvast, K.S.; Shtilman, M.I.; Argiris, A.; et al. Emerging aspects of nanotoxicology in health and disease: From agriculture and food sector to cancer therapeutics. Food Chem. Toxicol. 2016, 91, 42–57. [Google Scholar] [CrossRef] [PubMed]
- Dudefoi, W.; Terrisse, H.; Popa, A.F.; Gautron, E.; Humbert, B.; Ropers, M.H. Evaluation of the content of TiO2 nanoparticles in the coatings of chewing gums. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2018, 35, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Periasamy, V.S.; Athinarayanan, J.; Al-Hadi, A.M.; Juhaimi, F.A.; Mahmoud, M.H.; Alshatwi, A.A. Identification of titanium dioxide nanoparticles in food products: Induce intracellular oxidative stress mediated by TNF and CYP1A genes in human lung fibroblast cells. Environ. Toxicol. Pharmacol. 2015, 39, 176–186. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.J.; van Bemmel, G.; Herrera-Rivera, Z.; Helsper, H.P.; Marvin, H.J.; Weigel, S.; Tromp, P.C.; Oomen, A.G.; Rietveld, A.G.; Bouwmeester, H. Characterization of titanium dioxide nanoparticles in food products: Analytical methods to define nanoparticles. J. Agric. Food Chem. 2014, 62, 6285–6293. [Google Scholar] [CrossRef] [PubMed]
- Weir, A.; Westerhoff, P.; Fabricius, L.; Hristovski, K.; von Goetz, N. Titanium dioxide nanoparticles in food and personal care products. Environ. Sci. Technol. 2012, 46, 2242–2250. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Doudrick, K.; Bi, X.; Hristovski, K.; Herckes, P.; Westerhoff, P.; Kaegi, R. Characterization of food-grade titanium dioxide: The presence of nanosized particles. Environ. Sci. Technol. 2014, 48, 6391–6400. [Google Scholar] [CrossRef] [PubMed]
- Dekkers, S.; Krystek, P.; Peters, R.J.; Lankveld, D.P.; Bokkers, B.G.; van Hoeven-Arentzen, P.H.; Bouwmeester, H.; Oomen, A.G. Presence and risks of nanosilica in food products. Nanotoxicology 2011, 5, 393–405. [Google Scholar] [CrossRef] [PubMed]
- Heroult, J.; Nischwitz, V.; Bartczak, D.; Goenaga-Infante, H. The potential of asymmetric flow field-flow fractionation hyphenated to multiple detectors for the quantification and size estimation. Anal. Bioanal. Chem. 2014, 406, 3919–3927. [Google Scholar] [CrossRef] [PubMed]
- Peters, R.; Kramer, E.; Oomen, A.G.; Rivera, Z.E.; Oegema, G.; Tromp, P.C.; Fokkink, R.; Rietveld, A.; Marvin, H.J.; Weigel, S.; et al. Presence of nano-sized silica during in vitro digestion of foods containing silica as a food additive. ACS Nano 2012, 6, 2441–2451. [Google Scholar] [CrossRef] [PubMed]
- Verleysen, E.; Van Doren, E.; Waegeneers, N.; De Temmerman, P.J.; Abi Daoud Francisco, M.; Mast, J. TEM and SP-ICP-MS analysis of the release of silver nanoparticles from decoration of pastry. J. Agric. Food Chem. 2015, 63, 3570–3578. [Google Scholar] [CrossRef] [PubMed]
- Bettini, S.; Boutet-Robinet, E.; Cartier, C.; Comera, C.; Gaultier, E.; Dupuy, J.; Naud, N.; Taché, S.; Grysan, P.; Reguer, S.; et al. Oral exposure to food-grade TiO2 impairs intestinal and systemic immune homeostasis and initiates colon carcinogenesis in rats. Sci. Rep. 2017, 7, 40373. [Google Scholar] [CrossRef] [PubMed]
- Dorier, M.; Béal, D.; Marie-Desvergne, C.; Dubosson, M.; Barreau, F.; Houdeau, E.; Herlin-Boime, N.; Carrière, M. Continuous in vitro exposure of intestinal epithelial cells to E171 food additive causes oxidative stress, inducing oxidation of DNA bases but no endoplasmic reticulum stress. Nanotoxicology 2017, 11, 751–761. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Martucci, N.J.; Liu, Y.; Yoo, E.; Tako, E.; Mahler, G.J. Silicon dioxide nanoparticle exposure affects small intestine function in an in vitro model. Nanotoxicology 2018, 18, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Mahler, G.J.; Shuler, M.L.; Glahn, R.P. Characterization of Caco-2 and HT29-MTX cocultures in an in vitro digestion/cell culture model used to predict iron bioavailability. J. Nutr. Biochem. 2009, 20, 494–502. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific Opinion on the Re-Evaluation of Silver (E 174) as Food Additive. EFSA J. 2016, 14, 4364. [Google Scholar]
