Dirty Money: A Matter of Bacterial Survival, Adherence, and Toxicity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sources of Currency Used in This Study
2.2. Microbiome Preparation and Inoculation
2.3. Bacterial Cultures Used
2.4. Extraction of Bacteria from Coins
2.5. Extraction of Bacteria from Banknotes
2.6. Scanning Electron Microscopy (SEM) of Banknote Surfaces
3. Results and Discussion
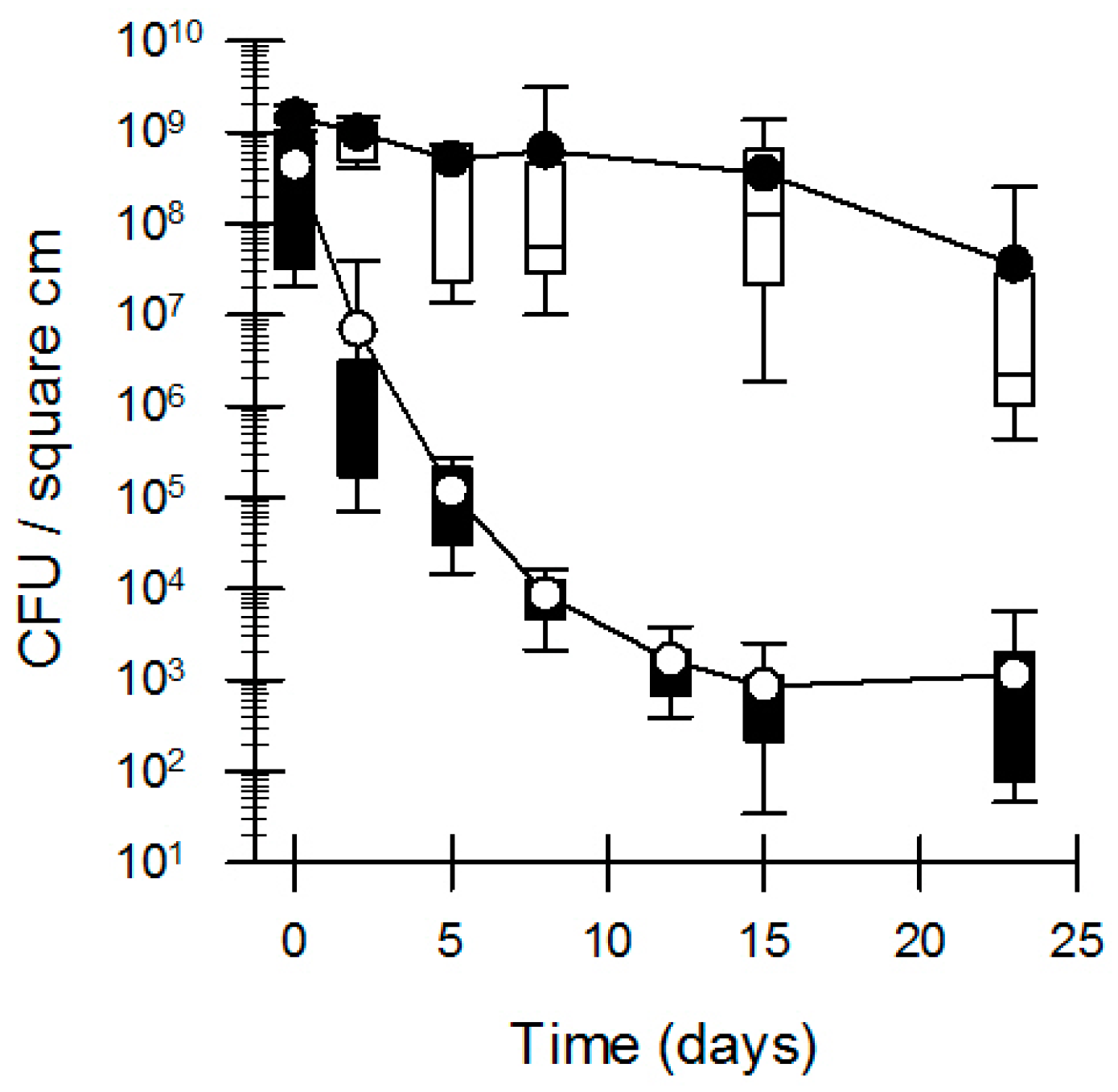
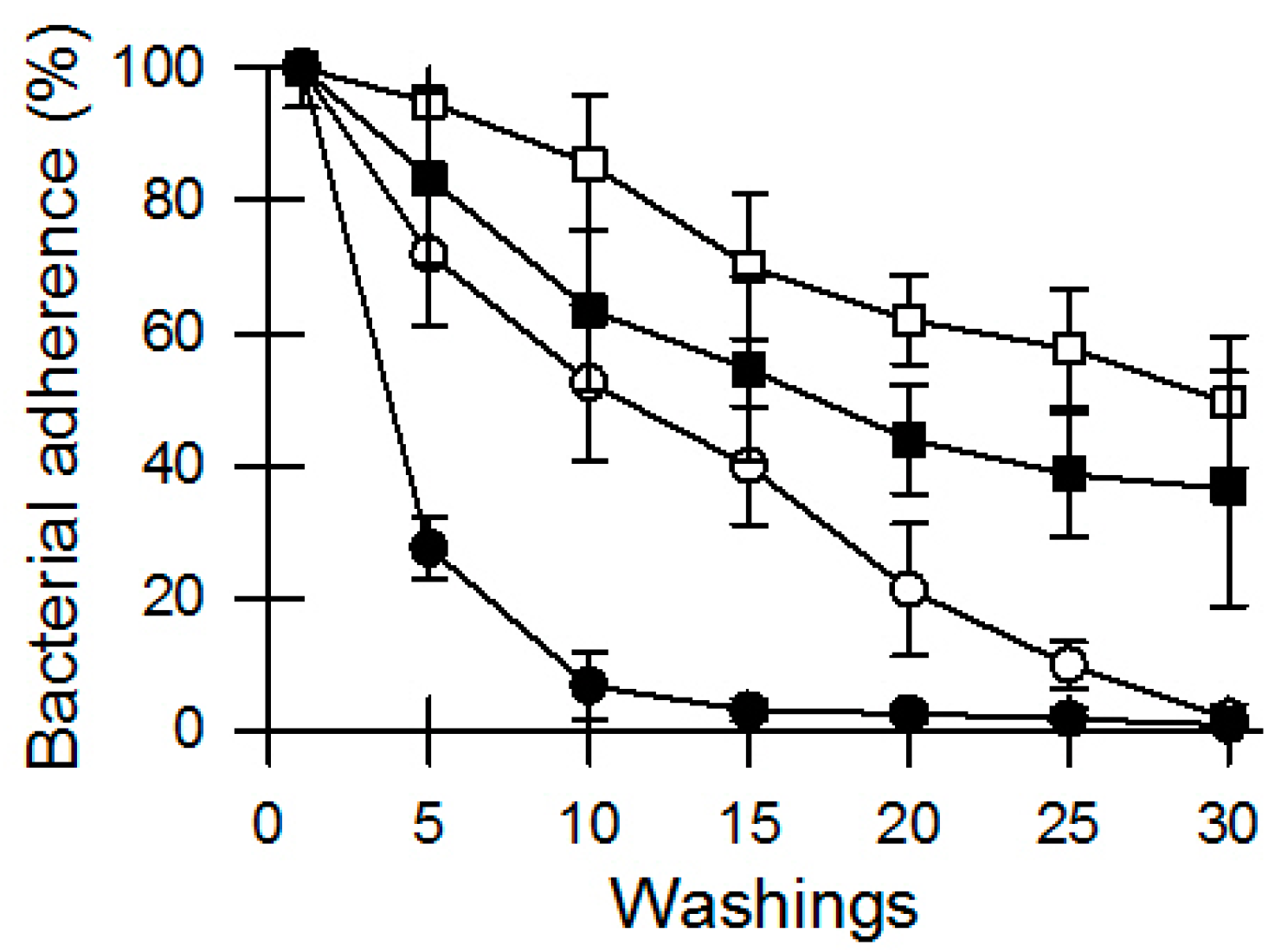
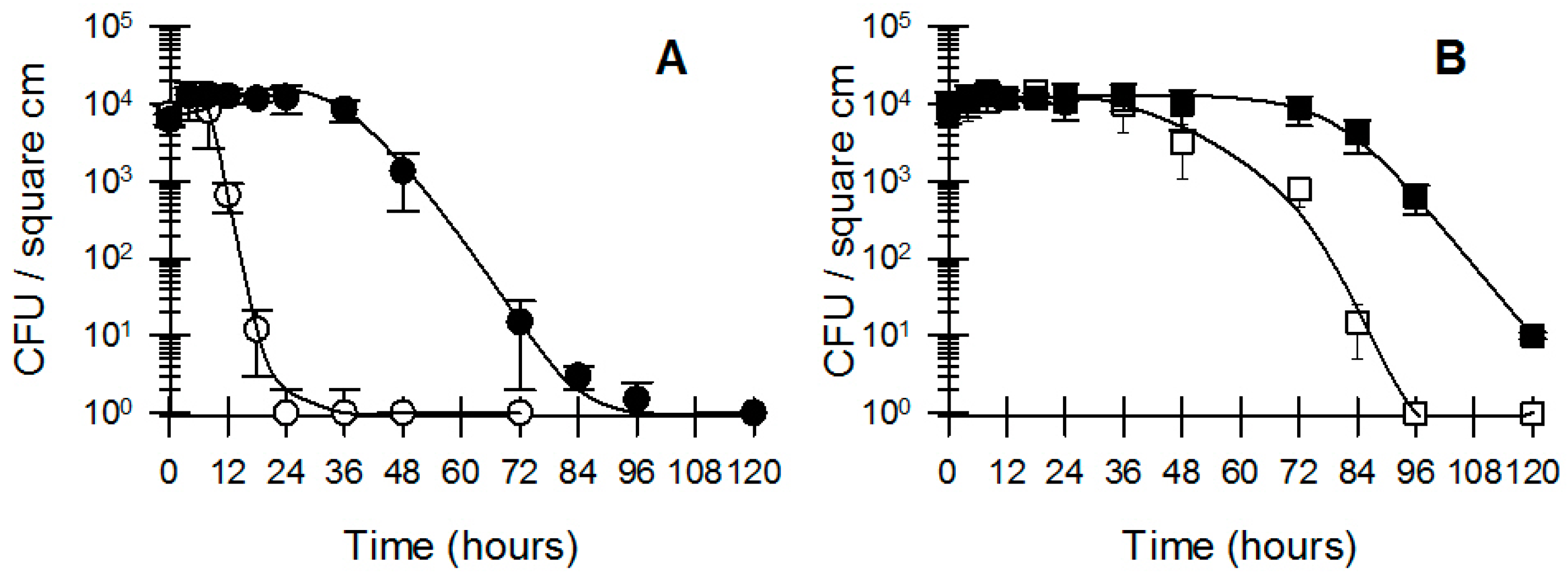
3.1. Bacterial Survival and Adherence to Banknotes
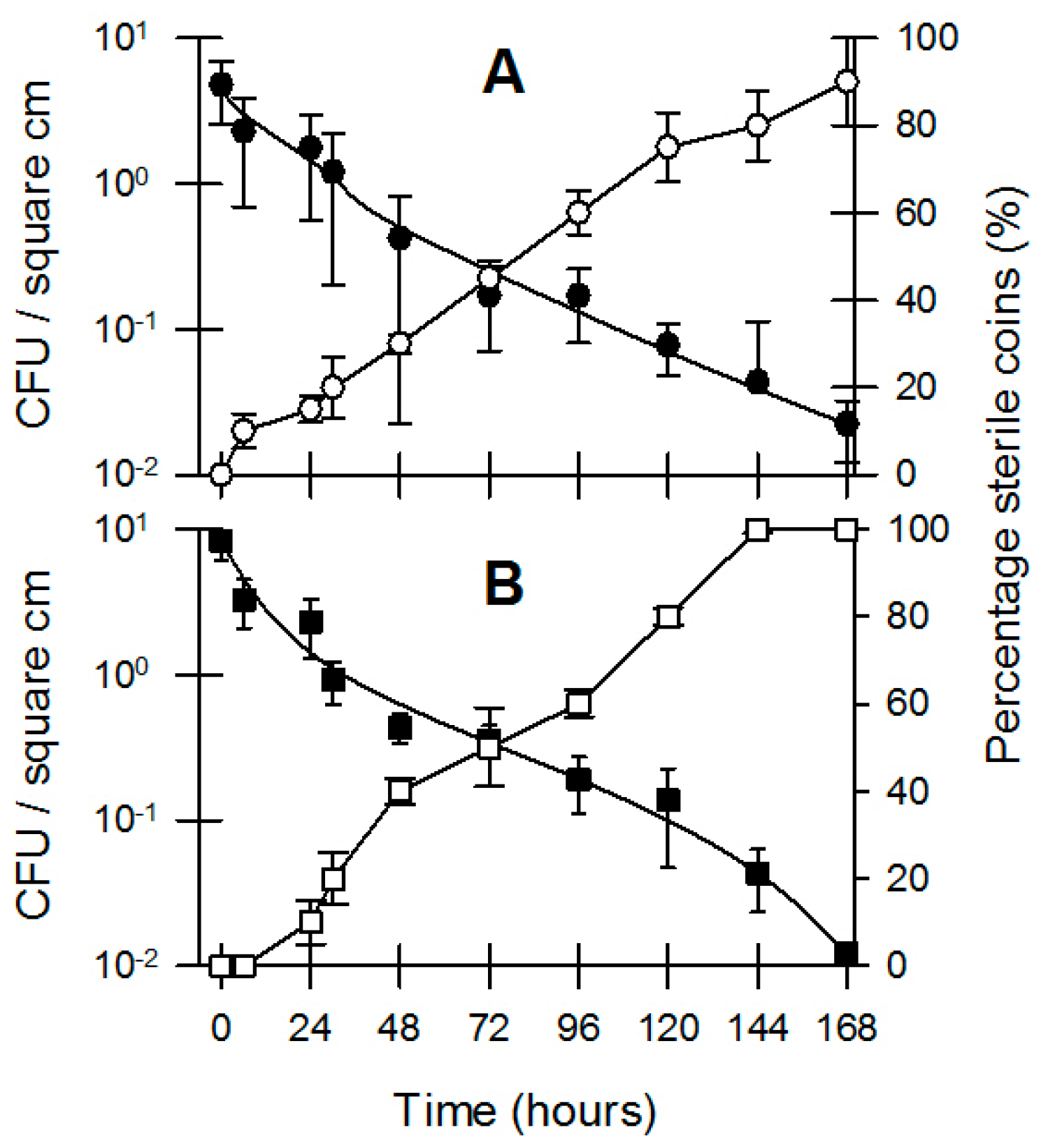
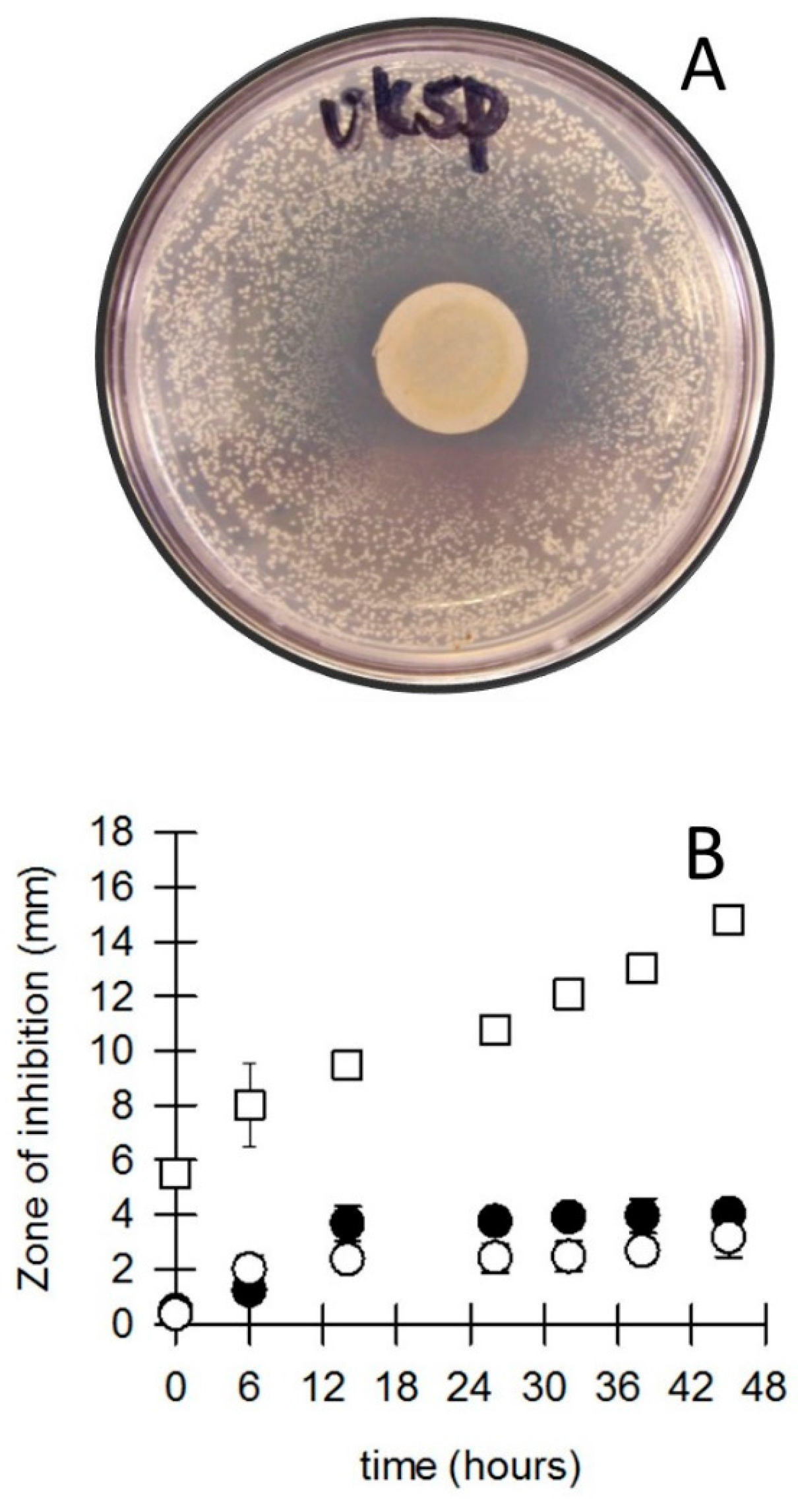
3.2. Bacterial Survival and Inhibition by Coins
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dodgson, M.; Gann, D.; Wladawsky-Berger, I.; Sultan, N.; George, G. Managing digital money. Acad. Manag. J. 2015, 58, 325–333. [Google Scholar] [CrossRef]
- Koblitz, N.; Menezes, A.J. Cryptocash, cryptocurrencies, and cryptocontracts. Des. Code Cryptogr. 2016, 78, 87–102. [Google Scholar] [CrossRef]
- Ward, C.B.; Tanner, F.W. Bacteria on subsidiary coins and currency. Am. J. Med. Sci. 1921, 162, 585–592. [Google Scholar] [CrossRef]
- Mihaéloff, S. The transference of pathogenic organisms on paper money and on coins. Ann. d’Hyg. Pub. Indust. et Sociale 1940, 18, 226–233. (In French) [Google Scholar]
- Warsa, R. Coins and paper money as potential vectors of pathogenic germs. Doc. Med. Geogr. Trop. 1952, 4, 139–174. (In French) [Google Scholar]
