Retrospective Single-Center Study on the Epidemiological Characteristics of Influenza B Infections in Korea (2007–2024): Analysis of Sex, Age, and Seasonal Patterns
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Data Collection
2.2. Testing Procedure
2.3. Data Preprocessing
2.4. Data Analysis
3. Results
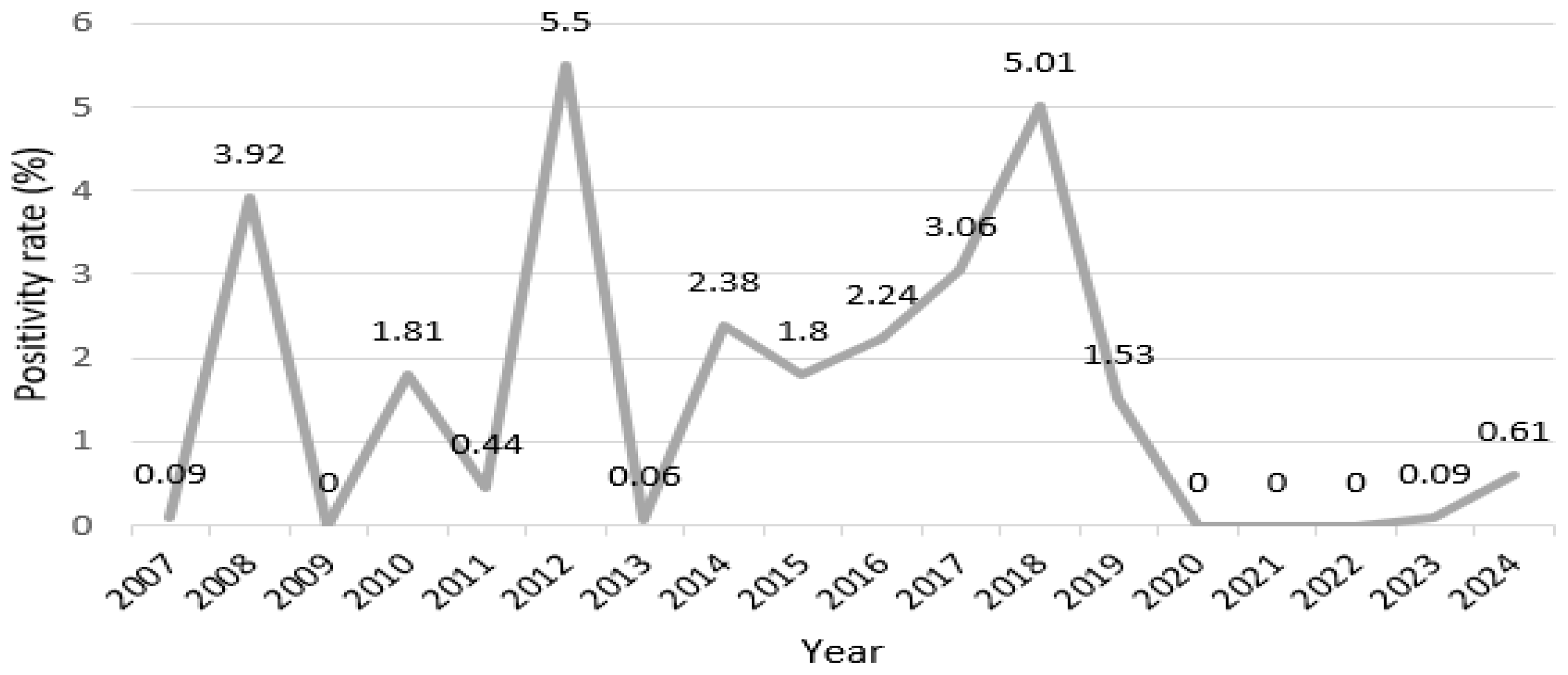
3.1. Annual Incidence Trends
3.2. Seasonal Patterns
3.3. Sex Analysis
3.4. Age Group Differences
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| RT-PCR | Real-time polymerase chain reaction |
| CT | Cycle threshold |
References
- Baumeister, E.; Duque, J.; Varela, T.; Palekar, R.; Couto, P.; Savy, V.; Giovacchini, C.; Haynes, A.K.; Rha, B.; Arriola, C.S.; et al. Timing of respiratory syncytial virus and influenza epidemic activity in five regions of Argentina, 2007-2016. Influenza Other Respir. Viruses 2019, 13, 10–17. [Google Scholar] [CrossRef]
- Kessler, S.; Harder, T.C.; Schwemmle, M.; Ciminski, K. Influenza A viruses and zoonotic events—Are we creating our reservoirs? Viruses 2021, 13, 2250. [Google Scholar] [CrossRef]
- Wolff, T.; Veit, M. Influenza B, C, and D Viruses (Orthomyxoviridae). In Encyclopedia of Virology; Elsevier: Amsterdam, The Netherlands, 2021; pp. 561–574. [Google Scholar] [CrossRef]
- Uyeki, T.M. High-risk groups for influenza complications. JAMA 2020, 324, 2334. [Google Scholar] [CrossRef]
- Zheng, Y.; Wang, K.; Zhang, L.; Wang, L. Study on the relationship between the incidence of influenza and climate indicators and the prediction of influenza incidence. Environ. Sci. Pollut. Res. Int. 2021, 28, 473–481. [Google Scholar] [CrossRef]
- Li, S.; Xue, Z.; Feng, Y.; Zhou, X.; Qi, Y.; Feng, N.; Li, Y. Epidemiological characteristics of eleven common respiratory viral infections in children. BMC Pediatr. 2024, 24, 827. [Google Scholar] [CrossRef]
- Ali, S.T.; Cowling, B.J. Influenza virus: Tracking, predicting, and forecasting. Ann. Rev. Public Health 2021, 42, 43–57. [Google Scholar] [CrossRef]
- Domnich, A.; Bruzzone, B.; Trombetta, C.S.; De Pace, V.D.; Ricucci, V.; Varesano, S.; Garzillo, G.; Ogliastro, M.; Orsi, A.; Icardi, G. Rapid differential diagnosis of SARS-CoV-2, influenza A/B and respiratory syncytial viruses: Validation of a novel RT-PCR assay. J. Clin. Virol. 2023, 161, 105402. [Google Scholar] [CrossRef]
- Mancini, F.; Barbanti, F.; Scaturro, M.; Fontana, S.; Di Martino, A.D.; Marsili, G.; Puzelli, S.; Calzoletti, L.; Facchini, M.; Di Mario, G.; et al. Multiplex real-time reverse-transcription polymerase chain reaction assays for diagnostic testing of severe acute respiratory syndrome coronavirus 2 and seasonal influenza viruses: A challenge of the Phase 3 pandemic setting. J. Infect. Dis. 2021, 223, 765–774. [Google Scholar] [CrossRef]
- Zhang, Y.; Ye, C.; Yu, J.; Zhu, W.; Wang, Y.; Li, Z.; Xu, Z.; Cheng, J.; Wang, N.; Hao, L.; et al. The complex associations of climate variability with seasonal influenza A and B virus transmission in subtropical Shanghai, China. Sci. Total Environ. 2020, 701, 134607. [Google Scholar] [CrossRef]
