Experience in Implementing Colonization Screening in a Multidisciplinary County Clinical Hospital in Romania
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Population
2.3. Laboratory Studies
2.4. Statistical Analyses
3. Results
3.1. Screening Results
3.2. Other Key Findings
3.3. Detailed Interpretation of the Screening Results by Department
4. Discussion
The Role of Host and Environmental Flora in Hospital-Acquired Infections
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Trimester of the Year | Total | |||
|---|---|---|---|---|
| 1 | 2 | |||
| Department | Allergology Department | 8 | 3 | 11 |
| 1st Anesthesia and Intensive Care Unit Clinic | 205 | 215 | 420 | |
| Anesthesia and Intensive Care Unit Dedicated to Neurosurgical, Plastic Surgery, and Other Surgical Departments | 39 | 24 | 63 | |
| Infectious Diseases Clinic | 65 | 91 | 156 | |
| 1st Cardiology Clinic | 3 | 7 | 10 | |
| 2nd Cardiology Clinic | 6 | 4 | 10 | |
| 1st Surgical Clinic—Thoracic Surgery Department | 3 | 3 | 6 | |
| 2nd Surgical Clinic—Vascular Surgery Department | 2 | 4 | 6 | |
| 1st Surgical Clinic | 1 | 1 | 2 | |
| 2nd Surgical Clinic | 4 | 4 | 8 | |
| Diabetes Clinic | 1 | 0 | 1 | |
| Gastroenterology Clinic | 2 | 5 | 7 | |
| Haematology Clinic | 6 | 19 | 25 | |
| 1st Internal Medicine Clinic | 150 | 156 | 306 | |
| 2nd Internal Medicine Clinic | 85 | 136 | 221 | |
| Neurosurgical Clinic | 5 | 3 | 8 | |
| Neurology Clinic | 0 | 23 | 23 | |
| Obstetrics and Gynecology Clinic | 0 | 1 | 1 | |
| Oncology Clinic | 3 | 2 | 5 | |
| Orthopedics and Traumatology Clinic | 42 | 54 | 96 | |
| 1st Medical Rehabilitation Clinic | 1 | 5 | 6 | |
| Medical Rehabilitation Clinic—Neurological Department | 5 | 24 | 29 | |
| Medical Rehabilitation Clinic—Orthopedics and Traumatology Department | 3 | 13 | 16 | |
| Rheumatology Clinic | 14 | 5 | 19 | |
| Cardiology Intensive Care Department | 9 | 45 | 54 | |
| Urology Clinic | 4 | 4 | 8 | |
| 2nd Anesthesia and Intensive Care Unit Clinic | 0 | 2 | 2 | |
| Nephrology and Dialysis Clinic | 0 | 3 | 3 | |
| Total | 666 | 856 | 1522 | |
| Department | Type of Swab | Total | |||||
|---|---|---|---|---|---|---|---|
| Nasal Swab | Axillary Swabs | Inguinal Swabs | Rectal/Perianal Swabs | ||||
| Nephrology and Dialysis Clinic | Trimester of the year | 2 | 3 | 2 | 1 | 24 | 30 |
| Total | 3 | 2 | 1 | 24 | 30 | ||
| 2nd Anesthesia and Intensive Care Unit Clinic | Trimester of the year | 2 | 2 | 2 | 16 | 20 | |
| Total | 2 | 2 | 16 | 20 | |||
| Urology Clinic | Trimester of the year | 1 | 4 | 1 | 3 | 28 | 36 |
| 2 | 5 | 4 | 1 | 40 | 50 | ||
| Total | 9 | 5 | 4 | 68 | 86 | ||
| Cardiology Intensive Care Department | Trimester of the year | 1 | 10 | 6 | 4 | 71 | 91 |
| 2 | 49 | 6 | 43 | 392 | 490 | ||
| Total | 59 | 12 | 47 | 463 | 581 | ||
| Rheumatology Clinic | Trimester of the year | 1 | 16 | 16 | 123 | 155 | |
| 2 | 5 | 5 | 40 | 50 | |||
| Total | 21 | 21 | 163 | 205 | |||
| Medical Rehabilitation Clinic—Orthopedics and Traumatology Department | Trimester of the year | 1 | 3 | 1 | 2 | 22 | 28 |
