High Disinfectant Tolerance in Pseudomonas spp. Biofilm Aids the Survival of Listeria monocytogenes
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling, Isolation, and Identification of Isolates
2.2. Biofilm-Forming Capability Testing by Microtiter Biofilm Assay
2.3. Antimicrobial Susceptibility Testing in Microtiter Plates
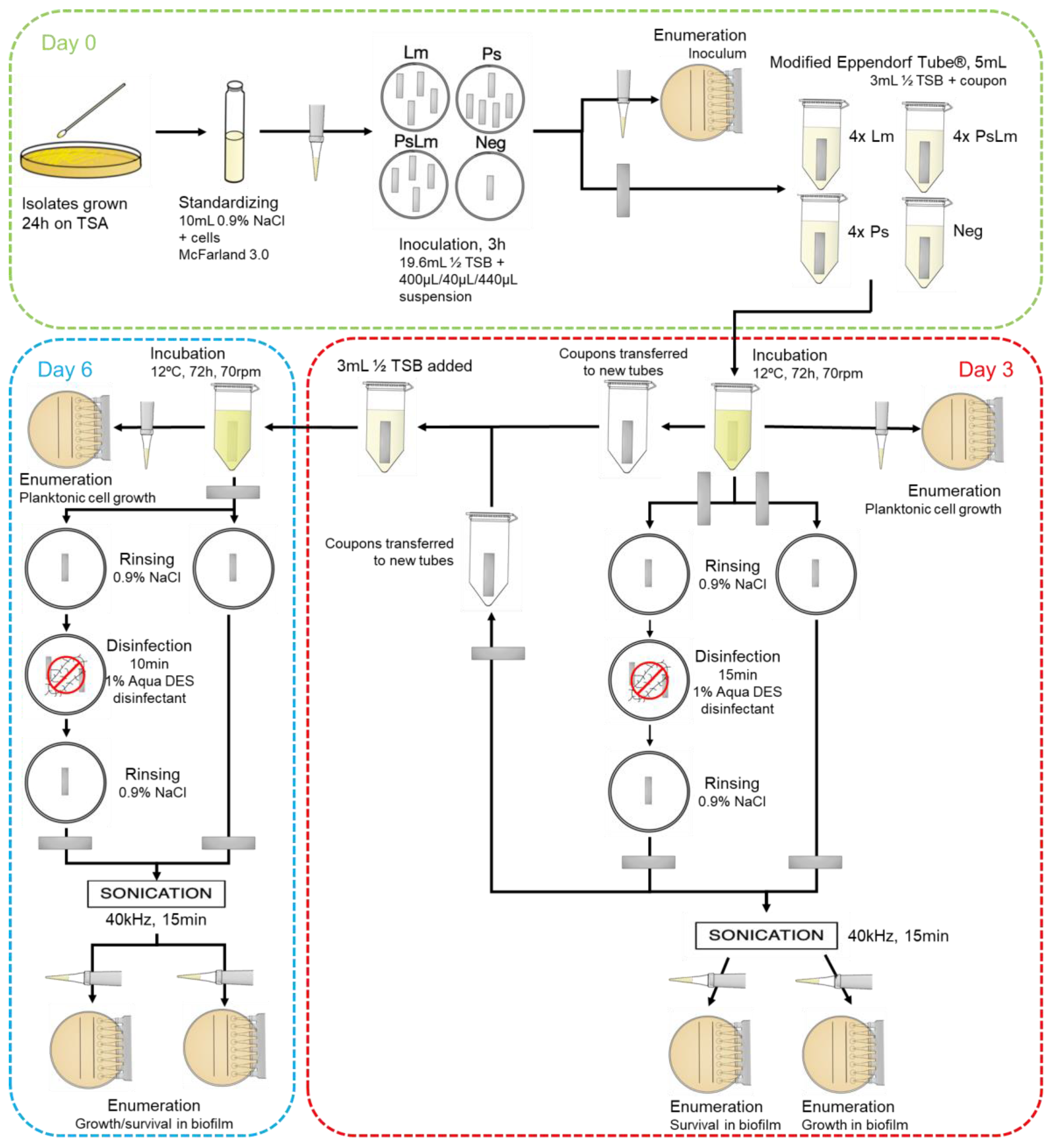
2.4. Survival of Listeria Monocytogenes in Multi-Species Pseudomonas Biofilm
2.5. Biofilm Disinfection: Survival and Growth
3. Results
3.1. Biofilm-Forming Capabilities
3.2. Antimicrobial Susceptibility in Selected Isolates
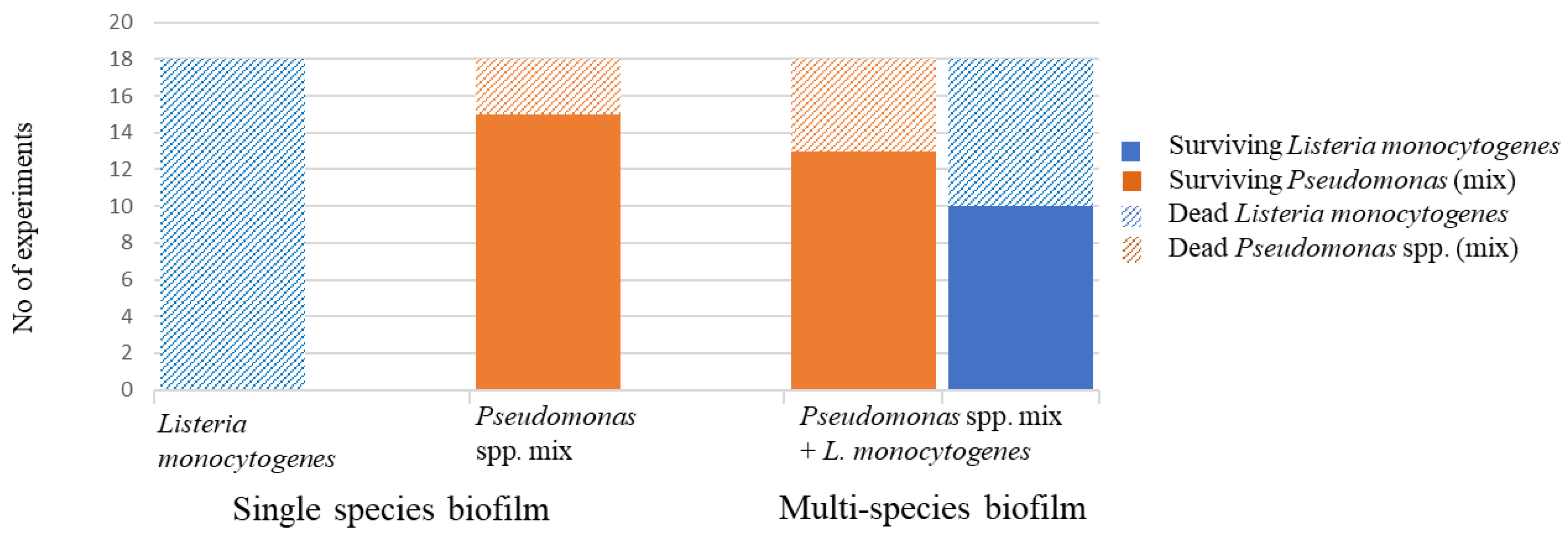
3.3. Survival and Growth Dynamics of L. monocytogenes and Pseudomonas spp. in Multi-Species Biofilm
3.4. Survival in Disinfected Multi-Species Biofilm
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Palleroni, N.J. Pseudomonas. In Bergey’s Manual of Systematics of Archea and Bacteria; Trujillo, M.E., Dedysh, S., DeVos, P., Hedlund, B., Kämpfer, P., Rainey, F.A., Whitman, W.B., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2015. [Google Scholar]
- Peix, A.; Ramírez-Bahena, M.H.; Velázquez, E. The current status on the taxonomy of Pseudomonas revisited: An update. Infect. Genet. Evol. 2018, 57, 106–116. [Google Scholar] [CrossRef]
- Kumar, H.; Franzetti, L.; Kaushal, A.; Kumar, D. Pseudomonas fluorescens: A potential food spoiler and challenges and advances in its detection. Ann. Microbiol. 2019, 69, 873–883. [Google Scholar] [CrossRef]
- Moore, E.R.B.; Tindall, B.J.; Martins Dos Santos, V.A.P.; Pieper, D.H.; Ramos, J.-L.; Palleroni, N.J. Nonmedical: Pseudomonas. In The Prokaryotes: A Handbook on the Biology of Bacteria Volume 6: Proteobacteria: Gamma Subclass; Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K.-H., Stackebrandt, E., Eds.; Springer: New York, NY, USA, 2006; pp. 646–703. [Google Scholar]
- Heir, E.; Moen, B.; Åsli, A.W.; Sunde, M.; Langsrud, S. Antibiotic resistance and phylogeny of Pseudomonas spp. isolated over three decades from chicken meat in the Norwegian food chain. Microorganisms 2021, 9, 207. [Google Scholar] [CrossRef]