- Kuria, J.K.N.; Wahome, R.G.; Jobalamin, M.; Kariuki, S.M. Profile of bacteria and fungi on money coins. East Afr. Med. J. 2009, 86, 151–156. [Google Scholar] [CrossRef] [PubMed]
- Vriesekoop, F.; Russell, C.; Alvarez-Mayorga, B.; Aidoo, K.; Yuan, Q.P.; Scannell, A.; Beumer, R.R.; Jiang, X.; Barro, N.; Otokunefor, K.; et al. Dirty money: An investigation into the hygiene status of some of the world’s currencies as obtained from food outlets. Foodborne Pathog. Dis. 2010, 7, 1497–1502. [Google Scholar] [CrossRef] [PubMed]
- Gedik, H.; Voss, T.A.; Voss, A. Money and transmission of bacteria. Antimicrob. Resist. Infect. Control 2013, 2, 1–4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Su, G.L.S.; Coden, C.; Cruz, A.; Jose, R.; Victorino, A.; Sison, M.A.; De Guzman, T. Bacteriological and parasitological assessment of currencies obtained in selected markets of Metro Manila. Asian Pac. J. Trop. Dis. 2015, 5, 468–470. [Google Scholar] [CrossRef]
- Yalowitz, M.; Brook, I. The recovery of bacteria from the handpiece of a high school telephone. J. Environ. Health 2002, 65, 18–20. [Google Scholar]
- Otter, J.A.; French, G.L. Bacterial contamination on touch surfaces in the public transport system and in public areas of a hospital in London. Lett. Appl. Microbiol. 2009, 49, 803–805. [Google Scholar] [CrossRef] [PubMed]
- Ayan, S. The hidden danger for the users of playgrounds and sport complexes. Life Sci. J. 2013, 10, 2884–2890. [Google Scholar]
