Molecular Detection and Phylogenetic Analysis of Anaplasma spp. in Ticks Collected from Grasslands near Livestock Farms in Gyeonggi and Gangwon Provinces, Republic of Korea
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Tick Collection and Species Identification
2.3. DNA Extraction and PCR Detection
2.4. Sequencing and Phylogenetic Analyses
2.5. Statistical Analysis
3. Results
3.1. Identification of Ticks
3.2. Infection Rate of Anaplasma spp. in Ticks
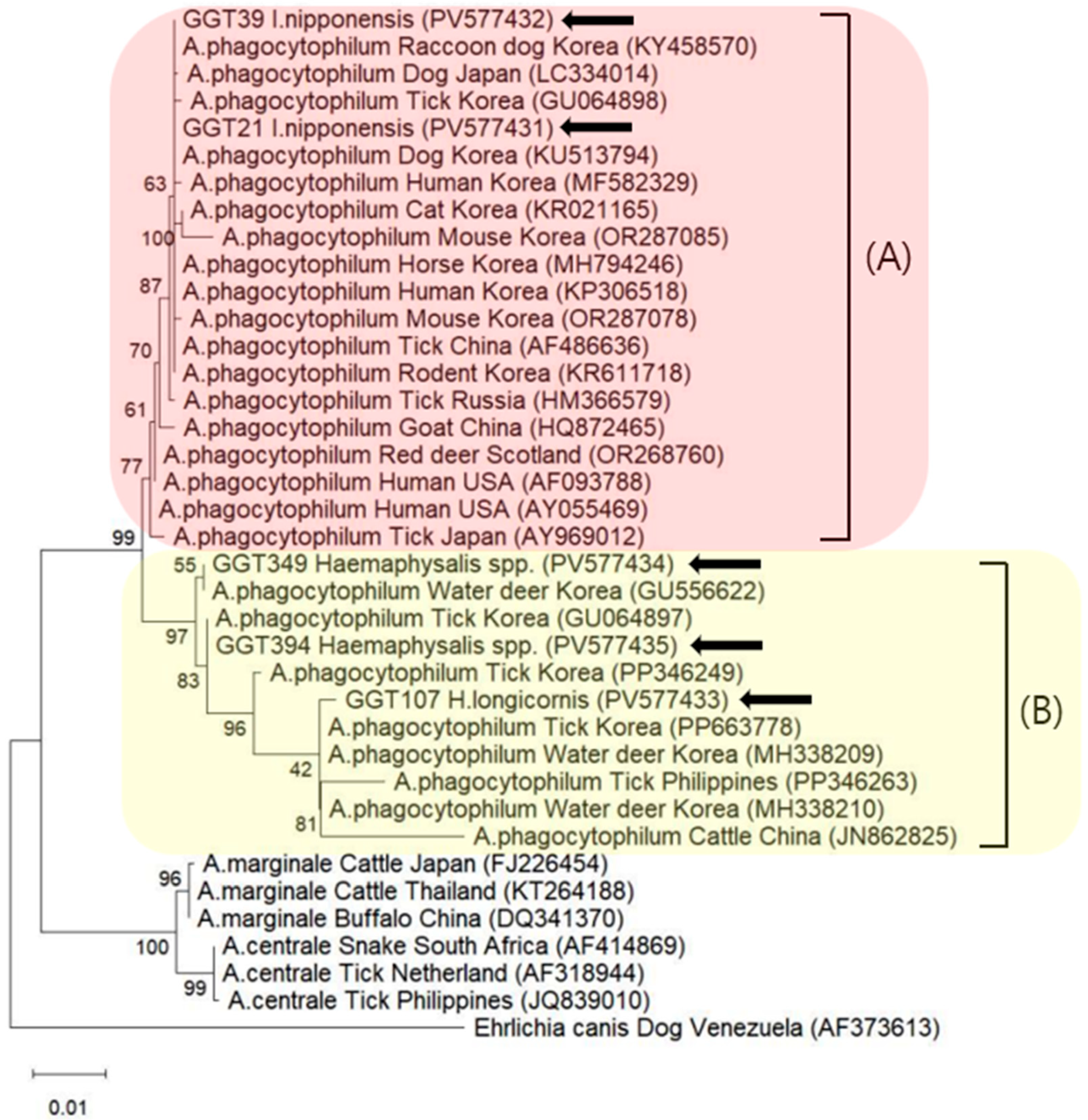
3.3. Molecular and Phylogenetic Analyses
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Madison-Antenucci, S.; Kramer, L.D.; Gebhardt, L.L.; Kauffman, E. Emerging tick-borne diseases. Clin. Microbiol. Rev. 2020, 33, e00083-18. [Google Scholar] [CrossRef]
- Rosenberg, R.; Lindsey, N.P.; Fischer, M.; Gregory, C.J.; Hinckley, A.F.; Mead, P.S.; Paz-Bailey, G.; Waterman, S.H.; Drexler, N.A.; Kersh, G.J.; et al. Vital signs: Trends in reported vector-borne disease cases—United States and territories, 2004–2016. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 496–501. [Google Scholar] [CrossRef]
- Battilani, M.; De Arcangeli, S.; Balboni, A.; Dondi, F. Genetic diversity and molecular epidemiology of Anaplasma. Infect. Genet. Evol. 2017, 49, 195–211. [Google Scholar] [CrossRef] [PubMed]
- Woldehiwet, Z. The natural history of Anaplasma phagocytophilum. Vet. Parasitol. 2010, 167, 108–122. [Google Scholar] [CrossRef] [PubMed]
- Dumler, J.S.; Choi, K.S.; Garcia-Garcia, J.C.; Barat, N.S.; Scorpio, D.G.; Garyu, J.W.; Grab, D.J.; Bakken, J.S. Human granulocytic anaplasmosis and Anaplasma phagocytophilum. Emerg. Infect. Dis. 2005, 11, 1828–1834. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.M.; Dumler, J.S.; Bakken, J.S.; Walker, D.H. Identification of a granulocytotropic Ehrlichia species as the etiologic agent of human disease. J. Clin. Microbiol. 1994, 32, 589–595. [Google Scholar] [CrossRef]
- Petrovec, M.; Lotric Furlan, S.; Zupanc, T.A.; Strle, F.; Brouqui, P.; Roux, V.; Dumler, J.S. Human disease in Europe caused by a granulocytic Ehrlichia species. J. Clin. Microbiol. 1997, 35, 1556–1559. [Google Scholar] [CrossRef]
- Zhang, L.; Cui, F.; Wang, L.; Zhang, L.; Zhang, J.; Yang, S.; Han, J. Anaplasma phagocytophilum and Ehrlichia chaffeensis in Yiyuan County, Shandong. Infect. Dis. Inf. 2009, 1, 21–25. [Google Scholar] [CrossRef]
- Ohashi, N.; Gaowa; Wuritu; Kawamori, F.; Wu, D.; Yoshikawa, Y.; Chiya, S.; Fukunaga, K.; Funato, T.; Shiojiri, M.; et al. Human granulocytic anaplasmosis, Japan. Emerg. Infect. Dis. 2013, 19, 289–292. [Google Scholar] [CrossRef]