- Quintieri, L.; Fanelli, F.; Caputo, L. Antibiotic resistant Pseudomonas spp. spoilers in fresh dairy products: An underestimated risk and the control strategies. Foods 2019, 8, 372. [Google Scholar] [CrossRef]
- Langsrud, S.; Sundheim, G.; Borgmann-Strahsen, R. Intrinsic and acquired resistance to quaternary ammonium compounds in food-related Pseudomonas spp. J. Appl. Microbiol. 2003, 95, 874–882. [Google Scholar] [CrossRef]
- Mann, E.E.; Wozniak, D.J. Pseudomonas biofilm matrix composition and niche biology. FEMS Microbiol. Rev. 2012, 36, 893–916. [Google Scholar] [CrossRef] [PubMed]
- Tolker-Nielsen, T.; Søren, M. The Biofilm Lifestyle of Pseudomonads. In Pseudomonas; Ramos, J.-L., Ed.; Springer: Boston, MA, USA, 2004; pp. 547–571. [Google Scholar]
- Stellato, G.; Utter, D.R.; Voorhis, A.; De Angelis, M.; Eren, A.M.; Ercolini, D. A few Pseudomonas oligotypes dominate in the meat and dairy processing environment. Front. Microbiol. 2017, 8, 264. [Google Scholar] [CrossRef]
- Møretrø, T.; Moen, B.; Heir, E.; Hansen, A.Å.; Langsrud, S. Contamination of salmon fillets and processing plants with spoilage bacteria. Int. J. Food Microbiol. 2016, 237, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Thomassen, G.M.B.; Krych, L.; Knøchel, S.; Mehli, L. Bacterial community development and diversity during the first year of production in a new salmon processing plant. Food Microbiol. 2023, 109, 104138. [Google Scholar] [CrossRef] [PubMed]
- Molin, G.; Ternström, A. Numerical taxonomy of psychrotrophic pseudomonads. J. Gen. Microbiol. 1982, 128, 1249–1264. [Google Scholar] [CrossRef] [PubMed]
- Boziaris, I.S.; Parlapani, F.F. Chapter 3—Specific Spoilage Organisms (SSOs) in Fish. In The Microbiological Quality of Food; Woodhead Publishing: Sawston, UK, 2017; pp. 61–98. [Google Scholar]
