Microbiome Research in Greece: A Comprehensive Bibliometric Study
Abstract
1. Introduction
1.1. Definitions of Microbiome
1.2. Gut Microbiome and Health
2. Materials and Methods
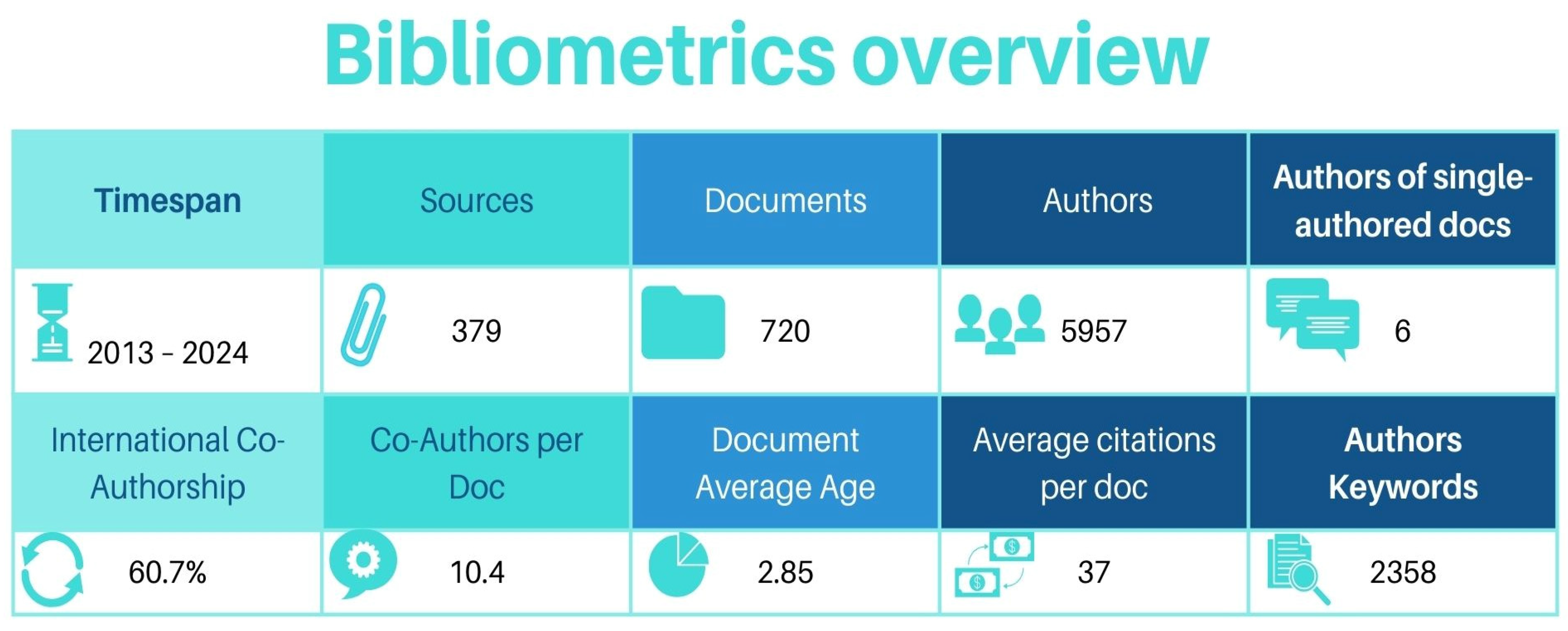
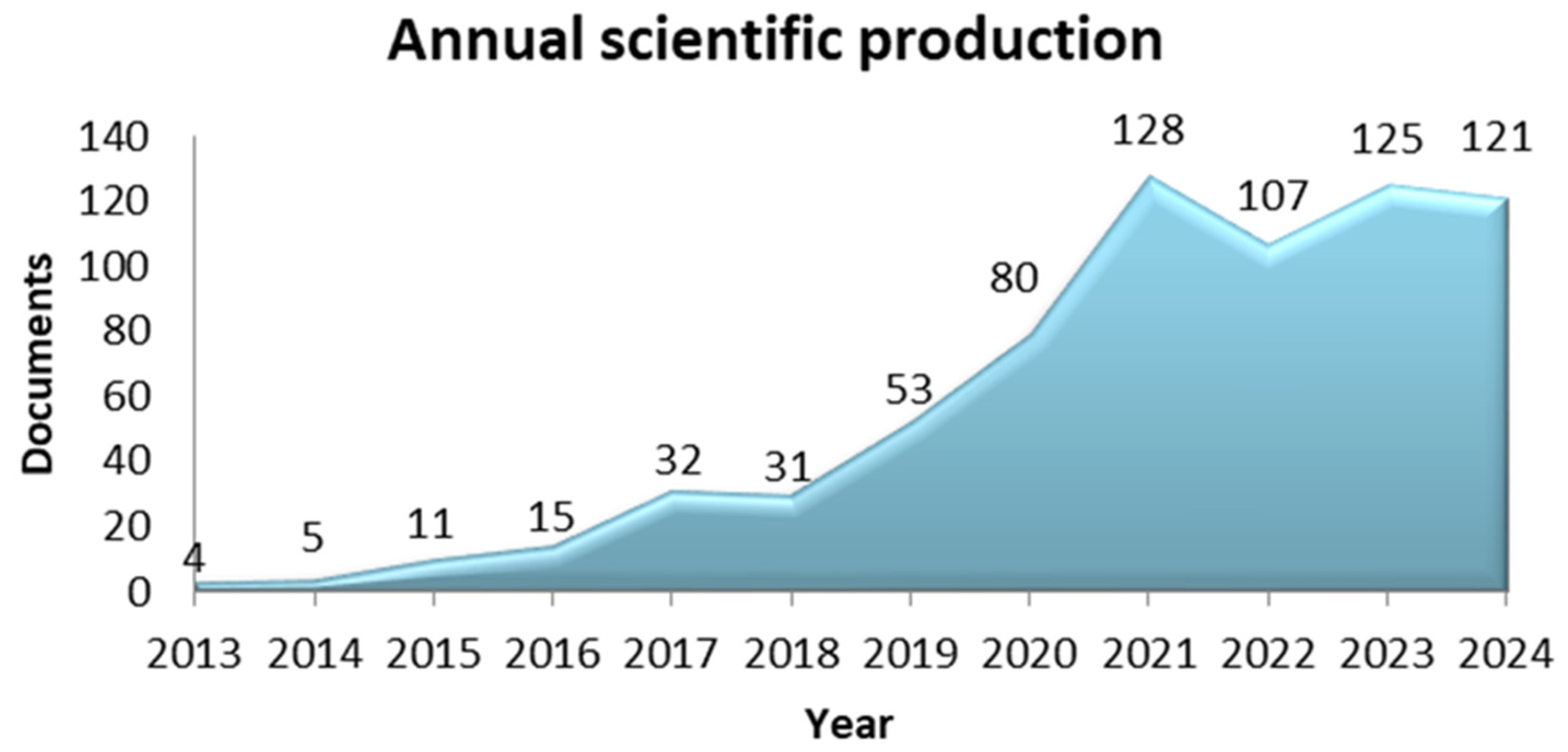
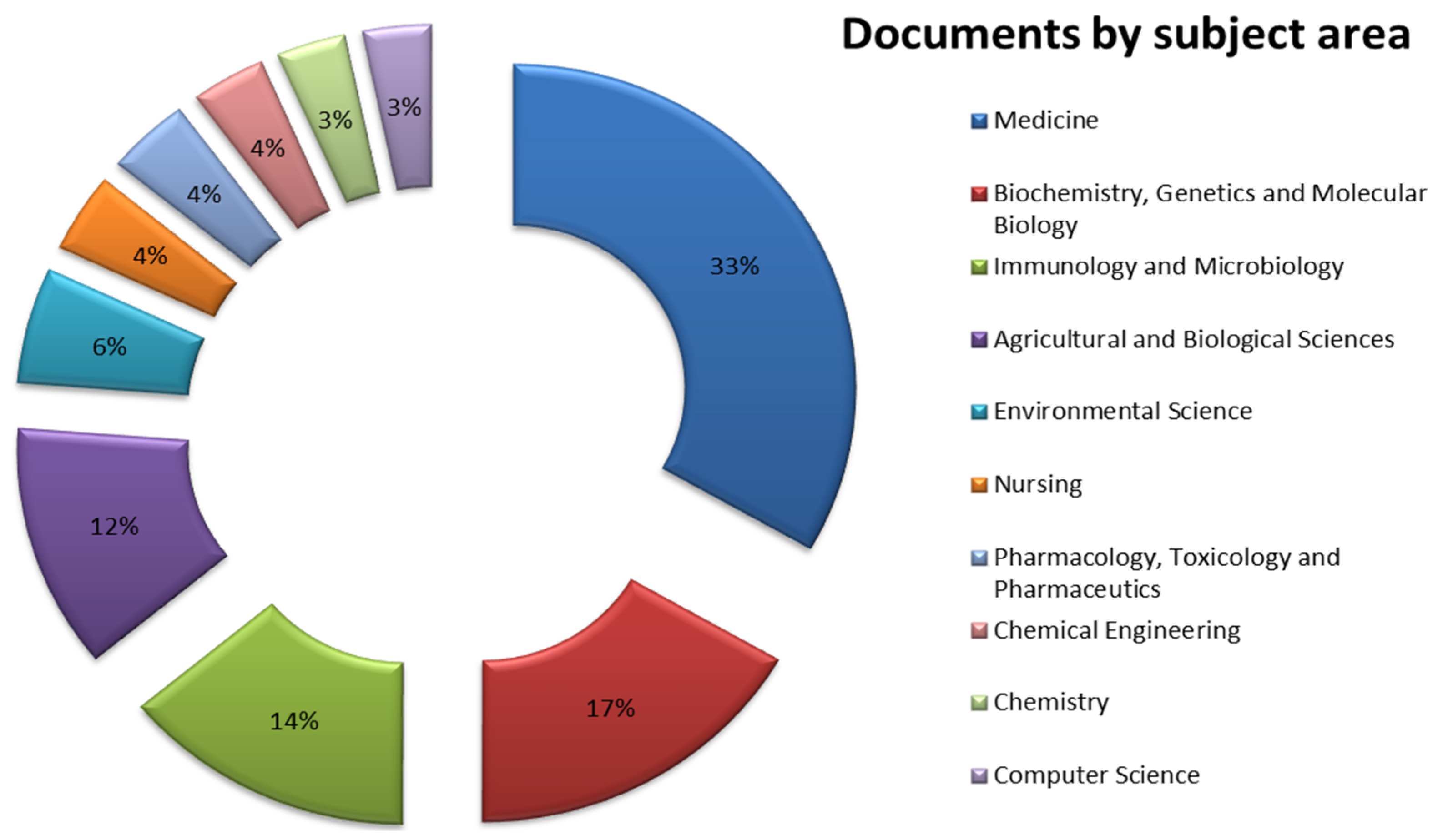
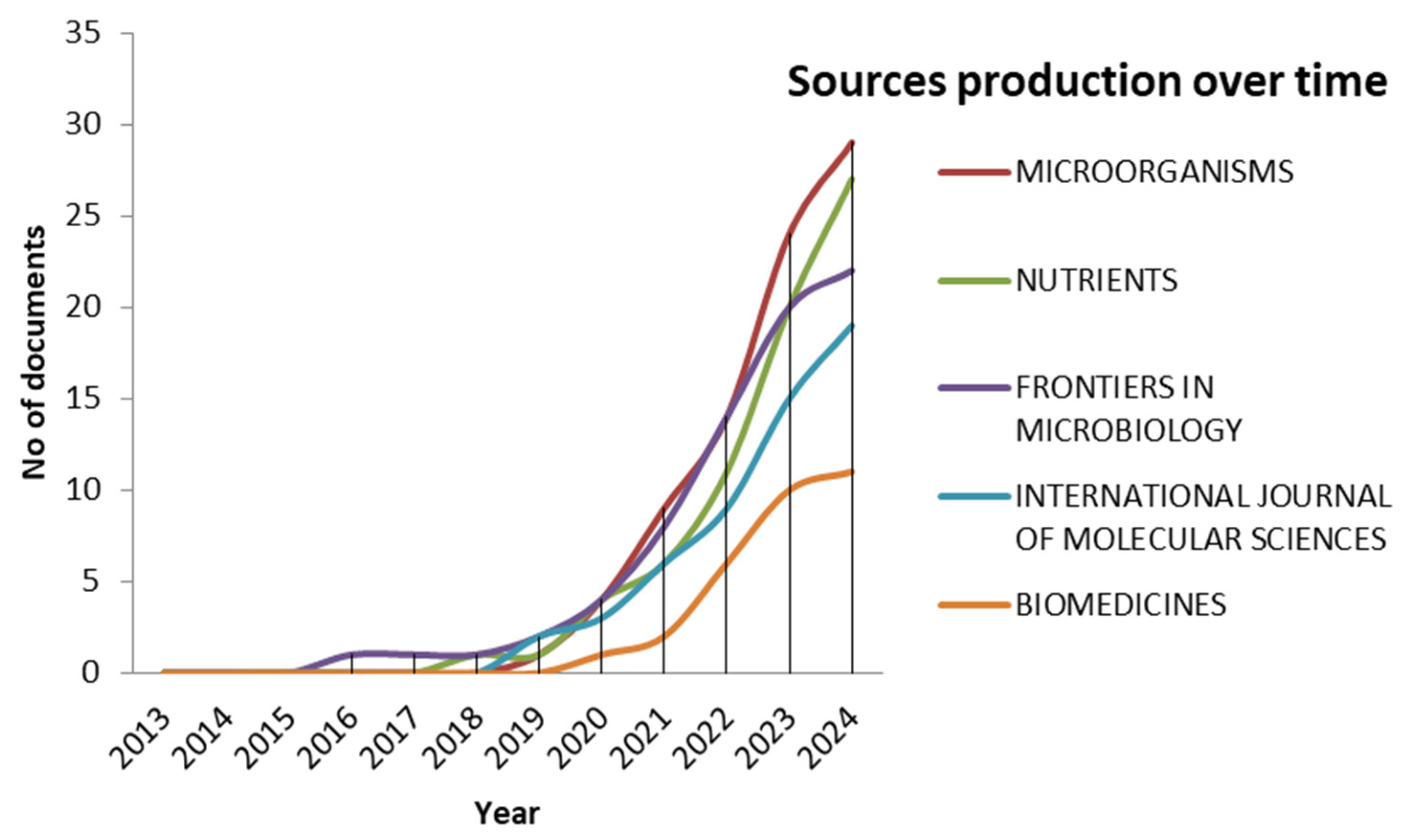
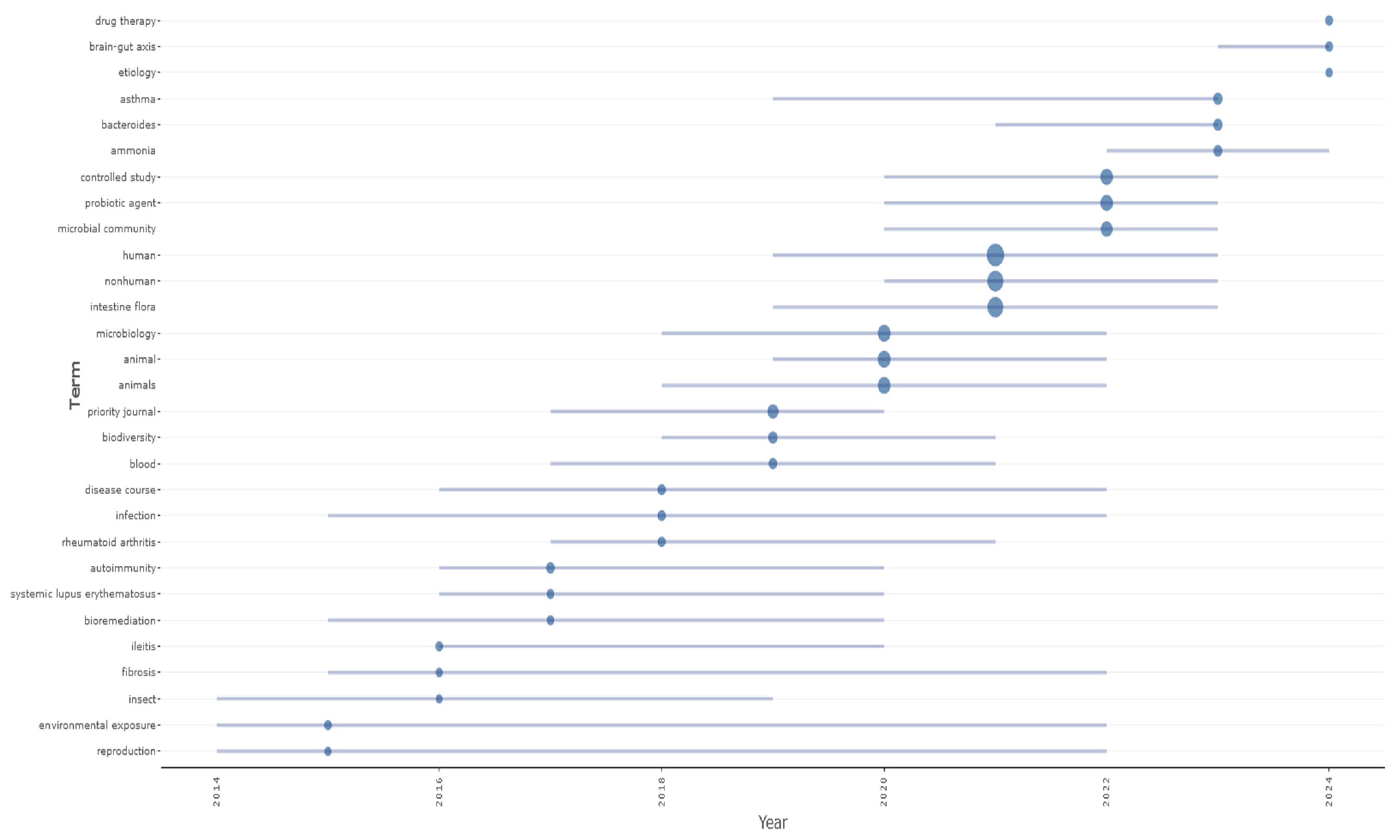
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Konopka, A. What is microbial community ecology? ISME J. 2009, 3, 1223–1230. [Google Scholar] [PubMed]
- Whipps, J.; Lewis, K.; Cooke, R. Mycoparasitism and plant disease control. In Fungi in Biological Control Systems; Burge, M., Ed.; Manchester University Press: Manchester, UK, 1988; pp. 161–187. [Google Scholar]
- Lederberg, J.; McCray, A.T. Ome Sweet Omics—A genealogical treasury of words. Scientist 2001, 15, 8. [Google Scholar]
- Marchesi, J.R.; Ravel, J. The vocabulary of microbiome research: A proposal. Microbiome 2015, 3, 31. [Google Scholar]
- Proctor, L. Priorities for the next 10 years of human microbiome research. Nature 2019, 569, 623–625. [Google Scholar] [CrossRef] [PubMed]
- Berg, G.; Rybakova, D.; Fischer, D.; Cernava, T.; Vergès, M.C.; Charles, T.; Chen, X.; Cocolin, L.; Eversole, K.; Corral, G.H.; et al. Microbiome definition revisited: Old concepts and new challenges. Microbiome 2020, 8, 103. [Google Scholar]
- Lloyd-Price, J.; Mahurkar, A.; Rahnavard, G.; Crabtree, J.; Orvis, J.; Hall, A.B.; Brantley, A.; Creasy, H.H.; McCracken, C.; Giglio, M.M.; et al. Strains, functions, and dynamics in the expanded Human Microbiome Project. Nature 2017, 550, 61–66. [Google Scholar] [PubMed]
- Clarke, G.; Stilling, R.M.; Kennedy, P.J.; Stanton, C.; Cryan, J.F.; Dinan, T.G. Minireview: Gut microbiota—The neglected endocrine organ. Mol. Endocrinol. 2014, 28, 1221–1238. [Google Scholar]
- Salvucci, E. The human-microbiome superorganism and its modulation to restore health. Int. J. Food Sci. Nutr. 2019, 70, 781–795. [Google Scholar]
- Huang, H.; Fang, M.; Jostins, L.; Mirkov, M.U.; Boucher, G.; Anderson, C.A.; Andersen, V.; Cleyen, I.; Cortes, A.; Crins, F.; et al. Fine-mapping inflammatory bowel disease loci to single-variant resolution. Nature 2017, 547, 173–178. [Google Scholar]
