Clinically Defined Lymphogranuloma Venereum among US Veterans with Human Immunodeficiency Virus, 2016–2023
Abstract
1. Introduction
2. Materials and Methods
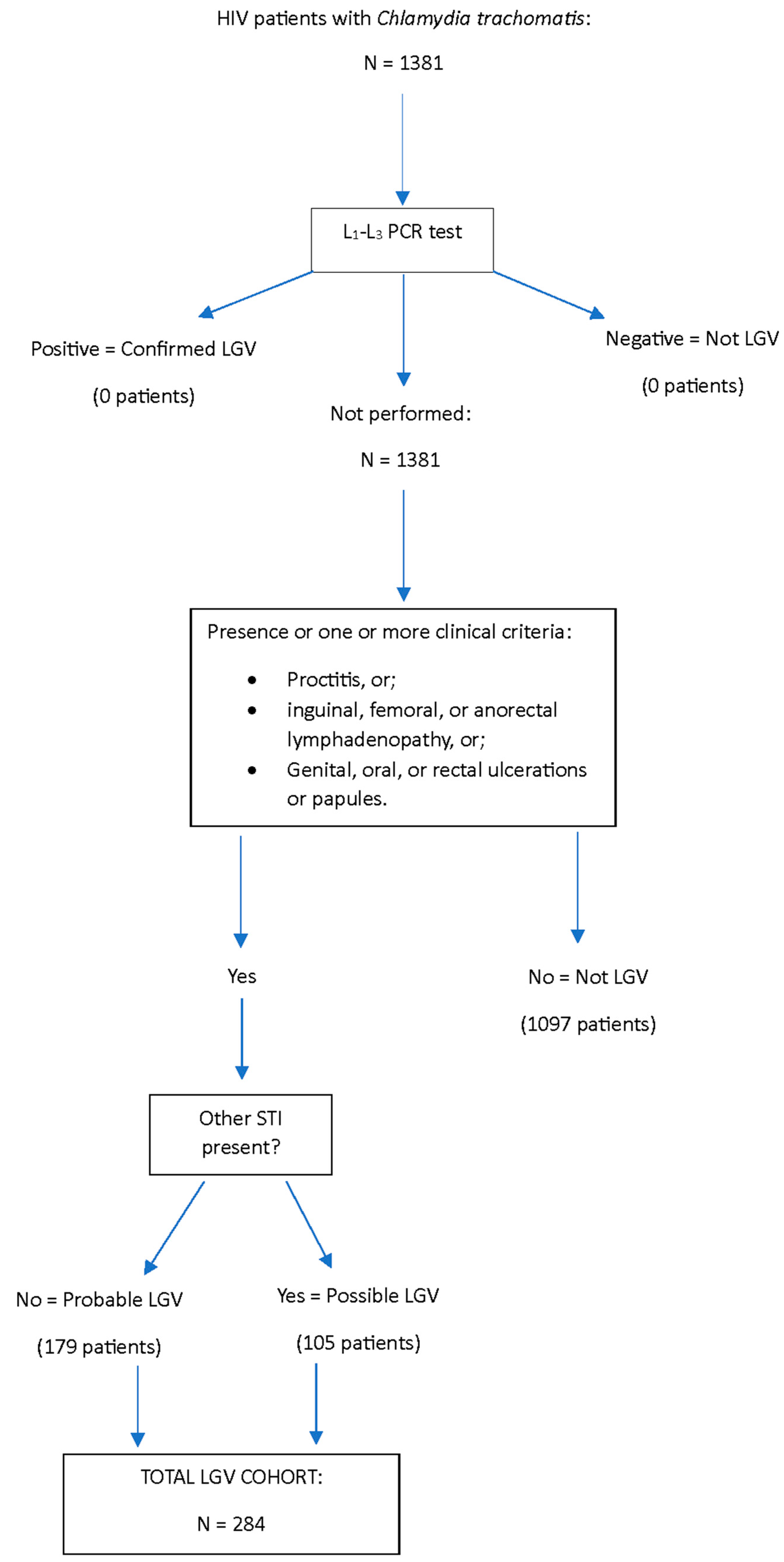
2.1. Data Sources and Study Cohort
- Presence of proctitis as evidenced by rectal bleeding, mucoid, and/or hemopurulent rectal discharge, abdominal pain, anal pain, pain when passing stools, constipation, and/or tenesmus. In later stages, rectal fibrosis and/or strictures and anal fistulae;
- Presence of inguinal, femoral, or anorectal lymphadenopathy or generalized lymphedema of genital organs;
- Genital, oral, or rectal ulcerations or papules.