- Benn, T.; Cavanagh, B.; Hristovski, K.; Posner, J.D.; Westerhoff, P. The release of nanosilver from consumer products used in the home. J. Environ. Qual. 2010, 39, 1875–1882. [Google Scholar] [CrossRef] [PubMed]
- Echegoyen, Y.; Nerín, C. Nanoparticle release from nano-silver antimicrobial food containers. Food Chem. Toxicol. 2013, 62, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Hannon, J.C.; Kerry, J.P.; Cruz-Romero, M.; Azlin-Hasim, S.; Morris, M.; Cummins, E. Assessment of the migration potential of nanosilver from nanoparticle-coated low-density polyethylene food packaging into food simulants. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2016, 33, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, F.; Kurdestany, A.H. The impact of silver nano particles on growth performance, lymphoid organs and oxidative stress indicators in broiler chicks. Glob. Vet. 2010, 5, 366–370. [Google Scholar]
- Pineda, L.; Chwalibog, A.; Sawosz, E.; Lauridsen, C.; Engberg, R.; Elnif, J.; Hotowy, A.; Sawosz, F.; Gao, Y.; Ali, A.; et al. Effect of silver nanoparticles on growth performance, metabolism and microbial profile of broiler chickens. Arch. Anim. Nutr. 2012, 66, 416–429. [Google Scholar] [CrossRef] [PubMed]
- Fondevila, M.; Herrer, R.; Casallasa, M.C.; Abecia, L.; Ducha, J.J. Silver nanoparticles as a potential antimicrobial additive for weaned pigs. Anim. Feed Sci. Technol. 2009, 150, 259–269. [Google Scholar] [CrossRef]
- Georgantzopoulou, A.; Serchi, T.; Cambier, S.; Leclercq, C.C.; Renaut, J.; Shao, J.; Kruszewski, M.; Lentzen, E.; Grysan, P.; Eswara, S.; et al. Effects of silver nanoparticles and ions on a co-culture model for the gastrointestinal epithelium. Part. Fibre Toxicol. 2016, 13, 9. [Google Scholar] [CrossRef] [PubMed]
- Jeong, G.N.; Jo, U.B.; Ryu, H.Y.; Kim, Y.S.; Song, K.S.; Yu, I.J. Histochemical study of intestinal mucins after administration of silver nanoparticles in Sprague-Dawley rats. Arch. Toxicol. 2010, 84, 63–69. [Google Scholar] [CrossRef] [PubMed]
| Heavy Metal | Experimental Model | Experimental Design | Main Effects * | References |
|---|---|---|---|---|
| Arsenic | in vitro HT29-MTX cells | As(III) 1 µM As(V) 10 µM; 5, 15, and 21 days post-seeding; 24 h | iAs accumulation in mucus ↓ arsenic cellular uptake | [66] |
| Mercury | in vitro Caco-2/HT29-MTX cells vs Caco-2 cells | inorganic mercury (Hg(II)) 3.5 µM, methylmercury (MeHg) 3.5 µM; 2 h | Hg accumulation in mucus ↓ apparent permeability in Caco-2/HT29-MTX | [68] |
| Cadmium | in vivo Rats | 15 µg/mL in drinking water; 30 days | ↓ gastric mucin content Negative correlation mucin/cadmium in gastric mucosa | [71] |
| Cadmium | in vivo Rats | 15 µg/mL in drinking water; 30 days | ↓ gastric mucin content ↓ gastric mucus thickness | [72] |
| Cadmium | in vivo Mice | 20 and 100 mg/kg; 21 days | ↓ colonic mucus thickness ↓ lactobacilli and bifidobacteria | [73] |
| Lead | in vivo Zebrafish | 10 and 30 µg/L; 7 days | ↑ mucus secretion | [75] |
| Lead | in vivo Mice Perinatal exposure | 32 µg/mL in drinking water (gestation and lactation) | ↓ A. muciniphila ↑ Desulfovibrio in adult mice | [76] |
| Pesticide | Experimental Model | Experimental Design | Main Effects * | References |
|---|---|---|---|---|
| Monocrotophos (MCP) | in vivo Mice | 0.45, 0.9, and 1.8 mg/kg bw/day; 30 days | ↑ number of goblet cells and goblet cell hypertrophy in the jejunum | [80] |
| Chlorpyrifos (CPF) | in vivo Female rats exposed from gestation to weaning of the pups | 1 and 5 mg/kg bw/day; weaning (D21)/adulthood (D60) | ↓ Muc2 expression | [82] |
| Imazalil (IMZ) | in vivo Zebrafish | 100 and 1000 µg/L; 1, 7, and 21 days | ↓ mucus secretion | [87] |
| Imazalil (IMZ) | in vivo Mice | 25, 50, and 100 mg/kg bw/day; 28 days | ↓ Bifidobacterium and Lactobacillus↑ Deltaproteobacteria and Desulfovibrio | [88] |