- Heo, E.J.; Park, J.H.; Koo, J.R.; Park, M.S.; Park, M.Y.; Dumler, J.S.; Chae, J.S. Serologic and molecular detection of Ehrlichia chaffeensis and Anaplasma phagocytophila (human granulocytic ehrlichiosis agent) in Korean patients. J. Clin. Microbiol. 2002, 40, 3082–3085. [Google Scholar] [CrossRef]
- Lee, S.J.; Kim, H.H.; Kim, J.Y.; Yoo, J.E.; Gill, B.C. Laboratory-based diagnostic test results for human granulocytic anaplasmosis in 2020. Public Health Wkly. Rep. PHWR 2021, 14, 2773–2780. [Google Scholar]
- Dzięgiel, B.; Adaszek, Ł.; Winiarczyk, S. Wild animals as reservoirs of Anaplasma phagocytophilum for humans. Prz. Epidemiol. 2016, 70, 428–435. [Google Scholar]
- Massung, R.F.; Slater, K.; Owens, J.H.; Nicholson, W.L.; Mather, T.N.; Solberg, V.B.; Olson, J.G. Nested PCR assay for detection of granulocytic ehrlichiae. J. Clin. Microbiol. 1998, 36, 1090–1095. [Google Scholar] [CrossRef]
- Massung, R.F.; Levin, M.L.; Munderloh, U.G.; Silverman, D.J.; Lynch, M.J.; Gaywee, J.K.; Kurtti, T.J. Isolation and propagation of the Ap-Variant 1 strain of Anaplasma phagocytophilum in a tick cell line. J. Clin. Microbiol. 2007, 45, 2138–2143. [Google Scholar] [CrossRef]
- Seo, J.Y.; Kim, Y.J.; Kim, S.Y.; Lee, H.I. Molecular detection of Anaplasma, Ehrlichia and Rickettsia pathogens in ticks collected from humans in the Republic of Korea, 2021. Pathogens 2023, 12, 802. [Google Scholar] [CrossRef]
- Kim, Y.-J.; Seo, J.Y.; Kim, S.Y.; Lee, H.I. Molecular Detection of Anaplasma phagocytophilum and Ehrlichia Species in Ticks Removed from Humans in the Republic of Korea. Microorganisms 2022, 10, 1224. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Lee, Y.J.; Kwak, D.; Seo, M.G. Nationwide survey of vector-borne diseases in rodents and mites in Korea: Anaplasma, Ehrlichia, and Rickettsia. Animals 2024, 14, 2950. [Google Scholar] [CrossRef]
- Kim, M.; Cho, S.; Park, G.; Kim, J.; Rieu, M.; Noh, K.T.; Ha, S.; Park, Q.; Kim, D.H.; Han, S.; et al. Molecular detection of Anplasma phagocytophilum, Borrelia theileri, and Severe fever with thrombocytopenia syndrome virus (SFTSV) in military working dogs and ticks collected from the Republic of Korea Army garrisons in Gangwon Province in 2021–2022. Entomol. Res. 2024, 54, e12762. [Google Scholar] [CrossRef]
- Yamaguti, N.; Tipton, V.J.; Keegan, H.L.; Toshioka, S. Ticks of Japan, Korea, and the Ryukyu islands. Brigh. Young Univ. Sci. Bull. Biol. Ser. 1971, 15, 1. [Google Scholar] [CrossRef]
- Barlough, J.E.; Madigan, J.E.; Derock, E.; Bigornia, L. Nested Polymerase Chain Reaction for Detection of Ehrlichia equi Genomic DNA in Horses and Ticks (Ixodes pacificus). Vet. Parasitol. 1996, 63, 319–329. [Google Scholar] [CrossRef]