- Grice, E.A.; Segre, J.A. The skin microbiome. Nat. Rev. Microbiol. 2011, 9, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Edmonds-Wilson, S.L.; Nurinova, N.I.; Zapka, C.A.; Fierer, N.; Wilson, M. Review of human hand microbiome research. J. Dermatol. Sci. 2015, 80, 3–12. [Google Scholar] [CrossRef] [PubMed]
- Dréno, B.; Araviiskaia, E.; Berardesca, E.; Gontijo, G.; Sanchez Viera, M.; Xiang, L.F.; Martin, R.; Bieber, T. Microbiome in healthy skin, update for dermatologists. J. Adv. Dermatol. Venereol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Larson, E.L.; Strom, M.S.; Evans, C.A. Analysis of three variables in sampling solutions used to assay bacteria of hands: Type of solution, use of antiseptic neutralizers, and solution temperature. J. Clin. Microbiol. 1980, 12, 355–360. [Google Scholar] [PubMed]
- Burton, M.; Cobb, E.; Donachie, P.; Judah, G.; Curtis, V.; Schmidt, W.P. The effect of handwashing with water or soap on bacterial contamination of hands. Int. J. Environ. Res. Public Health 2011, 8, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Lopez, G.U.; Gerba, C.P.; Tamimi, A.H.; Kitajima, M.; Maxwell, S.L.; Rose, J.B. Transfer efficiency of bacteria and viruses from porous and nonporous fomites to fingers under different relative humidity conditions. Appl. Environ. Microbiol. 2013, 79, 5728–5734. [Google Scholar] [CrossRef] [PubMed]
