Abstract
For the safe use of microbiome-based solutions in agriculture, the genome sequencing of strains composing the inoculum is mandatory to avoid the spread of virulence and multidrug resistance genes carried by them through horizontal gene transfer to other bacteria in the environment. Moreover, the annotated genomes can enable the design of specific primers to trace the inoculum into the soil and provide insights into the molecular and genetic mechanisms of plant growth promotion and biocontrol activity. In the present work, the genome sequences of some members of beneficial microbial consortia that have previously been tested in greenhouse and field trials as promising biofertilizers for maize, tomato and wheat crops have been determined. Strains belong to well-known plant-growth-promoting bacterial genera such as Bacillus, Burkholderia, Pseudomonas and Rahnella. The genome size of strains ranged from 4.5 to 7.5 Mbp, carrying many genes spanning from 4402 to 6697, and a GC content of 0.04% to 3.3%. The annotation of the genomes revealed the presence of genes that are implicated in functions related to antagonism, pathogenesis and other secondary metabolites possibly involved in plant growth promotion and gene clusters for protection against oxidative damage, confirming the plant-growth-promoting (PGP) activity of selected strains. All the target genomes were found to possess at least 3000 different PGP traits, belonging to the categories of nitrogen acquisition, colonization for plant-derived substrate usage, quorum sensing response for biofilm formation and, to a lesser extent, bacterial fitness and root colonization. No genes putatively involved in pathogenesis were identified. Overall, our study suggests the safe application of selected strains as “plant probiotics” for sustainable agriculture.
1. Introduction
Plant-growth-promoting (PGP) bacteria are a taxonomically heterogeneous group of bacteria that can enhance plant growth and protect plants from disease and abiotic stress [1]. They are normally found within the plant-associated microbiota, as members of the rhizosphere or the phyllosphere (the habitats surrounding roots and leaves, respectively) or as endophytes of different plant organs. These bacteria can contribute to the host plant’s health and yield multiple abilities from the absorption of nutrients to the regulation of the host immune response [2]. They provide protection against pathogens, additional metabolic capabilities, essential nutrients and even phytohormone modulation [3].
In recent years, an ever-increasing interest in plant-associated bacteria has been manifested by researchers but also growers, consumers and policymakers. The effectiveness of PGP bacterial inoculants in contributing to a net increase in crop productivity coupled with a decrease in the use of agrochemicals has been demonstrated [4]. Interest in scouting plant-associated microbiota and culturing novel PGP bacteria is growing [5]. One of the main concerns with the utilization of inoculants composed of a single bacterial strain is that, while they may be effective under in vitro testing, they often fail in tests in field conditions, potentially due to competition with indigenous soil and plant microbiota, as well as variable environmental conditions [6,7]. Recently, innovative solutions have been proposed such as the production of microbial consortia composed of different species, which can have complementary and beneficial activities with respect to the plant and which can ensure enough persistence in the environment to produce reliable plant-growth-promoting effects [8,9,10]. Such microbial consortia, when mirroring the complexity and services of a native microbial community, are sometimes referred to as synthetic communities (SynComs) [11,12], and they comprise several members trying to mimic the core functions of a healthy native microbial community [13]. The choice of plant-growth-promoting microorganism (PGPM) is fundamental to developing efficient SynComs to be used as biofertilizers in agriculture, capable of boosting the growth of crop plants and exerting their biocontrol activity against phytopathogens. In many cases, SynComs have been tested under controlled conditions in pot experiments, providing the opportunity to model the complexity of plant–microbe interactions and of the soil and rhizosphere microbiome [14,15,16]. However, the discrepancy between laboratory results and performance on the field is often considerable, resulting in unsuccessful attempts at microbial application [17]. In order to mitigate the risk of failure of PGPM application in the field, a proposal for guidelines pertaining to the design and implementation of field trials was put forth [18]. The objective was to ensure reproducibility of the results, thereby preventing potential issues that could arise in the context of a future large-scale application.
The advent of next-generation sequencing (NGS) has facilitated the whole-genome sequencing (WGS) of several PGPRs, mainly isolated from crop species, such as Pseudomonas sp., Bacillus sp., Paenibacillus polymyxa and Klebsiella sp. [19,20,21,22,23,24]. Recently, the whole-genome study on endophytic Bacillus velezensis strains isolated from cultivated maize revealed the presence of numerous genes with significant roles in PGP and biocontrol activity against phytopathogens and unveiled multiple secondary metabolite biosynthetic gene clusters (BGCs) [25]. Also, a comprehensive WGS of B. licheniformis YB06, isolated from the rhizosphere soil of healthy Codonopsis pilosula plants, revealed a diverse array of genes linked to induced systemic resistance (ISR) and PGP traits [26]. Genome mining performed on a large panel of B. ambifaria strains revealed the presence of the biosynthetic gene cluster responsible for the production of cepacin, a metabolite involved in biocontrol activity against Pythium damping-off disease [27].
The identification of the potential pathways and genes of the soil/rhizosphere microorganisms related to plant growth promotion enables a better understanding of the molecular mechanisms of PGP and biocontrol activity, which is fundamental to developing more efficient biofertilizers. Furthermore, WGS offers the highest level of bacterial strain discrimination, facilitating the development of tools for traceability into the soil of SynComs and more targeted risk assessment to exclude any potential risk associated with scaling-up and commercial application [28,29]. Within the frame of the Horizon 2020 SIMBA project (Sustainable Innovation of Microbiome Applications in Food System), aimed at using beneficial microorganisms along the entire food production value chain, SynComs were developed as sustainable biofertilizers [9,30]. The efficacy and reproducibility of their application were evaluated in the greenhouse and under field conditions on various farms in Germany and Italy [31,32,33,34]. These experimental trials were conducted under both organic and conventional cultivation conditions, taking into account a variety of important crops like maize, wheat and tomatoes. In the present study, the whole-genome sequencing and annotation of some members of SIMBA SynComs, which had not previously been sequenced, and which had been successfully used in greenhouse and field studies, were performed, i.e., Bacillus licheniformis PS141, Bacillus velezensis BV84, Burkholderia ambifaria MCI 7, Pseudomonas granadensis A23/T3c, Pseudomonas fluorescens DR54 and Rahnella aquatilis BB23/T4d. In this study, we reported the genome sequencing of these strains, which allowed us to putatively identify the genetic basis of some plant-growth-promoting and biocontrol traits. Additionally, virulence and antibiotic resistance determinants were also inspected to evaluate the safety and risk to public health of such biofertilizers.