| 2 | 24 | 8 | 16 | 192 | 240 | ||
| Total | 27 | 9 | 18 | 214 | 268 | ||
| Medical Rehabilitation Clinic—Neurological Department | Trimester of the year | 1 | 5 | 0 | 5 | 36 | 46 |
| 2 | 40 | 22 | 19 | 328 | 409 | ||
| Total | 45 | 22 | 24 | 364 | 455 | ||
| 1st Medical Rehabilitation Clinic | Trimester of the year | 1 | 1 | 0 | 1 | 7 | 9 |
| 2 | 6 | 4 | 3 | 48 | 61 | ||
| Total | 7 | 4 | 4 | 55 | 70 | ||
| Orthopedics and Traumatology Clinic | Trimester of the year | 1 | 44 | 5 | 39 | 242 | 330 |
| 2 | 55 | 7 | 50 | 288 | 400 | ||
| Total | 99 | 12 | 89 | 530 | 730 | ||
| Oncology Clinic | Trimester of the year | 1 | 3 | 2 | 1 | 22 | 28 |
| 2 | 2 | 1 | 1 | 16 | 20 | ||
| Total | 5 | 3 | 2 | 38 | 48 | ||
| Obstetrics and Gynecology Clinic | Trimester of the year | 2 | 1 | 1 | 8 | 10 | |
| Total | 1 | 1 | 8 | 10 | |||
| Neurology Clinic | Trimester of the year | 2 | 26 | 16 | 10 | 208 | 260 |
| Total | 26 | 16 | 10 | 208 | 260 | ||
| Neurosurgical Clinic | Trimester of the year | 1 | 5 | 3 | 2 | 35 | 45 |
| 2 | 3 | 1 | 2 | 24 | 30 | ||
| Total | 8 | 4 | 4 | 59 | 75 | ||
| 2nd Internal Medicine Clinic | Trimester of the year | 1 | 101 | 1 | 100 | 728 | 930 |
| 2 | 159 | 0 | 159 | 1272 | 1590 | ||
| Total | 260 | 1 | 259 | 2000 | 2520 | ||
| 1st Internal Medicine Clinic | Trimester of the year | 1 | 155 | 149 | 6 | 1111 | 1674 |
| 2 | 167 | 165 | 2 | 1340 | 1421 | ||
| Total | 322 | 314 | 8 | 2451 | 3095 | ||
| Haematology Clinic | Trimester of the year | 1 | 7 | 7 | 51 | 65 | |
| 2 | 21 | 21 | 168 | 210 | |||
| Total | 28 | 28 | 219 | 275 | |||
| Gastroenterology Clinic | Trimester of the year | 1 | 2 | 2 | 14 | 18 | |
| 2 | 6 | 6 | 48 | 60 | |||
| Total | 8 | 8 | 62 | 78 | |||
| Diabetes Clinic | Trimester of the year | 1 | 1 | 1 | 7 | 9 | |
| Total | 1 | 1 | 7 | 9 | |||
| 2nd Surgical Clinic | Trimester of the year | 1 | 4 | 4 | 28 | 36 | |
| 2 | 5 | 5 | 40 | 50 | |||
| Total | 9 | 9 | 68 | 86 | |||
| 1st Surgical Clinic | Trimester of the year | 1 | 1 | 1 | 0 | 7 | 9 |
| 2 | 1 | 0 | 1 | 8 | 10 | ||
| Total | 2 | 1 | 1 | 15 | 19 | ||
| 2nd Surgical Clinic—Vascular Surgery Department | Trimester of the year | 1 | 3 | 3 | 21 | 27 | |
| 2 | 4 | 4 | 32 | 40 | |||
| Total | 7 | 7 | 53 | 67 | |||
| 1st Surgical Clinic—Thoracic Surgery Department | Trimester of the year | 1 | 3 | 3 | 23 | 29 | |
| 2 | 4 | 4 | 32 | 40 | |||
| Total | 7 | 7 | 55 | 69 | |||
| 2nd Cardiology Clinic | Trimester of the year | 1 | 6 | 3 | 3 | 42 | 54 |
| 2 | 4 | 3 | 1 | 32 | 40 | ||
| Total | 10 | 6 | 4 | 74 | 94 | ||
| 1st Cardiology Clinic | Trimester of the year | 1 | 3 | 2 | 1 | 21 | 27 |
| 2 | 7 | 5 | 2 | 56 | 70 | ||
| Total | 10 | 7 | 3 | 77 | 97 | ||
| Infectious Diseases Clinic | Trimester of the year | 1 | 81 | 5 | 76 | 577 | 739 |
| 2 | 99 | 1 | 98 | 792 | 990 | ||
| Total | 180 | 6 | 174 | 1369 | 1729 | ||
| Anesthesia and Intensive Care Unit Dedicated to Neurosurgical, Plastic Surgery, and Other Surgical Departments | Trimester of the year | 1 | 96 | 7 | 69 | 545 | 697 |
| 2 | 95 | 12 | 83 | 766 | 956 | ||
| Total | 171 | 19 | 152 | 1311 | 1653 | ||