2.2. Analysis Methods
2.3. Ethics Statement and Disclaimer
3. Results
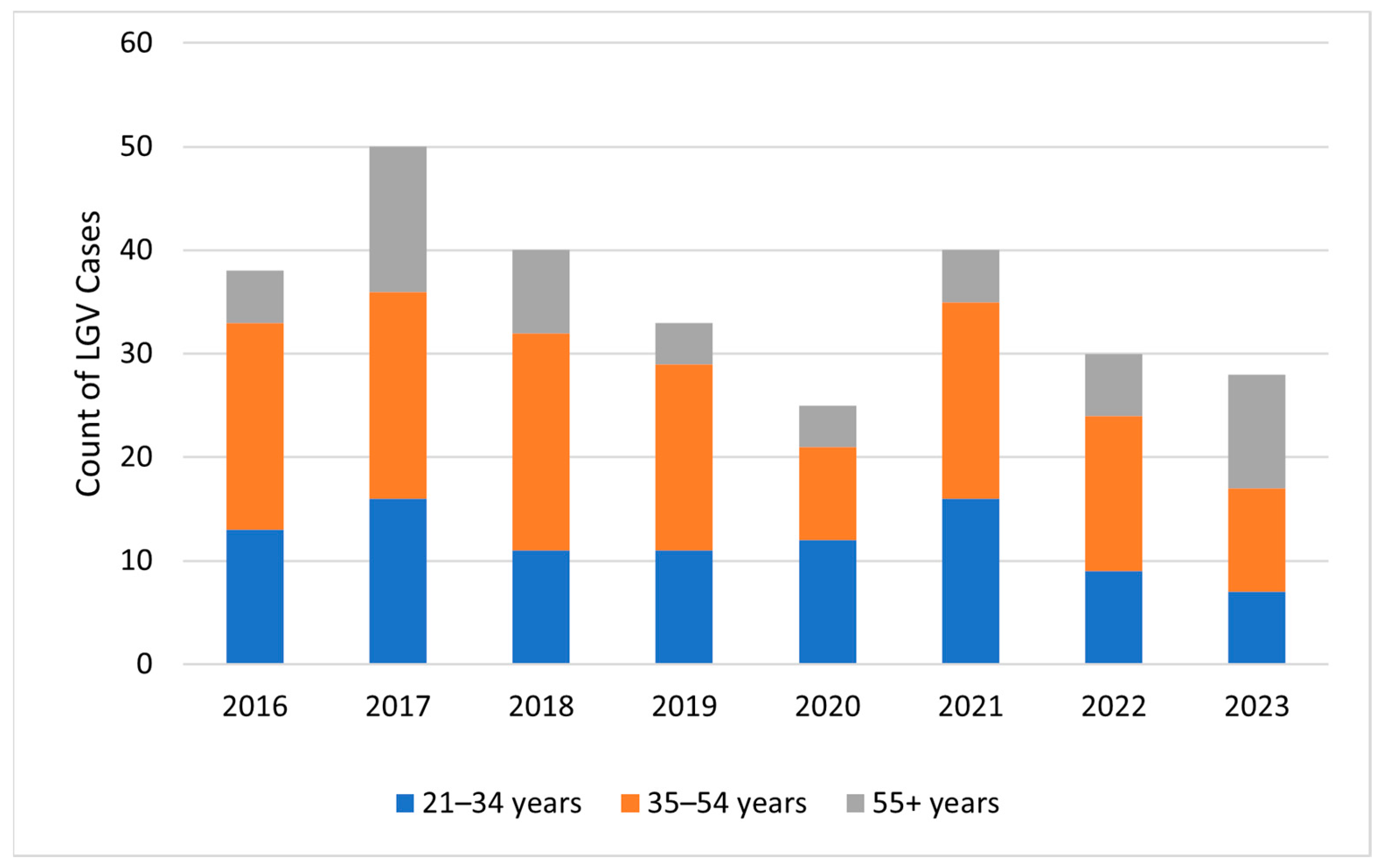
3.1. Characteristics of LGV Clinical Criteria-Positive Cases
3.2. Risk Factors for LGV
3.3. Comparison of Standard vs. Non-Standard Treatment Regimens and Follow-Up for LGV
3.4. Utility of ICD-10-Coded Encounters for Identification of LGV Cases
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Workowski, K.A.; Bachmann, L.H.; Chan, P.A.; Johnston, C.M.; Muzny, C.A.; Park, I.; Reno, H.; Zenilman, J.M.; Bolan, G.A. Sexually Transmitted Infections Treatment Guidelines, 2021. MMWR Recomm. Rep. 2021, 70, 1–187. [Google Scholar] [CrossRef] [PubMed]
- de Voux, A.; Kent, J.B.; Macomber, K.; Krzanowski, K.; Jackson, D.; Starr, T.; Johnson, S.; Richmond, D.; Crane, L.R.; Cohn, J.; et al. Notes from the Field: Cluster of Lymphogranuloma Venereum Cases Among Men Who Have Sex with Men-Michigan, August 2015–April 2016. MMWR Morb. Mortal Wkly. Rep. 2016, 65, 920–921. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rawla, P.; Thandra, K.C.; Limaiem, F. Lymphogranuloma Venereum; StatPearls: Treasure Island, FL, USA, 2024. [Google Scholar]
- Ceovic, R.; Gulin, S.J. Lymphogranuloma venereum: Diagnostic and treatment challenges. Infect. Drug Resist. 2015, 8, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Castro, J.G.; Ponomareva, O.; Alcaide, M.; Rosa-Cunha, I.; Dilanchian, P.; Willis, D. Increase in lymphogranuloma venereum cases in South Florida. J. Int. Assoc. Physicians AIDS Care 2012, 11, 220–222. [Google Scholar] [CrossRef] [PubMed]