- Morgan, X.C.; Tickle, T.L.; Sokol, H.; Gevers, D.; Devaney, K.L.; Ward, D.V.; Reyes, J.A.; Shah, S.A.; Leleiko, N.; Snapper, S.B.; et al. Dysfunction of the intestinal microbiome in inflammatory bowel disease and treatment. Genome Biol. 2012, 13, R79. [Google Scholar]
- Hall, A.B.; Yassour, M.; Sauk, J.; Garner, A.; Jiang, X.; Arthur, T.; Lagoudas, G.K.; Vatanen, T.; Fornelos, N.; Wilson, R.; et al. A novel Ruminococcus gnavus clade enriched in inflammatory bowel disease patients. Genome Med. 2017, 9, 103. [Google Scholar]
- Lloyd-Price, J.; Arze, C.; Ananthakrishnan, A.N.; Schirmer, M.; Avila-Pachero, J.; Poon, T.W.; Andrews, E.; Ajami, J.N.; Bonham, K.S.; Brislawn, C.J.; et al. Multi-omics of the gut microbial ecosystem in inflammatory bowel diseases. Nature 2019, 569, 655–662. [Google Scholar] [PubMed]
- Yao, H.; Wan, J.Y.; Wang, C.Z.; Li, L.; Wang, J.; Li, Y.; Huang, W.H.; Zeng, J.; Wang, Q.; Yuan, C.S. Bibliometric analysis of research on the role of intestinal microbiota in obesity. PeerJ 2018, 6, e5091. [Google Scholar] [CrossRef] [PubMed]
- Duranti, S.; Ferrario, C.; Van Sinderen, D.; Ventura, M.; Turroni, F. Obesity and microbiota: An example of an intricate relationship. Genes Nutr. 2017, 12, 18. [Google Scholar] [PubMed]
- Kvit, K.B.; Kharchenko, N.V. Gut microbiota changes as a risk factor for obesity. Wiad. Lek. 2017, 70, 231–235. [Google Scholar]
- Feng, W.W.; Zhao, T.; Mao, G.H.; Wang, W.; Feng, Y.; Fang, L.; Zjeng, D.; Wu, H.; Dun, J.; Yang, L.; et al. Type 2 diabetic rats on a diet supplemented with chromium malate show improved glycometabolism, glycometabolism-related enzyme levels, and lipid metabolism. PLoS ONE 2015, 10, e0125952. [Google Scholar]
- Ozturk, Y. Etiology and comorbidities of childhood obesity. Turk. J. Gastroenterol. 2017, 28, 149–151. [Google Scholar]
- Wang, H.Y.; Li, Q.; Deng, W.W.; Siaw, O.E.; Wang, Q.; Wang, S.; Wanf, S.; Cao, X.; Xu, X.; Yu, J. Self-nano emulsifying drug delivery system of trans-cinnamic acid: Formulation development and pharmacodynamic evaluation in alloxan-induced type 2 diabetic rat model. Drug Dev. Res. 2015, 76, 82–93. [Google Scholar]
