Inhibitory Potential of Cannabis Biomass Extracts on Livestock-Associated Staphylococcal and Streptococcal Pathogens
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of Plant Extracts
2.2.2. Determination of Cannabinoid Contents in the Extracts
2.2.3. Bacterial Strains and Culture Media
2.2.4. Antibacterial Assay
Data Analysis
3. Results
3.1. Minimum Inhibitory Concetration of Cannabis Extracts
3.2. Contents of Cannabinoids in Extracts
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- HealthforAnimals. Animal Health and Sustainability: A Global Data Analysis; Oxford Analytica: Oxford, UK, 2023; pp. 1–54. Available online: https://healthforanimals.org/resources/publications/publications/full-report-animal-health-and-sustainability-a-global-data-analysis/ (accessed on 8 December 2024).
- Mesquita, A.A.; Rocha, C.M.B.M.; Bruhn, F.R.P.; Custódio, D.A.C.; Braz, M.S.; Pinto, S.M.; Silva, D.B.; Costa, G.M. Staphylococcus aureus and Streptococcus agalactiae: Prevalence, resistance to antimicrobials, and their relationship with the milk quality of dairy cattle herds in Minas Gerais state, Brazil. Pesqui. Vet. Bras. 2019, 39, 308–316. [Google Scholar] [CrossRef]
- Taponen, S.; Liski, E.; Heikkilä, A.M.; Pyörälä, S. Factors associated with intramammary infection in dairy cows caused by coagulase-negative staphylococci, Staphylococcus aureus, Streptococcus uberis, Streptococcus dysgalactiae, Corynebacterium bovis, or Escherichia coli. J. Dairy Sci. 2017, 100, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Saro, J.; Stádník, L.; Bláhová, P.; Huguet, S.; Brožová, H.; Ducháček, J. A decision support system based on disease scoring enables dairy farmers to proactively improve herd health. Czech J. Anim. Sci. 2024, 69, 165–177. [Google Scholar] [CrossRef]
- Cheng, W.N.; Han, S.G. Bovine mastitis: Risk factors, therapeutic strategies, and alternative treatments—A review. Asian-Australas. J. Anim. Sci. 2020, 33, 1699–1713. [Google Scholar] [CrossRef] [PubMed]
- Guy, R.; Coelho, J.; Blakey, E.; Broughton, K.; Lamagni, T. Laboratory Surveillance of Pyogenic and Non-Pyogenic Streptococcal Bacteraemia in England (2022); UK Health Security Agency: London, UK, 2023; pp. 1–30. Available online: https://www.gov.uk/government/publications/pyogenic-and-non-pyogenic-streptococcal-bacteraemia-annual-data-from-voluntary-surveillance (accessed on 8 December 2024).