| 1st Anesthesia and Intensive Care Unit Clinic | Trimester of the year | 2 | 313 | 308 | 5 | 2234 | 2860 |
| 1 | 340 | 337 | 4 | 2720 | 3401 | ||
| Total | 653 | 645 | 9 | 4954 | 6261 | ||
| Allergology Department | Trimester of the year | 1 | 8 | 7 | 1 | 57 | 73 |
| 2 | 3 | 2 | 1 | 24 | 30 | ||
| Total | 11 | 9 | 2 | 81 | 103 | ||
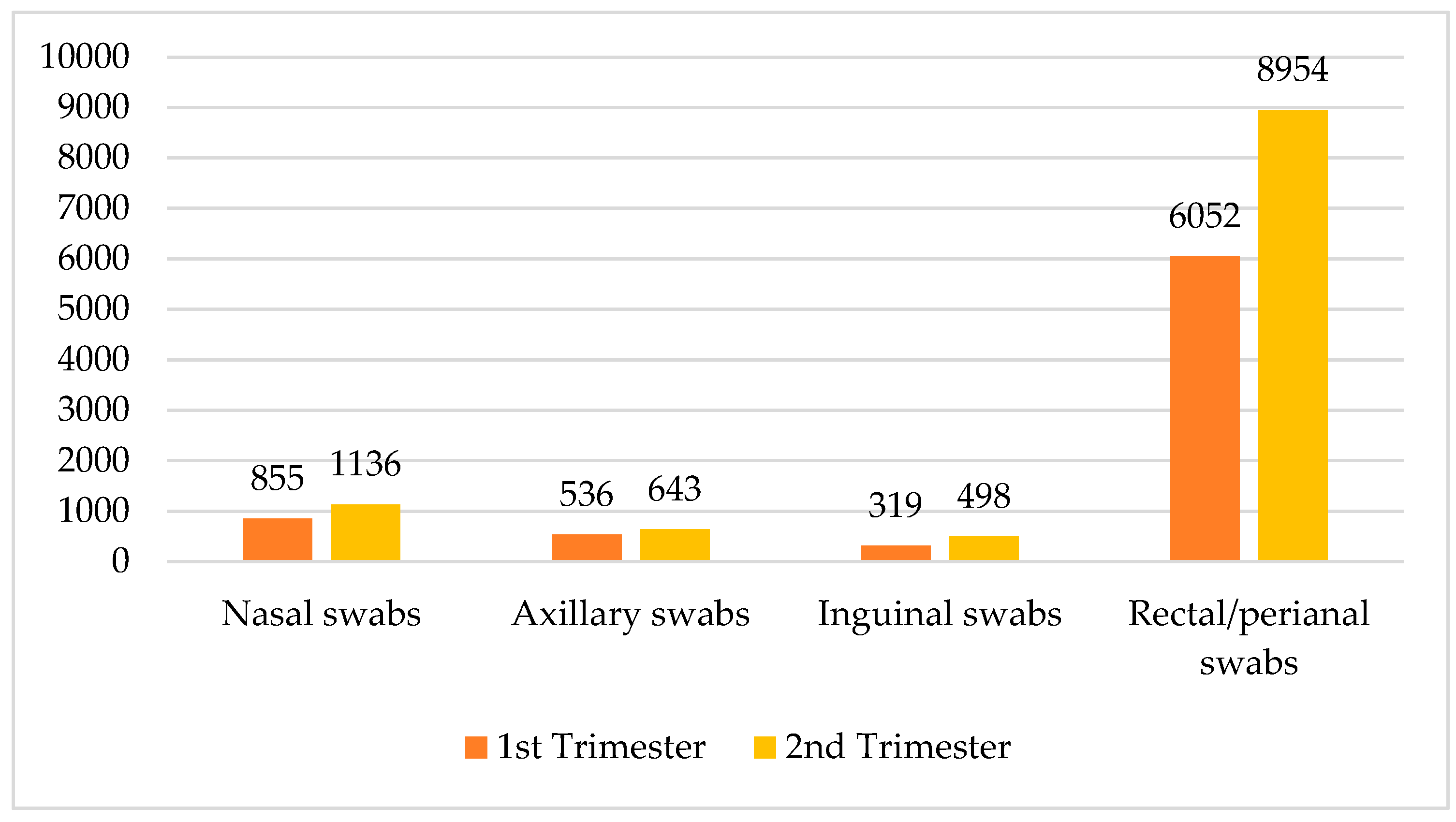
| Total | Trimester of the year | 1 | 855 | 536 | 319 | 6052 | 7762 |
| 2 | 1136 | 643 | 498 | 8954 | 11,231 | ||
| Total | 1991 | 1179 | 817 | 15,006 | 18,993 | ||

| Type of Swab | Total | |||||
|---|---|---|---|---|---|---|
| Nasal Swab | Axillary Swabs | Inguinal Swabs | Rectal/Perianal Swabs | |||
| Screening Result | Axillary Swabs-screening MRSA-positive | 0 | 55 | 0 | 0 | 55 |
| Axillary Swabs-screening MRSA-negative | 0 | 1124 | 0 | 0 | 1124 | |
| Inguinal Swab-screening MRSA-positive | 0 | 0 | 75 | 0 | 75 | |
| Inguinal swabs-screening MRSA-negative | 0 | 0 | 742 | 0 | 742 | |
| Proteeae group bacteria-MDR-positive | 0 | 0 | 0 | 33 | 33 | |
| KESC bacteria group-MDR-negative | 0 | 0 | 0 | 3435 | 3435 | |
| KESC bacteria group-MDR-positive | 0 | 0 | 0 | 491 | 491 | |
| Proteeae group bacteria-MDR-negative | 0 | 0 | 0 | 1931 | 1931 | |
| Nasal Swab-screening MRSA-positive | 141 | 0 | 0 | 0 | 141 | |
| Nasal Swab-screening MRSA-negative | 1850 | 0 | 0 | 0 | 1850 | |
| E. faecium-VRE-positive | 0 | 0 | 0 | 157 | 157 | |
| E. faecium-VRE-negative | 0 | 0 | 0 | 1806 | 1806 | |
| E. faecalis-positive | 0 | 0 | 0 | 4 | 4 | |
| E. faecalis-negative | 0 | 0 | 0 | 1959 | 1959 | |
| E. coli-ESBL-positive | 0 | 0 | 0 | 342 | 342 | |
| E. coli-ESBL-negative | 0 | 0 | 0 | 3584 | 3584 | |
| AB-MDR-positive | 0 | 0 | 0 | 62 | 62 | |
| AB-MDR-negative | 0 | 0 | 0 | 1202 | 1202 | |
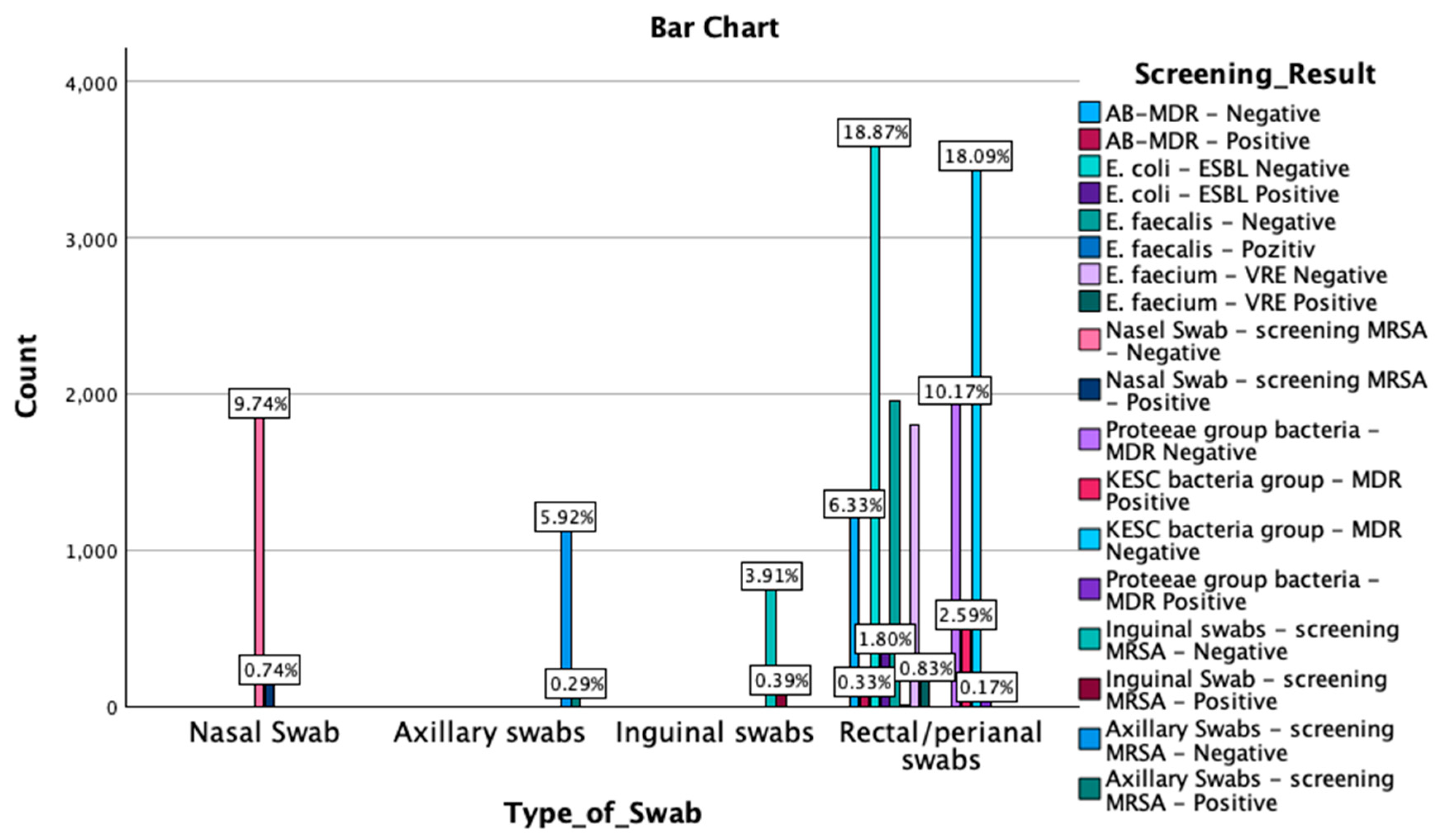
| Total | 1991 | 1179 | 817 | 15,006 | 18,993 | |
| Count | AB-MDR-Negative | AB-MDR-Positive | E. coli-Negative | E. coli-Positive | E. faecalis-Negative | E. faecalis-Positive | E. faecium-Negative | E. faecium-Positive | Nasal Swab-Screening Methicillin-Resistant Staphylococcus aureus-Negative | Nasal Swab-Screening Methicillin-Resistant Staphylococcus aureus-Positive | Proteeae Group Bacteria-MDR-Negative | KESC Bacteria Group-MDR–Positive | KESC Bacteria Group-MDR-Negative | Proteeae Group Bacteria-MDR-Positive | Inguinal Swab-Screening Methicillin-Resistant Staphylococcus aureus-Negative | Inguinal Swab-Screening Methicillin-Resistant Staphylococcus aureus-Positive | Axillary Swab-Screening Methicillin-Resistant Staphylococcus aureus-Negative | Axillary Swab-Screening Methicillin-Resistant Staphylococcus aureus-Positive | Total | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Department | Allergology Department | 4 | 0 | 17 | 5 | 11 | 0 | 8 | 3 | 11 | 0 | 11 | 0 | 22 | 0 | 2 | 0 | 9 | 0 | 103 |
| 1st Anesthesia and Intensive Care Unit Clinic | 368 | 15 | 1210 | 96 | 651 | 2 | 614 | 39 | 591 | 62 | 644 | 180 | 1126 | 9 | 7 | 2 | 609 | 36 | 6261 | |