2. Materials and Methods
2.1. Bacterial Strains and Culture Conditions
A total of six strains were examined belonging to the species Bacillus licheniformis, Bacillus velezensis, Burkholderia ambifaria, Pseudomonas fluorescens, Pseudomonas granadensis and Rahnella aqualitis. The bacterial strains were isolated from soil, rhizosphere, or plant tissues. The isolation features and relevant information concerning their PGP activity have been previously reported [9] and are summarized in Table 1. The strains were recovered from glycerol stocks stored at −80 °C and streaked onto nutrient agar (NA) plates. After microbial growth at 28 °C for 24–48 h, 3–4 isolated colonies were transferred to 4 mL of nutrient broth (NB) medium and incubated overnight at 28 °C and 200 rpm.

Table 1.
Plant-growth-promoting microorganisms used in this study. The species names were verified according to the latest classification in the NCBI Taxonomy database.
2.2. Genome Sequences and Annotation
Genomic DNA from each microbial strain was extracted with sodium dodecyl sulfate proteinase K lysis buffer, followed by treatment with cetyltrimethylammonium bromide (CTAB) as described in Current Protocols for Molecular Biology [43]. The integrity of genomic DNA was evaluated through analysis on a 1% agarose gel and the quantity was determined through the use of a NanoDrop (NanoDrop Technologies, Wilmington, DE, USA) and a Qubit fluorometer (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA). Paired-end sequencing was conducted at IGA Technology Services (Udine, Italy) using the HiSeq 2000 Illumina platform with a 2 × 150 approach. For almost all species, drafts of the sequenced genomes of other strains were sequenced at 100× coverage; the strain Bacillus velezensis BV84, for which a sequenced genome was not available, was sequenced at a greater depth (200× coverage). The sequences were assembled with SPAdes [44]. Annotation and COG assignment were performed by Prokka 1.4.0 [45] from the Galaxy webserver Orione [46] (https://galaxyproject.org/use/orione/ (accessed on 12 September 2021)).
2.3. Genome Characterization and Mining
The taxonomic identification of the strains from the genome sequence was performed using the Type (Strain) Genome Server [47]. The dDDH values were computed using the Genome BLAST Distance Phylogeny approach. Formula d4 from [48] was used since it is independent of genome length and is thus robust against the use of incomplete draft genomes. Secondary metabolite biosynthesis gene clusters in bacterial genomes have been identified by means of the AntiSMASH software tool [49] (https://antismash.secondarymetabolites.org/ (accessed on 1 October 2021)). The presence of type 3 secretion systems was analyzed by means of the integrated prediction pipeline for bacterial type 3 secreted effectors (T3Sepp) [50]. To perform pangenome analysis, the pipeline Roary [51] was used and core and dispensable/unique gene fractions against close relatives of each strain were identified.
The genomes were further annotated with RAST [52,53,54] and the predicted amino acid sequences of all strains were mapped against the plant growth promotion traits’ ontology with the PGPT-Pred tool [55,56,57], available on the web platform for plant-associated bacteria, PLaBAse (https://plabase.cs.uni-tuebingen.de/pb/plabase.php (accessed on 1 September 2024)).
2.4. Data Availability
Sequences are deposited in the NCBI database under BioProject ID PRJNA1018344 at the SRA Experiment SRX25324655.
3. Results
3.1. Genomic Sequencing Report
The genome size of the six sequenced bacterial strains of the SynCom ranged from 4.5 to 7.5 Mbp (Table 2). The number of genes spanned from 4402 to 6697, in agreement with the corresponding genome sizes.

Table 2.
Genome sequencing statistics and annotation.
For each genome, in silico DNA–DNA hybridization (DDH) and genome-based phylogeny were computed to allow a robust taxonomic classification of the strains [48]. The results of the taxonomic assignment based on dDDH values are reported in Table 3. Phylogenetic trees for each strain are shown in Supplementary Figure S1.

Table 3.
Taxonomic classification of strains.
3.2. Prediction of Functions Related to Antagonism, Plant Growth Promotion and Virulence
The presence of functions related to antagonism, pathogenesis and other secondary metabolites that may be involved in plant growth promotion (i.e., siderophores) was inspected on the whole dataset of six strains by searching for secondary metabolite gene clusters and type 3 secretion systems. The results are reported in Table 4. As expected, many nonribosomal peptide synthetase (NRPS) gene clusters and genes involved in the biosynthesis of relevant secondary metabolites were found; in particular, for strains belonging to the Bacillus, Pseudomonas and Burkholderia genera, we identified genes coding for antibiotics, surfactants and siderophores. Most notably, NRPSs were found in all strains. B. licheniformis PS141 and B. velezensis BV84 were found harboring the largest number of NRPSs. Concerning PGP abilities, siderophore production gene clusters were found for P. granadensis A23/T3c, P. fluorescens DR54 and R. aquatilis BB23/T4d. Finally, several strains contained gene clusters for protection against oxidative damage (as for aryl polyene and carotenoid production) [58]. Finally, no strain was found to be harboring genes related to T3SSs (type 3 secretion systems).

Table 4.
Presence of gene clusters linked to secondary metabolites and type 3 secretion systems.
Pangenome analysis of the six strains was performed by selecting four panels of strains with various degrees of phylogenetic relatedness (Supplementary Table S1 and Figure S1). This analysis, after the identification of unique genes for each strain (Supplementary Dataset S1), can facilitate the selection of gene candidates for primer design to allow the traceability of bioinoculants in the environment.
