The Role of Gut Dysbiosis in the Loss of Intestinal Immune Cell Functions and Viral Pathogenesis
Abstract
1. Introduction
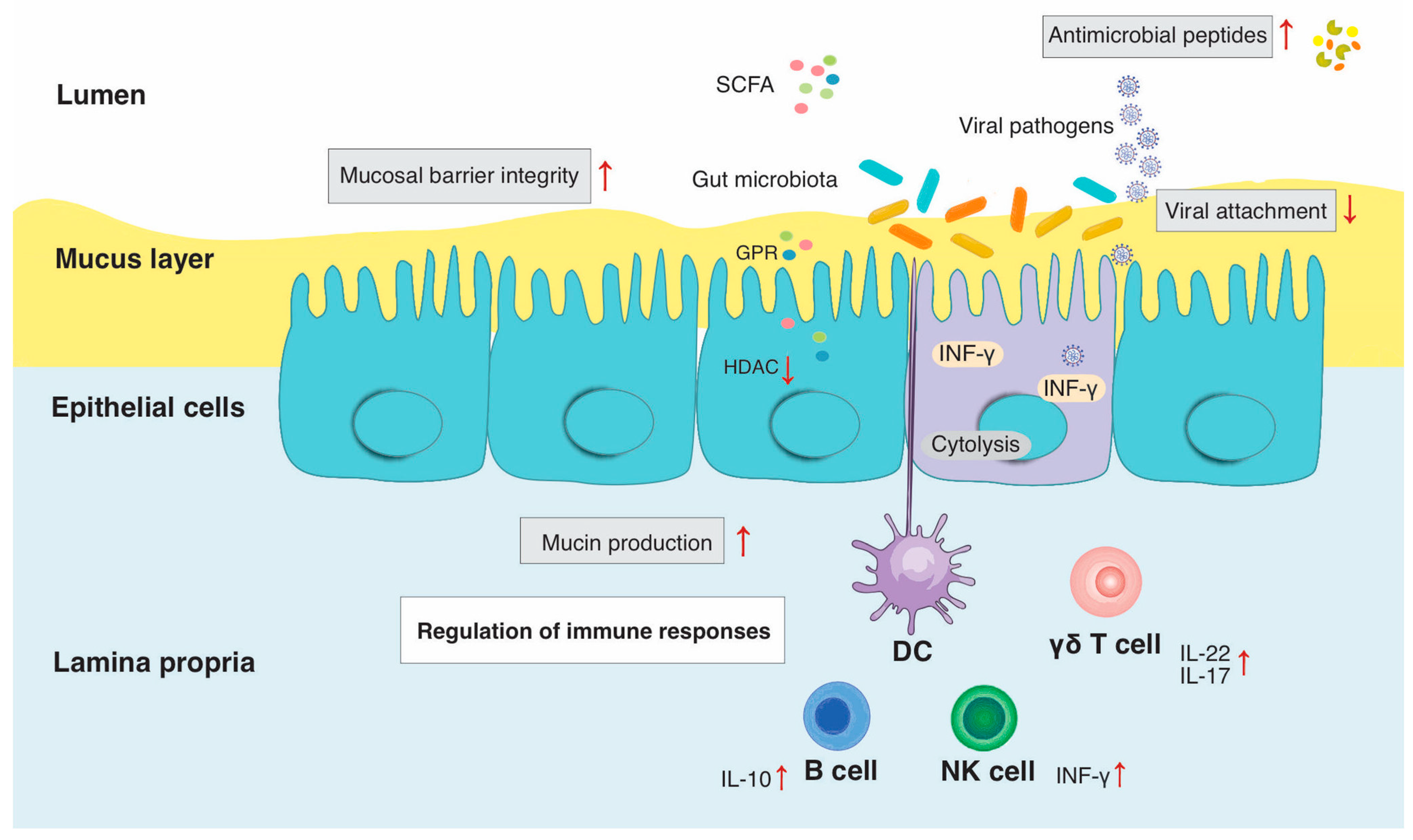
2. Impact of Gut Microbiome on the Host Immune System under Steady State
3. Role of Intestinal Immune Cells during Viral Dysbiosis
4. Gut Dysbiosis during Viral Infections
4.1. HIV/SIV
4.2. Influenza
4.3. SARS-CoV2
5. Role of Host Microbiota in Susceptibility to Viral Infection and Pathogenesis
5.1. Microbiome–Gut–Brain Axis in Viral Neuropathogenesis
5.2. Cardiovascular Disease Associated with Dysbiosis during Chronic Viral Infections
5.3. Fatty Liver Disease Due to Viral Dysbiosis
6. Microbiome-Based Therapies in Viral Infections
7. Conclusions and Future Directions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef] [PubMed]
- Morais, L.H.; Schreiber, H.L., IV; Mazmanian, S.K. The gut microbiota–brain axis in behaviour and brain disorders. Nat. Rev. Microbiol. 2021, 19, 241–255. [Google Scholar] [CrossRef]
- Fakharian, F.; Asgari, B.; Nabavi-Rad, A.; Sadeghi, A.; Soleimani, N.; Yadegar, A.; Zali, M.R. The interplay between Helicobacter pylori and the gut microbiota: An emerging driver influencing the immune system homeostasis and gastric carcinogenesis. Front. Cell. Infect. Microbiol. 2022, 12, 953718. [Google Scholar] [CrossRef]
- Durack, J.; Lynch, S.V. The gut microbiome: Relationships with disease and opportunities for therapy. J. Exp. Med. 2019, 216, 20–40. [Google Scholar] [CrossRef] [PubMed]
- Pascale, A.; Marchesi, N.; Govoni, S.; Coppola, A.; Gazzaruso, C. The role of gut microbiota in obesity, diabetes mellitus, and effect of metformin: New insights into old diseases. Curr. Opin. Pharmacol. 2019, 49, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Ying, J. Gut Microbiota and Immunotherapy. Front. Microbiol. 2022, 13, 2383. [Google Scholar] [CrossRef] [PubMed]
- Wilkins, L.J.; Monga, M.; Miller, A.W. Defining dysbiosis for a cluster of chronic diseases. Sci. Rep. 2019, 9, 12918. [Google Scholar] [CrossRef]
- Clerbaux, L.-A.; Fillipovska, J.; Muñoz, A.; Petrillo, M.; Coecke, S.; Amorim, M.-J.; Grenga, L. Mechanisms Leading to Gut Dysbiosis in COVID-19: Current Evidence and Uncertainties Based on Adverse Outcome Pathways. J. Clin. Med. 2022, 11, 5400. [Google Scholar] [CrossRef]
- DeGruttola, A.K.; Low, D.; Mizoguchi, A.; Mizoguchi, E. Current understanding of dysbiosis in disease in human and animal models. Inflamm. Bowel Dis. 2016, 22, 1137–1150. [Google Scholar] [CrossRef]
- Zhang, X.; Pan, Z. Influence of microbiota on immunity and immunotherapy for gastric and esophageal cancers. Gastroenterol. Rep. 2020, 8, 206–214. [Google Scholar] [CrossRef]
- den Besten, G.; van Eunen, K.; Groen, A.K.; Venema, K.; Reijngoud, D.J.; Bakker, B.M. The role of short-chain fatty acids in the interplay between diet, gut microbiota, and host energy metabolism. J. Lipid Res. 2013, 54, 2325–2340. [Google Scholar] [CrossRef] [PubMed]
- Thakur, A.; Mikkelsen, H.; Jungersen, G. Intracellular pathogens: Host immunity and microbial persistence strategies. J. Immunol. Res. 2019, 2019, 1356540. [Google Scholar] [CrossRef] [PubMed]
- Duan, T.; Du, Y.; Xing, C.; Wang, H.Y.; Wang, R.-F. Toll-like receptor signaling and its role in cell-mediated immunity. Front. Immunol. 2022, 13, 812774. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Chan, N.; Shao, H.; Born, W.K.; Kaplan, H.J. γδ T Cells Activated in Different Inflammatory Environments Are Functionally Distinct. J. Immunol. 2022, 208, 1224–1231. [Google Scholar] [CrossRef] [PubMed]
- Merino, K.M.; Jazwinski, S.M.; Rout, N. Th17-type immunity and inflammation of aging. Aging 2021, 13, 13378. [Google Scholar] [CrossRef] [PubMed]
- Bowcutt, R.; Forman, R.; Glymenaki, M.; Carding, S.R.; Else, K.J.; Cruickshank, S.M. Heterogeneity across the murine small and large intestine. World J. Gastroenterol. 2014, 20, 15216–15232. [Google Scholar] [CrossRef]
- Chen, Y.; Cui, W.; Li, X.; Yang, H. Interaction Between Commensal Bacteria, Immune Response and the Intestinal Barrier in Inflammatory Bowel Disease. Front. Immunol. 2021, 12, 761981. [Google Scholar] [CrossRef]
- Gubatan, J.; Holman, D.R.; Puntasecca, C.J.; Polevoi, D.; Rubin, S.J.; Rogalla, S. Antimicrobial peptides and the gut microbiome in inflammatory bowel disease. World J. Gastroenterol. 2021, 27, 7402–7422. [Google Scholar] [CrossRef]
- Kany, S.; Vollrath, J.T.; Relja, B. Cytokines in inflammatory disease. Int. J. Mol. Sci. 2019, 20, 6008. [Google Scholar] [CrossRef]
- Cianci, R.; Franza, L.; Schinzari, G.; Rossi, E.; Ianiro, G.; Tortora, G.; Gasbarrini, A.; Gambassi, G.; Cammarota, G. The interplay between immunity and microbiota at intestinal immunological niche: The case of cancer. Int. J. Mol. Sci. 2019, 20, 501. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, Y. Microbiota-Associated Metabolites and Related Immunoregulation in Colorectal Cancer. Cancers 2021, 13, 4054. [Google Scholar] [CrossRef]
- Magliocca, G.; Mone, P.; Di Iorio, B.R.; Heidland, A.; Marzocco, S. Short-chain fatty acids in chronic kidney disease: Focus on inflammation and oxidative stress regulation. Int. J. Mol. Sci. 2022, 23, 5354. [Google Scholar] [CrossRef]