- De Morais Teixeira, E.; Corrêa, A.C.; Manzoli, A.; de Lima Leite, F.; de Oliveira, C.R.; Mattoso, L.H.C. Cellulose nanofibers from white and naturally colored cotton fibers. Cellulose 2010, 17, 595–606. [Google Scholar] [CrossRef]
- Bledzki, A.K.; Gassan, J. Composites reinforced with cellulose based fibres. Prog. Polym. Sci. 1999, 24, 221–274. [Google Scholar] [CrossRef]
- Collings, T.; Milner, D. The identification of oriental paper-making fibres. Pap. Conserv. 1978, 3, 51–79. [Google Scholar] [CrossRef]
- Ericsson, H.; Hogman, C.; Wickman, K. A paper disk method for determination of bacterial sensitivity to chemotherapeutic and antibiotic agents. Scand. J. Clin. Lab. Investig. 1953, 6, 23–36. [Google Scholar]
- Bauer, A.W.; Kirby, W.M.M.; Sherris, J.C.T.; Turck, M. Antibiotic susceptibility testing by a standardized single disk method. Am. J. Clin. Pathol. 1966, 45, 493–496. [Google Scholar] [PubMed]
- Prasai, T.; Yami, K.D.; Joshi, D.R. Microbial load on paper/polymer currency and coins. Nepal J. Sci. Technol. 2008, 9, 105–109. [Google Scholar] [CrossRef]
- Dalen, M.B.; Ibrahim, A.Q.; Adamu, H.M.; Egbere, O.J. Studies on some quality parameters of the Nigerian polymer and paper bank notes. Br. J. Appl. Sci. Technol. 2014, 4, 2607–2619. [Google Scholar] [CrossRef]
- Jiang, X.; Doyle, M.P. Fate of Escherichia coli O157: H7 and Salmonella enteritidis on currency. J. Food Prot. 1999, 62, 805–807. [Google Scholar] [PubMed]
