Antifungal Peptides of the AFP Family Revisited: Are These Cannibal Toxins?
Abstract
1. Introduction
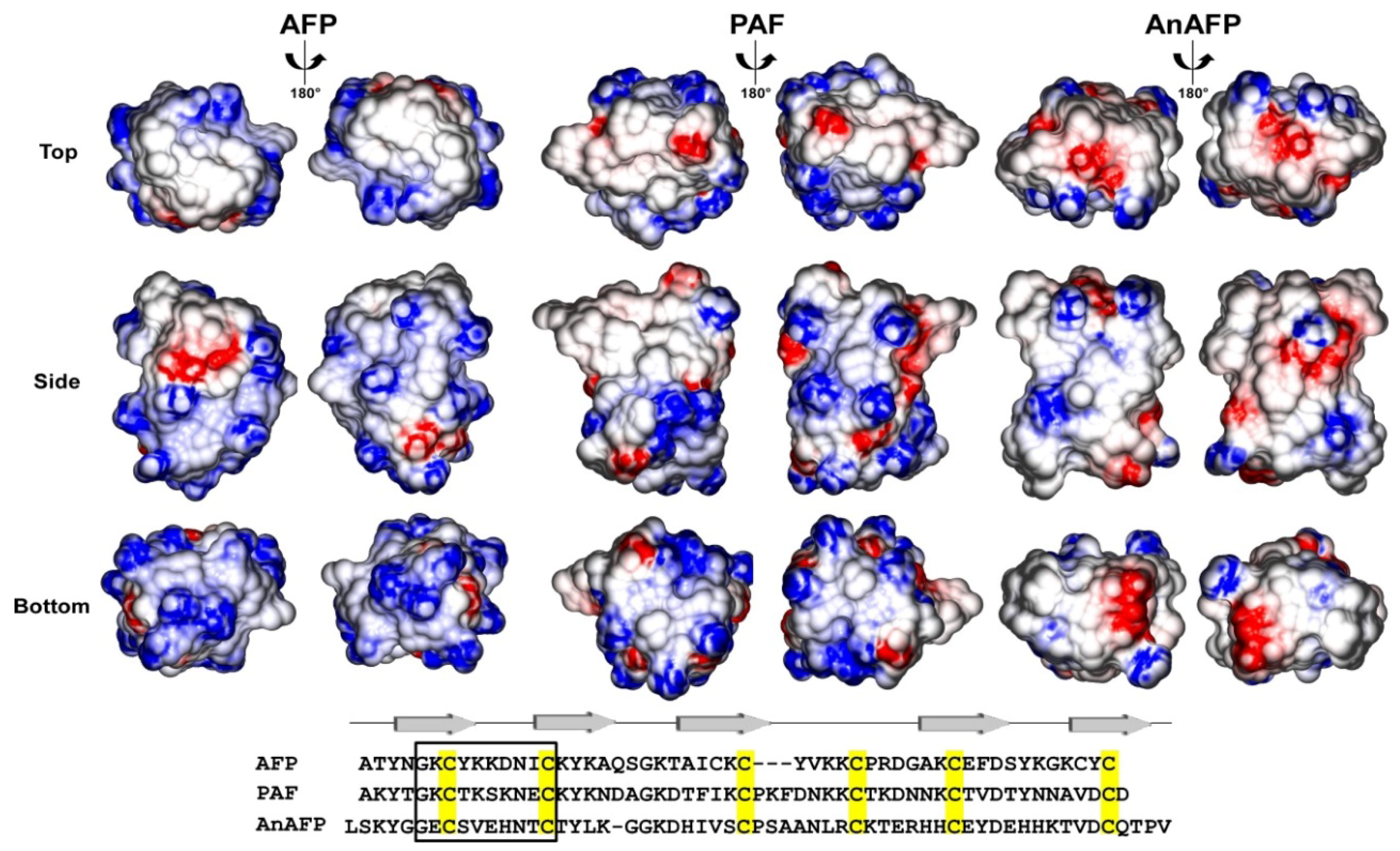
2. Antifungal Modes of Action of AFPs: Similar, but Not the Same
3. Gene Regulation of AFP-Encoding Genes: From the General to the Particular
3.1. The anafp Gene as a Paradigmatic Example
- the choice of carbon or nitrogen source does not impact anafp expression;
- carbon limitation and starvation strongly stimulate anafp expression, suggesting that anafp is under control of the carbon catabolite repressor CreA;
- the expression profile of anafp is similar to the expression profile of the early starvation response genes atg1 (predicted Ser/Thr kinase involved in autophagy) and nagA (predicted autolytic β-1,6-N-acetylglucosaminidase);
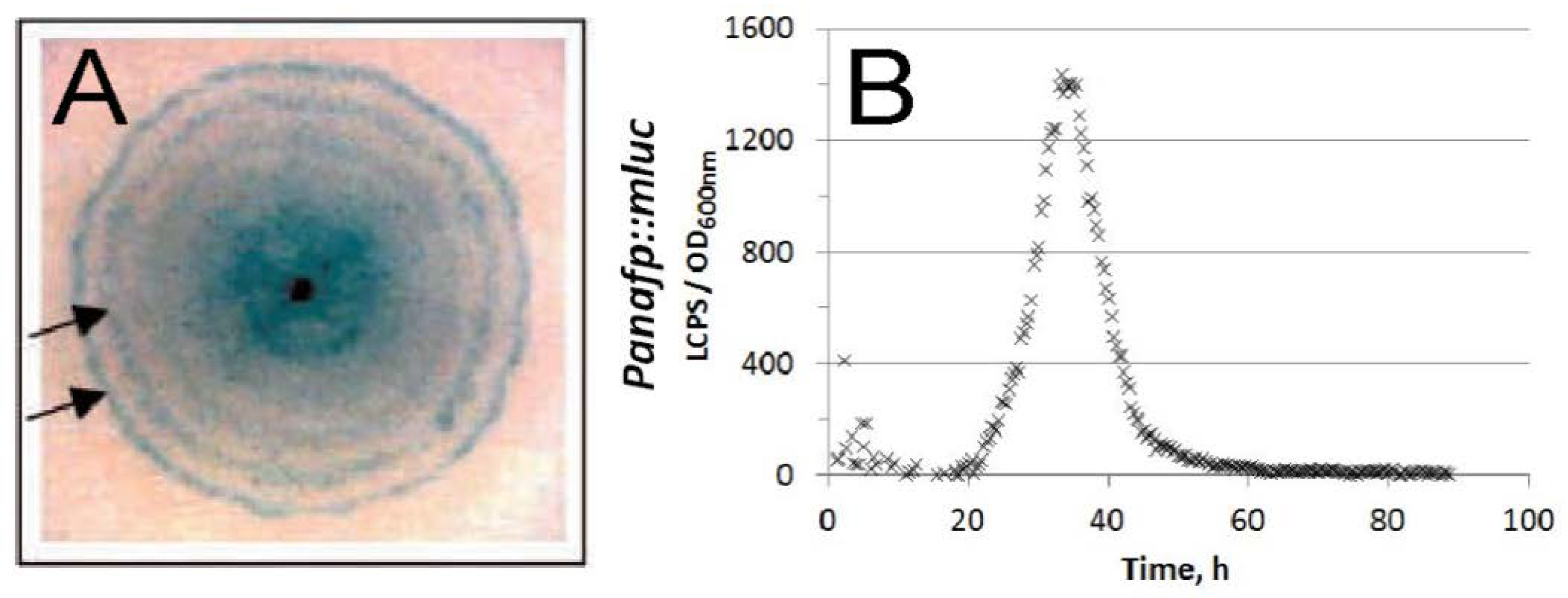
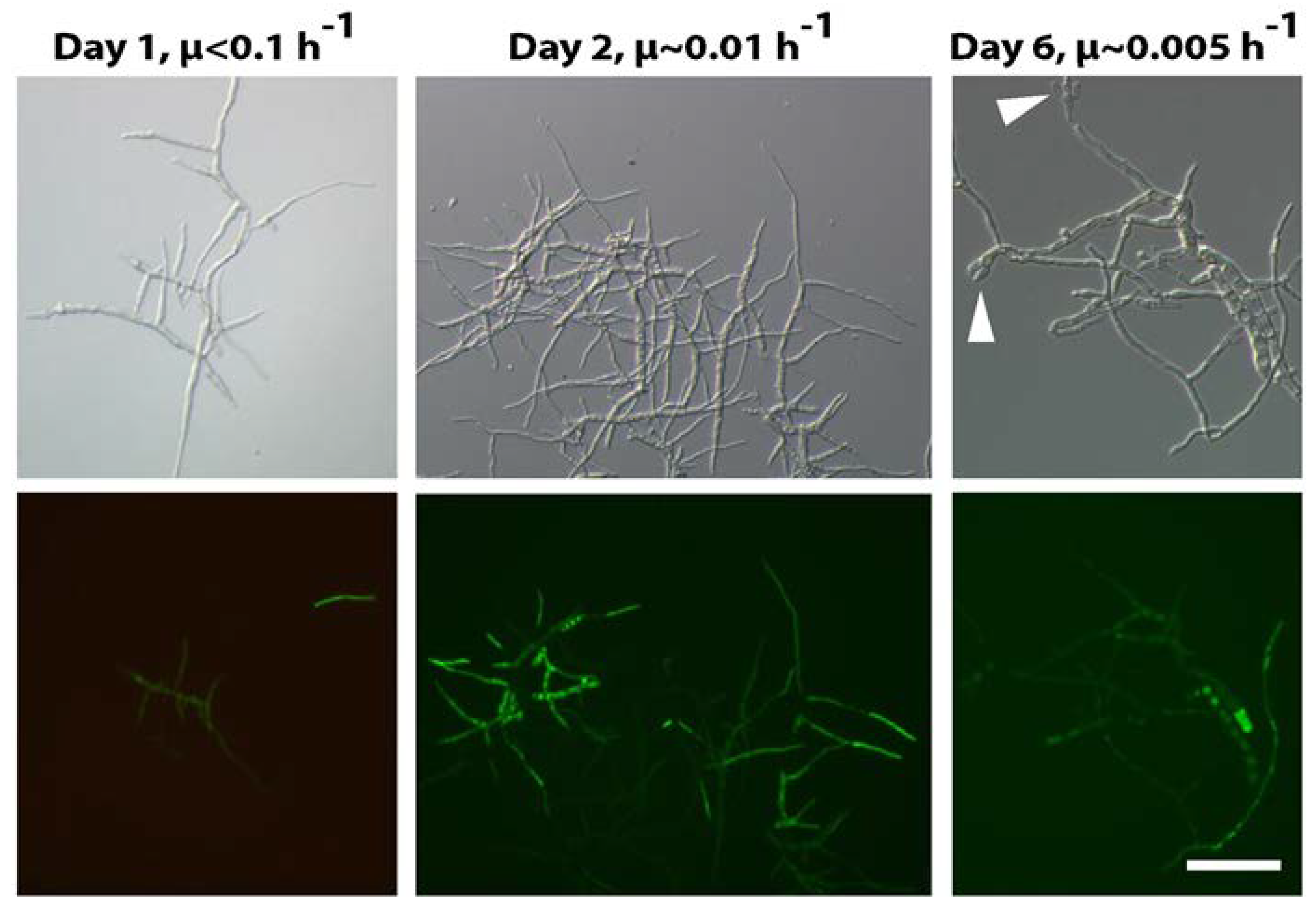
- anafp, nagA, and atg1 expression peaked at 16 h after carbon depletion and gradually decreased at 60 h and 140 h post-carbon depletion in submerged batch cultures. The expression peaks were paralleled with the appearance of a second hyphal population with reduced hyphal diameters (1 µm in diameter instead of 3 µm);
- under severe carbon and energy limitation resulting in very low growth rates (about 0.005 h−1), additionally to anafp, nagA, and atg1, other genes involved in nutrient mobilization, autophagy, N-acetylglucosamine metabolism, and carbohydrate transport are strongly upregulated;