- Alves-Barroco, C.; Roma-Rodrigues, C.; Raposo, L.R.; Brás, C.; Diniz, M.; Caço, J.; Costa, P.M.; Santos-Sanches, I.; Fernandes, A.R. Streptococcus dysgalactiae subsp. dysgalactiae isolated from milk of the bovine udder as emerging pathogens: In vitro and in vivo infection of human cells and zebrafish as biological models. MicrobiologyOpen 2019, 8, e00623. [Google Scholar] [CrossRef]
- Rao, S.; Linke, L.; Magnuson, R.; Jauch, L.; Hyatt, D.R. Antimicrobial resistance and genetic diversity of Staphylococcus aureus collected from livestock, poultry and humans. One Health 2022, 15, 100407. [Google Scholar] [CrossRef] [PubMed]
- Vaou, N.; Stavropoulou, E.; Voidarou, C.; Tsigalou, C.; Bezirtzoglou, E. Towards Advances in Medicinal Plant Antimicrobial Activity: A Review Study on Challenges and Future Perspectives. Microorganisms 2021, 9, 2041. [Google Scholar] [CrossRef] [PubMed]
- Cakir, M.; Karatas, T.; Yildirim, S. Protective effects of green tea (Camellia sinensis) extract against cypermethrin-induced neurotoxicity in rainbow trout (Oncorhynchus mykiss) brain tissues. Czech J. Anim. Sci. 2024, 69, 29–37. [Google Scholar] [CrossRef]
- Sionov, R.V.; Steinberg, D. Anti-Microbial Activity of Phytocannabinoids and Endocannabinoids in the Light of Their Physiological and Pathophysiological Roles. Biomedicines 2022, 10, 631. [Google Scholar] [CrossRef]
- Lanzoni, D.; Skrivanova, E.; Pinotti, L.; Rebucci, R.; Baldi, A.; Giromini, C. Review: Nutritional aspects of hemp-based products and their effects on health and performance of monogastric animals. Animal 2024, 18, 101058. [Google Scholar] [CrossRef] [PubMed]
- Bonini, S.A.; Premoli, M.; Tambaro, S.; Kumar, A.; Maccarinelli, G.; Memo, M.; Mastinu, A. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. J. Ethnopharmacol. 2018, 227, 300–315. [Google Scholar] [CrossRef] [PubMed]
- Alfei, S.; Schito, G.C.; Schito, A.M. Synthetic Pathways to Non-Psychotropic Phytocannabinoids as Promising Molecules to Develop Novel Antibiotics: A Review. Pharmaceutics 2023, 15, 1889. [Google Scholar] [CrossRef]
- Czauderna, M.; Taubner, T.; Wojtak, W. Comparative Study of Gas and Liquid Chromatography Methods for the Determination of Underivatised Neutral and Acidic Cannabinoids and Cholesterol. Molecules 2024, 29, 2165. [Google Scholar] [CrossRef] [PubMed]
- Rondevaldova, J.; Hummelova, J.; Tauchen, J.; Kokoska, L. In Vitro Antistaphylococcal Synergistic Effect of Isoflavone Metabolite Demethyltexasin with Amoxicillin and Oxacillin. Microb. Drug Resist. 2018, 24, 24–29. [Google Scholar] [CrossRef] [PubMed]
- CLSI standard M07; Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically. 11th ed. Clinical Laboratory Standards Institute: Wayne, PA, USA, 2018.