- Zhang, S.; Gang, X.; Yang, S.; Cui, M.; Sun, L.; Li, Z.; Wang, G. The alterations in and the role of the Th17/Treg balance in metabolic diseases. Front. Immunol. 2021, 12, 678355. [Google Scholar] [CrossRef] [PubMed]
- Jacobse, J.; Li, J.; Rings, E.H.; Samsom, J.N.; Goettel, J.A. Intestinal regulatory T cells as specialized tissue-restricted immune cells in intestinal immune homeostasis and disease. Front. Immunol. 2021, 12, 716499. [Google Scholar] [CrossRef] [PubMed]
- Venegas, D.P.; De La Fuente, M.K.; Landskron, G.; González, M.J.; Quera, R.; Dijkstra, G.; Harmsen, H.J.; Faber, K.N.; Hermoso, M.A. Short chain fatty acids (SCFAs)-mediated gut epithelial and immune regulation and its relevance for inflammatory bowel diseases. Front. Immunol 2019, 10, 277. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Wu, W.; Liu, Z.; Cong, Y. Microbiota metabolite short chain fatty acids, GPCR, and inflammatory bowel diseases. J. Gastroenterol. 2017, 52, 1–8. [Google Scholar] [CrossRef]
- Recharla, N.; Geesala, R.; Shi, X.Z. Gut Microbial Metabolite Butyrate and Its Therapeutic Role in Inflammatory Bowel Disease: A Literature Review. Nutrients 2023, 15, 2275. [Google Scholar] [CrossRef]
- Bach Knudsen, K.E.; Laerke, H.N.; Hedemann, M.S.; Nielsen, T.S.; Ingerslev, A.K.; Gundelund Nielsen, D.S.; Theil, P.K.; Purup, S.; Hald, S.; Schioldan, A.G.; et al. Impact of Diet-Modulated Butyrate Production on Intestinal Barrier Function and Inflammation. Nutrients 2018, 10, 1499. [Google Scholar] [CrossRef]
- Ishizaka, A.; Koga, M.; Mizutani, T.; Parbie, P.K.; Prawisuda, D.; Yusa, N.; Sedohara, A.; Kikuchi, T.; Ikeuchi, K.; Adachi, E. Unique gut microbiome in HIV patients on antiretroviral therapy (ART) suggests association with chronic inflammation. Microbiol. Spectr. 2021, 9, e00708–e00721. [Google Scholar] [CrossRef]
- Sencio, V.; Barthelemy, A.; Tavares, L.P.; Machado, M.G.; Soulard, D.; Cuinat, C.; Queiroz-Junior, C.M.; Noordine, M.-L.; Salomé-Desnoulez, S.; Deryuter, L. Gut dysbiosis during influenza contributes to pulmonary pneumococcal superinfection through altered short-chain fatty acid production. Cell Rep. 2020, 30, 2934–2947.e2936. [Google Scholar] [CrossRef]
- Bernard-Raichon, L.; Venzon, M.; Klein, J.; Axelrad, J.E.; Zhang, C.; Sullivan, A.P.; Hussey, G.A.; Casanovas-Massana, A.; Noval, M.G.; Valero-Jimenez, A.M. Gut microbiome dysbiosis in antibiotic-treated COVID-19 patients is associated with microbial translocation and bacteremia. Nat. Commun. 2022, 13, 5926. [Google Scholar] [CrossRef]
- Zhu, W.; Xu, X.; Leger, A.S.; Almaghrabi, F.; Yamane, R.H.; Caspi, R.R. Toll-like receptor 2 signaling in γδ T cells is required for immune responses to commensal bacteria at the ocular surface. Investig. Ophthalmol. Vis. Sci. 2021, 62, 2237. [Google Scholar]
- Walker, E.M.; Slisarenko, N.; Gerrets, G.L.; Grasperge, B.F.; Mattison, J.A.; Kissinger, P.J.; Welsh, D.A.; Veazey, R.S.; Jazwinski, S.M.; Rout, N. Dysregulation of IL-17/IL-22 Effector functions in blood and gut mucosal gamma delta T cells correlates with increase in circulating leaky gut and inflammatory markers during cART-treated chronic SIV infection in macaques. Front. Immunol. 2021, 12, 647398. [Google Scholar] [CrossRef] [PubMed]
- Hedges, J.F.; Lubick, K.J.; Jutila, M.A. γδ T cells respond directly to pathogen-associated molecular patterns. J. Immunol. 2005, 174, 6045–6053. [Google Scholar] [CrossRef] [PubMed]
- Sencio, V.; Gallerand, A.; Gomes Machado, M.; Deruyter, L.; Heumel, S.; Soulard, D.; Barthelemy, J.; Cuinat, C.; Vieira, A.T.; Barthelemy, A. Influenza virus infection impairs the gut’s barrier properties and favors secondary enteric bacterial infection through reduced production of short-chain fatty acids. Infect. Immun. 2021, 89, e0073420. [Google Scholar] [CrossRef] [PubMed]
- Sencio, V.; Machelart, A.; Robil, C.; Benech, N.; Hoffmann, E.; Galbert, C.; Deryuter, L.; Heumel, S.; Hantute-Ghesquier, A.; Flourens, A. Alteration of the gut microbiota following SARS-CoV-2 infection correlates with disease severity in hamsters. Gut Microbes 2022, 14, 2018900. [Google Scholar] [CrossRef]
- Vodnar, D.-C.; Mitrea, L.; Teleky, B.-E.; Szabo, K.; Călinoiu, L.-F.; Nemeş, S.-A.; Martău, G.-A. Coronavirus disease (COVID-19) caused by (SARS-CoV-2) infections: A real challenge for human gut microbiota. Front. Cell. Infect. Microbiol. 2020, 10, 575559. [Google Scholar] [CrossRef]
- Ragonnaud, E.; Biragyn, A. Gut microbiota as the key controllers of “healthy” aging of elderly people. Immun. Ageing 2021, 18, 2. [Google Scholar] [CrossRef]
- Rocafort, M.; Noguera-Julian, M.; Rivera, J.; Pastor, L.; Guillén, Y.; Langhorst, J.; Parera, M.; Mandomando, I.; Carrillo, J.; Urrea, V. Evolution of the gut microbiome following acute HIV-1 infection. Microbiome 2019, 7, 73. [Google Scholar] [CrossRef]
- Tanes, C.; Walker, E.M.; Slisarenko, N.; Gerrets, G.L.; Grasperge, B.F.; Qin, X.; Jazwinski, S.M.; Bushman, F.D.; Bittinger, K.; Rout, N. Gut microbiome changes associated with epithelial barrier damage and systemic inflammation during antiretroviral therapy of chronic SIV infection. Viruses 2021, 13, 1567. [Google Scholar] [CrossRef]