- Seo, M.-G.; Ouh, I.-O.; Lee, H.; Geraldino, P.J.L.; Rhee, M.H.; Kwon, O.-D.; Kwak, D. Differential identification of Anaplasma in cattle and potential of cattle to serve as reservoirs of Anaplasma capra, an emerging tick-borne zoonotic pathogen. Vet. Microbiol. 2018, 226, 15–22. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef]
- Seo, M.G.; Noh, B.E.; Lee, H.S.; Kim, T.K.; Song, B.G.; Lee, H.I. Nationwide temporal and geographical distribution of tick populations and phylogenetic analysis of severe fever with thrombocytopenia syndrome virus in ticks in Korea, 2020. Microorganisms 2021, 9, 1630. [Google Scholar] [CrossRef]
- St. John, H.K.; Masuoka, P.; Jiang, J.; Takhampunya, R.; Klein, T.A.; Kim, H.C.; Chong, S.T.; Song, J.W.; Kim, Y.J.; Farris, C.M.; et al. Geographic distribution and modeling of ticks in the Republic of Korea and the application of tick models towards understanding the distribution of associated pathogenic agents. Ticks Tick Borne Dis. 2021, 12, 101686. [Google Scholar] [CrossRef]
- Alkathiri, B.; Ahn, K.; Lee, H.; Cho, Y.S.; Youn, S.Y.; Seo, M.G.; Kwak, D.; Shin, S.; Lee, S.H. Molecular epidemiology of Theileria species in ticks and its potential threat to livestock in the Republic of Korea. Acta Trop. 2023, 238, 106780. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.G.; Lee, H.; Alkathiri, B.; Ahn, K.; Lee, S.H.; Shin, S.; Bae, S.; Kim, K.T.; Jang, M.; Lee, S.K.; et al. Tick populations and molecular analysis of Anaplasma species in ticks from the Republic of Korea. Microorganisms 2023, 11, 820. [Google Scholar] [CrossRef]
- Liveris, D.; Aguero-Rosenfeld, M.E.; Daniels, T.J.; Karpathy, S.; Paddock, C.; Adish, S.; Keesing, F.; Ostfeld, R.S.; Wormser, G.P.; Schwartz, I. A new genetic approach to distinguish strains of Anaplasma phagocytophilum that appear not to cause human disease. Ticks Tick Borne Dis. 2021, 12, 101659. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Shin, N.R.; Kim, C.M.; Park, S.; Yun, N.R.; Kim, D.M.; Jung, D.S. First identification of Anaplasma phagocytophilum in both a biting tick Ixodes nipponensis and a patient in Korea: A case report. BMC Infect. Dis. 2020, 20, 826. [Google Scholar] [CrossRef]
- Oh, J.Y.; Moon, B.C.; Bae, B.K.; Shin, E.H.; Ko, Y.H.; Kim, Y.J.; Park, J.H.; Park, B.K.; Yoo, J.Y.; Chae, J.S. Genetic identification and phylogenetic analysis of Anaplasma and Ehrlichia species in Haemaphysalis longicornis collected from Jeju Island, Korea. J. Bacteriol. Virol. 2009, 39, 257–267. [Google Scholar] [CrossRef]
- Howell, J.M.; Ueti, M.W.; Palmer, G.H.; Scoles, G.A.; Knowles, D.P. Transovarial transmission efficiency of Babesia bovis tick stages acquired by Rhipicephalus (Boophilus) microplus during acute infection. J. Clin. Microbiol. 2007, 45, 3155–3159. [Google Scholar] [CrossRef]