- Cos, P.; Vlietinck, A.J.; Berghe, D.V.; Maes, L. Anti-infective potential of natural products: How to develop a stronger in vitro ‘proof-of-concept’. J. Ethnopharmacol. 2006, 106, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Ali, E.M.M.; Almagboul, A.Z.I.; Khogali, S.M.E.; Gergeir, U.M.A. Antimicrobial activity of Cannabis sativa L. Chin. Med. 2012, 3, 61–64. [Google Scholar] [CrossRef]
- Kaur, S.; Sharma, C.; Chaudhry, S.; Aman, R. Antimicrobial Potential of Three Common Weeds of Kurukshetra: An in vitro Study. Res. J. Microbiol. 2015, 10, 280. [Google Scholar] [CrossRef]
- Giselle, F.; Azucena, I.; Dalila, O.; Florencia, F.; Facundo, R.; Giulia, M.; Sandra, F.; Maggi, M.; Ramirez, C.L. Antibacterial activity of cannabis (Cannabis sativa L.) female inflorescence and root extract against Paenibacillus larvae, causal agent of American foulbrood. Biocatal. Agric. Biotechnol. 2023, 47, 102575. [Google Scholar] [CrossRef]
- Skala, T.; Kahánková, Z.; Tauchen, J.; Janatová, A.; Klouček, P.; Hubka, V.; Fraňková, A. Medical cannabis dimethyl ether, ethanol and butane extracts inhibit the in vitro growth of bacteria and dermatophytes causing common skin diseases. Front. Microbiol. 2022, 13, 953092. [Google Scholar] [CrossRef]
- EFSA Panel on Additives and Products or Substances used in Animal Feed. Scientific Opinion on the safety of hemp (Cannabis genus) for use as animal feed (FEEDAP). EFSA J. 2011, 9, 2011. [Google Scholar] [CrossRef]
- Aqawi, M.; Sionov, R.V.; Gallily, R.; Friedman, M.; Steinberg, D. Anti-Bacterial Properties of Cannabigerol Toward Streptococcus mutans. Front. Microbiol. 2021, 12, 656471. [Google Scholar] [CrossRef] [PubMed]
- van Klingeren, B.; ten Ham, M. Antibacterial activity of Δ9-tetrahydrocannabinol and cannabidiol. Antonie van Leeuwenhoek 1976, 42, 9–12. [Google Scholar] [CrossRef]
- Blaskovich, M.A.T.; Kavanagh, A.M.; Elliott, A.G.; Zhang, B.; Ramu, S.; Amado, M.; Lowe, G.J.; Hinton, A.O.; Pham, D.M.T.; Zuegg, J.; et al. The antimicrobial potential of cannabidiol. Commun. Biol. 2021, 4, 7. [Google Scholar] [CrossRef]
- Glivar, T.; Eržen, J.; Kreft, S.; Zagožen, M.; Čerenak, A.; Čeh, B.; Tavčar Benković, E. Cannabinoid content in industrial hemp (Cannabis sativa L.) varieties grown in Slovenia. Ind. Crops Prod. 2020, 145, 112082. [Google Scholar] [CrossRef]
| Bacterium | Strain | Cultivars of Cannabis Sativa/MIC (µg/mL) | ||||
|---|---|---|---|---|---|---|
| Bialobrzeskie | Felina 32 | Futura 75 | Mixed | Santhica 27 | ||