- Lazar, V.; Ditu, L.-M.; Pircalabioru, G.G.; Gheorghe, I.; Curutiu, C.; Holban, A.M.; Picu, A.; Petcu, L.; Chifiriuc, M.C. Aspects of gut microbiota and immune system interactions in infectious diseases, immunopathology, and cancer. Front. Immunol. 2018, 9, 1830. [Google Scholar] [CrossRef]
- Li, Y.; Ye, Z.; Zhu, J.; Fang, S.; Meng, L.; Zhou, C. Effects of gut microbiota on host adaptive immunity under immune homeostasis and tumor pathology state. Front. Immunol. 2022, 13, 844335. [Google Scholar] [CrossRef]
- Wiertsema, S.P.; van Bergenhenegouwen, J.; Garssen, J.; Knippels, L.M. The interplay between the gut microbiome and the immune system in the context of infectious diseases throughout life and the role of nutrition in optimizing treatment strategies. Nutrients 2021, 13, 886. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Tan, H.; Kaiko, G.E. Role of the intestinal epithelium and its interaction with the microbiota in food allergy. Front. Immunol. 2020, 11, 604054. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Bris, R.; Saez, A.; Herrero-Fernandez, B.; Rius, C.; Sanchez-Martinez, H.; Gonzalez-Granado, J.M. CD4 T-Cell Subsets and the Pathophysiology of Inflammatory Bowel Disease. Int. J. Mol. Sci. 2023, 24, 2696. [Google Scholar] [CrossRef] [PubMed]
- Cox, J.R.; Cruickshank, S.M.; Saunders, A.E. Maintenance of barrier tissue integrity by unconventional lymphocytes. Front. Immunol. 2021, 12, 670471. [Google Scholar] [CrossRef]
- Seo, G.-Y.; Giles, D.A.; Kronenberg, M. The role of innate lymphoid cells in response to microbes at mucosal surfaces. Mucosal Immunol. 2020, 13, 399–412. [Google Scholar] [CrossRef]
- Zhou, A.; Yuan, Y.; Yang, M.; Huang, Y.; Li, X.; Li, S.; Yang, S.; Tang, B. Crosstalk between the gut microbiota and epithelial cells under physiological and infectious conditions. Front. Cell. Infect. Microbiol. 2022, 12, 23. [Google Scholar] [CrossRef]
- Lee, J.Y.; Tsolis, R.M.; Baumler, A.J. The microbiome and gut homeostasis. Science 2022, 377, eabp9960. [Google Scholar] [CrossRef]
- Zhang, E.; Fang, M.; Jones, C.; Minze, L.J.; Xing, J.; Zhang, Z. Mechanisms involved in controlling RNA virus-induced intestinal inflammation. Cell. Mol. Life Sci. 2022, 79, 313. [Google Scholar] [CrossRef]
- Mantis, N.J.; Forbes, S.J. Secretory IgA: Arresting microbial pathogens at epithelial borders. Immunol. Investig. 2010, 39, 383–406. [Google Scholar] [CrossRef]
- Gury-BenAri, M.; Thaiss, C.A.; Serafini, N.; Winter, D.R.; Giladi, A.; Lara-Astiaso, D.; Levy, M.; Salame, T.M.; Weiner, A.; David, E. The spectrum and regulatory landscape of intestinal innate lymphoid cells are shaped by the microbiome. Cell 2016, 166, 1231–1246.e13. [Google Scholar] [CrossRef] [PubMed]
- Constantinides, M.G.; McDonald, B.D.; Verhoef, P.A.; Bendelac, A. A committed precursor to innate lymphoid cells. Nature 2014, 508, 397–401. [Google Scholar] [CrossRef] [PubMed]
- Korchagina, A.A.; Koroleva, E.; Tumanov, A.V. Innate lymphoid cell plasticity in mucosal infections. Microorganisms 2023, 11, 461. [Google Scholar] [CrossRef]
- Streicher, F.; Jouvenet, N. Stimulation of innate immunity by host and viral RNAs. Trends Immunol. 2019, 40, 1134–1148. [Google Scholar] [CrossRef]
- Caron, J.; Ridgley, L.A.; Bodman-Smith, M. How to train your dragon: Harnessing gamma delta T cells antiviral functions and trained immunity in a pandemic era. Front. Immunol. 2021, 12, 666983. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Liang, H.; Hong, K.; Li, H.; Peng, H.; Zhao, Y.; Jia, M.; Ruan, Y.; Shao, Y. The potential role of CD16+ Vγ2Vδ2 T cell-mediated antibody-dependent cell-mediated cytotoxicity in control of HIV type 1 disease. AIDS Res. Hum. Retrovir. 2013, 29, 1562–1570. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xiang, Z.; Feng, T.; Li, J.; Liu, Y.; Fan, Y.; Lu, Q.; Yin, Z.; Yu, M.; Shen, C. Human Vγ9Vδ2-T cells efficiently kill influenza virus-infected lung alveolar epithelial cells. Cell. Mol. Immunol. 2013, 10, 159–164. [Google Scholar] [CrossRef]
- Makaremi, S.; Asgarzadeh, A.; Kianfar, H.; Mohammadnia, A.; Asghariazar, V.; Safarzadeh, E. The role of IL-1 family of cytokines and receptors in pathogenesis of COVID-19. Inflamm. Res. 2022, 71, 923–947. [Google Scholar] [CrossRef]
- Rezende, R.M.; Cox, L.M.; Moreira, T.G.; Liu, S.; Boulenouar, S.; Dhang, F.; LeServe, D.S.; Nakagaki, B.N.; Lopes, J.R.; Tatematsu, B.K.; et al. Gamma-delta T cells modulate the microbiota and fecal micro-RNAs to maintain mucosal tolerance. Microbiome 2023, 11, 32. [Google Scholar] [CrossRef]
- Anand, N.; Gorantla, V.R.; Chidambaram, S.B. The Role of Gut Dysbiosis in the Pathophysiology of Neuropsychiatric Disorders. Cells 2023, 12, 54. [Google Scholar] [CrossRef]
- Crakes, K.R.; Jiang, G. Gut microbiome alterations during HIV/SIV infection: Implications for HIV cure. Front. Microbiol. 2019, 10, 1104. [Google Scholar] [CrossRef] [PubMed]