- Macé, S.; Cornet, J.; Chevalier, F.; Cardinal, M.; Pilet, M.-F.; Dousset, X.; Joffraud, J.-J. Characterisation of the spoilage microbiota in raw salmon (Salmo salar) steaks stored under vacuum or modified atmosphere packaging combining conventional methods and PCR–TTGE. Food Microbiol. 2012, 30, 164–172. [Google Scholar] [CrossRef]
- Bagge-Ravn, D.; Ng, Y.; Hjelm, M.; Christiansen, J.N.; Johansen, C.; Gram, L. The microbial ecology of processing equipment in different fish industries-analysis of the microflora during processing and following cleaning and disinfection. Int. J. Food Microbiol. 2003, 87, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Langsrud, S.; Moen, B.; Møretrø, T.; Løype, M.; Heir, E. Microbial dynamics in mixed culture biofilms of bacteria surviving sanitation of conveyor belts in salmon-processing plants. J. Appl. Microbiol. 2016, 120, 366–378. [Google Scholar] [CrossRef] [PubMed]
- Møretrø, T.; Langsrud, S. Residential bacteria on surfaces in the food industry and their implications for food safety and quality. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1022–1041. [Google Scholar] [CrossRef]
- Caraballo Guzmán, A.; González Hurtado, M.I.; Cuesta-Astroz, Y.; Torres, G. Metagenomic characterization of bacterial biofilm in four food processing plants in Colombia. Braz. J. Microbiol. 2020, 51, 1259–1267. [Google Scholar] [CrossRef] [PubMed]
- Cruz, C.D.; Fletcher, G.C. Assessing manufacturers’ recommended concentrations of commercial sanitizers on inactivation of Listeria monocytogenes. Food Control 2012, 26, 194–199. [Google Scholar] [CrossRef]
- Antonelli, M.; Turolla, A.; Mezzanotte, V.; Nurizzo, C. Peracetic acid for secondary effluent disinfection: A comprehensive performance assessment. Water Sci. Technol. 2013, 68, 2638–2644. [Google Scholar] [CrossRef]
- Freitas, B.d.O.; Leite, L.d.S.; Daniel, L.A. Chlorine and peracetic acid in decentralized wastewater treatment: Disinfection, oxidation and odor control. Process Saf. Environ. Prot. 2021, 146, 620–628. [Google Scholar] [CrossRef]