3.3. PGP Traits by Inferring Genome Sequences with PLaBAse Database
Mapping of the predicted amino acid sequences of all strains against the plant growth promotion traits’ ontology with the PGPT-Pred tool was performed. The total number of PGP traits identified in both the target and reference genomes is presented in Table 5. Interestingly, Rahnella aquatilis BB23/T4d exhibited the highest number of PGP traits among the target and reference genomes. Furthermore, Bacillus licheniformis PS141, Bacillus velezensis BV84, Pseudomonas granadensis A23/T3c and Rahnella aquatilis BB23/T4d were identified as having a greater number of PGP traits than those observed in the reference genomes.

Table 5.
Number of PGP traits found in the target and reference genomes.
The difference between the reference and target genomes is reported in Figure 1. It is noteworthy that P. granadensis A23/T3c and B. velezensis BV84 exhibited the highest frequency of PGP traits, exceeding that of the reference strains. Despite R. aquatilis BB23/T4d reporting the highest number of PGP traits (Table 5), the difference in frequency between the target strain and the reference is minimal. Conversely, P. fluorescens DR54 was found to report lower-frequency values of PGP traits compared to the reference. Still, all the target genomes reported in this work were found to possess at least 3000 different PGP traits (Table 5).
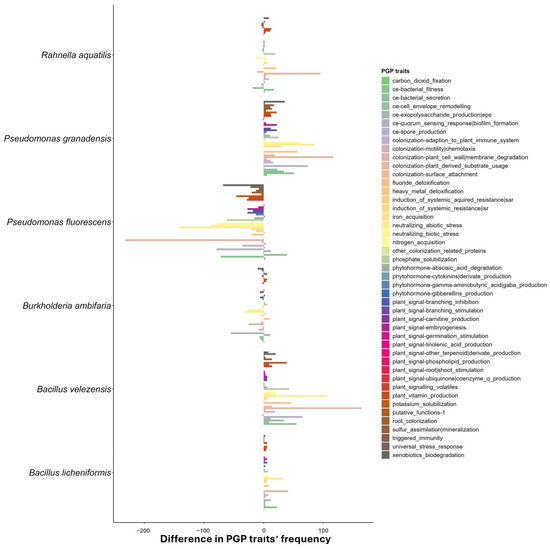
Figure 1.
The difference in frequency occurrence of PGP traits (from PLaBAse) between the genomes presented in this work and the reference genome for each of them. A positive value represents an increase in the frequency occurrence of that PGP trait in the target genome, compared to the reference. A negative value represents a decrease in the frequency of occurrence of that PGP trait in the target genome, compared to the reference.
Interestingly, the most prevalent PGP traits in the differential frequency plot (Figure 1) were identified as belonging to the categories of nitrogen acquisition, colonization for plant-derived substrate usage (e.g., indole-3 acetic acid degradation), quorum sensing response for biofilm formation and, to a lesser extent, bacterial fitness and root colonization.
A considerable number of PGP traits are grouped within these categories, as reported by Patz et al. [56].
4. Discussion
Whole-genome sequencing is an effective method to predict the safety of strains at the gene level, in terms of virulence and antibiotic resistance determinants, to identify genes that contribute to the beneficial activity of microbial strains and to select genetic markers that can enable tracing of the inoculum during field experiments. Under these premises, genome sequencing represents a relevant tool to allow the development of novel microbial inoculants.
All strains selected in this study were previously reported as PGPM strains (Table 1) and were selected as members of multifunctional microbial consortia (MC) according to their different modes of action, within the frame of the Horizon 2020 SIMBA project (Sustainable Innovation of Microbiome Applications in Food System) [10]. Their successful application in controlled environment conditions and open-field studies and the reproducibility of their efficacy on maize, wheat and tomato crops [10,31,32,33,34] allowed us to identify them as potential biofertilizers for sustainable agriculture. Despite the obvious agronomic advantages offered by biofertilization, it is mandatory to exclude any negative effects of their application on the behavior and genetic profile of soil microbial communities. It is well known that horizontal (or lateral) gene transfer (HGT) among bacterial species, one of the major mechanisms in bacterial evolution, could harbor virulence factors that could pose risks to human health [59]. In addition, whole-genome sequencing may be a helpful tool to track the establishment and/or persistence of specific microbes in the field and decipher the molecular and functional mechanisms of plant–microbe interaction [60].
Here, we investigated the WGS of six strains belonging to well-known plant-growth-promoting bacterial species including Bacillus velezensis, Bacillus licheniformis, Burkholderia ambifaria, Pseudomonas granadensis, Pseudomonas fluorescens and Rahnella aquatilis [9]. Genomic mining allowed for the exclusion of the presence of genes putatively involved in pathogenesis and identification of siderophore and NRPS gene clusters, which can explain the previous findings on strains’ abilities to possess multiple PGP traits and promote plant growth (see the references in Table 1). In addition, many gene clusters related to secondary metabolites involved in indirect mechanisms of plant growth have been detected in all sequenced genomes, suggesting a high potential of these strains as biocontrol agents. The presence of gene clusters involved in the protection against oxidative damage (as for aryl polyene and carotenoid production) [58] could contribute to strain survival in soil and in the bioinoculant formulation, as revealed in previous works performed in the greenhouse and in field trials [31,32,33,61].
Overall, genomic analysis revealed a broad spectrum of PGP genes in all strains, demonstrating significant functional diversity. The identification of PGP traits was conducted by inferring genome sequences using the PLaBAse database, and the highest number of PGP traits was detected in Rahnella aquatilis BB23/T4d, compared to the other strains. In a recent study [62], Rahnella aquatilis was found to improve plant growth and physiological parameters, indicating its potential for development as a biofertilizer to support a sustainable agricultural system [63,64]. The strain presented in this work, with its high number of PGP traits, proves essential for the consortium, as it was still detected one-month post-inoculation [61,65]. Concerning the other strains, they were found to harbor PGP traits, ranging from 3052 (Bacillus licheniformis PS141) to 3893 (B. ambifaria MCI 7). Genes associated with PGP traits such as “Plant Vitamin Production”, “Cell Envelope Remodeling”, “Universal Stress Response” and “Xenobiotics Biodegradation” are essential for rhizobacterial survival and plant nutrition, as they rely on plant exudates and contribute to plant growth by synthesizing metabolites and solubilizing nutrients, significantly impacting plant health and growth [66]. Also, the detection of genes related to quorum sensing and biofilm formation represents a favorable PGP trait for the establishment of strains on plant roots as a stage of the colonization process [65,67].