- Le Hingrat, Q.; Sette, P.; Xu, C.; Rahmberg, A.R.; Tarnus, L.; Annapureddy, H.; Kleinman, A.; Brocca-Cofano, E.; Sivanandham, R.; Sivanandham, S. Prolonged experimental CD4+ T-cell depletion does not cause disease progression in SIV-infected African green monkeys. Nat. Commun. 2023, 14, 979. [Google Scholar] [CrossRef]
- Alzahrani, J.; Hussain, T.; Simar, D.; Palchaudhuri, R.; Abdel-Mohsen, M.; Crowe, S.M.; Mbogo, G.W.; Palmer, C.S. Inflammatory and immunometabolic consequences of gut dysfunction in HIV: Parallels with IBD and implications for reservoir persistence and non-AIDS comorbidities. eBioMedicine 2019, 46, 522–531. [Google Scholar] [CrossRef] [PubMed]
- Mazzuti, L.; Turriziani, O.; Mezzaroma, I. The Many Faces of Immune Activation in HIV-1 Infection: A Multifactorial Interconnection. Biomedicines 2023, 11, 159. [Google Scholar] [CrossRef]
- Dillon, S.M.; Manuzak, J.A.; Leone, A.K.; Lee, E.J.; Rogers, L.M.; McCarter, M.D.; Wilson, C.C. HIV-1 infection of human intestinal lamina propria CD4+ T cells in vitro is enhanced by exposure to commensal Escherichia coli. J. Immunol. 2012, 189, 885–896. [Google Scholar] [CrossRef]
- Ling, B.; Mohan, M.; Lackner, A.A.; Green, L.C.; Marx, P.A.; Doyle, L.A.; Veazey, R.S. The large intestine as a major reservoir for simian immunodeficiency virus in macaques with long-term, nonprogressing infection. J. Infect. Dis. 2010, 202, 1846–1854. [Google Scholar] [CrossRef]
- Hensley-McBain, T.; Berard, A.R.; Manuzak, J.A.; Miller, C.J.; Zevin, A.S.; Polacino, P.; Gile, J.; Agricola, B.; Cameron, M.; Hu, S.-L. Intestinal damage precedes mucosal immune dysfunction in SIV infection. Mucosal Immunol. 2018, 11, 1429–1440. [Google Scholar] [CrossRef]
- Salinas, T.R.W.; Gosselin, A.; Marchand, L.R.; Gabriel, E.M.; Tastet, O.; Goulet, J.-P.; Zhang, Y.; Vlad, D.; Touil, H.; Routy, J.-P. IL-17A reprograms intestinal epithelial cells to facilitate HIV-1 replication and outgrowth in CD4+ T cells. iScience 2021, 24, 103225. [Google Scholar] [CrossRef]
- Vujkovic-Cvijin, I.; Dunham, R.M.; Iwai, S.; Maher, M.C.; Albright, R.G.; Broadhurst, M.J.; Hernandez, R.D.; Lederman, M.M.; Huang, Y.; Somsouk, M. Dysbiosis of the gut microbiota is associated with HIV disease progression and tryptophan catabolism. Sci. Transl. Med. 2013, 5, 193ra91. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhang, Y.; Cui, P.; Luo, L.; Chen, H.; Liang, B.; Jiang, J.; Ning, C.; Tian, L.; Zhong, X. Gut microbiome changes associated with HIV infection and sexual orientation. Front. Cell. Infect. Microbiol. 2020, 10, 434. [Google Scholar] [CrossRef]
- Parbie, P.K.; Mizutani, T.; Ishizaka, A.; Kawana-Tachikawa, A.; Runtuwene, L.R.; Seki, S.; Abana, C.Z.-Y.; Kushitor, D.; Bonney, E.Y.; Ofori, S.B. Dysbiotic fecal microbiome in HIV-1 infected individuals in Ghana. Front. Cell. Infect. Microbiol. 2021, 11, 646467. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.D.; Byrareddy, S.N. HIV-associated dysbiosis and immune recovery during antiretroviral therapy. Clin. Transl. Discov. 2022, 2, e58. [Google Scholar] [CrossRef]
- Ashuro, A.A.; Lobie, T.A.; Ye, D.-Q.; Leng, R.-X.; Li, B.-Z.; Pan, H.-F.; Fan, Y.-G. Review on the alteration of gut microbiota: The role of HIV infection and old age. AIDS Res. Hum. Retrovir. 2020, 36, 556–565. [Google Scholar] [CrossRef]
- Chachage, M. The gut-microbiome contribution to HIV-associated cardiovascular disease and metabolic disorders. Curr. Opin. Endocr. Metab. Res. 2021, 21, 100287. [Google Scholar] [CrossRef]
- Kaur, U.S.; Shet, A.; Rajnala, N.; Gopalan, B.P.; Moar, P.; Singh, B.P.; Chaturvedi, R.; Tandon, R. High Abundance of genus Prevotella in the gut of perinatally HIV-infected children is associated with IP-10 levels despite therapy. Sci. Rep. 2018, 8, 17679. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, A.J.; Shaffer, M.; Nusbacher, N.M.; Griesmer, C.; Fiorillo, S.; Schneider, J.M.; Preston Neff, C.; Li, S.X.; Fontenot, A.P.; Campbell, T. An exploration of Prevotella-rich microbiomes in HIV and men who have sex with men. Microbiome 2018, 6, 198. [Google Scholar] [CrossRef] [PubMed]
- Dubourg, G.; Lagier, J.C.; Hue, S.; Surenaud, M.; Bachar, D.; Robert, C.; Michelle, C.; Ravaux, I.; Mokhtari, S.; Million, M.; et al. Gut microbiota associated with HIV infection is significantly enriched in bacteria tolerant to oxygen. BMJ Open Gastroenterol. 2016, 3, e000080. [Google Scholar] [CrossRef] [PubMed]
- Littlefield, K.M.; Schneider, J.M.; Neff, C.P.; Soesanto, V.; Siebert, J.C.; Nusbacher, N.M.; Moreno-Huizar, N.; Cartwright, I.M.; Armstrong, A.J.; Colgen, S.P. Elevated inflammatory fecal immune factors in men who have sex with men with HIV associate with microbiome composition and gut barrier function. Front. Immunol. 2022, 13, 1072720. [Google Scholar] [CrossRef]
- Mudd, J.C.; Brenchley, J.M. Gut mucosal barrier dysfunction, microbial dysbiosis, and their role in HIV-1 disease progression. J. Infect. Dis. 2016, 214, S58–S66. [Google Scholar] [CrossRef]
- Li, S.X.; Armstrong, A.J.; Neff, C.P.; Shaffer, M.; Lozupone, C.A.; Palmer, B.E. Complexities of gut microbiome dysbiosis in the context of HIV infection and antiretroviral therapy. Clin. Pharmacol. Ther. 2016, 99, 600–611. [Google Scholar] [CrossRef]