- Reitler, R.; Marberg, K. The bactericidal effect of tin and its application to the treatment of typhoid fever. Trans. R. Soc. Trop. Med. Hyg. 1943, 36, 305–318. [Google Scholar] [CrossRef]
- Santo, C.E.; Morais, P.V.; Grass, G. Isolation and characterization of bacteria resistant to metallic copper surfaces. Appl. Environ. Microbiol. 2010, 76, 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- Santo, C.E.; Lam, E.W.; Elowsky, C.G.; Quaranta, D.; Domaille, D.W.; Chang, C.J.; Grass, G. Bacterial killing by dry metallic copper surfaces. Appl. Eviron. Microbiol. 2011, 77, 794–802. [Google Scholar] [CrossRef] [PubMed]
- Azam, A.; Ahmed, A.S.; Oves, M.; Khan, M.S.; Habib, S.S.; Memic, A. Antimicrobial activity of metal oxide nanoparticles against Gram-positive and Gram-negative bacteria: A comparative study. Int. J. Nanomed. 2012, 7, 6003–6009. [Google Scholar] [CrossRef] [PubMed]
- Flynn, S.L.; Szymanowski, J.E.; Fein, J.B. Modeling bacterial metal toxicity using a surface complexation approach. Chem. Geol. 2014, 374, 110–116. [Google Scholar] [CrossRef]
- Ericsson, B.H.; Tunevall, G.; Wickman, K. The paper disc method for determination of bacterial sensitivity to antibiotics: Relationship between the diameter of the zone of inhibition and the minimum inhibitory concentration. Scand. J. Clin. Lab. Investig. 1960, 12, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Babich, H.; Stotzky, G. Nickel toxicity to microbes: Effect of pH and implications for acid rain. Environ. Res. 1982, 29, 335–350. [Google Scholar] [CrossRef]