- anafp transcript levels are low in dormant conidia and young germlings, but increase about 15-fold and 60-fold in aerial hyphae and the vegetative mycelium, respectively. This expression profile is similar to those of genes encoding chitin-remodeling enzymes (ctcB, cfcI, and nagA);
- in A. niger wild-type colonies, anafp expression is highest in the center of a colony and gradually decreases towards its periphery;
- in A. niger deleted for FlbA (displaying a nonsporulating, slow-growing, and autolytic phenotype), anafp expression is strongly upregulated (5-fold). Note that FlbA is conserved in aspergilli and known to stop vegetative growth during the process of conidiation. Its main function is to activate the transcription factor BrlA in response to the extracellular signaling molecule FluG. BrlA, in turn, is the central regulator of asexual development in aspergilli [43];
- in A. niger deleted for BrlA (displaying a nonsporulating, slow-growing, but nonautolytic phenotype), anafp expression is upregulated as well (2-fold). However, induction of anafp expression precedes brlA expression in the wild type; that is, BrlA cannot be the first regulator of anafp;
- anafp expression is not induced upon cell wall stress (provoked by caspofungin, fenpropimorph, FK506, aureobasidin A, natamycin), secretion stress (induced by DTT, tunicamycin), or confrontation with Bacillus subtilis;
- anafp is not important for polar growth of A. niger, as morphology mutants (TORC2, RacA) do not show altered anafp expression;
- although AnAFP is a secreted protein, it becomes detectable in culture supernatant only at 140 h post-carbon depletion in the ΔflbA strain, although its transcription peaked at 16 h post-carbon depletion in wild-type (N402), ΔflbA, and ΔbrlA strains [44]. Similarly, a number of hydrolytic genes that displayed strong transcriptional upregulation during carbon starvation, including chitinases and mannanases, were not detectable in culture supernatants;
- the anafp promoter is activated during osmotic stress provoked by different salts including NaCl, CaCl2, KCl, MgCl2, and KH2PO4;
- the anafp promoter is activated in the presence of H2O2, but inhibited in the presence of menadione. Such an opposing effect of both oxidants is in good agreement with previous findings that autophagy-deficient A. niger strains deleted for atg1 (predicted Ser/Thr kinase) or atg8 (predicted autophagy-related ubiquitin modifier) are both more sensitive to H2O2, but less susceptible to menadione when compared to the wild-type strain [45].