| Anesthesia and Intensive Care Unit Dedicated to Neurosurgical, Plastic Surgery, and Other Surgical Departments | 83 | 26 | 312 | 32 | 171 | 0 | 145 | 26 | 150 | 21 | 168 | 92 | 252 | 4 | 122 | 30 | 17 | 2 | 1653 | |
| Infectious Diseases Clinic | 101 | 8 | 311 | 49 | 179 | 1 | 165 | 15 | 168 | 12 | 176 | 43 | 317 | 4 | 160 | 14 | 5 | 1 | 1729 | |
| 1st Cardiology Clinic | 7 | 0 | 19 | 1 | 10 | 0 | 10 | 0 | 9 | 1 | 10 | 1 | 19 | 0 | 3 | 0 | 7 | 0 | 97 | |
| 2nd Cardiology Clinic | 4 | 0 | 17 | 3 | 10 | 0 | 9 | 1 | 9 | 1 | 10 | 2 | 18 | 0 | 4 | 0 | 5 | 1 | 94 | |
| 1st Surgical Clinic—Thoracic Surgery Department | 6 | 0 | 11 | 3 | 7 | 0 | 5 | 2 | 7 | 0 | 5 | 7 | 7 | 2 | 0 | 0 | 7 | 0 | 69 | |
| 2nd Surgical Clinic—Vascular Surgery Department | 4 | 0 | 14 | 0 | 7 | 0 | 6 | 1 | 5 | 2 | 7 | 3 | 11 | 0 | 0 | 0 | 7 | 0 | 67 | |
| 1st Surgical Clinic | 1 | 0 | 4 | 0 | 2 | 0 | 2 | 0 | 2 | 0 | 2 | 1 | 3 | 0 | 1 | 0 | 1 | 0 | 19 | |
| 2nd Surgical Clinic | 5 | 0 | 17 | 1 | 9 | 0 | 6 | 3 | 7 | 2 | 9 | 7 | 11 | 0 | 0 | 0 | 9 | 0 | 86 | |
| Diabetes Clinic | 0 | 0 | 2 | 0 | 1 | 0 | 0 | 1 | 1 | 0 | 1 | 2 | 0 | 0 | 1 | 0 | 0 | 0 | 9 | |
| Gastroenterology Clinic | 6 | 0 | 15 | 1 | 8 | 0 | 8 | 0 | 8 | 0 | 8 | 5 | 11 | 0 | 0 | 0 | 7 | 1 | 78 | |
| Haematology Clinic | 23 | 0 | 50 | 6 | 28 | 0 | 23 | 5 | 28 | 0 | 28 | 10 | 46 | 0 | 0 | 0 | 28 | 0 | 275 | |
| 1st Internal Medicine Clinic | 189 | 5 | 586 | 58 | 322 | 1 | 300 | 23 | 302 | 20 | 317 | 38 | 606 | 6 | 8 | 0 | 302 | 12 | 3095 | |
| 2nd Internal Medicine Clinic | 179 | 1 | 472 | 48 | 260 | 0 | 239 | 21 | 246 | 14 | 255 | 51 | 469 | 5 | 236 | 23 | 0 | 1 | 2520 | |
| Neurosurgical Clinic | 3 | 0 | 15 | 1 | 8 | 0 | 6 | 2 | 8 | 0 | 8 | 2 | 14 | 0 | 4 | 0 | 4 | 0 | 75 | |
| Neurology Clinic | 24 | 2 | 47 | 5 | 26 | 0 | 26 | 0 | 24 | 2 | 25 | 13 | 39 | 1 | 8 | 2 | 15 | 1 | 260 | |
| Obstetrics and Gynecology Clinic | 1 | 0 | 2 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 1 | 0 | 2 | 0 | 1 | 0 | 0 | 0 | 10 | |
| Oncology Clinic | 3 | 0 | 8 | 2 | 5 | 0 | 5 | 0 | 5 | 0 | 5 | 0 | 10 | 0 | 2 | 0 | 3 | 0 | 48 | |
| Orthopedics and Traumatology Clinic | 47 | 0 | 128 | 10 | 69 | 0 | 65 | 4 | 97 | 2 | 69 | 9 | 129 | 0 | 86 | 3 | 12 | 0 | 730 | |
| 1st Medical Rehabilitation Clinic | 6 | 0 | 14 | 0 | 7 | 0 | 7 | 0 | 7 | 0 | 6 | 0 | 14 | 1 | 4 | 0 | 4 | 0 | 70 | |
| Medical Rehabilitation Clinic—Neurological Department | 39 | 3 | 88 | 4 | 46 | 0 | 44 | 2 | 45 | 0 | 45 | 4 | 88 | 1 | 24 | 0 | 22 | 0 | 455 | |
| Medical Rehabilitation Clinic—Orthopedics and Traumatology Department | 25 | 0 | 51 | 3 | 27 | 0 | 27 | 0 | 27 | 0 | 27 | 0 | 54 | 0 | 18 | 0 | 9 | 0 | 268 | |
| Rheumatology Clinic | 16 | 0 | 36 | 6 | 21 | 0 | 20 | 1 | 21 | 0 | 21 | 4 | 38 | 0 | 0 | 0 | 21 | 0 | 205 | |
| Cardiology Intensive Care Department | 49 | 1 | 111 | 7 | 59 | 0 | 52 | 7 | 57 | 2 | 59 | 13 | 105 | 0 | 46 | 1 | 12 | 0 | 581 | |
| Urology Clinic | 5 | 0 | 18 | 0 | 9 | 0 | 9 | 0 | 9 | 0 | 9 | 0 | 18 | 0 | 4 | 0 | 5 | 0 | 86 | |
| 2nd Anesthesia and Intensive Care Unit Clinic | 2 | 0 | 4 | 0 | 2 | 0 | 2 | 0 | 2 | 0 | 2 | 0 | 4 | 0 | 0 | 0 | 2 | 0 | 20 | |
