Culturing Toxic Benthic Blooms: The Fate of Natural Biofilms in a Microcosm System
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling
2.2. Laboratory Analysis
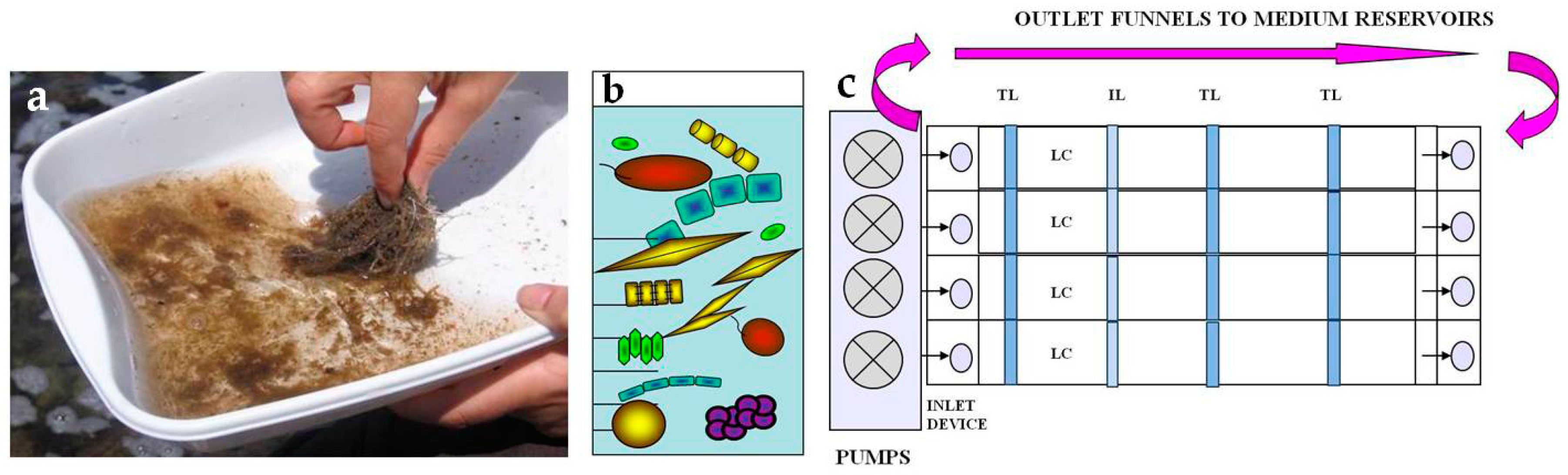
2.2.1. Preparation of Inocula
2.2.2. Counting of Ostreopsis cf. ovata Cells
2.3. Microcosm Cultures
- BiodiversityTo assess biodiversity of phototrophs in natural and cultured communities, samples scraped from randomly selected slides were fixed with formaldehyde at 2% and glutaraldehyde at 2.5% final concentrations and then stored at 4 °C. Fixed samples were examined using a ZEISS Axioskop light microscope equipped with differential interference contrast (DIC) at 40× and 100× objectives. Samples were also observed at light microscope after staining for 10 min with Alcian Blue (AB) 1% in HCl 0.5 N (pH 0.5) or in 3% acetic acid (pH 2.5) to evaluate the presence of sulfated and carboxylic polysaccharides in the extracellular matrix [30].
- BiomassCultured communities sampled at the three phases of development were scraped off the polycarbonate slides to evaluate the biomass. Biofilm biomass was determined as dry weight (DW) by oven drying samples at 60 °C for 72 h. Phototrophic biomass was assessed by determining Chl a concentration, extracted overnight in 90% acetone in the dark and then quantified spectrophotometrically according to [31].