- D’arc, M.; Furtado, C.; Siqueira, J.D.; Seuánez, H.N.; Ayouba, A.; Peeters, M.; Soares, M.A. Assessment of the gorilla gut virome in association with natural simian immunodeficiency virus infection. Retrovirology 2018, 15, 19. [Google Scholar] [CrossRef]
- Onabajo, O.O.; Mattapallil, J.J. Gut microbiome homeostasis and the CD4 T-follicular helper cell IgA axis in human immunodeficiency virus infection. Front. Immunol. 2021, 12, 657679. [Google Scholar] [CrossRef]
- Wang, J.; Li, F.; Wei, H.; Lian, Z.X.; Sun, R.; Tian, Z. Respiratory influenza virus infection induces intestinal immune injury via microbiota-mediated Th17 cell–dependent inflammation. J. Exp. Med. 2014, 211, 2397–2410. [Google Scholar] [CrossRef]
- Yildiz, S.; Mazel-Sanchez, B.; Kandasamy, M.; Manicassamy, B.; Schmolke, M. Influenza A virus infection impacts systemic microbiota dynamics and causes quantitative enteric dysbiosis. Microbiome 2018, 6, 9. [Google Scholar] [CrossRef] [PubMed]
- Deriu, E.; Boxx, G.M.; He, X.; Pan, C.; Benavidez, S.D.; Cen, L.; Rozengurt, N.; Shi, W.; Cheng, G. Influenza virus affects intestinal microbiota and secondary salmonella infection in the gut through type I interferons. PLoS Pathog. 2016, 12, e1005572. [Google Scholar] [CrossRef] [PubMed]
- Qin, N.; Zheng, B.; Yao, J.; Guo, L.; Zuo, J.; Wu, L.; Zhou, J.; Liu, L.; Guo, J.; Ni, S. Influence of H7N9 virus infection and associated treatment on human gut microbiota. Sci. Rep. 2015, 5, 14771. [Google Scholar] [CrossRef]
- Bartley, J.M.; Zhou, X.; Kuchel, G.A.; Weinstock, G.M.; Haynes, L. Impact of age, caloric restriction, and influenza infection on mouse gut microbiome: An exploratory study of the role of age-related microbiome changes on influenza responses. Front. Immunol. 2017, 8, 1164. [Google Scholar] [CrossRef]
- Groves, H.T.; Cuthbertson, L.; James, P.; Moffatt, M.F.; Cox, M.J.; Tregoning, J.S. Respiratory disease following viral lung infection alters the murine gut microbiota. Front. Immunol. 2018, 9, 182. [Google Scholar] [CrossRef]
- Luu, M.; Monning, H.; Visekruna, A. Exploring the molecular mechanisms underlying the protective effects of microbial SCFAs on intestinal tolerance and food allergy. Front. Immunol. 2020, 11, 1225. [Google Scholar] [CrossRef] [PubMed]
- Ichinohe, T.; Pang, I.K.; Kumamoto, Y.; Peaper, D.R.; Ho, J.H.; Murray, T.S.; Iwasaki, A. Microbiota regulates immune defense against respiratory tract influenza A virus infection. Proc. Natl. Acad. Sci. USA 2011, 108, 5354–5359. [Google Scholar] [CrossRef]
- Hanada, S.; Pirzadeh, M.; Carver, K.Y.; Deng, J.C. Respiratory viral infection-induced microbiome alterations and secondary bacterial pneumonia. Front. Immunol. 2018, 9, 2640. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.S.; Wang, J.; Yannie, P.J.; Ghosh, S. Intestinal barrier dysfunction, LPS translocation, and disease development. J. Endocr. Soc. 2020, 4, bvz039. [Google Scholar] [CrossRef]
- Mifsud, E.J.; Kuba, M.; Barr, I.G. Innate immune responses to influenza virus infections in the upper respiratory tract. Viruses 2021, 13, 2090. [Google Scholar] [CrossRef]
- Alqazlan, N.; Alizadeh, M.; Boodhoo, N.; Taha-Abdelaziz, K.; Nagy, E.; Bridle, B.; Sharif, S. Probiotic lactobacilli limit avian influenza virus subtype H9N2 replication in chicken cecal tonsil mononuclear cells. Vaccines 2020, 8, 605. [Google Scholar] [CrossRef]
- Shi, H.Y.; Zhu, X.; Li, W.L.; Mak, J.W.; Wong, S.H.; Zhu, S.T.; Guo, S.L.; Chan, F.K.; Zhang, S.T.; Ng, S.C. Modulation of gut microbiota protects against viral respiratory tract infections: A systematic review of animal and clinical studies. Eur. J. Nutr. 2021, 60, 4151–4174. [Google Scholar] [CrossRef]
- Azimirad, M.; Noori, M.; Raeisi, H.; Yadegar, A.; Shahrokh, S.; Asadzadeh Aghdaei, H.; Bentivegna, E.; Martelletti, P.; Petrosillo, N.; Zali, M.R. How does COVID-19 pandemic impact on incidence of Clostridioides difficile infection and exacerbation of its gastrointestinal symptoms? Front. Med. 2021, 8, 775063. [Google Scholar] [CrossRef]
- Jackson, C.B.; Farzan, M.; Chen, B.; Choe, H. Mechanisms of SARS-CoV-2 entry into cells. Nat. Rev. Mol. Cell Biol. 2022, 23, 3–20. [Google Scholar] [CrossRef]
- Ni, W.; Yang, X.; Yang, D.; Bao, J.; Li, R.; Xiao, Y.; Hou, C.; Wang, H.; Liu, J.; Yang, D. Role of angiotensin-converting enzyme 2 (ACE2) in COVID-19. Crit. Care 2020, 24, 422. [Google Scholar] [CrossRef] [PubMed]
- Alipoor, S.; Mortaz, E.; Jamaati, H.; Tabarsi, P.; Bayram, H.; Varahram, M.; Adcock, I. COVID-19: Molecular and cellular response. Front. Cell. Infect. Microbiol. 2021, 11, 563085. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Liu, J.; Zhang, D.; Xu, Z.; Ji, J.; Wen, C. Cytokine storm in COVID-19: The current evidence and treatment strategies. Front. Immunol. 2020, 11, 1708. [Google Scholar] [CrossRef]
- Yeoh, Y.K.; Zuo, T.; Lui, G.C.-Y.; Zhang, F.; Liu, Q.; Li, A.Y.; Chung, A.C.; Cheung, C.P.; Tso, E.Y.; Fung, K.S. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut 2021, 70, 698–706. [Google Scholar] [CrossRef]