- Shi, X.; Ma, T.; Sakandar, H.A.; Menghe, B.; Sun, Z. Gut microbiome and aging nexus and underlying mechanism. Appl. Microbiol. Biotechnol. 2022, 106, 5349–5358. [Google Scholar]
- Aleman, F.; Valenzano, D.R. Microbiome evolution during host aging. PLoS Pathog. 2019, 15, e1007727. [Google Scholar]
- Salvucci, E. Microbiome, holobiome, and the net of life. Crit. Rev. Microbiol. 2016, 42, 485–494. [Google Scholar] [PubMed]
- Van der Meulen, T.A.; Harmsen, H.J.M.; Bootsma, H.; Spijkervet, F.K.L.; Kroese, F.G.M.; Vissink, A. The microbiome systemic diseases connection. Oral Dis. 2016, 73, 14741–14750. [Google Scholar] [CrossRef]
- Bercik, P.; Collins, S.M.; Verdu, E.F. Microbes and the gut-brain axis. Neurogastroenterol. Motil. 2012, 24, 405–413. [Google Scholar]
- Foster, J.A.; McVey Neufeld, K.-A. Gut-brain axis: How the microbiome influences anxiety and depression. Trends Neurosci. 2013, 36, 305–312. [Google Scholar] [PubMed]
- Yang, Z.; Li, J.; Gui, X.; Shi, X.; Bao, Z.; Han, H.; Ming, D.L. Updated review of research on the gut microbiota and their relation to depression in animals and human beings. Mol. Psychiatry 2020, 25, 2759–2772. [Google Scholar]
- Cussotto, S.; Strain, C.R.; Fouhy, F.; Strain, R.G.; Peterson, V.L.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Differential effects of psychotropic drugs on microbiome composition and gastrointestinal function. Psychopharmacology 2019, 236, 1671–1685. [Google Scholar] [PubMed]
- Zhu, X.; Hu, J.; Deng, S.; Tan, Y.; Qiu, C.; Zhang, M.; Ni, X.; Lu, H.; Wang, Z.; Li, L. Bibliometric and visual analysis of research on the links between the gut microbiota and depression from 1999 to 2019. Front. Psychiatry 2021, 11, 587670. [Google Scholar]
- Rosenfeld, C.S. Microbiome disturbances and autism spectrum disorders. Drug Metab. Dispos. Biol. Fate Chem. 2015, 43, 1557–1571. [Google Scholar] [PubMed]
- Li, Q.; Han, Y.; Dy, A.B.C.; Hagerman, R.J. The gut microbiota and autism spectrum disorders. Front. Cell Neurosci. 2017, 11, 120. [Google Scholar]
- Strati, F.; Cavalieri, D.; Albanese, D.; Felice, C.D.; Donati, C.; Hayek, J.; Jousson, O.; Leoncini, S.; Renzi, D.; Calabro, A.; et al. New evidence on the altered gut microbiota in autism spectrum disorders. Microbiome 2017, 5, 24. [Google Scholar]