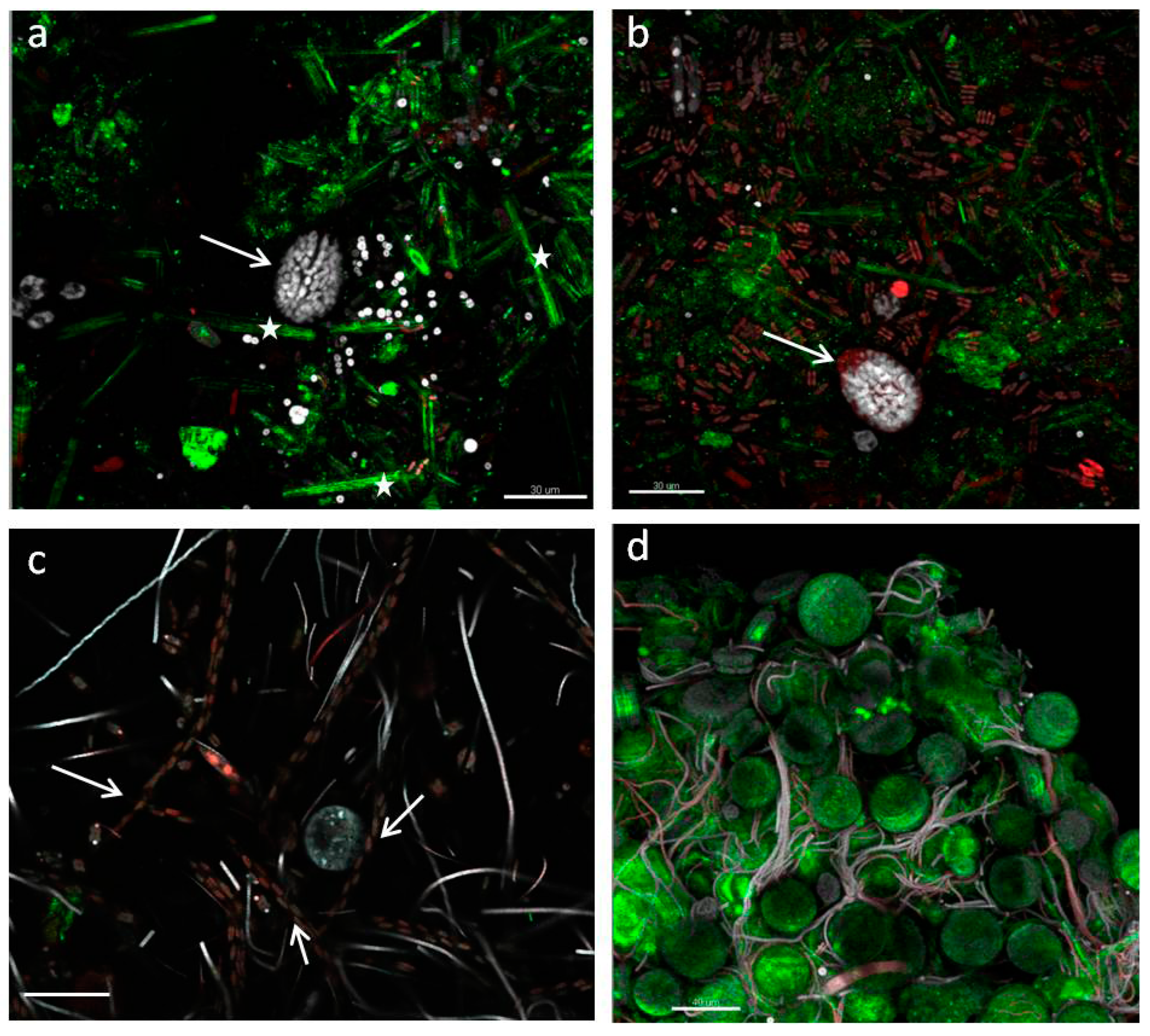
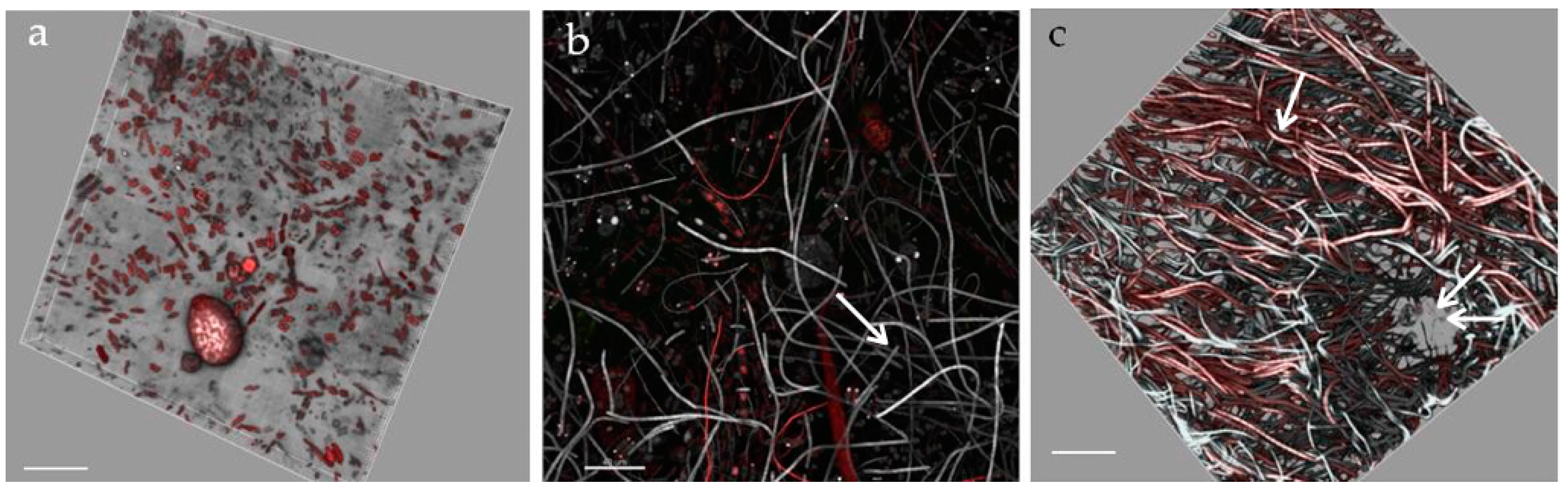
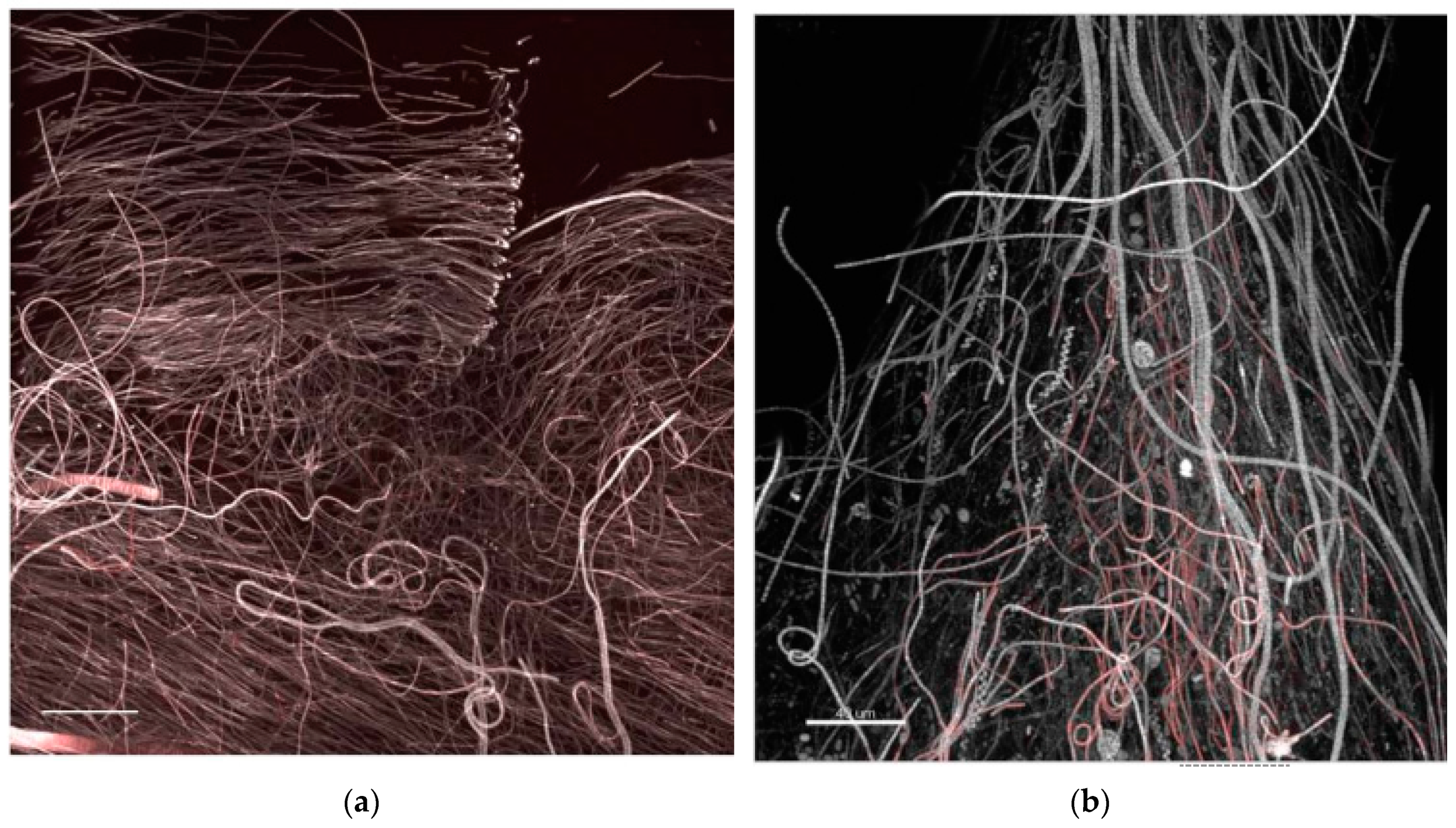
- StructureThe biofilm architecture and spatial distribution of phototrophs during the different phases of development was obtained by observing intact biofilm, after non-destructive, non-invasive sampling, at the confocal laser scanning microscope (CLSM) (Olympus FV1000, IX81 using Plan-Apochromatic 60× (NA 1.42, oil) objectives) at the Centre of Advanced Microscopy “P. Albertano”, Department of Biology, University of Rome “Tor Vergata”, Rome, Italy. CLSM was used in a multichannel mode, whereby the different channels mapped individual biofilm components. The excitation wavelengths were in the blue (488 nm, Ar), green (543 nm, Ar/HeNe), and red (636 nm, Ar/HeNe) regions. Data consisted of a set of two dimensional (2D), cross-sectional images in the x-y plane that were captured along the z-axis. The three-dimensional images were obtained through acquisitions in the XY plane, Z step size 0.5 µm, using the software package IMARIS 6.2.0 software (Bitplane AG, Zurich, Switzerland).
- Spectral analysesSpectral analysis (CLSM-SA) of different autofluorescence signals was also carried out on biofilm samples by using the lambda scan (λ scan) function of CLSM exciting specific regions of interest (1 µm2 ROIs) [32]. Lasers with the following excitation wavelengths: 488 nm; Ar, 543 nm; Ar/HeNe and red 635 nm; Ar/HeNe were used. Spectra were obtained for emission wavelengths ranging between 400 and 800 nm.
- Matrix exopolysaccharidesBound/capsular exopolysaccharides (CPS) were also extracted from mature communities using 0.1 M H2SO4 at 95 °C [33,34]. The carbohydrate fractions were measured spectrophotometrically using the phenol-sulphuric acid method [35]. In order to check the extraction efficiency of each eluent, the pellet obtained after the extraction was stained with Alcian Blue dyes, specific for acidic polysaccharides, to check localization of positive reaction in light microscopy [23]. CPS extracts were then analysed for their monosaccharide composition using RP-HPLC (reverse phase), using a Beckman Ultrasphere ODS (Octa Decyl Silane) column as reported in [36].