- Jinjarak, Y.; Ahmed, R.; Nair-Desai, S.; Xin, W.; Aizenman, J. Accounting for global COVID-19 diffusion patterns, January–April 2020. Econ. Disaster. Clim. Change 2020, 4, 515–559. [Google Scholar] [CrossRef]
- Lu, Y.; Wang, Y.; Shen, C.; Luo, J.; Yu, W. Decreased incidence of influenza during the COVID-19 pandemic. Int. J. Gen. Med. 2022, 15, 2957–2962. [Google Scholar] [CrossRef]
- Doroshenko, A.; Lee, N.; MacDonald, C.; Zelyas, N.; Asadi, L.; Kanji, J.N. Decline of influenza and respiratory viruses with COVID-19 public health measures: Alberta, Canada. Mayo Clin. Proc. 2021, 96, 3042–3052. [Google Scholar] [CrossRef]
- Zipfel, C.M.; Colizza, V.; Bansal, S. The missing season: The impacts of the COVID-19 pandemic on influenza. Vaccine 2021, 39, 3645–3648. [Google Scholar] [CrossRef]
- Yu, X.; Xu, C.; Huang, W.; Xu, X.; Xie, W.; Long, X. The incidence of influenza in children was decreased in the first flu season after COVID-19 pandemic in Wuhan. J. Infect. Public Health 2021, 14, 1279–1281. [Google Scholar] [CrossRef]
- Malosh, R.E.; McGovern, I.; Monto, A.S. Influenza during the 2010–2020 decade in the United States: Seasonal outbreaks and vaccine interventions. Clin. Infect. Dis. 2023, 76, 540–549. [Google Scholar] [CrossRef]
- Matsuki, E.; Kawamoto, S.; Morikawa, Y.; Yahagi, N. The impact of cold ambient temperature in the pattern of itnfluenza virus infection. Open Forum Infect. Dis. 2023, 10, ofad039. [Google Scholar] [CrossRef]
- Bulfone, T.C.; Malekinejad, M.; Rutherford, G.W.; Razani, N. Outdoor transmission of SARS-CoV-2 and other respiratory viruses: A systematic review. J. Infect. Dis. 2021, 223, 550–561. [Google Scholar] [CrossRef]
- Sun, H.C.; Pei, S.; Wang, L.; Sun, Y.Y.; Xu, X.K. The impact of spring festival travel on epidemic spreading in china. Viruses 2023, 15, 1527. [Google Scholar] [CrossRef]
- Ravelli, E.; Martinez, R.G. Environmental risk factors of airborne viral transmission: Humidity, Influenza and SARS-CoV-2 in the Netherlands. Spat. Spatiotemporal Epidemiol. 2022, 41, 100432. [Google Scholar] [CrossRef]
- Wang, J.; Tang, K.; Feng, K.; Lin, X.; Lv, W.; Chen, K.; Wang, F. Impact of temperature and relative humidity on the transmission of COVID-19: A modelling study in China and the United States. BMJ Open 2021, 11, e043863. [Google Scholar] [CrossRef]
- Slusky, D.J.G.; Zeckhauser, R.J. Sunlight and protection against influenza. Econ. Hum. Biol. 2021, 40, 100942. [Google Scholar] [CrossRef]
- Prada, G.I.; Băjenaru, O.L.; Chelu, G.C.; Matei-Lincă, C.M.; Nuţă, C.R.; Moscu, S.G. Protecting the elderly from influenza in the context of immune system senescence. Elderly aged 65 and over are vulnerable to influenza and its associated complications: Position paper by the Romanian Society of Gerontology and Geriatrics. J. Med. Life 2024, 17, 746–754. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kloc, M.; Ghobrial, R.M.; Kuchar, E.; Lewicki, S.; Kubiak, J.Z. Development of child immunity in the context of COVID-19 pandemic. Clin. Immunol. 2020, 217, 108510. [Google Scholar] [CrossRef]