- Zuo, T.; Zhang, F.; Lui, G.C.; Yeoh, Y.K.; Li, A.Y.; Zhan, H.; Wan, Y.; Chung, A.C.; Cheung, C.P.; Chen, N. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology 2020, 159, 944–955.e948. [Google Scholar] [CrossRef]
- Gou, W.; Fu, Y.; Yue, L.; Chen, G.-d.; Cai, X.; Shuai, M.; Xu, F.; Yi, X.; Chen, H.; Zhu, Y. Gut microbiota may underlie the predisposition of healthy individuals to COVID-19. medRxiv 2020. [Google Scholar] [CrossRef]
- Ribeiro, I.P.; Nascimento, L.G.d.; Tort, L.F.L.; Pereira, E.C.; Menezes, L.S.R.; Malta, F.C.; Oliveira, B.C.E.P.D.d.; Rodrigues, J.P.; Manso, P.P.d.A.; Pelajo, M. Infectious SARS-CoV-2 Particles from Rectal Swab Samples from COVID-19 Patients in Brazil. Viruses 2023, 15, 1152. [Google Scholar] [CrossRef]
- Qian, Q.; Fan, L.; Liu, W.; Li, J.; Yue, J.; Wang, M.; Ke, X.; Yin, Y.; Chen, Q.; Jiang, C. Direct evidence of active SARS-CoV-2 replication in the intestine. Clin. Infect. Dis. 2021, 73, 361–366. [Google Scholar] [CrossRef]
- Mizutani, T.; Ishizaka, A.; Koga, M.; Tsutsumi, T.; Yotsuyanagi, H. Role of microbiota in viral infections and pathological progression. Viruses 2022, 14, 950. [Google Scholar] [CrossRef]
- Torres, N.I.; Noll, K.S.; Xu, S.; Li, J.; Huang, Q.; Sinko, P.J.; Wachsman, M.B.; Chikindas, M.L. Safety, formulation and in vitro antiviral activity of the antimicrobial peptide subtilosin against herpes simplex virus type 1. Probiotics Antimicrob. Proteins 2013, 5, 26–35. [Google Scholar] [CrossRef]
- Quintana, V.M.; Torres, N.I.; Wachsman, M.B.; Sinko, P.J.; Castilla, V.; Chikindas, M. Antiherpes simplex virus type 2 activity of the antimicrobial peptide subtilosin. J. Appl. Microbiol. 2014, 117, 1253–1259. [Google Scholar] [CrossRef]
- Su, Y.; Zhang, B.; Su, L. CD4 detected from Lactobacillus helps understand the interaction between Lactobacillus and HIV. Microbiol. Res. 2013, 168, 273–277. [Google Scholar] [CrossRef]
- Botić, T.; Danø, T.; Weingartl, H.; Cencič, A. A novel eukaryotic cell culture model to study antiviral activity of potential probiotic bacteria. Int. J. Food Microbiol. 2007, 115, 227–234. [Google Scholar] [CrossRef]
- Alzahrani, O.R.; Hawsawi, Y.M.; Alanazi, A.D.; Alatwi, H.E.; Rather, I.A. In Vitro Evaluation of Leuconostoc mesenteroides Cell-Free-Supernatant GBUT-21 against SARS-CoV-2. Vaccines 2022, 10, 1581. [Google Scholar] [CrossRef]
- Jounai, K.; Ikado, K.; Sugimura, T.; Ano, Y.; Braun, J.; Fujiwara, D. Spherical lactic acid bacteria activate plasmacytoid dendritic cells immunomodulatory function via TLR9-dependent crosstalk with myeloid dendritic cells. PLoS ONE 2012, 7, e32588. [Google Scholar] [CrossRef]
- Satomi, S.; Kokubu, D.; Inoue, T.; Sugiyama, M.; Mizokami, M.; Suzuki, S.; Murata, K. Enterococcus casseliflavus KB1733 Isolated from a Traditional Japanese Pickle Induces Interferon-Lambda Production in Human Intestinal Epithelial Cells. Microorganisms 2022, 10, 827. [Google Scholar] [CrossRef]
- Kim, S.; Kim, M.-J.; Chung, H.J.; Kim, C.-H.; Yoon, J.-H.; Kim, H.J. Mitochondrial reactive oxygen species modulate innate immune response to influenza A virus in human nasal epithelium. Antivir. Res. 2015, 119, 78–83. [Google Scholar] [CrossRef]
- Ji, J.-j.; Sun, Q.-m.; Nie, D.-y.; Wang, Q.; Zhang, H.; Qin, F.-f.; Wang, Q.-s.; Lu, S.-f.; Pang, G.-m.; Lu, Z.-g. Probiotics protect against RSV infection by modulating the microbiota-alveolar-macrophage axis. Acta Pharmacol. Sin. 2021, 42, 1630–1641. [Google Scholar] [CrossRef]
- Bradley, K.C.; Finsterbusch, K.; Schnepf, D.; Crotta, S.; Llorian, M.; Davidson, S.; Fuchs, S.Y.; Staeheli, P.; Wack, A. Microbiota-driven tonic interferon signals in lung stromal cells protect from influenza virus infection. Cell Rep. 2019, 28, 245–256.e4. [Google Scholar] [CrossRef]
- Yitbarek, A.; Taha-Abdelaziz, K.; Hodgins, D.C.; Read, L.; Nagy, É.; Weese, J.S.; Caswell, J.L.; Parkinson, J.; Sharif, S. Gut microbiota-mediated protection against influenza virus subtype H9N2 in chickens is associated with modulation of the innate responses. Sci. Rep. 2018, 8, 13189. [Google Scholar] [CrossRef]
- Oh, J.E.; Kim, B.-C.; Chang, D.-H.; Kwon, M.; Lee, S.Y.; Kang, D.; Kim, J.Y.; Hwang, I.; Yu, J.-W.; Nakae, S. Dysbiosis-induced IL-33 contributes to impaired antiviral immunity in the genital mucosa. Proc. Natl. Acad. Sci. USA 2016, 113, E762–E771. [Google Scholar] [CrossRef]
- Gosmann, C.; Handley, S.A.; Farcasanu, M.; Abu-Ali, G.; Bowman, B.A.; Padavattan, N.; Desai, C.; Droit, L.; Moodley, A.; Dong, M. Lactobacillus-deficient cervicovaginal bacterial communities are associated with increased HIV acquisition in young South African women. Immunity 2017, 46, 29–37. [Google Scholar] [CrossRef]
- Ñahui Palomino, R.A.; Zicari, S.; Vanpouille, C.; Vitali, B.; Margolis, L. Vaginal Lactobacillus inhibits HIV-1 replication in human tissues ex vivo. Front. Microbiol. 2017, 8, 906. [Google Scholar] [CrossRef]
- Kerry, R.G.; Patra, J.K.; Gouda, S.; Park, Y.; Shin, H.-S.; Das, G. Benefaction of probiotics for human health: A review. J. Food Drug Anal. 2018, 26, 927–939. [Google Scholar] [CrossRef]
- Fulcher, J.A.; Li, F.; Tobin, N.H.; Zabih, S.; Elliott, J.; Clark, J.L.; D’Aquila, R.; Mustanski, B.; Kipke, M.D.; Shoptaw, S. Gut dysbiosis and inflammatory blood markers precede HIV with limited changes after early seroconversion. eBioMedicine 2022, 84, 104286. [Google Scholar] [CrossRef]