- ToxinsOstreopsis toxins were analysed in inocula and culture material by LC-MS (Orbitrap XL).MALDI-TOF mass spectrometry for cyanobacterial toxins was performed by AnagnosTec GmBH (Postdam, Germany) on mature biofilm samples.
3. Results and Discussion
3.1. Epibenthic Communities
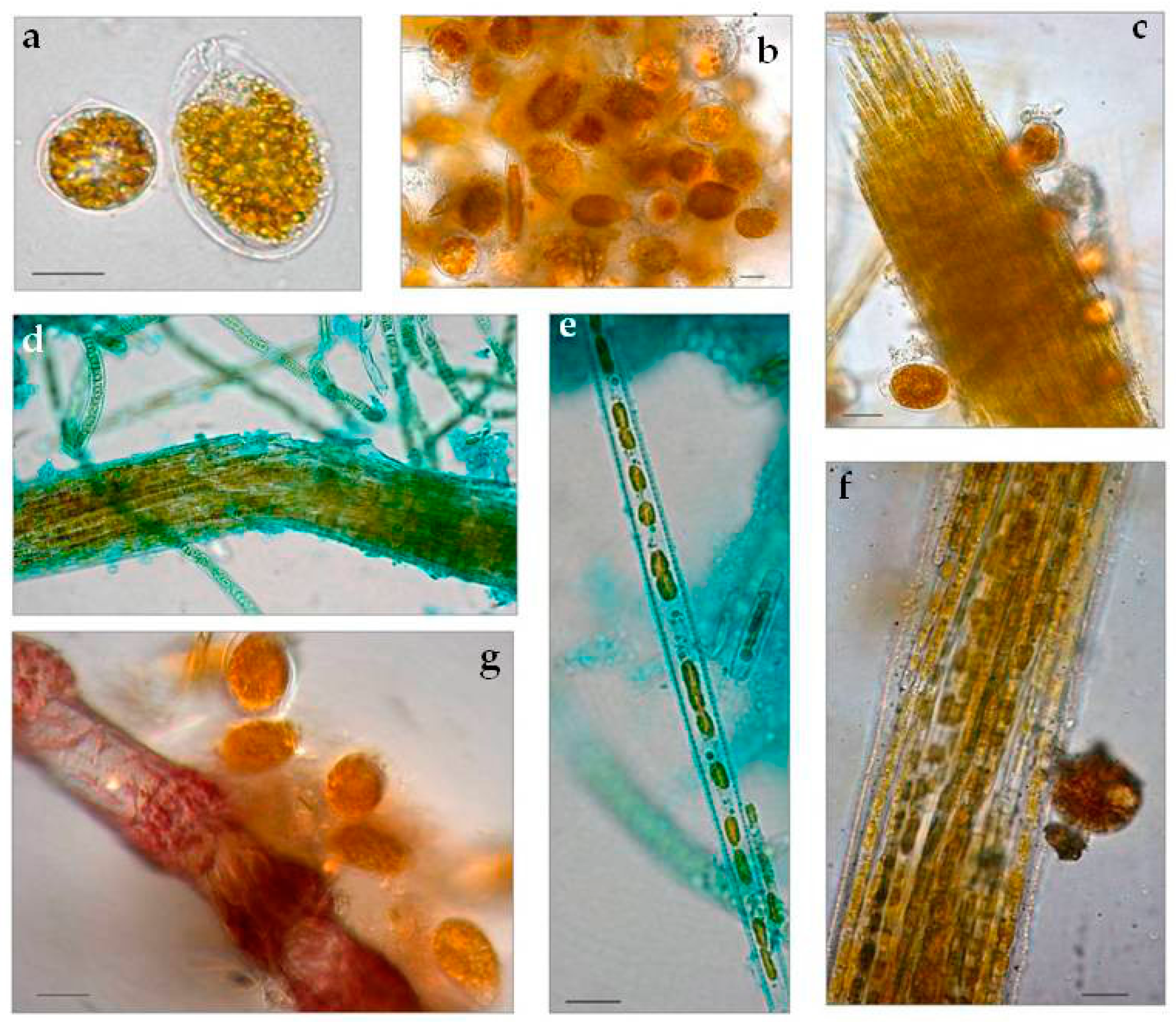
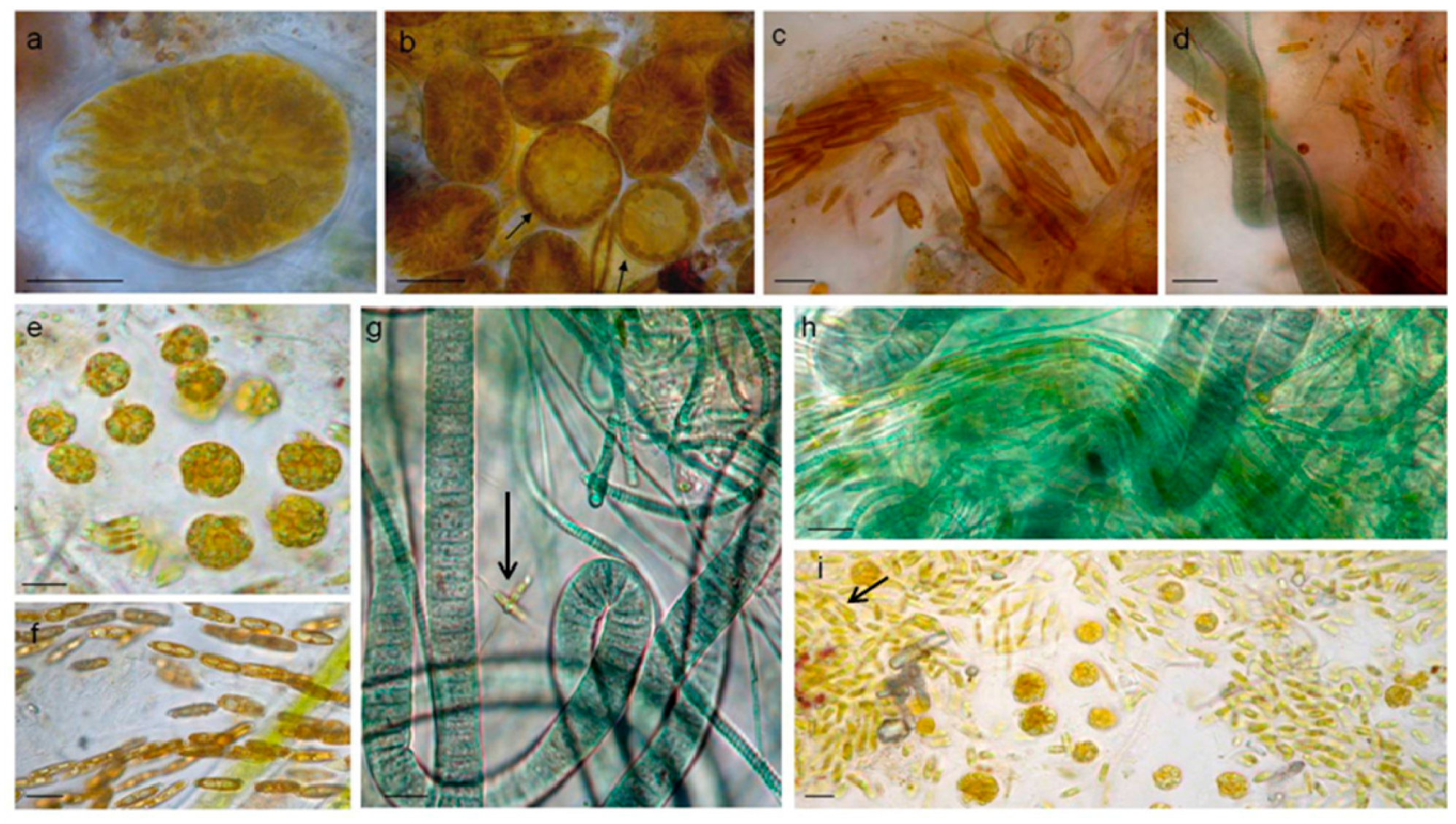
3.1.1. Taxonomic Composition
3.1.2. O. cf. ovata Abundance
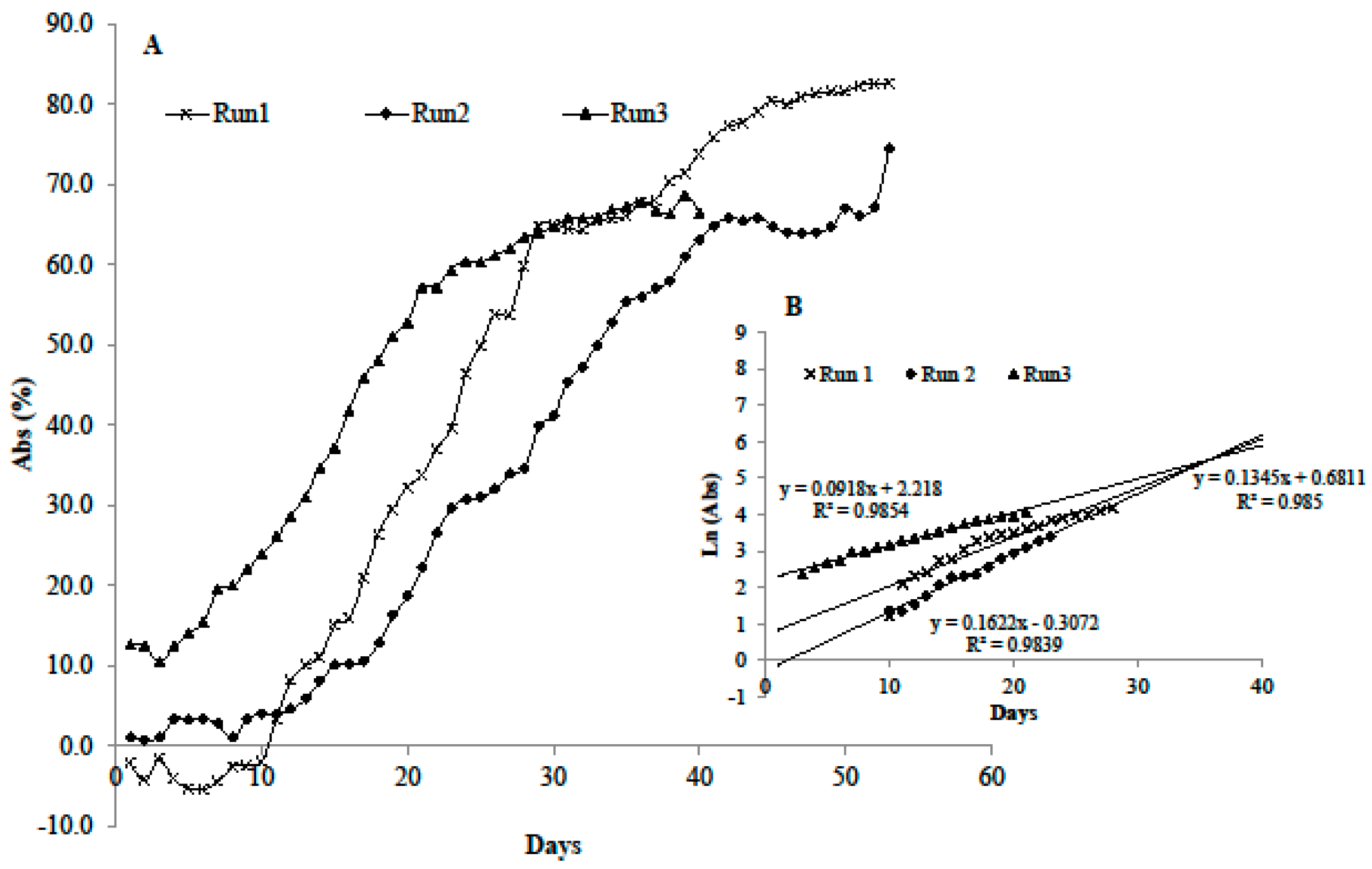
3.2. Growth in the Microcosm
3.3. Biofilm Observations
3.4. Biofilm Matrix
3.5. Biomass
3.6. Biofilm Structure
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Berdalet, E.; Tester, P.A.; Chinain, M.; Fraga, S.; Lemée, R.; Litaker, W.; Penna, A.; Usup, G.; Vila, M.; Zingone, A. Harmful algal blooms in benthic systems: Recent progress and future research. Oceanography 2017, 30, 36–45. [Google Scholar] [CrossRef]
- Accoroni, S.; Totti, C. The toxic benthic dinoflagellates of the genus Ostreopsis in temperate areas: A review. Adv. Oceanogr. Limnol. 2016, 7, 1–15. [Google Scholar] [CrossRef]
- Bianco, I.; Congestri, R.; Sangiorgi, V.; Albertano, P.; Zaottini, E. Fioriture di microalghe potenzialmente tossiche lungo le coste laziali. Biol. Mar. Mediterr. 2006, 13, 947–950. [Google Scholar]
- Simoni, F.; Gaddi, A.; Di Paolo, C.; Lepri, L. Harmful epiphytic dinoflagellate on Tyrrhenian Sea reefs. Harmful Algae News 2003, 24, 13–14. [Google Scholar]
- Penna, A.; Fraga, S.; Battocchi, C.; Casabianca, S.; Perini, F.; Capellacci, S.; Casabianca, A.; Riobo, P.; Giacobbe, M.; Totti, C.; et al. Genetic diversity of the genus Ostreopsis Schmidt: Phylogeographical considerations and molecular methodology applications for field detection in the Mediterranean Sea. Cryptogam. Algol. 2012, 33, 153–163. [Google Scholar] [CrossRef]
- Battocchi, C.; Totti, C.; Vila, M.; Masó, M.; Capellacci, S.; Accoroni, S.; Reñé, A.; Scardi, M.; Penna, A. Monitoring toxic microalgae Ostreopsis (dinoflagellate) species in coastal waters of the Mediterranean Sea using molecular PCR-based assay combined with light microscopy. Mar. Pollut. Bull. 2010, 60, 1074–1084. [Google Scholar] [CrossRef] [PubMed]
- Accoroni, S.; Romagnoli, T.; Pichierri, S.; Totti, C. Effects of the bloom of harmful benthic dinoflagellate Ostreopsis cf. ovata on the microphytobenthos community in the northern Adriatic Sea. Harmful Algae 2016, 55, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Mangialajo, L.; Ganzin, N.; Accoroni, S.; Asnaghi, V.; Blanfuné, A.; Cabrini, M.; Cattaneo-Vietti, R.; Chavanon, F.; Chiantore, M.; Cohu, S.; et al. Trends in Ostreopsis proliferation along the Northern Mediterranean coasts. Toxicon 2011, 57, 408–420. [Google Scholar] [CrossRef] [PubMed]