The data reported in this work could constitute the basis for developing a combined approach of phenotypic testing and genome sequencing of strains for the constitution of bioinoculant consortia. Prospectively, this approach can allow for the rational design of a SynCom formulation, which takes into account relevant genomic information, such as the putative biosynthetic pathways for relevant secondary metabolites and highlights the presence of a unique gene pool for each strain, which could be further used to design primers and develop molecular methods for biomonitoring (i.e., qPCR). In a recent study, based on in silico analysis and on the already published genomes of species composing the microbial consortium MC_C, developed within the frame of the Horizon 2020 SIMBA project, a real-time PCR (qtPCR) protocol was set up to follow the fate of PGPR strains under field conditions [61]. Results revealed that three out of the five bacterial species included in the MC_C consortium, i.e., Burkholderia ambifaria, Bacillus amyloliquefaciens and Rahnella aquatilis, were detected up to one month after inoculation, having been able to colonize and grow in the soil. In a previous work, from the whole-genome sequence of a B. velezens is strain NJAU-Z9, a strategy based on a real-time PCR method for directly monitoring of the target strain in the soil and the rhizosphere was developed [68]. Our annotated genomes will enable, therefore, the detection of specific DNA regions to be used for the design of unique and efficient quantitative primers for monitoring the target microorganisms composing beneficial microbial consortia, allowing the traceability of bioinoculants. Furthermore, the availability of the genome sequences of inoculated strains offers a strategy to evaluate the persistence of MC in the soil and/or rhizosphere, as well as the competitiveness of strains with indigenous microbiome members, by mapping metagenome sequence reads to the reference genomes [9].
5. Conclusions
In conclusion, the absence of pathogenicity traits, as well as the identification of genes that are likely responsible for the plant-growth-promoting and biocontrol features that were experimentally described, suggests that SIMBA microbial consortia represent good candidates for the scaling-up process and application of “plant probiotics” in sustainable agriculture. The exclusion of any pathogenic traits and the presence of a large plethora of PGP traits boost their use as microbial biofertilizers on a large scale. This study will enable us to proceed with the scaling-up and commercialization of SIMBA microbial consortia, which were highlighted as excellent innovations by the European Commission’s Innovation Radar team (https://simbaproject.eu/simba-partner-recognised-as-key-innovator/), (accessed on 12 April 2023). Our findings provide an essential contribution to build the necessary background for the safe application of microbiomes in food production.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/microorganisms12122562/s1, Table S1: Datasets of strains used for pangenome analysis; Figure S1: Genome-based phylogenies. Closest type strain genomes are included in each phylogeny as from Type (Strain) Genome Server pipeline [47]. Tree inferred with FastME 2.1.6.1 from GBDP distances calculated from genome sequences [69]. The branch lengths are scaled in terms of GBDP distance formula d5. The numbers above branches are GBDP pseudo-bootstrap support values > 60 % from 100 replications, with an average branch support of 91.4 %. The tree was rooted at the midpoint [70]; Supplementary dataset S1: A dataset containing a list of unique orthologs found in the genomes of the SynCom and pangenome statistics and results obtained. Refs. [69,70] can be found in Supplementary Materials.
Author Contributions
Conceptualization, A.B.; methodology, L.C., S.S., P.A., E.E., S.T., A.V., A.F. and G.A.; software, L.C. and A.V.; validation, L.C., A.B. and A.M.; formal analysis, L.C. and A.V.; investigation, A.B., L.C., S.T. and A.V.; resources, A.B.; data curation, A.B., L.C., S.T. and A.V.; writing—original draft preparation, A.B, L.C. and A.M.; writing—review and editing, A.B., A.M., A.V., L.C., S.S. and S.T.; visualization, A.B.; supervision, A.B.; project administration, A.B.; funding acquisition, A.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the European Union’s Horizon 2020 Research and Innovation Programme under grant agreement No. 818431 (SIMBA, Sustainable Innovation of Microbiome Applications in Food System). This output reflects only the authors’ views, and the Research Executive Agency (REA) of the EU cannot be held responsible for any use that may be made of the information contained therein. This project has also received funding from “Strengthening the MIRRI Italian Research Infrastructure for Sustainable Bioscience and Bioeconomy” SUS-MIRRI.IT project funded by the European Union—NextGeneration EU, PNRR—Mission 4 “Education and Research” Component 2: from research to business, Investment 3.1: Fund for the realization of an integrated system of research and innovation infrastructures—IR0000005 (D.M. Prot. n.120 del 21/06/2022), from the Italian project Creazione di un HUB italiano a supporto della partecipazione dell’Italia alla Global Soil Partnership ed alla rete di eccellenza europea sulla ricerca sul suolo–SOIL-HUB, granted by the Italian Ministry of Agricultural, Food and Forestry Policies MIPAAF (DM 37072 28/12/2018) CUP C52F18000200006, and from the European Union’s Horizon 2020 research and innovation programme under grant agreement No. 862695 (European Joint Programme SOIL).
Data Availability Statement
Sequences were deposited in the NCBI database under BioProject ID PRJNA1018344 at the SRA Experiment SRX25324655.
Acknowledgments
The authors are also grateful to the SIMBA partners (https://simbaproject.eu/about/consortium/ (accessed on 18 January 2024)) and, in particular, to the Coordinator of the SIMBA Project, Anne Pihlanto (Natural Resources Institute Finland (Luke)), for her helpful discussions.