- Hamann, C.R.; Hamann, D.; Hamann, C.; Thyssen, J.P.; Lidén, C. The cost of nickel allergy: A global investigation of coin composition and nickel and cobalt release. Contact Dermat. 2013, 68, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Oldfield, J.W. Electrochemical theory of galvanic corrosion. In Galvanic Corrosion; Hack, P.H., Ed.; American Society for Testing and Materials: Philadelphia, PA, USA, 1988; pp. 5–22. [Google Scholar]
- Havas, F. About the bacteriological state of notes and coins. Magy Allatorvosok Lapja 2000, 122, 501–503. [Google Scholar]
- Tolba, O.; Loughrey, A.; Goldsmith, C.E.; Millar, B.C.; Rooney, P.J.; Moore, J.E. Survival of epidemic strains of nosocomial-and community-acquired methicillin-resistant Staphylococcus aureus on coins. Am. J. Infect. Control 2007, 35, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Kalita, M.; Palusinska-Szysz, M.; Turska-Szewczuk, A.; Wdowiak-Wrobel, S.; Urbanik-Sypniewska, T. Isolation of cultivable microorganisms from Polish notes and coins. Pol. J. Microbiol. 2013, 62, 281–286. [Google Scholar] [PubMed]
- Amini, R.; Abdulamir, A.S.; Jahanshiri, F.; Shan, L.C.; Amini, Y.; Sekawi, Z.; Jalilian, F.A. Isolation and identification of methicillin-resistant Staphylococcus aureus from students’ coins. Afr. J. Biotechnol. 2012, 11, 11143–11149. [Google Scholar]