3.2. When and Where Is the anafp Gene Expressed?
4. A Small Interlude: Sporulation in Bacteria and the Importance of Cannibal Toxins
5. Are AFPs Cannibal Toxins?
- expression of these peptides is strongly repressed during vegetative growth;
- expression of these peptides is derepressed during environmental stress conditions favoring sporulation;
- expression of these peptides is under tight temporal and spatial control;
- overexpression of these peptides is detrimental for the producing strain and causes autolysis;
- expression of anafp, similarly to those of skf and sdp operons, occurs only in a subpopulation of cells;
- expression of these peptides is concomitant with expression of autophagic and autolytic proteins;
- expression of these peptides decreases when commitment to sporulation has been achieved;
- deletion of the respective peptide-encoding genes does not prevent sporulation, but might affect timing and/or efficacy of sporulation;
- sporulation causes death of a significant portion of the population, which releases nutrients to feed survivors;
- host cells are immune against their own toxins; that is, in MIC assays, they appear less sensitive to their own AMPs compared to alien AMPs;
- these peptides are membrane-interacting, whereby Skf and Sdp act specifically antibacterial and AFPs specifically antifungal;
- the primary function of these peptides could be to kill genetically identical siblings ( cannibalism); however, they can also function in the defense against other fungal (or bacterial) species.
6. Conclusions
Acknowledgments
Conflicts of Interest
References
- Wang, G.; Li, X.; Wang, Z. APD3: The antimicrobial peptide database as a tool for research and education. Nucleic Acids Research 44, D1087–D1093. 2006. Available online: http://aps.unmc.edu/AP (accessed on 23 April 2018).
- Yeaman, M.R.; Yount, N.Y. Unifying themes in host defence effector polypeptides. Nat. Rev. Microbiol. 2007, 5, 727–740. [Google Scholar] [CrossRef] [PubMed]
- Yount, N.Y.; Yeaman, M.R. Multidimensional signatures in antimicrobial peptides. Proc. Natl. Acad. Sci. USA 2004, 101, 7363–7368. [Google Scholar] [CrossRef] [PubMed]
- Marx, F.; Binder, U.; Leiter, E.; Pocsi, I. The Penicillium chrysogenum antifungal protein PAF, a promising tool for the development of new antifungal therapies and fungal cell biology studies. Cell. Mol. Life Sci. 2008, 65, 445–454. [Google Scholar] [CrossRef] [PubMed]
- Meyer, V. A small protein that fights fungi: AFP as a new promising antifungal agent of biotechnological value. Appl. Microbiol. Biotechnol. 2008, 78, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Palicz, Z.; Gall, T.; Leiter, E.; Kollar, S.; Kovacs, I.; Miszti-Blasius, K.; Pocsi, I.; Csernoch, L.; Szentesi, P. Application of a low molecular weight antifungal protein from Penicillium chrysogenum (PAF) to treat pulmonary aspergillosis in mice. Emerg. Microbes Infect. 2016, 5, e114. [Google Scholar] [CrossRef] [PubMed]
- Palicz, Z.; Jenes, A.; Gall, T.; Miszti-Blasius, K.; Kollar, S.; Kovacs, I.; Emri, M.; Marian, T.; Leiter, E.; Pocsi, I.; et al. In vivo application of a small molecular weight antifungal protein of Penicillium chrysogenum (PAF). Toxicol. Appl. Pharmacol. 2013, 269, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Meyer, V.; Andersen, M.R.; Brakhage, A.A.; Braus, G.H.; Caddick, M.X.; Cairns, T.C.; de Vries, R.P.; Haarmann, T.; Hansen, K.; Hertz-Fowler, C.; et al. Current challenges of research on filamentous fungi in relation to human welfare and a sustainable bio-economy: A white paper. Fungal Biol. Biotechnol. 2016, 3, 6. [Google Scholar] [CrossRef] [PubMed]
- Olson, B.H.; Goerner, G.L. Alpha Sarcin, a New Antitumor Agent. I. Isolation, Purification, Chemical Composition, and the Identity of a New Amino Acid. Appl. Microbiol. 1965, 13, 314–321. [Google Scholar] [PubMed]
- Paege, N.; Jung, S.; Schape, P.; Muller-Hagen, D.; Ouedraogo, J.P.; Heiderich, C.; Jedamzick, J.; Nitsche, B.M.; van den Hondel, C.A.; Ram, A.F.; et al. A Transcriptome Meta-Analysis Proposes Novel Biological Roles for the Antifungal Protein AnAFP in Aspergillus niger. PLoS ONE 2016, 11, e0165755. [Google Scholar] [CrossRef] [PubMed]
- Petrey, D.; Honig, B. GRASP2: Visualization, surface properties, and electrostatics of macromolecular structures and sequences. Methods Enzymol. 2003, 374, 492–509. [Google Scholar] [PubMed]