- Pritchard, A. Statistical bibliography or bibliometrics? J. Doc. 1969, 25, 348–349. [Google Scholar]
- Rousseau, R.; Egghe, L.; Guns, R. Becoming Metric-Wise: A Bibliometric Guide for Researchers; Chandos Publishing: Cambridge, MA, USA, 2018. [Google Scholar]
- De Bellis, N. Bibliometrics and Citation Analysis: From the Science Citation Index to Cybermetrics; Scarecrow Press: Lanham, MD, USA, 2009. [Google Scholar]
- Sarli, C.C.; Dubinsky, E.K.; Holmes, K.L. Beyond citation analysis: A model for assessment of research impact. J. Med. Libr. Assoc. 2010, 98, 17–23. [Google Scholar] [PubMed]
- Gusenbauer, M.; Haddaway, N.R. Which academic search systems are suitable for systematic reviews or meta-analyses? Evaluating retrieval qualities of Google Scholar, PubMed, and 26 other resources. Res. Syn. Meth. 2020, 11, 181–217. [Google Scholar]
- Yataganbaba, A.; Kurtbaş, I. A scientific approach with bibliometric analysis related to brick and tile drying: A review. Renew. Sustain. Energy Rev. 2016, 59, 206–224. [Google Scholar] [CrossRef]
- Martín-Martín, A.; Orduna-Malea, E.; Thelwall, M.; Delgado López-Cózar, E. Google Scholar, Web of Science, and Scopus: A systematic comparison of citations in 252 subject categories. J. Inf. 2018, 12, 1160–1177. [Google Scholar]
- Darko, A.; Chan, A.P.; Adabre, M.A.; Edwards, D.J.; Hosseini, M.R.; Ameyaw, E.E. Artificial intelligence in the AEC industry: Scientometric analysis and visualization of research activities. Autom. Constr. 2020, 112, 103081. [Google Scholar]
- Aria, M.; Cuccurullo, C. bibliometrix: An R-tool for comprehensive science mapping analysis. J. Informetr. 2017, 11, 959–975. [Google Scholar]
- Donthu, N.; Kumar, S.; Mukherjee, D.; Pandey, N.; Lim, W.M. How to conduct a bibliometric analysis: An overview and guidelines. J. Bus. Res. 2021, 133, 285–296. [Google Scholar] [CrossRef]
- Lim, W.M.; Kumar, S. Guidelines for interpreting the results of bibliometric analysis: A sensemaking approach. Glob. Bus. Organ. Excell. 2024, 43, 17–26. [Google Scholar] [CrossRef]
- Passas, I. Bibliometric Analysis: The Main Steps. Encyclopedia 2024, 4, 1014–1025. [Google Scholar] [CrossRef]