- Angelakis, E.; Azhar, E.I.; Bibi, F.; Yasir, M.; Al-Ghamdi, A.K.; Ashshi, A.M.; Elshemi, A.G.; Raoult, D. Paper money and coins as potential vectors of transmissible disease. Future Microbiol. 2014, 9, 249–261. [Google Scholar] [CrossRef] [PubMed]

| Notes & Composition | Cotton Gossypium spp | Linen Linum usitatissimum | Mitsumata Edgeworthia papyrifera | Abaca Musa textilis |
|---|---|---|---|---|
| Chinese and UK notes | 100% | |||
| USA notes | ~75% | ~25% | ||
| Japanese notes | ~67% | ~33% | ||
| Fibre length (mm) | 10–40 | 9–70 | 2–4 | 2–12 |
| Fibre width (μm) | 12–38 | 5–40 | 4–20 | 15–40 |
| Cellulose | 74–98 | 64.1 | 50 | 60 |
| Hemicellulose | 1–11 | 17 | 20 | 21 |
| Lignin | 1–16 | 2 | 4 | 12 |
| Coinage * | Zone of Inhibition (mm) | ||||||
|---|---|---|---|---|---|---|---|
| Currency | → Bacteria → → | S. aureus | S. Typhimurium | E. coli | L. monocytogenes | C. sakazakii | |
| ↓ Make-up ↓ | |||||||
| USA 1c | Copper plated zinc | 37.1 (3.7) | 30.9 (6.9) | 34.3 (7.1) | 37.3 (9.0) | 45.5 (3.8) | |
| UK £1 | 70% copper, 24.5% zinc, 5.5% nickel | 13.8 (1.7) | 3.9 (0.9) | 1.3 (0.8) | 2.8 (1.1) | 8.9 (1.1) | |
| AU $1; NZ $1 | 92% copper, 6% aluminium, 2% nickel | 4.9 (0.4) | 3.8 (1.1) | 2.5 (0.7) | 2.1 (0.7) | 7.6 (1.6) | |
| MEX 10c | 100% stainless steel | 12.2 (2.9) | 2.5 (0.6) | 1.7 (0.6) | 2.0 (0.7) | 0.0 | |
| NZ 50c | Nickel plated steel | 2.6 (0.5) | 4.2 (0.8) | 4.6 (0.9) | 3.9 (1.2) | 5.6 (0.56 | |
| UK 10p; USA 5c; AU 20c | 75% copper, 25% nickel | 8.8 (2.3) | 2.3 (0.6) | 2.3 (1.1) | 1.5 (1.1) | 18.4 (1.4) | |