- Garrigues, S.; Gandia, M.; Popa, C.; Borics, A.; Marx, F.; Coca, M.; Marcos, J.F.; Manzanares, P. Efficient production and characterization of the novel and highly active antifungal protein AfpB from Penicillium digitatum. Sci. Rep. 2017, 7, 14663. [Google Scholar] [CrossRef] [PubMed]
- Huber, A.; Hajdu, D.; Bratschun-Khan, D.; Gaspari, Z.; Varbanov, M.; Philippot, S.; Fizil, A.; Czajlik, A.; Kele, Z.; Sonderegger, C.; et al. New Antimicrobial Potential and Structural Properties of PAFB: A Cationic, Cysteine-Rich Protein from Penicillium chrysogenum Q176. Sci. Rep. 2018, 8, 1751. [Google Scholar] [CrossRef] [PubMed]
- Viragh, M.; Marton, A.; Vizler, C.; Toth, L.; Vagvolgyi, C.; Marx, F.; Galgoczy, L. Insight into the antifungal mechanism of Neosartorya fischeri antifungal protein. Protein Cell 2015, 6, 518–528. [Google Scholar] [CrossRef] [PubMed]
- Wnendt, S.; Ulbrich, N.; Stahl, U. Molecular cloning, sequence analysis and expression of the gene encoding an antifungal-protein from Aspergillus giganteus. Curr. Genet. 1994, 25, 519–523. [Google Scholar] [CrossRef] [PubMed]
- Campos-Olivas, R.; Bruix, M.; Santoro, J.; Lacadena, J.; del Pozo, A.M.; Gavilanes, J.G.; Rico, M. NMR solution structure of the antifungal protein from Aspergillus giganteus: Evidence for cysteine pairing isomerism. Biochemistry 1995, 34, 3009–3021. [Google Scholar] [CrossRef] [PubMed]
- Meyer, V.; Wedde, M.; Stahl, U. Transcriptional regulation of the Antifungal Protein in Aspergillus giganteus. Mol. Genet. Genom. 2002, 266, 747–757. [Google Scholar]
- Hagen, S.; Marx, F.; Ram, A.F.; Meyer, V. The antifungal protein AFP from Aspergillus giganteus inhibits chitin synthesis in sensitive fungi. Appl. Environ. Microbiol. 2007, 73, 2128–2134. [Google Scholar] [CrossRef] [PubMed]
- Theis, T.; Marx, F.; Salvenmoser, W.; Stahl, U.; Meyer, V. New insights into the target site and mode of action of the antifungal protein of Aspergillus giganteus. Res. Microbiol. 2005, 156, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Theis, T.; Wedde, M.; Meyer, V.; Stahl, U. The antifungal protein from Aspergillus giganteus causes membrane permeabilization. Antimicrob. Agents Chemother. 2003, 47, 588–593. [Google Scholar] [CrossRef] [PubMed]
- Martinez Del Pozo, A.; Lacadena, V.; Mancheno, J.M.; Olmo, N.; Onaderra, M.; Gavilanes, J.G. The antifungal protein AFP of Aspergillus giganteus is an oligonucleotide/oligosaccharide binding (OB) fold-containing protein that produces condensation of DNA. J. Biol. Chem. 2002, 277, 46179–46183. [Google Scholar] [CrossRef] [PubMed]
- Damveld, R.A.; Arentshorst, M.; Franken, A.; vanKuyk, P.A.; Klis, F.M.; van den Hondel, C.A.; Ram, A.F. The Aspergillus niger MADS-box transcription factor RlmA is required for cell wall reinforcement in response to cell wall stress. Mol. Microbiol. 2005, 58, 305–319. [Google Scholar] [CrossRef] [PubMed]
- Fujioka, T.; Mizutani, O.; Furukawa, K.; Sato, N.; Yoshimi, A.; Yamagata, Y.; Nakajima, T.; Abe, K. MpkA-Dependent and -independent cell wall integrity signaling in Aspergillus nidulans. Eukaryot. Cell 2007, 6, 1497–1510. [Google Scholar] [CrossRef] [PubMed]
- Levin, D.E. Cell wall integrity signaling in Saccharomyces cerevisiae. Microbiol. Mol. Biol. Rev. 2005, 69, 262–291. [Google Scholar] [CrossRef] [PubMed]
- Meyer, V.; Damveld, R.A.; Arentshorst, M.; Stahl, U.; van den Hondel, C.A.; Ram, A.F. Survival in the presence of antifungals: Genome-wide expression profiling of Aspergillus niger in response to sublethal concentrations of caspofungin and fenpropimorph. J. Biol. Chem. 2007, 282, 32935–32948. [Google Scholar] [CrossRef] [PubMed]
- Ouedraogo, J.P.; Hagen, S.; Spielvogel, A.; Engelhardt, S.; Meyer, V. Survival strategies of yeast and filamentous fungi against the antifungal protein AFP. J. Biol. Chem. 2011, 286, 13859–13868. [Google Scholar] [CrossRef] [PubMed]
- Binder, U.; Bencina, M.; Eigentler, A.; Meyer, V.; Marx, F. The Aspergillus giganteus antifungal protein AFPNN5353 activates the cell wall integrity pathway and perturbs calcium homeostasis. BMC Microbiol. 2011, 11, 209. [Google Scholar] [CrossRef] [PubMed]