| Nephrology and Dialysis Clinic | 2 | 1 | 5 | 1 | 3 | 0 | 2 | 1 | 3 | 0 | 3 | 4 | 2 | 0 | 1 | 0 | 2 | 0 | 30 | |
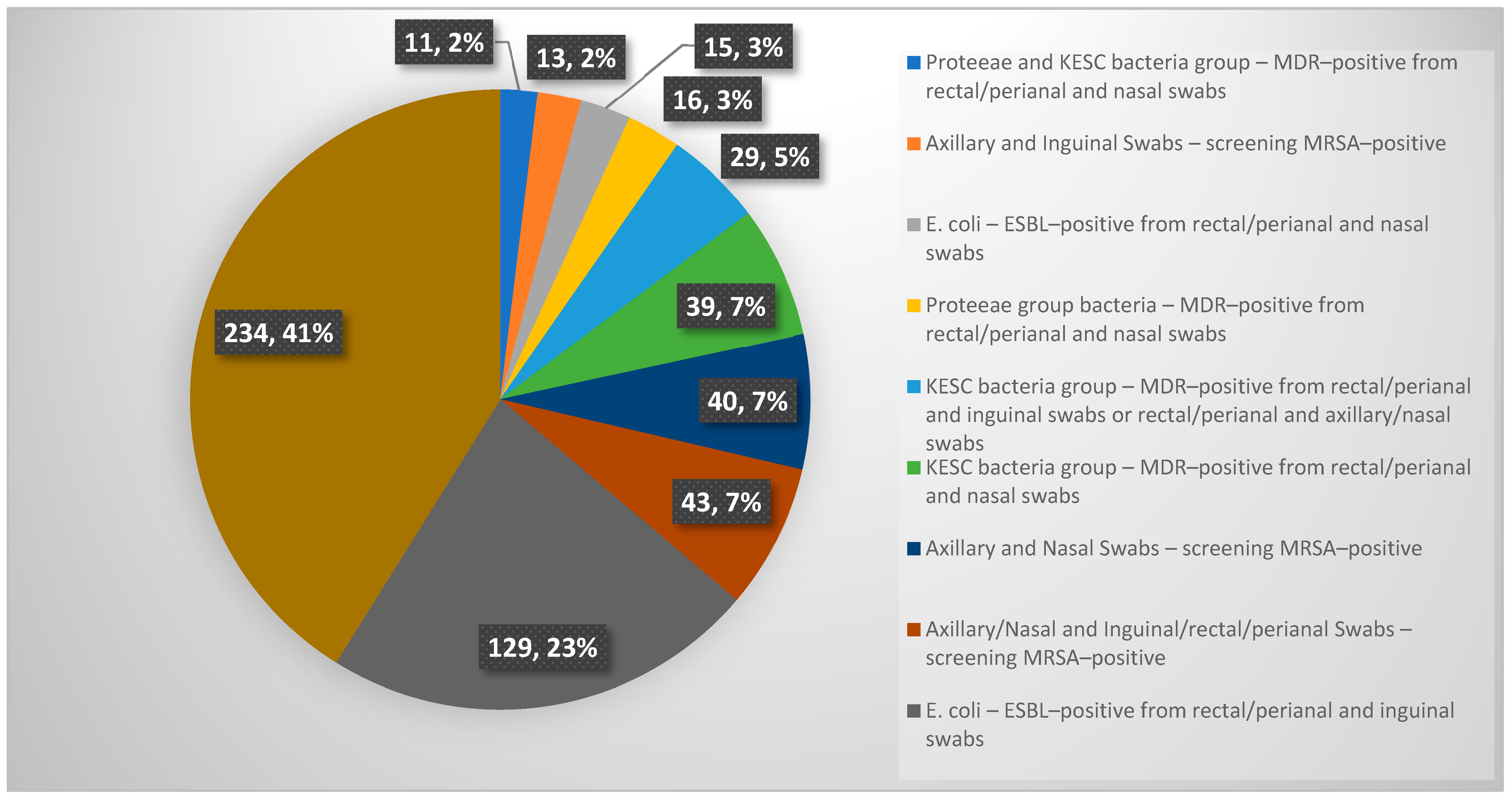
| Total | 1202 | 62 | 3584 | 342 | 1959 | 4 | 1806 | 157 | 1850 | 141 | 1931 | 491 | 3435 | 33 | 742 | 75 | 1124 | 55 | 18,993 | |
References
- Byrd, A.L.; Belkaid, Y.; Segre, J.A. The Human Skin Microbiome. Nat. Rev. Microbiol. 2018, 16, 143–155. [Google Scholar] [CrossRef] [PubMed]
- Reilly, J.S.; Stewart, S.; Christie, P.; Allardice, G.M.; Stari, T.; Matheson, A.; Masterton, R.; Gould, I.M.; Williams, C. Universal Screening for Meticillin-Resistant Staphylococcus Aureus in Acute Care: Risk Factors and Outcome from a Multicentre Study. J. Hosp. Infect. 2012, 80, 31–35. [Google Scholar] [CrossRef]
- Torres, K.; Sampathkumar, P. Predictors of Methicillin-Resistant Staphylococcus Aureus Colonization at Hospital Admission. Am. J. Infect. Control 2013, 41, 1043–1047. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Guidelines for the Prevention of Surgical Site Infection; WHO: Geneva, Switzerland, 2016; ISBN 9789241549882. [Google Scholar]
- Köck, R.; Mellmann, A.; Schaumburg, F.; Friedrich, A.W.; Kipp, F.; Becker, K. The Epidemiology of Methicillin-Resistant Staphylococcus Aureus (MRSA) in Germany. Dtsch. Arztebl. Int. 2011, 108, 761. [Google Scholar] [CrossRef] [PubMed]
- Jurke, A.; Daniels-Haardt, I.; Silvis, W.; Berends, M.S.; Glasner, C.; Becker, K.; Köck, R.; Friedrich, A.W. Changing Epidemiology of Meticillin-Resistant Staphylococcus Aureus in 42 Hospitals in the Dutch–German Border Region, 2012 to 2016: Results of the Search-and-Follow-Policy. Eurosurveillance 2019, 24, 1800244. [Google Scholar] [CrossRef]
- Piezzi, V.; Gasser, M.; Atkinson, A.; Kronenberg, A.; Vuichard-Gysin, D.; Harbarth, S.; Marschall, J.; Buetti, N. Increasing Proportion of Vancomycin Resistance among Enterococcal Bacteraemias in Switzerland: A 6-Year Nation-Wide Surveillance, 2013 to 2018. Eurosurveillance 2020, 25, 1900575. [Google Scholar] [CrossRef]
- Wassilew, N.; Seth-Smith, H.M.; Rolli, E.; Fietze, Y.; Casanova, C.; Führer, U.; Egli, A.; Marschall, J.; Buetti, N. Outbreak of Vancomycin-Resistant Enterococcus Faecium Clone ST796, Switzerland, December 2017 to April 2018. Eurosurveillance 2018, 23, 1800351. [Google Scholar] [CrossRef]
- Meschiari, M.; Kaleci, S.; Monte, M.D.; Dessilani, A.; Santoro, A.; Scialpi, F.; Franceschini, E.; Orlando, G.; Cervo, A.; Monica, M.; et al. Vancomycin Resistant Enterococcus Risk Factors for Hospital Colonization in Hematological Patients: A Matched Case-Control Study. Antimicrob. Resist. Infect. Control 2023, 12, 126. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Nosocomial Enterococci Resistant to Vancomycin—United States, 1989–1993. MMWR Morb. Mortal. Wkly. Rep. 1993, 42, 597–599. [Google Scholar]
- Giannella, M.; Trecarichi, E.M.; De Rosa, F.G.; Del Bono, V.; Bassetti, M.; Lewis, R.E.; Losito, A.R.; Corcione, S.; Saffioti, C.; Bartoletti, M.; et al. Risk Factors for Carbapenem-Resistant Klebsiella Pneumoniae Bloodstream Infection among Rectal Carriers: A Prospective Observational Multicentre Study. Clin. Microbiol. Infect. 2014, 20, 1357–1362. [Google Scholar] [CrossRef]