- Dong, Y.; Mo, X.; Hu, Y.; Qi, X.; Jiang, F.; Jiang, Z.; Tong, S. Epidemiology of COVID-19 among children in china. Pediatrics 2020, 145, e20200702. [Google Scholar] [CrossRef]
- Zhou, D.; Borsa, M.; Simon, A.K. Hallmarks and detection techniques of cellular senescence and cellular ageing in immune cells. Aging Cell 2021, 20, e13316. [Google Scholar] [CrossRef]
- Jones, E.; Sheng, J.; Carlson, J.; Wang, S. Aging-induced fragility of the immune system. J. Theor. Biol. 2021, 510, 110473. [Google Scholar] [CrossRef]
- Gustafson, C.E.; Kim, C.; Weyand, C.M.; Goronzy, J.J. Influence of immune aging on vaccine responses. J. Allergy Clin. Immunol. 2020, 145, 1309–1321. [Google Scholar] [CrossRef]
| Season | Positive | Negative | Positivity Rate (%) |
|---|---|---|---|
| Spring | 243 | 6148 | 3.95 |
| Summer | 5 | 4805 | 0.10 |
| Autumn | 1 | 5606 | 0.01 |
| Winter | 188 | 6288 | 2.98 |
| Sex | Positive | Negative | Positivity Rate (%) |
|---|---|---|---|
| Male | 238 | 13,723 | 1.70 |
| Female | 199 | 9124 | 2.13 |
| Age Group | Male (n = 238) | Female (n = 199) |
|---|---|---|
| Infants (0 years) | 13 (5.5%) | 10 (5.0%) |
| Children (1–19 years) | 144 (60.5%) | 121 (60.8%) |
| Adults (20–64 years) | 39 (16.3%) | 26 (13.1%) |
| Older adults (65 years and above) | 42 (17.6%) | 42 (21.1%) |
| Age Group | Total Individuals | Positive | Negative | Positivity Rate (%) |
|---|---|---|---|---|
| Infant (0 years) | 4556 | 23 | 4533 | 0.50 |
| Child (1–19 years) | 11,137 | 265 | 10,872 | 2.40 |
| Adult (20–64 years) | 2899 | 65 | 2834 | 2.24 |
| Elderly (65 years and above) | 4692 | 84 | 4608 | 1.79 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, J.S.; Chung, Y.N.; Kim, J.K. Retrospective Single-Center Study on the Epidemiological Characteristics of Influenza B Infections in Korea (2007–2024): Analysis of Sex, Age, and Seasonal Patterns. Microorganisms 2025, 13, 1141. https://doi.org/10.3390/microorganisms13051141
Han JS, Chung YN, Kim JK. Retrospective Single-Center Study on the Epidemiological Characteristics of Influenza B Infections in Korea (2007–2024): Analysis of Sex, Age, and Seasonal Patterns. Microorganisms. 2025; 13(5):1141. https://doi.org/10.3390/microorganisms13051141
Chicago/Turabian StyleHan, Jeong Su, Yoo Na Chung, and Jae Kyung Kim. 2025. "Retrospective Single-Center Study on the Epidemiological Characteristics of Influenza B Infections in Korea (2007–2024): Analysis of Sex, Age, and Seasonal Patterns" Microorganisms 13, no. 5: 1141. https://doi.org/10.3390/microorganisms13051141
APA StyleHan, J. S., Chung, Y. N., & Kim, J. K. (2025). Retrospective Single-Center Study on the Epidemiological Characteristics of Influenza B Infections in Korea (2007–2024): Analysis of Sex, Age, and Seasonal Patterns. Microorganisms, 13(5), 1141. https://doi.org/10.3390/microorganisms13051141