- Monarca, S.; Richardso, S.D.; Feretti, D.; Grottolo, M.; Thruston, A.D., Jr.; Zani, C.; Navazio, G.; Ragazzo, P.; Zerbini, I.; Alberti, A. Mutagenicity and disinfection by-products in surface drinking water disinfected with peracetic acid. Environ. Toxicol. Chem. 2002, 21, 309–318. [Google Scholar] [CrossRef]
- Poole, K.; Krebes, K.; McNally, C.; Neshat, S. Multiple antibiotic resistance in Pseudomonas aeruginosa: Evidence for involvement of an efflux operon. J. Bacteriol. 1993, 175, 7363–7372. [Google Scholar] [CrossRef]
- Ramos, J.L.; Duque, E.; Godoy, P.; Segura, A. Efflux pumps involved in toluene tolerance in Pseudomonas putida DOT-T1E. J. Bacteriol. 1998, 180, 3323–3329. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.; Liu, H.; Lan, T.; Dong, L.; Hu, H.; Zhao, S.; Zhang, Y.; Zheng, N.; Wang, J. Antibiotic resistance patterns of Pseudomonas spp. isolated from raw milk revealed by whole genome sequencing. Front. Microbiol. 2020, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Fernández Márquez, M.L.; Burgos, M.J.; Pulido, R.P.; Gálvez, A.; López, R.L. Biocide tolerance and antibiotic resistance in Salmonella isolates from hen eggshells. Foodborne Pathog. Dis. 2017, 14, 89–95. [Google Scholar] [CrossRef]
- Nasr, A.M.; Mostafa, M.S.; Arnaout, H.H.; Elshimy, A.A.A. The effect of exposure to sub-inhibitory concentrations of hypochlorite and quaternary ammonium compounds on antimicrobial susceptibility of Pseudomonas aeruginosa. Am. J. Infect. Control 2018, 46, e57–e63. [Google Scholar] [CrossRef] [PubMed]
- Wales, A.D.; Davies, R.H. Co-Selection of resistance to antibiotics, biocides and heavy metals, and its relevance to foodborne pathogens. Antibiotics 2015, 4, 567–604. [Google Scholar] [CrossRef]
- Thomassen, G.M.B.; Reiche, T.; Tennfjord, C.E.; Mehli, L. Antibiotic Resistance Properties among Pseudomonas spp. Associated with Salmon Processing Environments. Microorganisms 2022, 10, 1420. [Google Scholar] [CrossRef]
- NORM/NORM-VET 2020: Usage of Antimicrobial Agents and Occurence of Antimicrobial Resistance in Norway; Norwegian Institute of Public Health: Oslo, Norway, 2021.