- Cano, Á.; Gutiérrez-Gutiérrez, B.; Machuca, I.; Torre-Giménez, J.; Gracia-Ahufinger, I.; Natera, A.M.; Pérez-Nadales, E.; Castón, J.J.; Rodríguez-Baño, J.; Martínez-Martínez, L.; et al. Association between Timing of Colonization and Risk of Developing Klebsiella Pneumoniae Carbapenemase-Producing K. Pneumoniae Infection in Hospitalized Patients. Microbiol. Spectr. 2022, 10, e01970-12. [Google Scholar] [CrossRef]
- Khawcharoenporn, T.; Laichuthai, W. Subsequent Carbapenem-Resistant Enterobacteriaceae (CRE)–Associated Infections among Hospitalized Patients with CRE Colonization: Impact of Antibiotic Use and Other Factors. Infect. Control Hosp. Epidemiol. 2020, 41, 1084–1089. [Google Scholar] [CrossRef] [PubMed]
- Zheng, G.; Cai, J.; Deng, H.; Yang, H.; Xiong, W.; Chen, E.; Bai, H.; He, J. Development of a Risk Prediction Model for Subsequent Infection after Colonization with Carbapenem-Resistant Enterobacterales: A Retrospective Cohort Study. Antimicrob. Resist. Infect. Control 2024, 13, 46. [Google Scholar] [CrossRef] [PubMed]
- Alhmidi, H.; Cadnum, J.L.; Koganti, S.; Jencson, A.L.; Bonomo, R.A.; Wilson, B.M.; Mayer, J.; Samore, M.H.; Donskey, C.J. Shedding of Multidrug-Resistant Gram-Negative Bacilli by Colonized Patients during Procedures and Patient Care Activities. Am. J. Infect. Control 2020, 48, 1336–1340. [Google Scholar] [CrossRef] [PubMed]
- Richter, S.E.; Miller, L.; Needleman, J.; Uslan, D.Z.; Bell, D.; Watson, K.; Humphries, R.; McKinnell, J.A. Risk Factors for Development of Carbapenem Resistance Among Gram-Negative Rods. In Open Forum Infectious Diseases; Oxford University Press: New York, NY, USA, 2019; Volume 6. [Google Scholar] [CrossRef]
- Tang, S.S.L.; Chee, E.; Teo, J.Q.; Chlebicki, M.P.; Kwa, A.L.H. Incidence of a Subsequent Carbapenem-Resistant Enterobacteriaceae Infection after Previous Colonisation or Infection: A Prospective Cohort Study. Int. J. Antimicrob. Agents 2021, 57, 106340. [Google Scholar] [CrossRef]
- Borer, A.; Saidel-Odes, L.; Eskira, S.; Nativ, R.; Riesenberg, K.; Livshiz-Riven, I.; Schlaeffer, F.; Sherf, M.; Peled, N. Risk Factors for Developing Clinical Infection with Carbapenem-Resistant Klebsiella Pneumoniae in Hospital Patients Initially Only Colonized with Carbapenem-Resistant K Pneumoniae. Am. J. Infect. Control 2012, 40, 421–425. [Google Scholar] [CrossRef]
- Schechner, V.; Kotlovsky, T.; Kazma, M.; Mishali, H.; Schwartz, D.; Navon-Venezia, S.; Schwaber, M.J.; Carmeli, Y. Asymptomatic Rectal Carriage of BlaKPC Producing Carbapenem-Resistant Enterobacteriaceae: Who Is Prone to Become Clinically Infected? Clin. Microbiol. Infect. 2013, 19, 451–456. [Google Scholar] [CrossRef]
- Palacios-Baena, Z.R.; Oteo, J.; Conejo, C.; Larrosa, M.N.; Bou, G.; Fernández-Martínez, M.; González-López, J.J.; Pintado, V.; Martínez-Martínez, L.; Merino, M.; et al. Comprehensive Clinical and Epidemiological Assessment of Colonisation and Infection Due to Carbapenemase-Producing Enterobacteriaceae in Spain. J. Infect. 2016, 72, 152–160. [Google Scholar] [CrossRef]