- Meyer, V.; Technische Universität Berlin, Institute of Biotechnology, Department Applied and Molecular Microbiology, Gustav-Meyer-Allee 25, D-13355 Berlin, Germany. Wet lab experiments. Unpublished data. 2010. [Google Scholar]
- Hegedus, N.; Sigl, C.; Zadra, I.; Pocsi, I.; Marx, F. The paf gene product modulates asexual development in Penicillium chrysogenum. J. Basic Microbiol. 2011, 51, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Leiter, E.; Szappanos, H.; Oberparleiter, C.; Kaiserer, L.; Csernoch, L.; Pusztahelyi, T.; Emri, T.; Pocsi, I.; Salvenmoser, W.; Marx, F. Antifungal protein PAF severely affects the integrity of the plasma membrane of Aspergillus nidulans and induces an apoptosis-like phenotype. Antimicrob. Agents Chemother. 2005, 49, 2445–2453. [Google Scholar] [CrossRef] [PubMed]
- Binder, U.; Oberparleiter, C.; Meyer, V.; Marx, F. The antifungal protein PAF interferes with PKC/MPK and cAMP/PKA signalling of Aspergillus nidulans. Mol. Microbiol. 2010, 75, 294–307. [Google Scholar] [CrossRef] [PubMed]
- Oberparleiter, C.; Kaiserer, L.; Haas, H.; Ladurner, P.; Andratsch, M.; Marx, F. Active internalization of the Penicillium chrysogenum antifungal protein PAF in sensitive aspergilli. Antimicrob. Agents Chemother. 2003, 47, 3598–3601. [Google Scholar] [CrossRef] [PubMed]
- Binder, U.; Bencina, M.; Fizil, A.; Batta, G.; Chhillar, A.K.; Marx, F. Protein kinase A signaling and calcium ions are major players in PAF mediated toxicity against Aspergillus niger. FEBS Lett. 2015, 589, 1266–1271. [Google Scholar] [CrossRef] [PubMed]
- Marx, F. Small, basic antifungal proteins secreted from filamentous ascomycetes: A comparative study regarding expression, structure, function and potential application. Appl. Microbiol. Biotechnol. 2004, 65, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Adams, T.H.; Wieser, J.K.; Yu, J.H. Asexual sporulation in Aspergillus nidulans. Microbiol. Mol. Biol. Rev. 1998, 62, 35–54. [Google Scholar] [PubMed]
- Meyer, V.; Stahl, U. New insights in the regulation of the afp gene encoding the antifungal protein of Aspergillus giganteus. Curr. Genet. 2002, 42, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Garrigues, S.; Gandia, M.; Marcos, J.F. Occurrence and function of fungal antifungal proteins: A case study of the citrus postharvest pathogen Penicillium digitatum. Appl. Microbiol. Biotechnol. 2016, 100, 2243–2256. [Google Scholar] [CrossRef] [PubMed]
- Pel, H.J.; de Winde, J.H.; Archer, D.B.; Dyer, P.S.; Hofmann, G.; Schaap, P.J.; Turner, G.; de Vries, R.P.; Albang, R.; Albermann, K.; et al. Genome sequencing and analysis of the versatile cell factory Aspergillus niger CBS 513.88. Nat. Biotechnol. 2007, 25, 221–231. [Google Scholar] [CrossRef] [PubMed]
- Meyer, V.; Fiedler, M.; Nitsche, B.; King, R. The Cell Factory Aspergillus Enters the Big Data Era: Opportunities and Challenges for Optimising Product Formation. Adv. Biochem. Eng. Biotechnol. 2015, 149, 91–132. [Google Scholar] [PubMed]
- Nitsche, B.M.; Meyer, V. Transcriptomics of Industrial Filamentous Fungi: A New View on Regulation, Physiology, and Application. In Fungal Genomics, The Mycota XIII, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2014; Volume 13, pp. 209–232. [Google Scholar]
- Meyer, V.; Arentshorst, M.; Flitter, S.J.; Nitsche, B.M.; Kwon, M.J.; Reynaga-Pena, C.G.; Bartnicki-Garcia, S.; van den Hondel, C.A.; Ram, A.F. Reconstruction of signaling networks regulating fungal morphogenesis by transcriptomics. Eukaryot. Cell 2009, 8, 1677–1691. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, B.M.; Crabtree, J.; Cerqueira, G.C.; Meyer, V.; Ram, A.F.; Wortman, J.R. New resources for functional analysis of omics data for the genus Aspergillus. BMC Genom. 2011, 12, 486. [Google Scholar] [CrossRef] [PubMed]
- Krijgsheld, P.; Bleichrodt, R.; van Veluw, G.J.; Wang, F.; Muller, W.H.; Dijksterhuis, J.; Wosten, H.A. Development in Aspergillus. Stud. Mycol. 2013, 74, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Van Munster, J.M.; Nitsche, B.M.; Akeroyd, M.; Dijkhuizen, L.; van der Maarel, M.J.; Ram, A.F. Systems approaches to predict the functions of glycoside hydrolases during the life cycle of Aspergillus niger using developmental mutants brlA and flbA. PLoS ONE 2015, 10, e0116269. [Google Scholar] [CrossRef] [PubMed]