- O’Byrne, P.; MacPherson, P.; DeLaplante, S.; Metz, G.; Bourgault, A. Approach to lymphogranuloma venereum. Can. Fam. Physician 2016, 62, 554–558. [Google Scholar] [PubMed]
- Chi, K.H.; de Voux, A.; Morris, M.; Katz, S.S.; Pillay, A.; Danavall, D.; Bowden, K.E.; Gaynor, A.M.; Kersh, E.N. Detection of Lymphogranuloma Venereum-Associated Chlamydia trachomatis L2 Serovars in Remnant Rectal Specimens Collected from 7 US Public Health Laboratories. Sex. Transm. Dis. 2022, 49, e26–e28. [Google Scholar] [CrossRef] [PubMed]
- Childs, T.; Simms, I.; Alexander, S.; Eastick, K.; Hughes, G.; Field, N. Rapid increase in lymphogranuloma venereum in men who have sex with men, United Kingdom, 2003 to September 2015. Eurosurveillance 2015, 20, 30076. [Google Scholar] [CrossRef] [PubMed]
- de Vries, H.J.C.; de Barbeyrac, B.; de Vrieze, N.H.N.; Viset, J.D.; White, J.A.; Vall-Mayans, M.; Unemo, M. 2019 European guideline on the management of lymphogranuloma venereum. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 1821–1828. [Google Scholar] [CrossRef] [PubMed]
- White, J.A. Manifestations and management of lymphogranuloma venereum. Curr. Opin. Infect. Dis. 2009, 22, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Stoner, B.P.; Cohen, S.E. Lymphogranuloma Venereum 2015: Clinical Presentation, Diagnosis, and Treatment. Clin. Infect. Dis. 2015, 61 (Suppl. 8), S865–S873. [Google Scholar] [CrossRef] [PubMed]
- Council of State and Territorial Epidemiologists. Pubic Health Reporting and National Notification for Infection Caused by Chlamydia Trachomatis. Available online: https://cdn.ymaws.com/www.cste.org/resource/resmgr/ps/ps2021/21-ID-06_ChlamydiaLGV.pdf (accessed on 11 June 2024).