- Malfertheiner, P.; Megraud, F.; O’Morain, C.A.; Gisbert, J.P.; Kuipers, E.J.; Axon, A.T.; Bazzoli, F.; Gasbarrini, A.; Atherton, J.; Graham, D.Y.; et al. Management of Helicobacter pylori infection—The Maastricht V/Florence Consensus Report. Gut 2017, 66, 6–30. [Google Scholar] [CrossRef] [PubMed]
- Avgerinos, K.I.; Spyrou, N.; Mantzoros, C.S.; Dalamaga, M. Obesity and cancer risk: Emerging biological mechanisms and perspectives. Metabolism 2020, 92, 121–135. [Google Scholar] [CrossRef]
- Economou, V.; Gousia, P. Agriculture and food animals as a source of antimicrobial-resistant bacteria. Infect. Drug Resist. 2015, 8, 49–61. [Google Scholar]
- Petra, A.I.; Panagiotidou, S.; Hatziagelaki, E.; Stewart, J.M.; Conti, P.; Theoharides, T.C. Gut-microbiota-brain axis and its effect on neuropsychiatric disorders with suspected immune dysregulation. Clin. Ther. 2015, 37, 984–995. [Google Scholar] [CrossRef] [PubMed]
- Stefanis, C.; Giorgi, E.; Tselemponis, G.; Voidarou, C.; Skoufos, I.; Tzora, A.; Tsigalou, C.; Kourkoutas, Y.; Constantinidis, T.C.; Bezirtzoglou, E. Terroir in view of bibliometrics. Stats 2023, 6, 956–979. [Google Scholar] [CrossRef]
- Blondel, V.D.; Guillaume, J.L.; Lambiotte, R.; Lefebvre, E. Fast Unfolding of Communities in Large Networks. J. Stat. Mech. Theory Exp. 2008, 2008, P10008. [Google Scholar] [CrossRef]
- Kammerer, K.; Göster, M.; Reichert, M.; Pryss, R. Ambalytics: A Scalable and Distributed System Architecture Concept for Bibliometric Network Analyses. Future Internet 2021, 13, 203. [Google Scholar] [CrossRef]
- Dovrolis, N.; Kolios, G.; Spyrou, G.M.; Maroulakou, I. Computational profiling of the gut-brain axis: Microflora dysbiosis insights to neurological disorders. Brief Bioinform. 2019, 20, 825–841. [Google Scholar] [CrossRef]
- Agioutantis, P.; Koumandou, V. Bioenergetic diversity of the human gut microbiome. Meta. Gene. 2018, 16, 10–14. [Google Scholar] [CrossRef]
- Available online: https://www.scimagojr.com/countryrank.php?category=2726®ion=Western%20Europe&order=h&ord=desc&area=1300 (accessed on 28 January 2025).
- Available online: https://www.scimagojr.com/comparecountries.php?ids[]=gr&area=2400&area=2700 (accessed on 28 January 2025).