- Miranda, C.D.; Rojas, R. Occurrence of florfenicol resistance in bacteria associated with two Chilean salmon farms with different history of antibacterial usage. Aquaculture 2007, 266, 39–46. [Google Scholar] [CrossRef]
- Miranda, C.D.; Godoy, F.A.; Lee, M.R. Current status of the use of antibiotics and the antimicrobial resistance in the Chilean salmon farms. Front. Microbiol. 2018, 9, 1–14. [Google Scholar] [CrossRef]
- Wagner, E.M.; Pracser, N.; Thalguter, S.; Fischel, K.; Rammer, N.; Pospíšilová, L.; Alispahic, M.; Wagner, M.; Rychli, K. Identification of biofilm hotspots in a meat processing environment: Detection of spoilage bacteria in multi-species biofilms. Int. J. Food Microbiol. 2020, 328, 108668. [Google Scholar] [CrossRef]
- Bridier, A.; Sanchez-Vizuete, P.; Guilbaud, M.; Piard, J.C.; Naïtali, M.; Briandet, R. Biofilm-associated persistence of food-borne pathogens. Food Microbiol. 2015, 45, 167–178. [Google Scholar] [CrossRef]
- Giaouris, E.; Heir, E.; Desvaux, M.; Hébraud, M.; Møretrø, T.; Langsrud, S.; Doulgeraki, A.; Nychas, G.-J.; Kačániová, M.; Czaczyk, K.; et al. Intra- and inter-species interactions within biofilms of important foodborne bacterial pathogens. Front. Microbiol. 2015, 6, 841. [Google Scholar] [CrossRef] [PubMed]
- Fagerlund, A.; Møretrø, T.; Heir, E.; Briandet, R.; Langsrud, S. Cleaning and disinfection of biofilms composed of Listeria monocytogenes and background microbiota from meat processing surfaces. Appl. Environ. Microbiol. 2017, 83, e01046-17. [Google Scholar] [CrossRef]
- Thomassen, G.M.B.; Krych, L.; Knøchel, S.; Mehli, L. ON-rep-seq as a rapid and cost-effective alternative to whole-genome sequencing for species-level identification and strain-level discrimination of Listeria monocytogenes contamination in a salmon processing plant. MicrobiologyOpen 2021, 10, e1246. [Google Scholar] [CrossRef] [PubMed]
- Fagerlund, A.; Langsrud, S.; Schirmer, B.C.T.; Møretrø, T.; Heir, E. Genome analysis of Listeria monocytogenes sequence type 8 strains persisting in salmon and poultry processing environments and comparison with related strains. PLoS ONE 2016, 11, e0151117. [Google Scholar] [CrossRef]
- EFSA; ECDC. Multi-country outbreak of Listeria monocytogenes sequence type 8 infections linked to consumption of salmon products. EFSA Support. Publ. 2018, 15, 1496E. [Google Scholar] [CrossRef]
- McFarland, J. The Nephelometer: An instrument for estimating the number of bacteria in suspensions used for calculating the opsonic index and for vaccines. J. Am. Med. Assoc. 1907, 49, 1176–1178. [Google Scholar] [CrossRef]
- Harrigan, W.F. Laboratory Methods in Food Microbiology, 3rd ed.; Academic Press: Cambridge, MA, USA, 1998; p. 532. [Google Scholar]
- Møretrø, T.; Schirmer, B.C.T.; Heir, E.; Fagerlund, A.; Hjemli, P.; Langsrud, S. Tolerance of L. monocytogenes to quaternary ammonium compounds may enhance growth in the food industry. Int. J. Food Microbiol. 2017, 241, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Harrison, J.J.; Stremick, C.A.; Turner, R.J.; Allan, N.D.; Olson, M.E.; Ceri, H. Microtiter susceptibility testing of microbes growing on peg lids: A miniaturized biofilm model for high-throughput screening. Nat. Protoc. 2010, 5, 1236–1254. [Google Scholar] [CrossRef]
- Heir, E.; Møretrø, T.; Simensen, A.; Langsrud, S. Listeria monocytogenes strains show large variations in competitive growth in mixed culture biofilms and suspensions with bacteria from food processing environments. Int. J. Food Microbiol. 2018, 275, 46–55. [Google Scholar] [CrossRef]
- Maes, S.; Heyndrickx, M.; Vackier, T.; Steenackers, H.; Verplaetse, A.; Reu, K.D. Identification and spoilage potential of the remaining dominant microbiota on food contact surfaces after cleaning and disinfection in different food industries. J. Food Prot. 2019, 82, 262–275. [Google Scholar] [CrossRef]
- Liu, Y.-J.; Xie, J.; Zhao, L.-J.; Qian, Y.-F.; Zhao, Y.; Liu, X. Biofilm formation characteristics of Pseudomonas lundensis isolated from meat. J. Food Sci. 2015, 80, M2904–M2910. [Google Scholar] [CrossRef] [PubMed]
- Biesta-Peters, E.G.; Reij, M.W.; Joosten, H.; Gorris, L.G.M.; Zwietering, M.H. Comparison of two optical-density-based methods and a plate count method for estimation of growth parameters of Bacillus cereus. Appl. Environ. Microbiol. 2010, 76, 1399–1405. [Google Scholar] [CrossRef]
- Bland, R.; Brown, S.R.B.; Waite-Cusic, J.; Kovacevic, J. Probing antimicrobial resistance and sanitizer tolerance themes and their implications for the food industry through the Listeria monocytogenes lens. Compr. Rev. Food Sci. Food Saf. 2022, 21, 1777–1802. [Google Scholar] [CrossRef] [PubMed]
- EPA. roduct Performance Test Guidelines Ocspp 810.2300: Sanitizers for Use on Hard Surfaces—Efficacy Data Recommendations [EPA 712-C-07-091]. 2012. Available online: https://www.regulations.gov/document/EPA-HQ-OPPT-2009-0150-0022 (accessed on 11 July 2022).