- Pistocchi, R.; Pezzolesi, L.; Guidi, F.; Vanucci, S.; Guerrini, F.; Pinna, A. Inorganic nutrients uptake and organic phosphorus utilization by Ostreopsis cf. ovata. In Marine and Freshwater Harmful Algae, Proceedings of the 16th International Conference on Harmful Algae, Wellington, New Zealand, 27–31 October 2014; MacKenzie, A.L., Ed.; Cawthron Institute, International Society for the Study of Harmful Algae (ISSHA): Nelson, New Zealand, 2014; Volume 471, pp. 147–150. ISBN 978-87-990827-5-9. [Google Scholar]
- Accoroni, S.; Totti, C.; Razza, E.; Congestri, R.; Campanelli, A.; Marini, M.; Ellwood, N.T.W. Phosphatase activities of a microepiphytic community during a bloom of Ostreopsis cf. ovata in the northern Adriatic Sea. Water Res. 2017, 120, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Jauzein, C.; Couet, D.; Blasco, T.; Lemée, R. Uptake of dissolved inorganic and organic nitrogen by the benthic toxic dinoflagellate Ostreopsis cf. ovata. Harmful Algae 2017, 65, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Honsell, G.; Bonifacio, A.; De Bortoli, M.; Penna, A.; Battocchi, C.; Ciminiello, P.; Dell’Aversano, C.; Fattorusso, E.; Sosa, S.; Yasumoto, T.; et al. New insights on cytological and metabolic features of Ostreopsis cf. ovata Fukuyo (Dinophyceae): A multidisciplinary approach. PLoS ONE 2013, 8, e5791. [Google Scholar] [CrossRef]
- Barone, R. Behavioural trait of Ostreopsis ovata (Dinophyceae) in Mediterranean rock pools: The spider’s strategy. Harmful Algae News 2007, 33, 1–3. [Google Scholar]
- Escalera, L.; Benvenuto, G.; Scalco, E.; Zingone, A.; Montresor, M. Ultrastructural features of the benthic dinoflagellate Ostreopsis cf. ovata (Dinophyceae). Protist 2014, 165, 260–274. [Google Scholar] [CrossRef] [PubMed]
- Ciminiello, P.; Dell’Aversano, E.; Fattorusso, E.; Fiorino, M.; Tartaglione, L.; Grillo, C.; Melchiorre, N. Putative palytoxin and its new analogue, ovatoxin-a, in Ostreopsis ovata collected along the Ligurian coasts during the 2006 toxic outbreak. J. Am. Soc. Mass Spetr. 2008, 19, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Tubaro, A.; Durando, P.; Del Favero, G.; Ansaldi, F.; Icardi, G.; Deeds, J.R.; Sosa, S. Case definitions for human poisonings postulated to palytoxins exposure. Toxicon 2011, 57, 478–495. [Google Scholar] [CrossRef] [PubMed]