- Centers for Disease Control and Prevention. Guidance: Including Information on Lymphogranuloma Venereum in Chlamydia Case Notifications Provided to CDC. Available online: https://www.cdc.gov/std/program/forms/guidance-for-including-lgv-information-on-chlamydia-case-notifications.pdf (accessed on 11 June 2024).
- Sheinman, M.D.; Vinod, J. Lymphogranuloma Venereum Proctocolitis; StatPearls: Treasure Island, FL, USA, 2024. [Google Scholar]
- European Centre for Disease Prevention and Control. Lymphogranuloma Venereum Annual Epidemiological Report for 2022. Available online: https://www.ecdc.europa.eu/sites/default/files/documents/lymphogranuloma-venereum-annual-epidemiological-report-2022_0.pdf (accessed on 11 June 2024).
- Cole, M.J.; Field, N.; Pitt, R.; Amato-Gauci, A.J.; Begovac, J.; French, P.D.; Kese, D.; Klavs, I.; Zidovec Lepej, S.; Pocher, K.; et al. Substantial underdiagnosis of lymphogranuloma venereum in men who have sex with men in Europe: Preliminary findings from a multicentre surveillance pilot. Sex. Transm. Infect. 2020, 96, 137–142. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Veterans Affairs. National Center for Veterans Analysis and Statistics Pocket Cards. Available online: https://www.va.gov/vetdata/pocketcard/index.asp (accessed on 11 June 2024).
- Federal Register. System of Records: “VHA Corporate Data Warehouse-VA” (172VA10P2): National Archives and Records Administration. Available online: https://www.govinfo.gov/content/pkg/FR-2014-01-27/pdf/FR-2014-01-27.pdf (accessed on 11 June 2024).
- U.S. Census Bureau. 2010 Census Regions and Divisions of the United States. Available online: https://www.census.gov/geographies/reference-maps/2010/geo/2010-census-regions-and-divisions-of-the-united-states.html (accessed on 11 June 2024).
- Federal Register. Code of Federal Regulations, Title 38: Pensions, Bonuses, and Veterans’ Relief. Chapter 1. Part 3. Subpart A. §3.2. Available online: https://www.ecfr.gov/current/title-38/chapter-I/part-3#3.2 (accessed on 11 June 2024).
- Schirmer, P.; Sharma, A.; Lucero-Obusan, C.; Oda, G.; Holodniy, M. Trends in Follow-up Testing Among Patients Positive for Chlamydia and Gonorrhea in the Veterans Health Administration, 2013 to 2019. Sex. Transm. Dis. 2023, 50, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Prochazka, M.; Charles, H.; Allen, H.; Cole, M.; Hughes, G.; Sinka, K. Rapid Increase in Lymphogranuloma Venereum among HIV-Negative Men Who Have Sex with Men, England, 2019. Emerg. Infect. Dis. 2021, 27, 2695–2699. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. Men Who Have Sex with Men (MSM). Available online: https://www.cdc.gov/std/treatment-guidelines/msm.htm (accessed on 11 June 2024).