- World Health Organization. WHO Publishes List of Bacteria for Which New Antibiotics Are Urgently Needed; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, Research, and Development of New Antibiotics: The WHO Priority List of Antibiotic-Resistant Bacteria and Tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef]
- Birlutiu, R.M.; Stoica, C.I.; Russu, O.; Cismasiu, R.S.; Birlutiu, V. Positivity Trends of Bacterial Cultures from Cases of Acute and Chronic Periprosthetic Joint Infections. J. Clin. Med. 2022, 11, 2238. [Google Scholar] [CrossRef]
- Roman, M.D.; Bocea, B.-A.; Ion, N.-I.-C.; Vorovenci, A.E.; Dragomirescu, D.; Birlutiu, R.-M.; Birlutiu, V.; Fleaca, S.R. Are There Any Changes in the Causative Microorganisms Isolated in the Last Years from Hip and Knee Periprosthetic Joint Infections? Antimicrobial Susceptibility Test Results Analysis. Microorganisms 2023, 11, 116. [Google Scholar] [CrossRef] [PubMed]
- Crespo-Rivas, J.C.; Guisado-Gil, A.B.; Peñalva, G.; Rodríguez-Villodres, Á.; Martín-Gandul, C.; Pachón-Ibáñez, M.E.; Lepe, J.A.; Cisneros, J.M. Are Antimicrobial Stewardship Interventions Effective and Safe in Long-Term Care Facilities? A Systematic Review and Meta-Analysis. Clin. Microbiol. Infect. 2021, 27, 1431–1438. [Google Scholar] [CrossRef]
- Jump, R.L.P.; Olds, D.M.; Seifi, N.; Kypriotakis, G.; Jury, L.A.; Peron, E.P.; Hirsch, A.A.; Drawz, P.E.; Watts, B.; Bonomo, R.A.; et al. Effective Antimicrobial Stewardship in a Long-Term Care Facility through an Infectious Disease Consultation Service: Keeping a LID on Antibiotic Use. Infect. Control Hosp. Epidemiol. 2012, 33, 1185–1192. [Google Scholar] [CrossRef] [PubMed]
- Schneider, F.; Dureau, A.-F.; Hellé, S.; Betscha, C.; Senger, B.; Cremel, G.; Boulmedais, F.; Strub, J.-M.; Corti, A.; Meyer, N.; et al. A Pilot Study on Continuous Infusion of 4% Albumin in Critically Ill Patients. Crit. Care Explor. 2019, 1, e0044. [Google Scholar] [CrossRef]
- Safdar, N.; Bradley, E.A. The Risk of Infection after Nasal Colonization with Staphylococcus Aureus. Am. J. Med. 2008, 121, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Wohrley, J.D.; Bartlett, A.H. The Role of the Environment and Colonization in Healthcare-Associated Infections. In Healthcare-Associated Infections in Children; Springer International Publishing: Cham, Germany, 2019; pp. 17–36. [Google Scholar]

| Department | |||
|---|---|---|---|
| Frequency (n=) | Percent | Cumulative Percent | |
| Allergology Department | 11 | 0.7 | 0.7 |
| 1st Anesthesia and Intensive Care Unit Clinic | 420 | 27.6 | 28.3 |
| Anesthesia and Intensive Care Unit Dedicated to Neurosurgical, Plastic Surgery, and Other Surgical Departments | 63 | 4.1 | 32.5 |
| Infectious Diseases Clinic | 156 | 10.2 | 42.7 |
| 1st Cardiology Clinic | 10 | 0.7 | 43.4 |
| 2nd Cardiology Clinic | 10 | 0.7 | 44.0 |
| 1st Surgical Clinic—Thoracic Surgery Department | 6 | 0.4 | 44.4 |
| 2nd Surgical Clinic—Vascular Surgery Department | 6 | 0.4 | 44.8 |