- Vila, M.; Abós-Herràndiz, R.; Isern-Fontanet, J.; Àlvarez, J.; Berdalet, E. Establishing the link between Ostreopsis cf. ovata blooms and human health impacts using ecology and epidemiology. Sci. Mar. 2016, 80, 107–115. [Google Scholar]
- Gorbi, S.; Avio, G.C.; Benedetti, M.; Totti, C.; Accoroni, S.; Pichierri, S.; Bacchiocchi, S.; Orletti, R.; Graziosi, T.; Regoli, F. Effects of harmful dinoflagellate Ostreopsis cf. ovata exposure on immunological, histological and oxidative responses of mussels Mytilus galloprovincialis. Fish Shellfish Immunol. 2013, 35, 941–950. [Google Scholar] [PubMed]
- GEOHAB. Global Ecology and Oceanography of Harmful Algal Blooms. Core Research Project: Harmful Algal Blooms in Benthic Systems; Berdalet, E., Tester, P., Zingone, A., Eds.; IOC: Newark, NY, USA; SCOR: Paris, France, 2012; p. 64. [Google Scholar]
- Parsons, M.L.; Aligizaki, K.; Dechraoui Bottein, M.Y.; Fraga, S.; Morton, S.L.; Penna, A.; Rhodes, L. Gambierdiscus and Ostreopsis: Reassessment of the state of knowledge of their taxonomy, geography, ecophysiology, and toxicology. Harmful Algae 2012, 14, 107–129. [Google Scholar] [CrossRef]
- Accoroni, S.; Romagnoli, T.; Pichierri, S.; Totti, C. New insights on the life cycle stages of the toxic benthic dinoflagellate Ostreopsis cf. ovata. Harmful Algae 2014, 34, 7–16. [Google Scholar] [CrossRef]
- Scalco, E.; Brunet, C.; Marino, F.; Rossi, R.; Soprano, V.; Zingone, A.; Montresor, M. Growth and toxicity responses of Mediterranean Ostreopsis cf. ovata to seasonal irradiance and temperature conditions. Harmful Algae 2012, 17, 25–34. [Google Scholar]
- Di Pippo, F.; Bohn, A.; Congestri, R.; De Philippis, R.; Albertano, P. Capsular polysaccharides of cultured phototrophic biofilms. Biofouling 2009, 25, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Congestri, R.; Albertano, P. Benthic diatoms in biofilm culture. In The Diatom World, Cellular Origin, Life in Extreme Habitats and Astrobiology; Part 2; Seckbach, J., Kociolek, P., Eds.; Springer Science: Berlin, Germany, 2011; Volume 19, pp. 227–243. [Google Scholar]
- Di Pippo, F.; Ellwood, N.T.W.; Guzzon, A.; Bohn, A.; Congestri, R. Diversity and biomass accumulation in cultured phototrophic biofilms. Eur. J. Phycol. 2014, 49, 384–394. [Google Scholar] [CrossRef]