- Vakili, K.; Fathi, M.; Yaghoobpoor, S.; Sayehmiri, F.; Nazerian, Y.; Nazerian, A.; Mohamadkhani, A.; Khodabakhsh, P.; Réus, G.Z.; Hajibeygi, R. The contribution of gut-brain axis to development of neurological symptoms in COVID-19 recovered patients: A hypothesis and review of literature. Front. Cell. Infect. Microbiol. 2022, 12, 983089. [Google Scholar] [CrossRef]
- Rich, S.; Klann, E.; Bryant, V.; Richards, V.; Wijayabahu, A.; Bryant, K.; Mai, V.; Cook, R. A review of potential microbiome-gut-brain axis mediated neurocognitive conditions in persons living with HIV. Brain Behav. Immun. Health 2020, 9, 100168. [Google Scholar] [CrossRef]
- Fung, T.C.; Olson, C.A.; Hsiao, E.Y. Interactions between the microbiota, immune and nervous systems in health and disease. Nat. Neurosci. 2017, 20, 145–155. [Google Scholar] [CrossRef]
- Ray, S.; Sil, S.; Kannan, M.; Periyasamy, P.; Buch, S. Role of the gut-brain axis in HIV and drug abuse-mediated neuroinflammation. Adv. Drug Alcohol Res. 2023, 3, 11092. [Google Scholar] [CrossRef]
- Mudra Rakshasa-Loots, A.; Whalley, H.C.; Vera, J.H.; Cox, S.R. Neuroinflammation in HIV-associated depression: Evidence and future perspectives. Mol. Psychiatry 2022, 27, 3619–3632. [Google Scholar] [CrossRef]
- Mao, L.; Jin, H.; Wang, M.; Hu, Y.; Chen, S.; He, Q.; Chang, J.; Hong, C.; Zhou, Y.; Wang, D. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020, 77, 683–690. [Google Scholar] [CrossRef]
- Kobiyama, K.; Ley, K. Atherosclerosis: A chronic inflammatory disease with an autoimmune component. Circ. Res. 2018, 123, 1118–1120. [Google Scholar] [CrossRef]
- Li, L.; Peng, P.; Ding, N.; Jia, W.; Huang, C.; Tang, Y. Oxidative Stress, Inflammation, Gut Dysbiosis: What Can Polyphenols Do in Inflammatory Bowel Disease? Antioxidants 2023, 12, 967. [Google Scholar] [CrossRef] [PubMed]
- Novakovic, M.; Rout, A.; Kingsley, T.; Kirchoff, R.; Singh, A.; Verma, V.; Kant, R.; Chaudhary, R. Role of gut microbiota in cardiovascular diseases. World J. Cardiol. 2020, 12, 110. [Google Scholar] [CrossRef] [PubMed]
- Perkins, M.V.; Joseph, S.B.; Dittmer, D.P.; Mackman, N. Cardiovascular disease and thrombosis in HIV infection. Arterioscler. Thromb. Vasc. Biol. 2023, 43, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Srinivasa, S.; Fitch, K.V.; Lo, J.; Kadar, H.; Knight, R.; Wong, K.; Abbara, S.; Gauguier, D.; Capeau, J.; Boccara, F. Plaque burden in HIV-infected patients is associated with serum intestinal microbiota-generated trimethylamine. AIDS 2015, 29, 443. [Google Scholar] [CrossRef] [PubMed]
- Zilberman-Schapira, G.; Zmora, N.; Itav, S.; Bashiardes, S.; Elinav, H.; Elinav, E. The gut microbiome in human immunodeficiency virus infection. BMC Med. 2016, 14, 83. [Google Scholar] [CrossRef]
- Ullah Goraya, M.; Li, R.; Gu, L.; Deng, H.; Wang, G. Blood Stream Microbiota Dysbiosis Establishing New Research Standards in Cardio-Metabolic Diseases, A Meta-Analysis Study. Microorganisms 2023, 11, 777. [Google Scholar] [CrossRef]
- Medina-Leyte, D.J.; Zepeda-García, O.; Domínguez-Pérez, M.; González-Garrido, A.; Villarreal-Molina, T.; Jacobo-Albavera, L. Endothelial dysfunction, inflammation and coronary artery disease: Potential biomarkers and promising therapeutical approaches. Int. J. Mol. Sci. 2021, 22, 3850. [Google Scholar] [CrossRef]
- Fang, J.; Yu, C.-H.; Li, X.-J.; Yao, J.-M.; Fang, Z.-Y.; Yoon, S.-H.; Yu, W.-Y. Gut dysbiosis in nonalcoholic fatty liver disease: Pathogenesis, diagnosis, and therapeutic implications. Front. Cell. Infect. Microbiol. 2022, 12, 997018. [Google Scholar] [CrossRef]
- El-Mowafy, M.; Elgaml, A.; El-Mesery, M.; Sultan, S.; Ahmed, T.A.; Gomaa, A.I.; Aly, M.; Mottawea, W. Changes of gut-microbiota-liver axis in hepatitis C virus infection. Biology 2021, 10, 55. [Google Scholar] [CrossRef]
- Jian, Z.; Zeng, L.; Xu, T.; Sun, S.; Yan, S.; Zhao, S.; Su, Z.; Ge, C.; Zhang, Y.; Jia, J. The intestinal microbiome associated with lipid metabolism and obesity in humans and animals. J. Appl. Microbiol. 2022, 133, 2915–2930. [Google Scholar] [CrossRef]
- Han, W.; Huang, C.; Ji, Y.; Zhou, L.; Chen, J.; Hou, J. Alterations in the Gut Microbiota and Hepatitis-B-Virus Infection in Southern Chinese Patients with Coexisting Non-Alcoholic Fatty Liver Disease and Type-2 Diabetes Mellitus. Front. Med. 2021, 8, 2608. [Google Scholar] [CrossRef] [PubMed]
- Saltzman, E.T.; Palacios, T.; Thomsen, M.; Vitetta, L. Intestinal microbiome shifts, dysbiosis, inflammation, and non-alcoholic fatty liver disease. Front. Microbiol. 2018, 9, 61. [Google Scholar] [CrossRef]
- Seth, A.; Sherman, K.E. Fatty liver disease in persons with HIV infection. Top. Antivir. Med. 2019, 27, 75. [Google Scholar]
- Milic, J.; Barbieri, S.; Gozzi, L.; Brigo, A.; Beghé, B.; Verduri, A.; Bacca, E.; Iadisernia, V.; Cuomo, G.; Dolci, G. Metabolic-Associated Fatty Liver Disease Is Highly Prevalent in the Postacute COVID Syndrome. Open Forum Infect. Dis. 2022, 9, ofac003. [Google Scholar] [CrossRef] [PubMed]