Conflicts of Interest
Author Patrizia Ambrosino was employed by the company AGRIGES srl and author Enrico Ercole was employed by the company CCS-AOSTA srl. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
- Oleńska, E.; Małek, W.; Wójcik, M.; Swiecicka, I.; Thijs, S.; Vangronsveld, J. Beneficial Features of Plant Growth-Promoting Rhizobacteria for Improving Plant Growth and Health in Challenging Conditions: A Methodical Review. Sci. Total Environ. 2020, 743, 140682. [Google Scholar] [CrossRef] [PubMed]
- Khoso, M.A.; Wagan, S.; Alam, I.; Hussain, A.; Ali, Q.; Saha, S.; Poudel, T.R.; Manghwar, H.; Liu, F. Impact of Plant Growth-Promoting Rhizobacteria (PGPR) on Plant Nutrition and Root Characteristics: Current Perspective. Plant Stress 2024, 11, 100341. [Google Scholar] [CrossRef]
- Su, F.; Zhao, B.; Dhondt-Cordelier, S.; Vaillant-Gaveau, N. Plant-Growth-Promoting Rhizobacteria Modulate Carbohydrate Metabolism in Connection with Host Plant Defense Mechanism. Int. J. Mol. Sci. 2024, 25, 1465. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, J.; Liu, H.; Macdonald, C.A.; Singh, B.K. Application of Microbial Inoculants Significantly Enhances Crop Productivity: A Meta-Analysis of Studies from 2010 to 2020. J. Sustain. Agric. Environ. 2022, 1, 216–225. [Google Scholar] [CrossRef]
- Mapelli, F.; Mengoni, A.; Riva, V.; Borin, S. Bacterial Culturing Is Crucial to Boost Sustainable Agriculture. Trends Microbiolol. 2023, 31, 1–4. [Google Scholar] [CrossRef]
- Mącik, M.; Gryta, A.; Frąc, M. Biofertilizers in Agriculture: An Overview on Concepts, Strategies and Effects on Soil Microorganisms. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press; Elsevier Inc.: Cambridge, MA, USA, 2020; Volume 162, pp. 31–87. [Google Scholar] [CrossRef]
- Maitra, S.; Brestic, M.; Bhadra, P.; Shankar, T.; Praharaj, S.; Palai, J.B.; Shah, M.M.R.; Barek, V.; Ondrisik, P.; Skalický, M.; et al. Bioinoculants—Natural Biological Resources for Sustainable Plant Production. Microorganisms 2022, 10, 51. [Google Scholar] [CrossRef]
- Wang, C.; Li, Y.; Li, M.; Zhang, K.; Ma, W.; Zheng, L.; Xu, H.; Cui, B.; Liu, R.; Yang, Y.; et al. Functional Assembly of Root-Associated Microbial Consortia Improves Nutrient Efficiency and Yield in Soybean. J. Integr. Plant Biol. 2021, 63, 1021–1035. [Google Scholar] [CrossRef]
- Tabacchioni, S.; Passato, S.; Ambrosino, P.; Huang, L.; Caldara, M.; Cantale, C.; Hett, J.; Del Fiore, A.; Fiore, A.; Schlüter, A.; et al. Identification of Beneficial Microbial Consortia and Bioactive Compounds with Potential as Plant Biostimulants for a Sustainable Agriculture. Microorganisms 2021, 9, 426. [Google Scholar] [CrossRef]
- Jain, A.; Singh, S.; Kumar Sarma, B.; Bahadur Singh, H. Microbial Consortium-Mediated Reprogramming of Defence Network in Pea to Enhance Tolerance against Sclerotinia Sclerotiorum. J. Appl. Microbiol. 2012, 112, 537–550. [Google Scholar] [CrossRef]
- Ke, J.; Wang, B.; Yoshikuni, Y. Microbiome Engineering: Synthetic Biology of Plant-Associated Microbiomes in Sustainable Agriculture. Trends Biotechnol. 2021, 39, 244–261. [Google Scholar] [CrossRef]
- Shayanthan, A.; Ordoñez, P.A.C.; Oresnik, I.J. The Role of Synthetic Microbial Communities (SynCom) in Sustainable Agriculture. Front. Agron. 2022, 4, 896307. [Google Scholar] [CrossRef]
- Fagorzi, C.; Passeri, I.; Cangioli, L.; Vaccaro, F.; Mengoni, A. When Biodiversity Preservation Meets Biotechnology: The Challenge of Developing Synthetic Microbiota for Resilient Sustainable Crop Production. J. Sustain. Agric. Environ. 2023, 2, 5–15. [Google Scholar] [CrossRef]
- Vaccaro, F.; Cangioli, L.; Mengoni, A.; Fagorzi, C. Synthetic Plant Microbiota Challenges in Nonmodel Species. Trends Microbiol. 2022, 30, 922–924. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Baquerizo, M. Simplifying the Complexity of the Soil Microbiome to Guide the Development of Next-Generation SynComs. J. Sustain. Agric. Environ. 2022, 1, 9–15. [Google Scholar] [CrossRef]
- Vorholt, J.A.; Vogel, C.; Carlström, C.I.; Müller, D.B. Establishing Causality: Opportunities of Synthetic Communities for Plant Microbiome Research. Cell Host Microbe. 2017, 22, 142–155. [Google Scholar] [CrossRef]
- Sessitsch, A.; Pfaffenbichler, N.; Mitter, B. Microbiome Applications from Lab to Field: Facing Complexity. Trends Plant Sci. 2019, 24, 194–198. [Google Scholar] [CrossRef]
- Neuhoff, D.; Neumann, G.; Weinmann, M. Testing Plant Growth Promoting Microorganisms in the Field—A Proposal for Standards. Front. Plant Sci. 2024, 14, 1324665. [Google Scholar] [CrossRef]