- Zippel, B.; Rijstenbil, J.; Neu, T.R. A flow-lane incubator for studying freshwater and marine phototrophic biofilms. J. Microbiol. Meth. 2007, 70, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Totti, C.; Accoroni, S.; Cerino, F.; Cucchiari, E.; Romagnoli, T. Ostreopsis ovata bloom along the Conero Riviera (northern Adriatic Sea): Relationships with environmental conditions and substrata. Harmful Algae 2010, 9, 233–239. [Google Scholar] [CrossRef]
- Utermöhl, H. Zur Vervollkommnung der quantitativen Phytoplankton-Methodik. Mitt. Int. Ver. Theor. Angew. Limnol. 1958, 9, 1–38. [Google Scholar]
- Keller, M.D.; Selvin, R.C.; Claus, W.; Guillard, R.R.L. Media for the culture of oceanic ultraphytoplankton. J. Phycol. 1987, 23, 633–638. [Google Scholar] [CrossRef]
- Bellezza, S.; De Philippis, R.; Paradossi, G.; Albertano, P. Exopolysaccharides of two cyanobacterial strains from Roman hypogea. Geomicrobiol. J. 2006, 23, 301–310. [Google Scholar] [CrossRef]
- Jeffrey, S.W.; Humphrey, G.F. New spectrophotometric equations for determining chlorophylls a, b, c1 and c2 in higher plants, algae, and natural phytoplankton. Biochem. Physiol. Pflanz. 1975, 167, 191–194. [Google Scholar] [CrossRef]
- Bruno, L.; Romano, E.; Bellezza, S.; Urzi’, C.; Albertano, P. Confocal microscopy and diagnosis of biodeterioration caused by phototrophic biofilms. In Diagnosis for the Conservation and Valorization of Cultural Heritage; Campanella, L., Piccioli, C., Eds.; Museo Leonardiano: Naples, Italy, 2010; pp. 152–157. [Google Scholar]
- Barranguet, C.; Van Beuseom, S.A.M.; Veuger, B.; Neu, T.R.; Manders, E.M.M.; Sinke, J.J.; Admiraal, W. Studying undisturbed autotrophic biofilms: Still a technical challenge. Aquat. Microb. Ecol. 2004, 34, 1–9. [Google Scholar] [CrossRef]
- Barranguet, C.; Veuger, B.; van Beusekom, S.A.M.; Marvan, P.; Sinke, J.J.; Admiraal, W. Divergent composition of algal-bacterial biofilms developing under various external factors. Eur. J. Phycol. 2005, 40, 1–8. [Google Scholar] [CrossRef]
- Dubois, M. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Congestri, R.; Di Pippo, F.; De Philippis, R.; Paradossi, G.; Albertano, P. Seasonal succession of phototrophic biofilms in an Italian wastewater treatment plant: Biovolume, spatial structure and exopolysaccharides. Aquat. Microb. Ecol. 2006, 45, 301–312. [Google Scholar] [CrossRef]