- Miller, H.; Ferris, R.; Phelps, B. The effect of probiotics on CD4 counts among people living with HIV: A systematic review. Benef. Microbes 2016, 7, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Yeh, C.; Jin, Z.; Ding, L.; Liu, B.Y.; Zhang, L.; Dannelly, H.K. Prospective study of probiotic supplementation results in immune stimulation and improvement of upper respiratory infection rate. Synth. Syst. Biotechnol. 2018, 3, 113–120. [Google Scholar] [CrossRef]
- Fujii, T.; Jounai, K.; Horie, A.; Takahashi, H.; Suzuki, H.; Ohshio, K.; Fujiwara, D.; Yamamoto, N. Effects of heat-killed Lactococcus lactis subsp. lactis JCM 5805 on mucosal and systemic immune parameters, and antiviral reactions to influenza virus in healthy adults; a randomized controlled double-blind study. J. Funct. Foods 2017, 35, 513–521. [Google Scholar] [CrossRef]
- Chai, W.; Burwinkel, M.; Wang, Z.; Palissa, C.; Esch, B.; Twardziok, S.; Rieger, J.; Wrede, P.; Schmidt, M.F. Antiviral effects of a probiotic Enterococcus faecium strain against transmissible gastroenteritis coronavirus. Arch. Virol. 2013, 158, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.V.J.; Seo, B.J.; Mun, M.R.; Kim, C.-J.; Lee, I.; Kim, H.; Park, Y.-H. Putative probiotic Lactobacillus spp. from porcine gastrointestinal tract inhibit transmissible gastroenteritis coronavirus and enteric bacterial pathogens. Trop. Anim. Health Prod. 2010, 42, 1855–1860. [Google Scholar]
- Park, S.-Y.; Seo, G.S. Fecal microbiota transplantation: Is it safe? Clin. Endosc. 2021, 54, 157–160. [Google Scholar] [CrossRef]
- Serrano-Villar, S.; Talavera-Rodríguez, A.; Gosalbes, M.J.; Madrid, N.; Pérez-Molina, J.A.; Elliott, R.J.; Navia, B.; Lanza, V.F.; Vallejo, A.; Osman, M. Fecal microbiota transplantation in HIV: A pilot placebo-controlled study. Nat. Commun. 2021, 12, 1139. [Google Scholar] [CrossRef] [PubMed]
- Utay, N.S.; Monczor, A.N.; Somasunderam, A.; Lupo, S.; Jiang, Z.-D.; Alexander, A.S.; Finkelman, M.; Vigil, K.J.; Lake, J.E.; Hanson, B. Evaluation of six weekly oral fecal microbiota transplants in people with HIV. Pathog. Immun. 2020, 5, 364. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Chen, H.; Xu, L.; Li, S.; Yan, H.; Jiang, L.; Cheng, W.; Jiang, Z. Effects of Intestinal Microorganisms on Influenza-Infected Mice with Antibiotic-Induced Intestinal Dysbiosis, through the TLR7 Signaling Pathway. Front. Biosci.-Landmark 2023, 28, 43. [Google Scholar] [CrossRef]
- Michailidis, L.; Currier, A.C.; Le, M.; Flomenhoft, D.R. Adverse events of fecal microbiota transplantation: A meta-analysis of high-quality studies. Ann. Gastroenterol. 2021, 34, 802. [Google Scholar] [PubMed]
- Bilinski, J.; Winter, K.; Jasinski, M.; Szczes, A.; Bilinska, N.; Mullish, B.H.; Malecka-Panas, E.; Basak, G.W. Rapid resolution of COVID-19 after faecal microbiota transplantation. Gut 2022, 71, 230–232. [Google Scholar] [CrossRef]
- Cammarota, G.; Ianiro, G.; Tilg, H.; Rajilić-Stojanović, M.; Kump, P.; Satokari, R.; Sokol, H.; Arkkila, P.; Pintus, C.; Hart, A. European consensus conference on faecal microbiota transplantation in clinical practice. Gut 2017, 66, 569–580. [Google Scholar] [CrossRef]
- Mcilroy, J.R.; Segal, J.P.; Mullish, B.H.; Quraishi, M.N.; Gasbarrini, A.; Cammarota, G.; Ianiro, G. Current and future targets for faecal microbiota transplantation. Hum. Microbiome J. 2019, 11, 100045. [Google Scholar] [CrossRef]
- Hrncir, T.; Hrncirova, L.; Kverka, M.; Hromadka, R.; Machova, V.; Trckova, E.; Kostovcikova, K.; Kralickova, P.; Krejsek, J.; Tlaskalova-Hogenova, H. Gut microbiota and NAFLD: Pathogenetic mechanisms, microbiota signatures, and therapeutic interventions. Microorganisms 2021, 9, 957. [Google Scholar] [CrossRef]
- Xue, L.; Deng, Z.; Luo, W.; He, X.; Chen, Y. Effect of fecal microbiota transplantation on non-alcoholic fatty liver disease: A randomized clinical trial. Front. Cell. Infect. Microbiol. 2022, 12, 759306. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fakharian, F.; Thirugnanam, S.; Welsh, D.A.; Kim, W.-K.; Rappaport, J.; Bittinger, K.; Rout, N. The Role of Gut Dysbiosis in the Loss of Intestinal Immune Cell Functions and Viral Pathogenesis. Microorganisms 2023, 11, 1849. https://doi.org/10.3390/microorganisms11071849
Fakharian F, Thirugnanam S, Welsh DA, Kim W-K, Rappaport J, Bittinger K, Rout N. The Role of Gut Dysbiosis in the Loss of Intestinal Immune Cell Functions and Viral Pathogenesis. Microorganisms. 2023; 11(7):1849. https://doi.org/10.3390/microorganisms11071849
Chicago/Turabian StyleFakharian, Farzaneh, Siva Thirugnanam, David A. Welsh, Woong-Ki Kim, Jay Rappaport, Kyle Bittinger, and Namita Rout. 2023. "The Role of Gut Dysbiosis in the Loss of Intestinal Immune Cell Functions and Viral Pathogenesis" Microorganisms 11, no. 7: 1849. https://doi.org/10.3390/microorganisms11071849
APA StyleFakharian, F., Thirugnanam, S., Welsh, D. A., Kim, W.-K., Rappaport, J., Bittinger, K., & Rout, N. (2023). The Role of Gut Dysbiosis in the Loss of Intestinal Immune Cell Functions and Viral Pathogenesis. Microorganisms, 11(7), 1849. https://doi.org/10.3390/microorganisms11071849