- Liu, W.; Wang, Q.; Hou, J.; Tu, C.; Luo, Y.; Christie, P. Whole Genome Analysis of Halotolerant and Alkalotolerant Plant Growth-Promoting Rhizobacterium Klebsiella Sp. D5A. Sci. Rep. 2016, 6, 26710. [Google Scholar] [CrossRef]
- Gupta, A.; Gopal, M.; Thomas, G.V.; Manikandan, V.; Gajewski, J.; Thomas, G.; Seshagiri, S.; Schuster, S.C.; Rajesh, P.; Gupta, R. Whole Genome Sequencing and Analysis of Plant Growth Promoting Bacteria Isolated from the Rhizosphere of Plantation Crops Coconut, Cocoa and Arecanut. PLoS ONE 2014, 9, e104259. [Google Scholar] [CrossRef]
- Mingchao, M.; Wang, C.; Ding, Y.; Li, L.; Shen, D.; Jiang, X.; Guan, D.; Cao, F.; Chen, H.; Feng, R.; et al. Complete Genome Sequence of Paenibacillus Polymyxa SC2, a Strain of Plant Growth-Promoting Rhizobacterium with Broad-Spectrum Antimicrobial Activity. J. Bacteriol. 2011, 193, 311–312. [Google Scholar] [CrossRef]
- Khalifa, A.; Alsowayeh, N. Whole-Genome Sequence Insight into the Plant-Growth-Promoting Bacterium Priestia Filamentosa Strain AZC66 Obtained from Zygophyllum Coccineum Rhizosphere. Plants 2023, 12, 1944. [Google Scholar] [CrossRef] [PubMed]
- Song, J.Y.; Kim, H.A.; Kim, J.S.; Kim, S.Y.; Jeong, H.; Kang, S.G.; Kim, B.K.; Kwon, S.K.; Lee, C.H.; Yu, D.S.; et al. Genome Sequence of the Plant Growth-Promoting Rhizobacterium Bacillus sp. Strain JS. J. Bacteriol. 2012, 194, 3760–3761. [Google Scholar] [CrossRef] [PubMed]
- Mathimaran, N.; Srivastava, R.; Wiemken, A.; Sharma, A.K.; Boller, T. Genome Sequences of Two Plant Growth-Promoting Fluorescent Pseudomonas Strains, R62 and R81. J. Bacteriol. 2012, 194, 3760–3761. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ercole, T.G.; Kava, V.M.; Petters-Vandresen, D.A.L.; Nassif Gomes, M.E.; Aluizio, R.; Ribeiro, R.A.; Hungria, M.; Galli, L.V. Unlocking the Growth-Promoting and Antagonistic Power: A Comprehensive Whole Genome Study on Bacillus Velezensis Strains. Gene 2024, 927, 1486669. [Google Scholar] [CrossRef] [PubMed]
- Ni, S.; Wu, Y.; Zhu, N.; Leng, F.; Wang, Y. Bacillus LicheniformisYB06: A Rhizosphere–Genome-Wide Analysis and Plant Growth-Promoting Analysis of a Plant Growth-Promoting Rhizobacterium Isolated from Codonopsis Pilosula. Microorganisms 2024, 12, 1861. [Google Scholar] [CrossRef]
- Mullins, A.J.; Murray, J.A.H.; Bull, M.J.; Jenner, M.; Jones, C.; Webster, G.; Green, A.E.; Neill, D.R.; Connor, T.R.; Parkhill, J.; et al. Genome Mining Identifies Cepacin as a Plant-Protective Metabolite of the Biopesticidal Bacterium Burkholderia Ambifaria. Nat. Microbiol. 2019, 4, 996–1005. [Google Scholar] [CrossRef]
- Yapa, N.; Lakmali, D.; De Zoysa, K.S.; Silva, S.; Manawadu, C.; Herath, B.M.; Madhushan, A.; Perera, G.; Ratnayakae, O.; Kapilan, R.; et al. Biofertilizers: An Emerging Trend in Agricultural Sustainability. Chiang Mai J. Sci. 2022, 49, 1–33. [Google Scholar] [CrossRef]
- Mahdi, I.; Fahsi, N.; Hijri, M.; Sobeh, M. Antibiotic Resistance in Plant Growth Promoting Bacteria: A Comprehensive Review and Future Perspectives to Mitigate Potential Gene Invasion Risks. Front. Microbiol. 2022, 13, 999988. [Google Scholar] [CrossRef]
- Magarelli, R.A.; Trupo, M.; Ambrico, A.; Larocca, V.; Martino, M.; Palazzo, S.; Balducchi, R.; Joutsjoki, V.; Pihlanto, A.; Bevivino, A. Designing a Waste-Based Culture Medium for the Production of Plant Growth Promoting Microorganisms Based on Cladodes Juice from Opuntia Ficus-Indica Pruning. Fermentation 2022, 8, 225. [Google Scholar] [CrossRef]
- Hett, J.; Neuhoff, D.; Döring, T.F.; Masoero, G.; Ercole, E.; Bevivino, A. Effects of Multi-Species Microbial Inoculants on Early Wheat Growth and Litterbag Microbial Activity. Agronomy 2022, 12, 899. [Google Scholar] [CrossRef]
- Graziano, S.; Caldara, M.; Gullì, M.; Bevivino, A.; Maestri, E.; Marmiroli, N. A Metagenomic and Gene Expression Analysis in Wheat (T. durum) and Maize (Z. mays) Biofertilized with PGPM and Biochar. Int. J. Mol. Sci. 2022, 23, 10376. [Google Scholar] [CrossRef] [PubMed]
- Hett, J.; Döring, T.F.; Bevivino, A.; Neuhoff, D. Impact of Microbial Consortia on Organic Maize in a Temperate Climate Varies with Environment but Not with Fertilization. Eur. J. Agron. 2023, 144, 126743. [Google Scholar] [CrossRef]
- Caldara, M.; Gullì, M.; Graziano, S.; Riboni, N.; Maestri, E.; Mattarozzi, M.; Bianchi, F.; Careri, M.; Marmiroli, N. Microbial Consortia and Biochar as Sustainable Biofertilisers: Analysis of Their Impact on Wheat Growth and Production. Sci. Tot. Environ. 2024, 917, 170168. [Google Scholar] [CrossRef] [PubMed]
- Bevivino, A.; Dalmastri, C.; Tabacchioni, S.; Chiarini, L. Efficacy of Burkholderia Cepacia MCI 7 in Disease Suppression and Growth Promotion of Maize. Biol Fertil. Soils 2000, 31, 225–231. [Google Scholar] [CrossRef]