- Kersh, E.N.; Pillay, A.; de Voux, A.; Chen, C. Laboratory Processes for Confirmation of Lymphogranuloma Venereum Infection During a 2015 Investigation of a Cluster of Cases in the United States. Sex. Transm. Dis. 2017, 44, 691–694. [Google Scholar] [CrossRef] [PubMed]
- Bachmann, L.H.; Barbee, L.A.; Chan, P.; Reno, H.; Workowski, K.A.; Hoover, K.; Mermin, J.; Mena, L. CDC Clinical Guidelines on the Use of Doxycycline Postexposure Prophylaxis for Bacterial Sexually Transmitted Infection Prevention, United States, 2024. MMWR Recomm. Rep. 2024, 73, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Im, J.H.; Ko, Y.J.; Huh, K.; Yoon, C.G.; Rhee, C.; Kim, Y.E.; Go, D.S.; Kim, A.; Jung, Y.; et al. Complementing conventional infectious disease surveillance with national health insurance claims data in the Republic of Korea. Sci. Rep. 2019, 9, 8750. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Number (%) |
|---|---|
| Classification of LGV | |
| Probable | 179 (63.0) |
| Possible (other STIs present) | 105 (37.0) |
| Presenting clinical syndrome a | |
| Proctitis | 230 (81.0) |
| Genital, oral, or rectal ulcers | 71 (25.0) |
| Lymphadenopathy (inguinal, femoral, or anorectal) | 66 (23.2) |
| Sample site b | |
| Rectal | 234 (82.0) |
| Urine | 48 (16.9) |
| Pharynx | 12 (4.2) |
| Genital | 3 (1.1) |
| Cervix | 1 (0.4) |
| LGV Clinical Criteria Positive (n = 284) | LGV Clinical Criteria Negative (n = 1097) | ||
|---|---|---|---|
| N (%) | N (%) | p-Value a | |
| Characteristic | |||
| Age Tested (Median (IQR)) | 42 (33–53) | 42 (33–54) | 0.694 b |
| Age Group | |||
| 21–34 years | 96 (33.5) | 340 (31.0) | 0.322 |
| 35–54 years | 132 (46.5) | 491 (44.8) | |
| 55 years or older | 57 (20.1) | 266 (24.2) | |
| Birth Sex | |||
| Male | 284 (100.0) | 1066 (97.2) | 0.004 |
| Female | 0 (0.0) | 31 (2.8) | |
| Sexual Orientation | |||
| Bisexual/MSM/Transgender | 241 (84.9) | 670 (61.1) | <0.001 |
| Heterosexual | 18 (6.3) | 224 (20.4) | |
| Unknown/Not defined | 25 (8.8) | 203 (18.5) | |
| Race/Ethnicity | |||
| Non-Hispanic Black | 157 (55.3) | 613 (55.9) | 0.311 |
| Non-Hispanic White | 87 (30.6) | 281 (25.6) | |
| Hispanic/Latino | 23 (8.1) | 123 (11.2) | |
| Other Race/Ethnicity c | 6 (2.1) | 27 (2.5) | |
| Unknown | 11 (3.9) | 53 (4.8) | |
| Rurality of Residence | |||
| Urban residence | 264 (93.0) | 992 (90.4) | 0.186 |
| Rural residence | 20 (7.0) | 105 (9.6) | |
| Census Region of Testing Facility | |||
| Northeastern | 20 (7.0) | 68 (6.2) | 0.136 d |
| Midwestern | 23 (8.1) | 141 (12.9) | |
| Southern | 154 (54.2) | 599 (54.6) | |
| Western | 87 (30.6) | 288 (26.3) | |
| Puerto Rico | 0 (0.0) | 2 (0.2) | |
| Period of Military Service e | |||
| Persian Gulf War | 226 (79.6) | 845 (77.0) | 0.637 d |
| Vietnam Era | 58 (20.4) | 250 (22.8) | |
| Korean Conflict | 0 (0.0) | 2 (0.2) | |
| Concurrent STI f | |||
| GC positive | 61 (21.5) | 240 (21.9) | 0.885 |
| Syphilis positive | 53 (18.7) | 186 (17.0) | 0.498 |
| Other STI positive g | 12 (4.2) | 19 (1.7) | 0.011 |
| CD4 count, cells/µL h (Median (IQR)) | 627 (454–819) | 642 (447–867) | 0.442 |