| S. aureus | ATCC 29213 | 512 | 1024 | 1024 | 512 | 256 |
| ATCC 43300 | 256 | 1024 | 1024 | 512 | 256 | |
| SA1 | 256 | 512 | 512 | 256 | 128 | |
| SA3 | 256 | 512 | 512 | 256 | 128 | |
| Str. agalactiae | DSM 2134 | 128 | 256 | 256 | 128 | 128 |
| Str. dysgalactiae | DSM 20662 | 1024 | 2048 | 2048 | 1024 | 1024 |
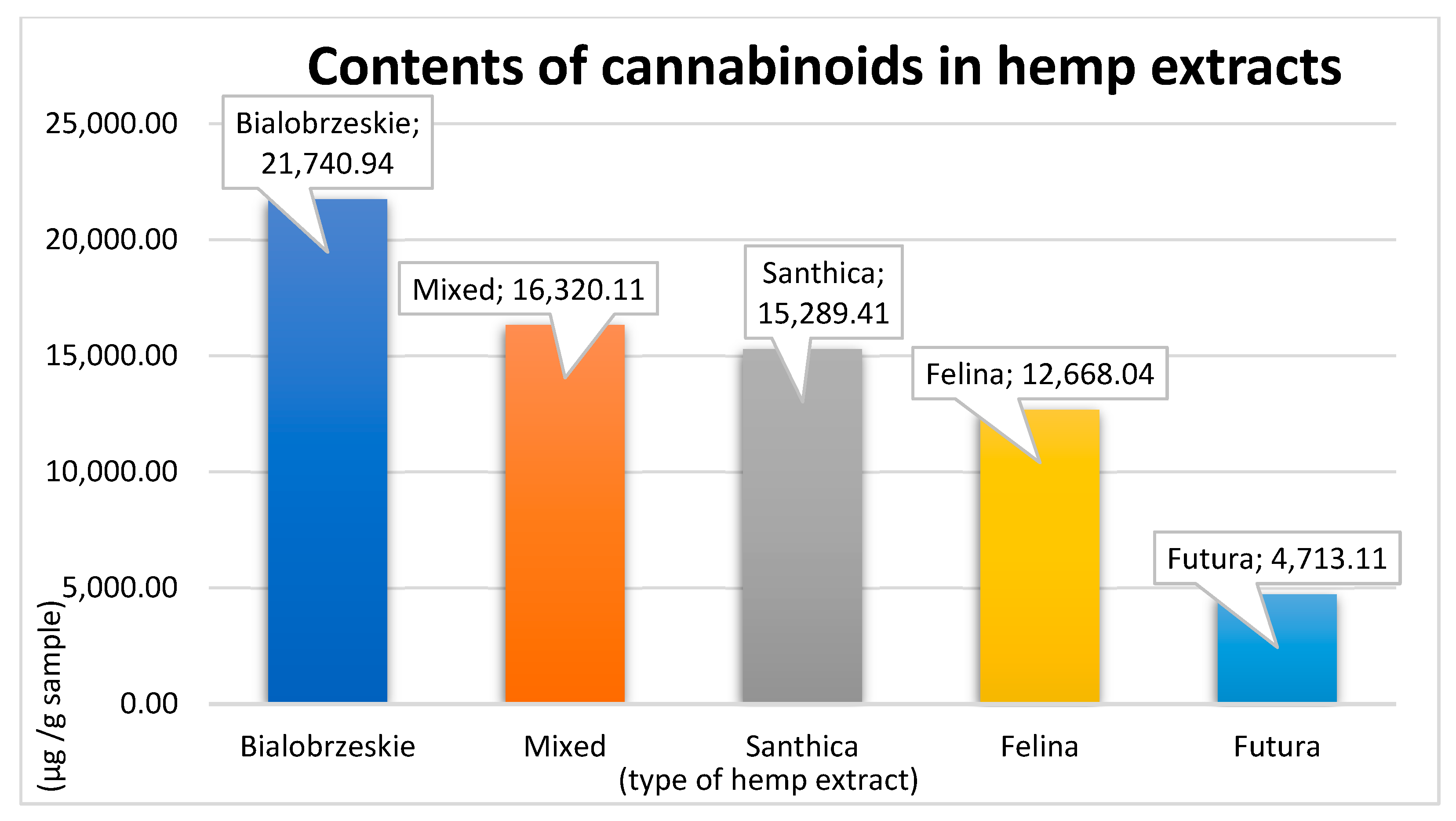
| Observed Cannabinoids | Contents of Cannabinoids in Extracts (µg/g Sample) | ||||
|---|---|---|---|---|---|
| Bialobrzeskie | Felina 32 | Futura 75 | Mixed | Santhica 27 | |
| CBDVA | 360.58 | 144.94 | 63.82 | 258.58 | ND |
| CBDV | ND | 8.65 | 14.90 | ND | ND |
| CBDA | 15,208.79 | 7404.60 | 2776.69 | 10,206.44 | 2650.84 |
| CBGA | 471.21 | 2727.43 | 213.81 | 2215.25 | 10,450.78 |
| CBG | 89.78 | 269.63 | 17.12 | 232.44 | 1163.19 |
| CBD | 2839.14 | 521.72 | 1257.34 | 1590.11 | 370.69 |
| THCV | ND | ND | ND | ND | ND |
| THCVA | ND | ND | ND | ND | ND |
| CBN | ND | ND | 15.60 | ND | ND |
| Δ9-THC | 513.27 | ND | 40.61 | 261.37 | 59.36 |
| Δ8-THC | ND | ND | ND | ND | ND |
| CBC | ND | 301.71 | 105.28 | ND | ND |
| THCA | 1350.45 | 314.58 | 53.32 | 746.26 | 194.74 |
| CBCA | 907.73 | 974.79 | 154.62 | 809.65 | 399.81 |
| Total value | 21,740.94 | 12,668.04 | 4713.11 | 16,320.11 | 15,289.41 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paulová, T.; Malíková, L.; Lanzoni, D.; Taubner, T.; Malík, M.; Houdková, M.; Pěchoučková, E. Inhibitory Potential of Cannabis Biomass Extracts on Livestock-Associated Staphylococcal and Streptococcal Pathogens. Microorganisms 2025, 13, 432. https://doi.org/10.3390/microorganisms13020432
Paulová T, Malíková L, Lanzoni D, Taubner T, Malík M, Houdková M, Pěchoučková E. Inhibitory Potential of Cannabis Biomass Extracts on Livestock-Associated Staphylococcal and Streptococcal Pathogens. Microorganisms. 2025; 13(2):432. https://doi.org/10.3390/microorganisms13020432
Chicago/Turabian StylePaulová, Tereza, Lucie Malíková, Davide Lanzoni, Tomáš Taubner, Matěj Malík, Markéta Houdková, and Eva Pěchoučková. 2025. "Inhibitory Potential of Cannabis Biomass Extracts on Livestock-Associated Staphylococcal and Streptococcal Pathogens" Microorganisms 13, no. 2: 432. https://doi.org/10.3390/microorganisms13020432
APA StylePaulová, T., Malíková, L., Lanzoni, D., Taubner, T., Malík, M., Houdková, M., & Pěchoučková, E. (2025). Inhibitory Potential of Cannabis Biomass Extracts on Livestock-Associated Staphylococcal and Streptococcal Pathogens. Microorganisms, 13(2), 432. https://doi.org/10.3390/microorganisms13020432