- Flandroy, L.; Poutahidis, T.; Berg, G.; Clarke, G.; Dao, M.C.; Decaestecker, E.; Frman, E.; Haahtela, T.; Massart, S.; Plovier, H.; et al. The impact of human activities and lifestyles on the interlinked microbiota and health of humans and of ecosystems. Sci. Total Environ. 2018, 627, 1018–1038. [Google Scholar] [CrossRef]
- Koutsokostas, C.; Merkouris, E.; Goulas, A.; Aidinopoulou, K.; Sini, N.; Dimaras, T.; Tsiptsios, D.; Mueller, C.; Nystazaki, M.; Tsakmakis, K. Gut microbes associated with neurodegenerative disorders: A comprehensive review of the literature. Microorganisms 2024, 12, 1735. [Google Scholar] [CrossRef] [PubMed]
- Tsiavos, A.; Antza, C.; Trakatelli, C.; Kotsis, V. The microbial perspective: A systematic literature review on hypertension and gut microbiota. Nutrients 2024, 16, 3698. [Google Scholar] [CrossRef] [PubMed]
- Rozani, S.; Lykoudis, P.M. The impact of intestinal and mammary microbiomes on breast cancer development: A review on the microbiota and oestrobolome roles in tumour microenvironments. Am. J. Surg. 2024, 237, 115795. [Google Scholar] [CrossRef]
- Bamicha, V.; Pergantis, P.; Drigas, A. The effect of gut microbiome, neurotransmitters, and digital insights in autism. Appl. Microbiol. 2024, 4, 1677–1701. [Google Scholar] [CrossRef]
- Kebede, V.; Ravizza, T.; Balosso, S.; Di Sapia, R.; Canali, L.; Soldi, S.; Galletti, S.; Papazlatani, C.; Karas, P.A.; Vasileiadis, S.; et al. Early treatment with rifaximin during epileptogenesis reverses gut alterations and reduces seizure duration in a mouse model of acquired epilepsy. Brain Behav. Immun. 2024, 119, 363–380. [Google Scholar] [CrossRef]
- Richards, E.M.; Pepine, C.J.; Raizada, M.K.; Kim, S. The gut, its microbiome, and hypertension. Curr. Hypertens. Rep. 2017, 19, 36. [Google Scholar] [CrossRef]
- Ferreira-Halder, C.V.; Faria, A.V.S.; Andrade, S.S. Action and function of Faecalibacterium prausnitzii in health and disease. Best Pract. Res. Clin. Gastroenterol. 2017, 31, 643–648. [Google Scholar] [CrossRef]
- Seneff, S.; Kyriakopoulos, A.M. Cancer, deuterium, and gut microbes: A novel perspective. Endocr. Metab. Sci. 2025, 17, 100215. [Google Scholar] [CrossRef]
- Nobili, A.; Pane, M.; Skvortsova, M.; Salem, M.B.; Morgenthaler, S.; Jamieson, E.; Di Stefano, M.; Barthelou, E.; Mamalaki, E.; Ramos-Garcia, V.; et al. Innovative biomarkers for obesity and type 1 diabetes based on Bifidobacterium and metabolomic profiling. Microorganisms 2024, 12, 931. [Google Scholar] [CrossRef]
- Stefanaki, C.; Rozou, P.; Efthymiou, V.; Xinias, I.; Mastorakos, G.; Bacopoulou, F.; Papagianni, M. Impact of probiotics on the glycemic control of pediatric and adolescent individuals with type 1 diabetes: A systematic review and meta-analysis. Nutrients 2024, 16, 2629. [Google Scholar] [CrossRef]
- Vallianou, N.G.; Stratigou, T.; Tsagarakis, S. Microbiome and diabetes: Where are we now? Diabetes Res. Clin. Pract. 2018, 146, 111–118. [Google Scholar]
- Galaris, A.; Fanidis, D.; Stylianaki, E.-A.; Harokopos, V.; Kalantzi, A.S.; Moulos, P.; Dimas, A.S.; Hatsis, P.; Aidinis, V. Obesity reshapes the microbial population structure along the gut-liver-lung axis in mice. Biomedicines 2022, 10, 494. [Google Scholar] [CrossRef] [PubMed]
- Vallianou, N.; Dalamaga, M.; Stratigou, T.; Karampela, I.; Tsigalou, C. Do antibiotics cause obesity through long-term alterations in the gut microbiome? A review of current evidence. Curr. Obes. Rep. 2021, 10, 244–262. [Google Scholar] [PubMed]
- Vetrani, C.; Di Nisio, A.; Paschou, S.A.; Barrea, L.; Muscogiuri, G.; Graziado, C.; Savastano, S.; Colao, A. From gut microbiota through low-grade inflammation to obesity: Key players and potential targets. Nutrients 2022, 14, 2103. [Google Scholar] [CrossRef]
- Wei, L.Q.; Cheong, I.H.; Yang, G.H.; Li, G.X.; Kozlakidis, Z.; Ding, L.; Liu, N.N.; Wang, H. The application of high-throughput technologies for the study of microbiome and cancer. Front. Genet. 2021, 12, 699793. [Google Scholar] [CrossRef]
- Faktor, J.; Henek, T.; Hernychova, L.; Singh, A.; Vojtesek, B.; Polom, J.; Bhatia, R.; Cuschieri, K.; Cruickshark, M.; Gurumurthy, M.; et al. Metaproteomic analysis from cervical biopsies and cytologies identifies proteinaceous biomarkers representing both human and microbial species. Talanta 2024, 278, 126460. [Google Scholar] [CrossRef]
- Vakirlis, N.; Kupczok, A. Large-scale investigation of species-specific orphan genes in the human gut microbiome elucidates their evolutionary origins. Genome Res. 2024, 34, 888–903. [Google Scholar] [PubMed]
- Cruz, A.K.; Alves, M.A.; Andresson, T.; Bayless, A.L.; Bloodsworth, K.J.; Bowden, J.A.; Bullock, A.; Burnet, C.M.; Neto, F.C.; Choy, A.; et al. Multiplatform metabolomic interlaboratory study of a whole human stool candidate reference material from omnivore and vegan donors. Metabolomics 2024, 20, 125. [Google Scholar]