- Nacamulli, C.; Bevivino, A.; Dalmastri, C.; Tabacchioni, S.; Chiarini, L. Perturbation of Maize Rhizosphere Microflora Following Seed Bacterization with Burkholderia Cepacia MCI 7. FEMS Microbiol. Ecol. 1997, 23, 183–193. [Google Scholar] [CrossRef]
- Nielsen, M.N.; Sørensen, J.; Fels, J.; Pedersen, H.C. Secondary Metabolite- and Endochitinase-Dependent Antagonism toward Plant-Pathogenic Microfungi of Pseudomonas Fluorescens Isolates from Sugar Beet Rhizosphere. Appl. Environm. Microbiol. 1998, 64, 3563–3569. [Google Scholar] [CrossRef]
- Thirup, L.; Johnsen, K.; Winding, A. Succession of Indigenous Pseudomonas Spp. and Actinomycetes on Barley Roots Affected by the Antagonistic Strain Pseudomonas Fluorescens DR54 and the Fungicide Imazalil. Appl. Environm. Microbiol. 2001, 67, 1147–1153. [Google Scholar] [CrossRef]
- Andersen, K.S.; Winding, A. Non-Target Effects of Bacterial Biological Control Agents on Soil Protozoa. Biol. Fertil. Soils 2004, 40, 230–236. [Google Scholar] [CrossRef]
- Johansen, A.; Knudsen, I.M.B.; Binnerup, S.J.; Winding, A.; Johansen, J.E.; Jensen, L.E.; Andersen, K.S.; Svenning, M.M.; Bonde, T.A. Non-Target Effects of the Microbial Control Agents Pseudomonas Fluorescens DR54 and Clonostachys Rosea IK726 in Soils Cropped with Barley Followed by Sugar Beet: A Greenhouse Assessment. Soil Biol. Biochem. 2005, 37, 2225–2239. [Google Scholar] [CrossRef]
- Sørensen, J.; Sjøholm, O. Multiparameter Flow Cytometry for Characterization of Physiological States in Pseudomonas Fluorescens DR54 Biocontrol Inoculants Under Dry Formulation and Long-Term Storage in Clay Carrier. In Molecular Microbial Ecology of the Rhizosphere; de Bruijn, F.J., Ed.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2013; Volume 2, pp. 943–950. [Google Scholar] [CrossRef]
- Chiarini, L.; Bevivino, A.; Tabacchioni, S.; Dalmastri, C. Inoculation of Burkholderia Cepacia, Pseudomonas Fluorescens and Enterobacter Sp. on Sorghum Bicolor: Root Colonization and Plant Growth Promotion of Dual Strain Inocula. Soil Biol. Biochem. 1998, 30, 81–87. [Google Scholar] [CrossRef]
- Wilson, K. Preparation of Genomic DNA from Bacteria. In Current Protocols in Molecular Biology; Ausubel, F.M., Brent, R., Kingston, R.E., Moore, D.D., Seidman, J.G., Smith, J.A., Struhl, K., Eds.; Wiley & Sons: New York, NY, USA, 1987; pp. 2.4.1–2.4.5. [Google Scholar] [CrossRef]
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.I.; Pham, S.; Prjibelski, A.D.; et al. SPAdes: A New Genome Assembly Algorithm and Its Applications to Single-Cell Sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef] [PubMed]
- Seemann, T. Prokka: Rapid Prokaryotic Genome Annotation. Bioinformatics 2014, 30, 2068–2069. [Google Scholar] [CrossRef] [PubMed]
- Cuccuru, G.; Orsini, M.; Pinna, A.; Sbardellati, A.; Soranzo, N.; Travaglione, A.; Uva, P.; Zanetti, G.; Fotia, G. Orione, a web-based framework for NGS analysis in microbiology. Bioinformatics 2014, 30, 1928–1929. [Google Scholar] [CrossRef] [PubMed]
- Meier-Kolthoff, J.P.; Göker, M. TYGS Is an Automated High-Throughput Platform for State-of-the-Art Genome-Based Taxonomy. Nat. Commun. 2019, 10, 2182. [Google Scholar] [CrossRef] [PubMed]
- Meier-Kolthoff, J.P.; Auch, A.F.; Klenk, H.P.; Göker, M. Genome Sequence-Based Species Delimitation with Confidence Intervals and Improved Distance Functions. BMC Bioinform. 2013, 14, 60. [Google Scholar] [CrossRef]
- Blin, K.; Shaw, S.; Kautsar, S.A.; Medema, M.H.; Weber, T. The AntiSMASH Database Version 3: Increased Taxonomic Coverage and New Query Features for Modular Enzymes. Nucleic Acids Res. 2021, 49, D639–D643. [Google Scholar] [CrossRef]
- Hui, X.; Chen, Z.; Lin, M.; Zhang, J.; Hu, Y.; Zeng, Y.; Cheng, X.; Ou-Yang, L.; Sun, M.; White, A.P.; et al. T3SEpp: An Integrated Prediction Pipeline for Bacterial Type III Secreted Effectors. mSystems 2020, 5, e00288-20. [Google Scholar] [CrossRef]
- Page, A.J.; Cummins, C.A.; Hunt, M.; Wong, V.K.; Reuter, S.; Holden, M.T.G.; Fookes, M.; Falush, D.; Keane, J.A.; Parkhill, J. Roary: Rapid Large-Scale Prokaryote Pan Genome Analysis. Bioinformatics 2015, 31, 3691–3693. [Google Scholar] [CrossRef]
- Aziz, R.K.; Bartels, D.; Best, A.; DeJongh, M.; Disz, T.; Edwards, R.A.; Formsma, K.; Gerdes, S.; Glass, E.M.; Kubal, M.; et al. The RAST Server: Rapid Annotations Using Subsystems Technology. BMC Genomics 2008, 9, 75. [Google Scholar] [CrossRef]