| Imazalil (IMZ) | in vivo Mice | 0.1, 0.5, and 2.5 mg/kg bw/day; 2, 5, and 15 weeks | ↓ colonic mucus secretion ↓ Muc2 expression (2 weeks) ↑ Muc2 expression (15 weeks) ↓ Muc1, Muc3 expression (5 weeks) ↑ Muc1, Muc3 expression (15 weeks) | [89] |
| Propamocarb (PM) | in vivo Mice | 3, 30, and 300 µg/mL in drinking water; 28 days | ↓ Oscillospira, Parabacteroides, Desulfovibrio, and Ruminococcus ↑ Bacteroides, Dehalobacterium, and Butyricimonas | [92] |
| Persistent Organic Pollutant | Experimental Model | Experimental Design | Main Effects * | References |
|---|---|---|---|---|
| 4-Nitrophenol (PNP) | in vivo Rats | 200 mg/kg single dose (1-d), repeated dose (3-d), repeated dose with recovery (6-d) | ↑ mucus secretion in the duodenum and jejunum; reversible | [95] |
| Benzo[a]pyrene (BaP) | in vivo Mice | 50 mg/kg bw/day; 28 days | ↓ Lactobacillaceae and Verrucomicrobiacea; changes in Mucispirillum and Ruminococcaceae; ↑ Bifidobacterium | [98] |
| Perfluorooctane sulfonate (PFOS) | in vivo Mice | 2 mg/kg in drinking water; infection with Citrobacter rodentium | ↓ Muc1, Muc2, but not Muc3 in the colon 11 days p.i. ↓ mucin content in the colon 11 days p.i.; ↑ Escherichia coli; ↓ Lactobacillus | [103] |
| Emulsifier | Experimental Model | Experimental Design | Main Effects * | References |
|---|---|---|---|---|
| Polysorbate-80 | in vivo Rats | 1% (w/v) single-pass in situ perfusion | ↑ mucus secretion; reversible | [105] |
| Carboxymethylcellulose (CMC)/polysorbate-80 | in vivo Mice | 1% (v/v) P80/ 1% (w/v) CMC in drinking water or chow; 12 weeks | ↓ colonic mucus thickness; no changes in Muc2 expression ↑ R. gnavus and A. muciniphila | [106] |
| Glycerol monolaurate | in vivo Mice | 150 mg/kg bw; 8 weeks | ↓ A. muciniphila | [107] |
| Carrageenan | in vivo Mice | 20 µg/mL in drinking water; 6 weeks | ↓ A. muciniphila | [108] |
| Nanomaterial | Experimental Model | NPs Size | Experimental Design | Main Effects * | References |
|---|---|---|---|---|---|
| Titanium dioxide (TiO2) TiO2-NPs | in vitro Caco-2/HT29-MTX cells vs Caco-2 cells | 12 nm | 50 µg/mL; 6, 24, and 48 h | ↑ cell uptake in Caco-2/HT29-MTX cells | [26] |
| TiO2 food-grade (E171) and TiO2-NPs | in vitro Caco-2/HT29-MTX cells vs Caco-2 cells | E171: 118 nmTiO2-NPs: 12 and 24 nm | Acute exposure: 1–200 µg/mL; 6, 24, and 48 hChronic exposure: 1–100 µg/mL; 3 weeks | ↑ cell accumulation for E171 vs TiO2-NPs; no mucus-mediated trapping | [127] |
| Silicon dioxide (SiO2-NPs) | in vitro Caco-2/HT29-MTX cells | 20–30 nm | 0.0002, 0.02, and 2 µg/mL; 4 h (acute) and 5 days (repeated) | ↓ transepithelial electrical resistance | [128] |
| Silver (AgNPs) | in vitro Caco-2/HT29-MTX cells vs Caco-2 cells | 20 and 200 nm | 10–100 µg/mL; 24 h | Mucus trapping for 200-nm AgNPs; ↓ cell-NPs interaction, IL-8 release, and ROS production | [137] |
| Silver (AgNPs) | in vivo Rats | 60 nm | 30, 300, and 1000 mg/kg/day; 28 days | ↑ mucus release in the ileum and rectum; changes in mucin composition | [138] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gillois, K.; Lévêque, M.; Théodorou, V.; Robert, H.; Mercier-Bonin, M. Mucus: An Underestimated Gut Target for Environmental Pollutants and Food Additives. Microorganisms 2018, 6, 53. https://doi.org/10.3390/microorganisms6020053
Gillois K, Lévêque M, Théodorou V, Robert H, Mercier-Bonin M. Mucus: An Underestimated Gut Target for Environmental Pollutants and Food Additives. Microorganisms. 2018; 6(2):53. https://doi.org/10.3390/microorganisms6020053
Chicago/Turabian StyleGillois, Kévin, Mathilde Lévêque, Vassilia Théodorou, Hervé Robert, and Muriel Mercier-Bonin. 2018. "Mucus: An Underestimated Gut Target for Environmental Pollutants and Food Additives" Microorganisms 6, no. 2: 53. https://doi.org/10.3390/microorganisms6020053
APA StyleGillois, K., Lévêque, M., Théodorou, V., Robert, H., & Mercier-Bonin, M. (2018). Mucus: An Underestimated Gut Target for Environmental Pollutants and Food Additives. Microorganisms, 6(2), 53. https://doi.org/10.3390/microorganisms6020053