| Standard Treatment a (n = 124) | Non-Standard Treatment (n = 160) | ||
|---|---|---|---|
| N (%) | N (%) | p-Value d | |
| Characteristic | |||
| Classification of LGV | |||
| Probable | 90 (72.6) | 89 (55.6) | 0.003 |
| Possible (other STIs present) | 34 (27.4) | 71 (44.4) | |
| Sample Type | |||
| Rectal | 108 (87.1) | 126 (78.8) | 0.067 |
| Nonrectal | 16 (12.9) | 34 (21.3) | |
| Presenting Clinical Syndrome | |||
| Proctitis | 106 (85.5) | 124 (77.5) | 0.089 |
| Genital, oral, or rectal ulcers | 29 (23.4) | 42 (26.3) | 0.581 |
| Lymphadenopathy (inguinal, femoral, or anorectal) | 30 (24.2) | 27 (16.9) | 0.127 |
| Hospitalization | |||
| Yes | 23 (18.5) | 140 (12.5) | 0.158 |
| No | 101 (81.5) | 20 (87.5) | |
| Median Length of Stay (days) | 3 (IQR 2–6) | 2 (IQR 1–4) | 0.162 |
| Time Between CT Testing and Result Completion | |||
| 0–3 days | 59 (47.6) | 87 (54.4) | 0.381 |
| 4–7 days | 57 (46.0) | 67 (41.9) | |
| >7 days | 8 (6.5) | 6 (3.8) | |
| Timing of Treatment Initiation | |||
| Within ≤7 days of + CT test | 111 (89.5) | 146 (91.3) | 0.621 |
| >7 days of + CT test | 13 (10.5) | 14 (8.8) | |
| CT Follow-up Testing Within 3–12 months of + CT Test | |||
| Yes | 88 (71.0) | 122 (76.3) | 0.315 |
| No | 36 (29.0) | 38 (23.8) | |
| CT Follow-up Test Result (n = 210 tested) | |||
| Positive | 6 (6.8) | 16 (13.1) | 0.207 |
| Negative | 82 (93.2) | 106 (86.9) | |
| Standard Treatment Regimens: (n = 124) | No. (%) |
| Doxycycline 100 mg orally 2 times/day for 21 days | 121 (97.6) |
| Azithromycin 1 g orally once weekly for 3 weeks | 3 (2.4) |
| Non-standard treatment regimens: (n = 160) | |
| Doxycycline 100 mg orally 2 times/day for 7 days | 55 (34.4) |
| Doxycycline 100 mg orally 2 times/day for >7 days but <21 days | 19 (11.9) |
| Minocycline 100 mg orally 2 times/day for 21 days | 2 (1.3) |
| Azithromycin 1 g orally one time | 78 (48.8) |
| Azithromycin 500 mg orally 2 times/day for 5 days | 2 (1.3) |
| No treatment * | 4 (2.5) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oda, G.; Chung, J.; Lucero-Obusan, C.; Holodniy, M. Clinically Defined Lymphogranuloma Venereum among US Veterans with Human Immunodeficiency Virus, 2016–2023. Microorganisms 2024, 12, 1327. https://doi.org/10.3390/microorganisms12071327
Oda G, Chung J, Lucero-Obusan C, Holodniy M. Clinically Defined Lymphogranuloma Venereum among US Veterans with Human Immunodeficiency Virus, 2016–2023. Microorganisms. 2024; 12(7):1327. https://doi.org/10.3390/microorganisms12071327
Chicago/Turabian StyleOda, Gina, Joyce Chung, Cynthia Lucero-Obusan, and Mark Holodniy. 2024. "Clinically Defined Lymphogranuloma Venereum among US Veterans with Human Immunodeficiency Virus, 2016–2023" Microorganisms 12, no. 7: 1327. https://doi.org/10.3390/microorganisms12071327
APA StyleOda, G., Chung, J., Lucero-Obusan, C., & Holodniy, M. (2024). Clinically Defined Lymphogranuloma Venereum among US Veterans with Human Immunodeficiency Virus, 2016–2023. Microorganisms, 12(7), 1327. https://doi.org/10.3390/microorganisms12071327