- Overbeek, R.; Olson, R.; Pusch, G.D.; Olsen, G.J.; Davis, J.J.; Disz, T.; Edwards, R.A.; Gerdes, S.; Parrello, B.; Shukla, M.; et al. The SEED and the Rapid Annotation of Microbial Genomes Using Subsystems Technology (RAST). Nucleic Acids Res. 2014, 42, D206–D214. [Google Scholar] [CrossRef]
- Brettin, T.; Davis, J.J.; Disz, T.; Edwards, R.A.; Gerdes, S.; Olsen, G.J.; Olson, R.; Overbeek, R.; Parrello, B.; Pusch, G.D.; et al. RASTtk: A Modular and Extensible Implementation of the RAST Algorithm for Building Custom Annotation Pipelines and Annotating Batches of Genomes. Sci. Rep. 2015, 5, 8365. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, S.; Kuzmanović, N.; Patz, S.; Lohwasser, U.; Bunk, B.; Spröer, C.; Lorenz, M.; Elhady, A.; Frühling, A.; Neumann-Schaal, M.; et al. Two New Rhizobiales Species Isolated from Root Nodules of Common Sainfoin (Onobrychis Viciifolia) Show Different Plant Colonization Strategies. Microbiol. Spectr. 2022, 10, e0109922. [Google Scholar] [CrossRef] [PubMed]
- Patz, S.; Gautam, A.; Becker, S.; Ruppel, S.; Rodríguez-Palenzuela, P.; Huson, D.H. PLaBAse: A Comprehensive Web Resource for Analyzing the Plant Growth-Promoting Potential of Plant-Associated Bacteria. bioRxiv 2021. [Google Scholar] [CrossRef]
- Sascha, P.; Rauh, M.; Gautam, A.; Huson, D.H. MgPGPT: Metagenomic Analysis of Plant Growth-Promoting Traits. bioRxiv 2024. [Google Scholar] [CrossRef]
- Schöner, T.A.; Gassel, S.; Osawa, A.; Tobias, N.J.; Okuno, Y.; Sakakibara, Y.; Shindo, K.; Sandmann, G.; Bode, H.B. Aryl Polyenes, a Highly Abundant Class of Bacterial Natural Products, Are Functionally Related to Antioxidative Carotenoids. Chem. Bio. Chem. 2016, 17, 247–253. [Google Scholar] [CrossRef]
- Popa, O.; Dagan, T. Trends and Barriers to Lateral Gene Transfer in Prokaryotes. Curr. Opin. Microbiol. 2011, 145, 615–623. [Google Scholar] [CrossRef]
- Rilling, J.I.; Acuña, J.J.; Nannipieri, P.; Cassan, F.; Maruyama, F.; Jorquera, M.A. Current Opinion and Perspectives on the Methods for Tracking and Monitoring Plant Growth-Promoting Bacteria. Soil Biol. Biochem. 2019, 130, 205–219. [Google Scholar] [CrossRef]
- Iosa, I.; Agrimonti, C.; Marmiroli, N. Real-Time PCR (QtPCR) to Discover the Fate of Plant Growth-Promoting Rhizobacteria (PGPR) in Agricultural Soils. Microorganisms 2024, 12, 1002. [Google Scholar] [CrossRef]
- Rana, K.L.; Negi, R.; Sharma, B.; Yadav, A.; Devi, R.; Kaur, T.; Shreaz, S.; Rustagi, S.; Rai, A.K.; Singh, S.; et al. Potential Effect of Novel Endophytic Nitrogen Fixing Diverse Species of Rahnella on Growth Promotion of Wheat (Triticum aestivum L.). J. Crop Sci. Biotechnol. 2024, 27, 605–615. [Google Scholar] [CrossRef]
- Peng, J.; Xu, Z.; Li, L.; Zhao, B.; Guo, Y. Disruption of the Sensor Kinase PhoQ Gene Decreases Acid Resistance in Plant Growth-Promoting Rhizobacterium Rahnella Aquatilis HX2. J. Appl. Microbiol. 2023, 134, lxad009. [Google Scholar] [CrossRef]
- Li, G.E.; Kong, W.L.; Wu, X.Q.; Ma, S.B. Phytase-Producing Rahnella Aquatilis Jz-Gx1 Promotes Seed Germination and Growth in Corn (Zea mays L.). Microorganisms 2021, 9, 1647. [Google Scholar] [CrossRef] [PubMed]
- Podile, A.R.; Vukanti, R.V.N.R.; Sravani, A.; Kalam, S.; Dutta, S.; Durgeshwar, P.; Papa Rao, V. Root Colonization and Quorum Sensing Are the Driving Forces of Plant Growth Promoting Rhizobacteria (PGPR) for Growth Promotion. Proc. Indian Natl. Sci. Acad. 2014, 80, 407–413. [Google Scholar] [CrossRef]
- Pellegrinetti, T.A.; Monteiro, G.G.T.N.; Lemos, L.N.; Santos, R.A.C.d.; Barros, A.G.; Mendes, L.W. PGPg_finder: A Comprehensive and User-Friendly Pipeline for Identifying Plant Growth-Promoting Genes in Genomic and Metagenomic Data. Rhizosphere 2024, 30, 100905. [Google Scholar] [CrossRef]
- Źuñiga, A.; Donoso, R.A.; Ruiz, D.; Ruz, G.A.; Gonźalez, B. Quorum-Sensing Systems in the Plant Growth-Promoting Bacterium Paraburkholderia Phytofirmans PsJN Exhibit Cross-Regulation and Are Involved in Biofilm Formation. Mol. Plant Microbe Interact. 2017, 30, 557–565. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Gao, X.; Wang, S.; Zhu, C.; Li, R.; Shen, Q. Application of Bacillus Velezensis NJAU-Z9 Enhanced Plant Growth Associated with Efficient Rhizospheric Colonization Monitored by QPCR with Primers Designed from the Whole Genome Sequence. Curr. Microbiol. 2018, 75, 1574–1583. [Google Scholar] [CrossRef]
- Lefort, V.; Desper, R.; Gascuel, O. FastME 2.0: A comprehensive, accurate, and fast distance-based phylogeny inference program. Mol. Biol. Evol. 2015, 32, 2798–2800. [Google Scholar] [CrossRef]
- Farris, J.S. Estimating Phylogenetic Trees from Distance Matrices. Am. Nat. 1972, 106, 645–668. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).