| 1st Surgical Clinic | 2 | 0.1 | 44.9 |
| 2nd Surgical Clinic | 8 | 0.5 | 45.5 |
| Diabetes Clinic | 1 | 0.1 | 45.5 |
| Gastroenterology Clinic | 7 | 0.5 | 46.0 |
| Haematology Clinic | 25 | 1.6 | 47.6 |
| 1st Internal Medicine Clinic | 306 | 20.1 | 67.7 |
| 2nd Internal Medicine Clinic | 221 | 14.5 | 82.3 |
| Neurosurgical Clinic | 8 | 0.5 | 82.8 |
| Neurology Clinic | 23 | 1.5 | 84.3 |
| Obstetrics and Gynecology Clinic | 1 | 0.1 | 84.4 |
| Oncology Clinic | 5 | 0.3 | 84.7 |
| Orthopedics and Traumatology Clinic | 96 | 6.3 | 91.0 |
| 1st Medical Rehabilitation Clinic | 6 | 0.4 | 91.4 |
| Medical Rehabilitation Clinic—Neurological Department | 29 | 1.9 | 93.3 |
| Medical Rehabilitation Clinic—Orthopedics and Traumatology Department | 16 | 1.1 | 94.3 |
| Rheumatology Clinic | 19 | 1.2 | 95.6 |
| Cardiology Intensive Care Department | 54 | 3.5 | 99.1 |
| Urology Clinic | 8 | 0.5 | 99.7 |
| 2nd Anesthesia and Intensive Care Unit Clinic | 2 | 0.1 | 99.8 |
| Nephrology and Dialysis Clinic | 3 | 0.2 | 100.0 |
| Total | 1522 | 100.0 | |
| Screening Result | ||
|---|---|---|
| Frequency (n=) | Percent (%) | |
| AB-MDR-negative | 1202 | 6.3 |
| AB-MDR-positive | 62 | 0.3 |
| E. coli-ESBL-negative | 3584 | 18.9 |
| E. coli-ESBL-positive | 342 | 1.8 |
| E. faecalis-negative | 1959 | 10.3 |
| E. faecalis-positive | 4 | 0 |
| E. faecium-VRE-negative | 1806 | 9.5 |
| E. faecium-VRE-positive | 157 | 0.8 |
| Nasal swab-screening MRSA-negative | 1850 | 9.7 |
| Nasal swab-screening MRSA-positive | 141 | 0.7 |
| Proteeae group bacteria-MDR-negative | 1931 | 10.2 |
| KESC bacteria group-MDR-positive | 491 | 2.6 |
| KESC bacteria group-MDR-negative | 3435 | 18.1 |
| Proteeae group bacteria-MDR-positive | 33 | 0.2 |
| Inguinal swab-screening MRSA-negative | 742 | 3.9 |
| Inguinal swab-screening MRSA-positive | 75 | 0.4 |
| Axillary swab-screening MRSA-negative | 1124 | 5.9 |
| Axillary swab-screening MRSA-positive | 55 | 0.3 |
| Total | 18,993 | 100.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Birlutiu, V.; Birlutiu, R.-M.; Ene, R.; Rusu, D. Experience in Implementing Colonization Screening in a Multidisciplinary County Clinical Hospital in Romania. Microorganisms 2025, 13, 775. https://doi.org/10.3390/microorganisms13040775
Birlutiu V, Birlutiu R-M, Ene R, Rusu D. Experience in Implementing Colonization Screening in a Multidisciplinary County Clinical Hospital in Romania. Microorganisms. 2025; 13(4):775. https://doi.org/10.3390/microorganisms13040775
Chicago/Turabian StyleBirlutiu, Victoria, Rares-Mircea Birlutiu, Razvan Ene, and Dana Rusu. 2025. "Experience in Implementing Colonization Screening in a Multidisciplinary County Clinical Hospital in Romania" Microorganisms 13, no. 4: 775. https://doi.org/10.3390/microorganisms13040775
APA StyleBirlutiu, V., Birlutiu, R.-M., Ene, R., & Rusu, D. (2025). Experience in Implementing Colonization Screening in a Multidisciplinary County Clinical Hospital in Romania. Microorganisms, 13(4), 775. https://doi.org/10.3390/microorganisms13040775