- Gioula, G.; Melidou, A.; Siasios, P.; Minti, F.; Malisiovas, N. 16S rRNA deep sequencing for the characterization of healthy human pharyngeal microbiome. Hippokratia 2018, 22, 29–36. [Google Scholar]
- Tsiouris, C.G.; Kelesi, M.; Vasilopoulos, G.; Kalemikerakis, I.; Papageorgiou, E.G. The efficacy of probiotics as pharmacological treatment of cutaneous wounds: Meta-analysis of animal studies. Eur. J. Pharm. Sci. 2017, 104, 230–239. [Google Scholar]
- Konstantinidis, T.; Tsigalou, C.; Karvelas, A.; Stavropoulou, E.; Voidarou, C.; Bezirtzoglou, E. Effects of antibiotics upon the gut microbiome: A review of the literature. Biomedicines 2020, 8, 502. [Google Scholar] [CrossRef] [PubMed]
- Nash, V.; Ranadheera, C.S.; Georgousopoulou, E.N.; Mellor, D.D.; Panagiotakos, D.P.; McKune, A.J.; Kellet, J.; Naumovski, N. The effects of grape and red wine polyphenols on gut microbiota—A systematic review. Food Res. Int. 2018, 113, 277–287. [Google Scholar]
- Baliou, S.; Adamaki, M.; Spandidos, D.A.; Kyriakopoulos, A.M.; Christodoulou, I.; Zoumpourlis, V. The microbiome, its molecular mechanisms and its potential as a therapeutic strategy against colorectal carcinogenesis: A review. World Acad. Sci. J. 2019, 1, 3–19. [Google Scholar] [CrossRef]
- Tatsika, S.; Karamanoli, K.; Karayanni, H.; Genitsaris, S. Metagenomic characterization of bacterial communities on ready-to-eat vegetables and effects of household washing on their diversity and composition. Pathogens 2019, 8, 37. [Google Scholar] [CrossRef] [PubMed]
- Stavridou, E.; Giannakis, I.; Karamichali, I.; Kamou, N.N.; Lagiotis, G.; Madesis, P.; Emmanouil, C.; Kungolos, A.; Nianiou-Obeitat, I.; Lagopodi, A.L. Biosolid-amended soil enhances defense responses in tomato based on metagenomic profile and expression of pathogenesis-related genes. Plants 2021, 10, 2789. [Google Scholar] [CrossRef] [PubMed]
- Papadimitriou, K.; Anastasiou, R.; Georgalaki, M.; Bounenni, R.; Paximadaki, A.; Charmpi, C.; Alaxandraski, V.; Kazou, M.; Tsakalidou, E. Comparison of the microbiome of artisanal homemade and industrial Feta cheese through amplicon sequencing and shotgun metagenomics. Microorganisms 2022, 10, 1073. [Google Scholar] [CrossRef]
- Karapantzou, I.; Mitropoulou, G.; Prapa, I.; Papanikolaou, D.; Charovas, V.; Kourkoutas, Y. Physicochemical changes and microbiome associations during vermicomposting of winery waste. Sustainability 2023, 15, 7484. [Google Scholar] [CrossRef]
- Samieri, C.; Yassine, H.N.; Melo van Lent, D.; Lefevre-Arbogast, S.; van de Rest, O.; Bowman, G.L.; Scarmeas, N. Personalized nutrition for dementia prevention. Alzheimer’s Dement. 2022, 18, 1424–1437. [Google Scholar] [CrossRef]
- Vinarov, Z.; Abdallah, M.; Agundez, J.A.G.; Allegaetr, K.; Basit, A.W.; Braeckmans, M.; Ceaulemans, J.; Corseti, M.; Griffin, B.T.; Grimm, M.; et al. Impact of gastrointestinal tract variability on oral drug absorption and pharmacokinetics: An UNGAP review. Eur. J. Pharm. Sci. 2021, 162, 105812. [Google Scholar]
- Grammatikopoulou, M.G.; Nigdelis, M.P.; Goulis, D.G. Weight gain in midlife women: Understanding drivers and underlying mechanisms. Curr. Opin. Endocr. Metab. Res. 2022, 27, 100406. [Google Scholar] [CrossRef]
- Sakkas, H.; Bozidis, P.; Touzios, C.; Kolios, D.; Athanasiou, G.; Athanasopoulou, E.; Gerou, I.; Gartzonika, C. Nutritional status and the influence of the vegan diet on the gut microbiota and human health. Medicina 2020, 56, 88. [Google Scholar] [CrossRef] [PubMed]
- Pessach, I.; Tsirigotis, P.; Nagler, A. The gastrointestinal tract: Properties and role in allogeneic hematopoietic stem cell transplantation. Expert Rev. Hematol. 2017, 10, 315–326. [Google Scholar] [CrossRef]
- Hamer, M.; Stamatakis, E. Metabolically healthy obesity and risk of all-cause and cardiovascular disease mortality. J. Clin. Endocrinol. Metab. 2012, 97, 2482–2488. [Google Scholar] [PubMed]
- Handakas, E.; Keski-Rahkonen, P.; Chatzi, L.; Alfano, R.; Roumelioti, T.; Plusquin, M.; Maitre, L.; Ruchiardi, L.; Brescianini, S.; Scalbert, A.; et al. Cord blood metabolic signatures predictive of childhood overweight and rapid growth. Int. J. Obes. 2021, 45, 2252–2260. [Google Scholar] [CrossRef]
- Tanaka, T.; Talegawkar, S.A.; Jin, Y.; Candia, J.; Qu, T.; Moaddel, R.; Simonsick, E.M.; Ferrucci, L. Metabolomic profile of different dietary patterns and their association with frailty index in community-dwelling older men and women. Nutrients 2022, 14, 2237. [Google Scholar] [CrossRef] [PubMed]
- Papandreou, C.; Papagiannopoulos, C.; Koutsonida, M.; Kanellopoulou, A.; Markozannes, G.; Polychronidis, G.; Tzakos, A.G.; Fragkiadakis, G.A.; Evangelou, E.; Ntzani, E.; et al. Mediterranean diet-related metabolite profiles and cognitive performance. Clin. Nutr. 2023, 42, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Navajas-Porras, B.; Pérez-Burillo, S.; Hinojosa-Nogueira, D.; Douros, K.; Pastoriza, S.; Rufian-Henares, J.A. The gut microbiota of obese children releases lower antioxidant capacity from food than that of lean children. Nutrients 2022, 14, 2829. [Google Scholar] [CrossRef]