- Nitsche, B.M.; Welzen, A.M.B.; Lamers, G.; Meyer, V.; Ram, A.F. Autophagy promotes survival in aging submerged cultures of the filamentous fungus Aspergillus niger. Appl. Microbiol. Biotechnol. 2013, 97, 8205–8218. [Google Scholar] [CrossRef] [PubMed]
- Drysdale, M.R.; Kolze, S.E.; Kelly, J.M. The Aspergillus niger carbon catabolite repressor encoding gene, creA. Gene 1993, 130, 241–245. [Google Scholar] [CrossRef]
- Clutterbuck, A.J. A mutational analysis of conidial development in Aspergillus nidulans. Genetics 1969, 63, 317–327. [Google Scholar] [PubMed]
- Hu, P.; Wang, Y.; Zhou, J.; Pan, Y.; Liu, G. acstuA, which encodes an APSES transcription regulator, is involved in conidiation, cephalosporin biosynthesis and cell wall integrity of Acremonium chrysogenum. Fungal Genet. Biol. 2015, 83, 26–40. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, D.C.; Doedt, T.; Chiang, L.Y.; Kim, H.S.; Chen, D.; Nierman, W.C.; Filler, S.G. The Aspergillus fumigatus StuA protein governs the up-regulation of a discrete transcriptional program during the acquisition of developmental competence. Mol. Biol. Cell 2005, 16, 5866–5879. [Google Scholar] [CrossRef] [PubMed]
- Sigl, C.; Haas, H.; Specht, T.; Pfaller, K.; Kurnsteiner, H.; Zadra, I. Among developmental regulators, StuA but not BrlA is essential for penicillin V production in Penicillium chrysogenum. Appl. Environ. Microbiol. 2011, 77, 972–982. [Google Scholar] [CrossRef] [PubMed]
- Paege, N.; Jung, S.; Meyer, V. Gene co-expression network analyses predict several transcription factors controlling anafp expression in Aspergillus niger. In preparation.
- Nitsche, B.M.; Jorgensen, T.R.; Akeroyd, M.; Meyer, V.; Ram, A.F. The carbon starvation response of Aspergillus niger during submerged cultivation: Insights from the transcriptome and secretome. BMC Genom. 2012, 13, 380. [Google Scholar] [CrossRef] [PubMed]
- Emri, T.; Vekony, V.; Gila, B.; Nagy, F.; Forgacs, K.; Pocsi, I. Autolytic hydrolases affect sexual and asexual development of Aspergillus nidulans. Folia Microbiol. (Praha) 2018. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Hu, X.; Xia, Y.; Xiao, G.; Zheng, P.; Wang, C. Linkage of oxidative stress and mitochondrial dysfunctions to spontaneous culture degeneration in Aspergillus nidulans. Mol. Cell. Proteom. 2014, 13, 449–461. [Google Scholar] [CrossRef] [PubMed]
- Pocsi, I.; Leiter, E.; Kwon, N.J.; Shin, K.S.; Kwon, G.S.; Pusztahelyi, T.; Emri, T.; Abuknesha, R.A.; Price, R.G.; Yu, J.H. Asexual sporulation signalling regulates autolysis of Aspergillus nidulans via modulating the chitinase ChiB production. J. Appl. Microbiol. 2009, 107, 514–523. [Google Scholar] [CrossRef] [PubMed]
- Thrane, C.; Kaufmann, U.; Stummann, B.M.; Olsson, S. Activation of caspase-like activity and poly (ADP-ribose) polymerase degradation during sporulation in Aspergillus nidulans. Fungal Genet. Biol. 2004, 41, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Wanka, F.; Arentshorst, M.; Cairns, T.C.; Jorgensen, T.; Ram, A.F.; Meyer, V. Highly active promoters and native secretion signals for protein production during extremely low growth rates in Aspergillus niger. Microb. Cell Fact. 2016, 15, 145. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, B.; Hegedus, N.; Balint, M.; Szabo, Z.; Emri, T.; Kiss, G.; Antal, M.; Pocsi, I.; Leiter, E. Penicillium antifungal protein (PAF) is involved in the apoptotic and autophagic processes of the producer Penicillium chrysogenum. Acta Microbiol. Immunol. Hung. 2014, 61, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Pastor, J.E. Cannibalism: A social behavior in sporulating Bacillus subtilis. FEMS Microbiol. Rev. 2011, 35, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Dworkin, J.; Losick, R. Developmental commitment in a bacterium. Cell 2005, 121, 401–409. [Google Scholar] [CrossRef] [PubMed]
- De Jong, I.G.; Haccou, P.; Kuipers, O.P. Bet hedging or not? A guide to proper classification of microbial survival strategies. Bioessays 2011, 33, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.T.; Yang, Y.L.; Xu, Y.; Lamsa, A.; Haste, N.M.; Yang, J.Y.; Ng, J.; Gonzalez, D.; Ellermeier, C.D.; Straight, P.D.; et al. Imaging mass spectrometry of intraspecies metabolic exchange revealed the cannibalistic factors of Bacillus subtilis. Proc. Natl. Acad. Sci. USA 2010, 107, 16286–16290. [Google Scholar] [CrossRef] [PubMed]