| UK 20p | 84% copper, 16% nickel | 7.9 (2.1) | 2.2 (1.0) | 2.0 (0.6) | 1.8 (0.9) | 16.4 (1.7) | |
| USA 10c; 25c | 92% copper, 8% nickel | 6.6 (1.4) | 1.8 (0.3) | 2.3 (1.1) | 1.9 (0.4) | 13.1 (1.6) | |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vriesekoop, F.; Chen, J.; Oldaker, J.; Besnard, F.; Smith, R.; Leversha, W.; Smith-Arnold, C.; Worrall, J.; Rufray, E.; Yuan, Q.; et al. Dirty Money: A Matter of Bacterial Survival, Adherence, and Toxicity. Microorganisms 2016, 4, 42. https://doi.org/10.3390/microorganisms4040042
Vriesekoop F, Chen J, Oldaker J, Besnard F, Smith R, Leversha W, Smith-Arnold C, Worrall J, Rufray E, Yuan Q, et al. Dirty Money: A Matter of Bacterial Survival, Adherence, and Toxicity. Microorganisms. 2016; 4(4):42. https://doi.org/10.3390/microorganisms4040042
Chicago/Turabian StyleVriesekoop, Frank, Jing Chen, Jenna Oldaker, Flavien Besnard, Reece Smith, William Leversha, Cheralee Smith-Arnold, Julie Worrall, Emily Rufray, Qipeng Yuan, and et al. 2016. "Dirty Money: A Matter of Bacterial Survival, Adherence, and Toxicity" Microorganisms 4, no. 4: 42. https://doi.org/10.3390/microorganisms4040042
APA StyleVriesekoop, F., Chen, J., Oldaker, J., Besnard, F., Smith, R., Leversha, W., Smith-Arnold, C., Worrall, J., Rufray, E., Yuan, Q., Liang, H., Scannell, A., & Russell, C. (2016). Dirty Money: A Matter of Bacterial Survival, Adherence, and Toxicity. Microorganisms, 4(4), 42. https://doi.org/10.3390/microorganisms4040042