- Moore, T.C.; Pulscher, L.A.; Caddell, L.; von Fricken, M.E.; Anderson, B.D.; Gonchigoo, B.; Gray, G.C. Evidence for transovarial transmission of tick-borne rickettsiae circulating in Northern Mongolia. PLoS Negl. Trop. Dis. 2018, 12, e0006696. [Google Scholar] [CrossRef] [PubMed]
- Ahn, K.; Alkathiri, B.; Lee, S.H.; Lee, H.; Kwak, D.; Cho, Y.S.; Lee, H.S.; Youn, S.; Yoo, M.S.; Kim, J.; et al. Molecular detection of Anaplasma phagocytophilum in field-collected Haemaphysalis larvae in the Republic of Korea. Parasites Vectors 2025, 18, 67. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, H.; Xu, B.; Zhang, Z.; Jin, Y.; Li, W.; Lu, Q.; Li, L.; Chang, L.; Zhang, X.; et al. Rural residents in China are at increased risk of exposure to tick-borne pathogens Anaplasma phagocytophilum and Ehrlichia chaffeensis. Biomed. Res. Int. 2014, 2014, 313867. [Google Scholar] [CrossRef]

| Tick Species | Stage | No. of Collected Ticks by Region (%) | p-Value 2 | ||
|---|---|---|---|---|---|
| Gyeonggi | Gangwon | Total | |||
| Haemaphysalis spp. 1 | Larva | 753 | 1013 | 1766 (40.91) | - |
| Haemaphysalis longicornis | Nymph | 1007 | 1415 | 2422 | <0.001 |
| Adult male | 7 | 7 | 14 | ||
| Adult female | 28 | 24 | 52 | ||
| Subtotal | 1042 | 1446 | 2488 (57.65) | ||
| Haemaphysalisflava | Nymph | 9 | 39 | 48 | <0.001 |
| Adult male | 1 | 0 | 1 | ||
| Adult female | 2 | 2 | 4 | ||
| Subtotal | 12 | 41 | 53 (1.23) | ||
| Ixodes nipponensis | Adult male | 5 | 1 | 6 | 0.011 |
| Adult female | 2 | 0 | 2 | ||
| Subtotal | 7 | 1 | 8 (0.19) | ||
| Amblyomma testudinarium | Nymph | 0 | 1 | 1 (0.02) | - |
| Total | 1814 | 2502 | 4316 | ||
| Month | Locality | No. of Tested Ticks | No. of Tick Pools | Tick Species | Stage | No. of Positive Pools (MIR 1) | Total No. of Positive Pool (MIR 2) |
|---|---|---|---|---|---|---|---|
| 4 | Gyeonggi | 365 | 46 | I. nipponensis | Male | 1 (0.27) | 3 (0.32) |
| Gangwon | 579 | 68 | I. nipponensis | Male | 1 (0.17) | ||
| H. longicornis | Nymph | 1 (0.17) | |||||
| 5 | Gyeonggi | 282 | 33 | - | - | - | - |
| Gangwon | 373 | 42 | - | - | - | ||
| 6 | Gyeonggi | 220 | 28 | - | - | - | - |
| Gangwon | 294 | 35 | - | - | - | ||
| 7 | Gyeonggi | 165 | 33 | - | - | - | - |
| Gangwon | 240 | 43 | - | - | - | ||
| 8 | Gyeonggi | 205 | 15 | 1 (0.21) | |||
| Gangwon | 263 | 18 | Haemaphysalis spp. | Larva | 1 (0.38) | ||
| 9 | Gyeonggi | 252 | 8 | - | - | - | - |
| Gangwon | 394 | 12 | - | - | - | ||
| 10 | Gyeonggi | 325 | 8 | - | - | - | 1 (0.15) |
| Gangwon | 359 | 11 | Haemaphysalis spp. | Larva | 1 (0.28) | ||
| Total | Gyeonggi | 1814 | 171 | - | - | 1 (0.06) | 5 (0.12) |
| Gangwon | 2502 | 229 | - | - | 4 (0.16) |