- Carnicer, O.; Guallar, C.; Andree, K.B.; Diogène, J.; Fernández-Tejedor, M. Ostreopsis cf. ovata dynamics in the NW Mediterranean Sea in relation to biotic and abiotic factors. Environ. Res. 2015, 143, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Krueger, B.P.; Lampoura, S.S.; van Stokkum, I.H.M.; Papagiannakis, E.; Salverda, J.M.; Gradinaru, C.C.; Rutkauskas, D.; Hiller, R.G.; van Grondelle, R. Energy Transfer in the Peridinin Chlorophyll-a Protein of Amphidinium carterae studied by Polarized Transient Absorption and Target Analysis. Biophys. J. 2001, 80, 2843–2855. [Google Scholar] [CrossRef]
- Lamastra, F.R.; De Angelis, R.; Antonucci, A.; Salvatori, D.; Prosposito, P.; Casalboni, M.; Congestri, R.; Melino, S.; Nanni, F. Polymer composite random lasers based on diatom frustules as scatterers. RSC Adv. 2014, 4, 61809–61816. [Google Scholar] [CrossRef]
- De Angelis, R.; Melino, S.; Prosposito, P.; Casalboni, M.; Lamastra, F.R.; Nanni, F.; Bruno, L.; Congestri, R. The diatom Staurosirella pinnata for photoactive material production. PLoS ONE 2016, 11, e0165571. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.K.; Singh, R.P.; Patel, V. Isolation and Characterization of Exopolysaccharide Secreted by a Toxic Dinoflagellate, Amphidinium carterae Hulburt 1957 and Its Probable Role in Harmful Algal Blooms (HABs). Microb. Ecol. 2011, 62, 518–527. [Google Scholar] [CrossRef] [PubMed]




| Parameters | Site 1 | Site 2 |
|---|---|---|
| Temperature (°C) | 27.7 | 27.0 |
| O2 (%) | 91.0 | 101.5 |
| Salinity (‰) | 30.0 | 37.0 |
| pH | 7.8 | 8.2 |
| Host substrata and FW | 4 bryozoan colonies (28.3 g Run 1, 72.8 g Run 2) | 2 Cystoseira sp. thalli (95 g Run 3) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Pippo, F.; Congestri, R. Culturing Toxic Benthic Blooms: The Fate of Natural Biofilms in a Microcosm System. Microorganisms 2017, 5, 46. https://doi.org/10.3390/microorganisms5030046
Di Pippo F, Congestri R. Culturing Toxic Benthic Blooms: The Fate of Natural Biofilms in a Microcosm System. Microorganisms. 2017; 5(3):46. https://doi.org/10.3390/microorganisms5030046
Chicago/Turabian StyleDi Pippo, Francesca, and Roberta Congestri. 2017. "Culturing Toxic Benthic Blooms: The Fate of Natural Biofilms in a Microcosm System" Microorganisms 5, no. 3: 46. https://doi.org/10.3390/microorganisms5030046
APA StyleDi Pippo, F., & Congestri, R. (2017). Culturing Toxic Benthic Blooms: The Fate of Natural Biofilms in a Microcosm System. Microorganisms, 5(3), 46. https://doi.org/10.3390/microorganisms5030046