- Piggot, P.J.; Hilbert, D.W. Sporulation of Bacillus subtilis. Curr. Opin. Microbiol. 2004, 7, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Branda, S.S.; Gonzalez-Pastor, J.E.; Ben-Yehuda, S.; Losick, R.; Kolter, R. Fruiting body formation by Bacillus subtilis. Proc. Natl. Acad. Sci. USA 2001, 98, 11621–11626. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Qu, L.J.; Gu, H.; Chen, Z. A 3.1-kb genomic fragment of Bacillus subtilis encodes the protein inhibiting growth of Xanthomonas oryzae pv. oryzae. J. Appl. Microbiol. 2001, 91, 1044–1050. [Google Scholar] [CrossRef] [PubMed]
- Nonejuie, P.; Trial, R.M.; Newton, G.L.; Lamsa, A.; Perera, V.R.; Aguilar, J.; Liu, W.T.; Dorrestein, P.C.; Pogliano, J.; Pogliano, K. Application of bacterial cytological profiling to crude natural product extracts reveals the antibacterial arsenal of Bacillus subtilis. J. Antibiot. (Tokyo) 2016, 69, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Lamsa, A.; Liu, W.T.; Dorrestein, P.C.; Pogliano, K. The Bacillus subtilis cannibalism toxin SDP collapses the proton motive force and induces autolysis. Mol. Microbiol. 2012, 84, 486–500. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.K.; Kwon, N.J.; Lee, I.S.; Jung, S.; Kim, S.C.; Yu, J.H. Negative regulation and developmental competence in Aspergillus. Sci. Rep. 2016, 6, 28874. [Google Scholar] [CrossRef] [PubMed]
- Adams, T.H.; Boylan, M.T.; Timberlake, W.E. brlA is necessary and sufficient to direct conidiophore development in Aspergillus nidulans. Cell 1988, 54, 353–362. [Google Scholar] [CrossRef]
- DߣSouza, C.A.; Lee, B.N.; Adams, T.H. Characterization of the role of the FluG protein in asexual development of Aspergillus nidulans. Genetics 2001, 158, 1027–1036. [Google Scholar] [PubMed]
- Garzia, A.; Etxebeste, O.; Herrero-Garcia, E.; Ugalde, U.; Espeso, E.A. The concerted action of bZip and cMyb transcription factors FlbB and FlbD induces brlA expression and asexual development in Aspergillus nidulans. Mol. Microbiol. 2010, 75, 1314–1324. [Google Scholar] [CrossRef] [PubMed]
- Kwon, N.J.; Garzia, A.; Espeso, E.A.; Ugalde, U.; Yu, J.H. FlbC is a putative nuclear C2H2 transcription factor regulating development in Aspergillus nidulans. Mol. Microbiol. 2010, 77, 1203–1219. [Google Scholar] [CrossRef] [PubMed]
- Kwon, N.J.; Shin, K.S.; Yu, J.H. Characterization of the developmental regulator FlbE in Aspergillus fumigatus and Aspergillus nidulans. Fungal Genet. Biol. 2010, 47, 981–993. [Google Scholar] [CrossRef] [PubMed]
- Hicks, J.K.; Yu, J.H.; Keller, N.P.; Adams, T.H. Aspergillus sporulation and mycotoxin production both require inactivation of the FadA G alpha protein-dependent signaling pathway. EMBO J. 1997, 16, 4916–4923. [Google Scholar] [CrossRef] [PubMed]
- Durgan, J.; Florey, O. Cancer cell cannibalism: Multiple triggers emerge for entosis. Biochim. Biophys. Acta 2018, 1865, 831–841. [Google Scholar] [CrossRef] [PubMed]
- Hamann, J.C.; Surcel, A.; Chen, R.; Teragawa, C.; Albeck, J.G.; Robinson, D.N.; Overholtzer, M. Entosis Is Induced by Glucose Starvation. Cell Rep. 2017, 20, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.; Gallo, R.L. AMPed up immunity: How antimicrobial peptides have multiple roles in immune defense. Trends Immunol. 2009, 30, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Mishra, B.; Lau, K.; Lushnikova, T.; Golla, R.; Wang, X. Antimicrobial peptides in 2014. Pharmaceuticals (Basel) 2015, 8, 123–150. [Google Scholar] [CrossRef] [PubMed]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meyer, V.; Jung, S. Antifungal Peptides of the AFP Family Revisited: Are These Cannibal Toxins? Microorganisms 2018, 6, 50. https://doi.org/10.3390/microorganisms6020050
Meyer V, Jung S. Antifungal Peptides of the AFP Family Revisited: Are These Cannibal Toxins? Microorganisms. 2018; 6(2):50. https://doi.org/10.3390/microorganisms6020050
Chicago/Turabian StyleMeyer, Vera, and Sascha Jung. 2018. "Antifungal Peptides of the AFP Family Revisited: Are These Cannibal Toxins?" Microorganisms 6, no. 2: 50. https://doi.org/10.3390/microorganisms6020050
APA StyleMeyer, V., & Jung, S. (2018). Antifungal Peptides of the AFP Family Revisited: Are These Cannibal Toxins? Microorganisms, 6(2), 50. https://doi.org/10.3390/microorganisms6020050