| Tick Species | Stage | No. of Tested Ticks (Pool) | No. of Positive Tick Pools by Region (%) | MIR 1 | p-Value 2 | ||
|---|---|---|---|---|---|---|---|
| Gyeonggi | Gangwon | Total | |||||
| Haemaphysalis spp. | Larva | 1766 (53) | 0/17 | 2/36 (5.56) | 2/53 (3.77) | 0.11 | <0.001 |
| H. longicornis | Nymph | 2422 (249) | 0/105 | 1/144 (0.69) | 1/249 (0.40) | 0.04 | |
| Adult male | 14 (14) | 0/7 | 0/7 | 0/14 | 0 | ||
| Adult female | 52 (52) | 0/28 | 0/24 | 0/52 | 0 | ||
| sub total | 2488 (315) | 0/140 | 1/175 (0.57) | 1/315 (0.32) | 0.04 | ||
| H. flava | Nymph | 48 (18) | 0/4 | 0/14 | 0/18 | 0 | |
| Adult male | 1 (1) | 0/1 | 0 | 0/1 | 0 | ||
| Adult female | 4 (4) | 0/2 | 0/2 | 0/4 | 0 | ||
| sub total | 53 (23) | 0/7 | 0/16 | 0/23 | 0 | ||
| I. nipponensis | Adult male | 6 (6) | 1/5 (20.00) | 1/1 (100) | 2/6 (33.33) | 33.33 | |
| Adult female | 2 (2) | 0/2 | 0 | 0/2 | 0 | ||
| sub total | 8 (8) | 1/7 (14.29) | 1/1 (100) | 2/8 (25.00) | 25.00 | ||
| A. testudinarium | Nymph | 1 (1) | 0 | 0/1 | 0/1 | 0 | |
| Total | 4316 (400) | 1/171 (0.58) | 4/229 (1.75) | 5/400 (1.25) | 0.12 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, H.; Jo, M.; Choi, Y.; Lee, H.; Youn, S.; Yoo, M.-S.; Lee, H.-S.; Kim, J.-M.; Nazim, K.; Yun, E.H.; et al. Molecular Detection and Phylogenetic Analysis of Anaplasma spp. in Ticks Collected from Grasslands near Livestock Farms in Gyeonggi and Gangwon Provinces, Republic of Korea. Microorganisms 2025, 13, 2008. https://doi.org/10.3390/microorganisms13092008
Kim H, Jo M, Choi Y, Lee H, Youn S, Yoo M-S, Lee H-S, Kim J-M, Nazim K, Yun EH, et al. Molecular Detection and Phylogenetic Analysis of Anaplasma spp. in Ticks Collected from Grasslands near Livestock Farms in Gyeonggi and Gangwon Provinces, Republic of Korea. Microorganisms. 2025; 13(9):2008. https://doi.org/10.3390/microorganisms13092008
Chicago/Turabian StyleKim, Hyeyeon, Myungji Jo, Younhyoung Choi, Haeseung Lee, SoYoun Youn, Mi-Sun Yoo, Hyang-Sim Lee, Jae-Myung Kim, Kaifa Nazim, Eun Hee Yun, and et al. 2025. "Molecular Detection and Phylogenetic Analysis of Anaplasma spp. in Ticks Collected from Grasslands near Livestock Farms in Gyeonggi and Gangwon Provinces, Republic of Korea" Microorganisms 13, no. 9: 2008. https://doi.org/10.3390/microorganisms13092008
APA StyleKim, H., Jo, M., Choi, Y., Lee, H., Youn, S., Yoo, M.-S., Lee, H.-S., Kim, J.-M., Nazim, K., Yun, E. H., Seo, M.-G., Park, S.-J., Rhee, M. H., Lee, S.-H., Shin, S., & Kwak, D. (2025). Molecular Detection and Phylogenetic Analysis of Anaplasma spp. in Ticks Collected from Grasslands near Livestock Farms in Gyeonggi and Gangwon Provinces, Republic of Korea. Microorganisms, 13(9), 2008. https://doi.org/10.3390/microorganisms13092008