- Yang, J.J.; Shu, X.O.; Herrington, D.M.; Moore, S.C.; Meyer, K.A.; Ose, J.; Menni, C.; Palmer, N.D.; Eliassen, H.; Harada, S.; et al. Circulating trimethylamine N-oxide in association with diet and cardiometabolic biomarkers: An international pooled analysis. Am. J. Clin. Nutr. 2021, 113, 1145–1156. [Google Scholar] [CrossRef]
- Andrews, R.R.; Anderson, K.R.; Fry, J.L. Sex-specific variation in metabolic responses to diet. Nutrients 2024, 16, 2921. [Google Scholar] [CrossRef]
- Tsoukalas, D.; Sarandi, E.; Fragoulakis, V.; Xenidis, S.; Mhliopoulou, M.; Charta, M.; Paramera, E.; Papakonstantinou, E.; Tsatsakis, A. Metabolomics-based treatment for chronic diseases: Results from a multidisciplinary clinical study. BMJ Nutr. Prev. Health 2024, 7, e000883. [Google Scholar] [CrossRef]
- Ye, C.; Li, Z.; Yuan, L.; Wu, K.; Zhu, C. Association between gut microbiota and biological aging: A two-sample Mendelian randomization study. Microorganisms 2024, 12, 370. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Tilves, C.; Kim, H.; Tanaka, T.; Spira, A.P.; Chia, C.W.; Talegawkar, S.A.; Ferrucci, L.; Mueller, N.T. Plant-based diets and the gut microbiome: Findings from the Baltimore Longitudinal Study of Aging. Am. J. Clin. Nutr. 2024, 119, 628–638. [Google Scholar] [CrossRef] [PubMed]
- Olalekan, S.O.; Bakare, O.O.; Osonuga, I.O.; Faponle, A.S.; Adegbesan, B.O.; Ezima, E.N. Gut microbiota-derived metabolites: Implications for metabolic syndrome and therapeutic interventions. Egypt J. Intern. Med. 2024, 36, 72. [Google Scholar] [CrossRef]
- Sheng, L.; Jena, P.K.; Hu, Y.; Wan, Y.Y. Age-specific microbiota in altering host inflammatory and metabolic signaling as well as metabolome based on sex. Hepatobiliary Surg. Nutr. 2021, 10, 31–48. [Google Scholar] [CrossRef]
- Lagoumintzis, G.; Patrinos, G.P. Triangulating nutrigenomics, metabolomics, and microbiomics toward personalized nutrition and healthy living. Hum. Genom. 2023, 17, 109. [Google Scholar] [CrossRef]







| Conceptual Framework | Definition |
|---|---|
| Ecology |
|
| Organism–host interactions |
|
| Genomic methods |
|
| Combinative definitions |
|
| Title | Authors | Source | Year | Citations |
|---|---|---|---|---|
| Management of Helicobacter pylori infection-the Maastricht V/Florence consensus report | Malfertheier P. et al. | Gut, 66(1), pp. 6–30 | 2017 | 2338 |
| Microbiome definition re-visited: old concepts and new challenges | Berg G. et al. | Microbiome, 8(1), 103 | 2020 | 1263 |
| Obesity and cancer risk: Emerging biological mechanisms and perspectives | Avgerinos K.I. et al. | Metabolism: Clinical and Experimental, 92, pp. 121–135 | 2019 | 921 |
| Agriculture and food animals as a source of antimicrobial-resistant bacteria | Economou V. and Gousia P. | Infection and Drug Resistance, 8, pp. 49–61 | 2015 | 549 |
| Gut-Microbiota-Brain Axis and Its Effect on Neuropsychiatric Disorders with Suspected Immune Dysregulation | Petra A.I. et al. | Clinical Therapeutics, 37(5), pp. 984–995 | 2015 | 467 |
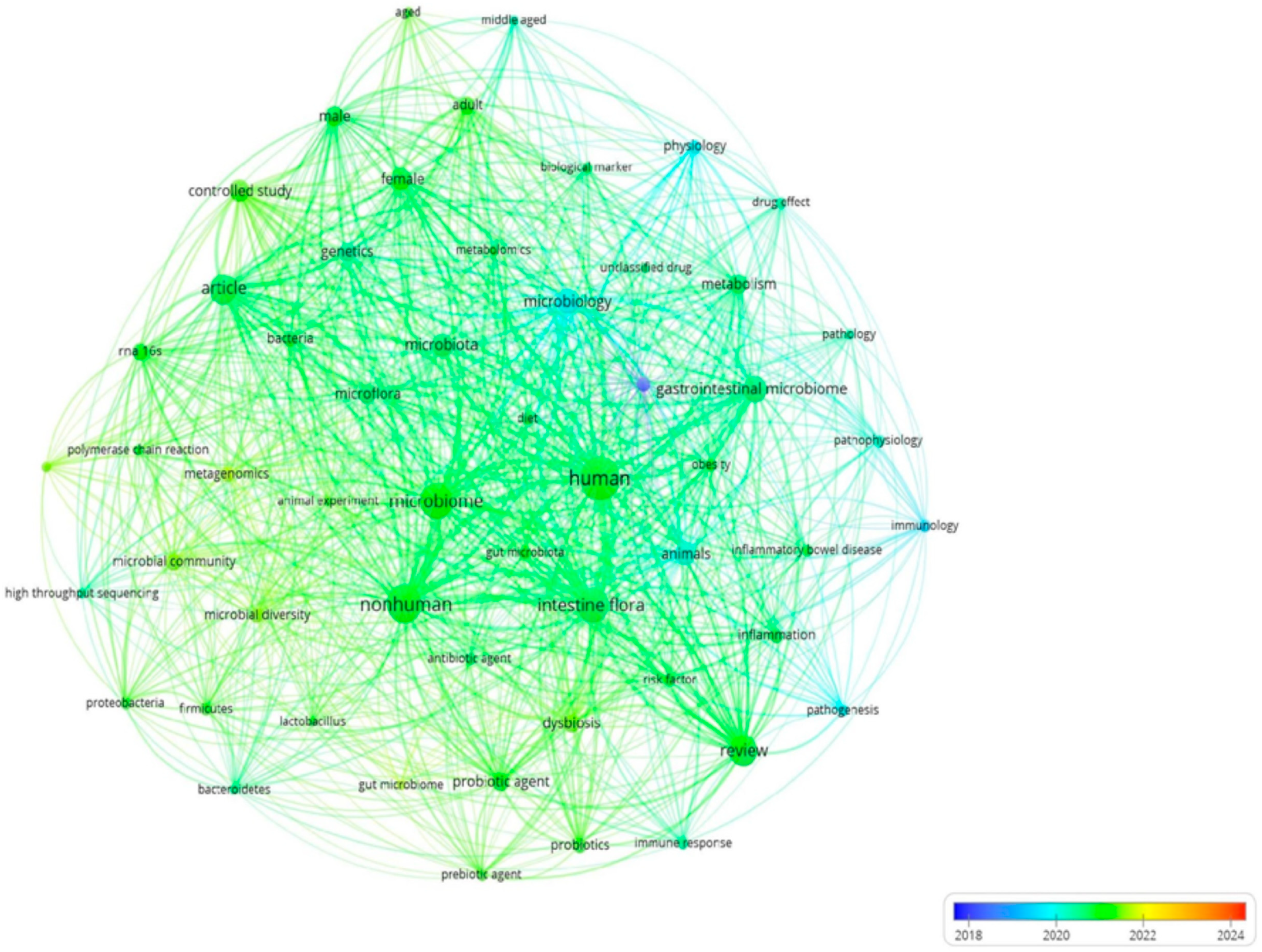
| Cluster Identification | Keywords |
|---|---|
| Red | human, intestine flora, review, gastrointestinal microbiome, microbiology, animals, metabolism, dysbiosis, probiotic agent, priority journal, probiotics, physiology, inflammation, obesity, gut microbiota, risk factor, immunology, immune response, pathogenesis, prebiotic agent, inflammatory bowel disease, pathophysiology, antibiotic agent, drug effect, pathology, gut microbiome, unclassified drug |
| Green | nonhuman, microbiome, article, microbiota, controlled study, microflora, genetics, microbial community, rna 16s, bacteria, microbial diversity, metagenomics, Firmicutes, Proteobacteria, high-throughput sequencing, lactobacillus, animal experiment, polymerase chain reaction, Bacteroides, DNA extraction |
| Blue | female, male, adult, biological marker, diet, middle-aged, metabolomics, aged |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stefanis, C.; Tsigalou, C.; Bezirtzoglou, I.; Voidarou, C.; Stavropoulou, E. Microbiome Research in Greece: A Comprehensive Bibliometric Study. Microorganisms 2025, 13, 725. https://doi.org/10.3390/microorganisms13040725
Stefanis C, Tsigalou C, Bezirtzoglou I, Voidarou C, Stavropoulou E. Microbiome Research in Greece: A Comprehensive Bibliometric Study. Microorganisms. 2025; 13(4):725. https://doi.org/10.3390/microorganisms13040725
Chicago/Turabian StyleStefanis, Christos, Christina Tsigalou, Ioanna Bezirtzoglou, Chrysoula (Chrysa) Voidarou, and Elisavet Stavropoulou. 2025. "Microbiome Research in Greece: A Comprehensive Bibliometric Study" Microorganisms 13, no. 4: 725. https://doi.org/10.3390/microorganisms13040725
APA StyleStefanis, C., Tsigalou, C., Bezirtzoglou, I., Voidarou, C., & Stavropoulou, E. (2025). Microbiome Research in Greece: A Comprehensive Bibliometric Study. Microorganisms, 13(4), 725. https://doi.org/10.3390/microorganisms13040725