- Bridier, A.; Briandet, R.; Thomas, V.; Dubois-Brissonnet, F. Resistance of bacterial biofilms to disinfectants: A review. Biofouling 2011, 27, 1017–1032. [Google Scholar] [CrossRef] [PubMed]
- Kverme, K.O.; Haugland, G.T.; Hannisdal, R.; Kallekleiv, M.; Colquhoun, D.J.; Lunestad, B.T.; Wergeland, H.I.; Samuelsen, O.B. Pharmacokinetics of florfenicol in lumpfish (Cyclopterus lumpus L.) after a single oral administration. Aquaculture 2019, 512, 734279. [Google Scholar] [CrossRef]
- Samuelsen, O.B.; Hjeltnes, B.; Glette, J. Efficacy of orally administered florfenicol in the treatment of furunculosis in Atlantic salmon. J. Aquat. Anim. Health 1998, 10, 56–61. [Google Scholar] [CrossRef]
- Adesoji, A.T.; Call, D.R. Molecular analysis of florfenicol-resistant bacteria isolated from drinking water distribution systems in Southwestern Nigeria. J. Glob. Antimicrob. Resist. 2020, 23, 340–344. [Google Scholar] [CrossRef]
- Ho, S.P.; Hsu, T.Y.; Che, M.H.; Wang, W.S. Antibacterial effect of chloramphenicol, thiamphenicol and florfenicol against aquatic animal bacteria. J. Vet. Med. Sci. 2000, 62, 479–485. [Google Scholar] [CrossRef]
- Ceri, H.; Olson, M.E.; Stremick, C.; Read, R.R.; Morck, D.; Buret, A. The Calgary biofilm device: New technology for rapid determination of antibiotic susceptibilities of bacterial biofilms. J. Clin. Microbiol. 1999, 37, 1771–1776. [Google Scholar] [CrossRef]
- Olson, M.E.; Ceri, H.; Morck, D.W.; Buret, A.G.; Read, R.R. Biofilm bacteria: Formation and comparative susceptibility to antibiotics. Can. J. Vet. Res. 2002, 66, 86–92. [Google Scholar]



| Isolate ID | Predicted Species | Biofilm Capability | Aqua Des Foam PAA (%) | Florfenicol (µg/mL) | ||||
|---|---|---|---|---|---|---|---|---|
| MIC | MBC | MBEC | MIC | MBC | MBEC | |||
| LJP042 | P. fluorescens | Strong | 2.0 | 0.5 | 2.0 | >2400 | 600 | >2400 |
| LJP316 | P. fluorescens | Medium | 0.25 | 0.25 | 0.5 | 300 | 300 | 2400 |
| LJP321 | P. fluorescens | Medium | 0.25 | 0.25 | 0.5 | 300 | 300 | 2400 |
| LJP760 | P. putida | Low | 0.25 | 1.0 | 2.0 | 150 | 150 | >2400 |
| MF4624 | L. monocytogenes | - | - | 0.06 | - | - | 4.68 | - |
| LJP040 | P. fluorescens | No | 0.25 | 0.5 | 2.0 | 2400 | 300 | 600 |
| LJP788 | P. lundensis | Low | 0.25 | 1.0 | 2.0 | 300 | 300 | >2400 |
| LJP863 | Pseudomonas sp. | Low | 0.06 | 0.25 | 1.00 | 19 | 75 | >2400 |
| LJP906 | P. fluorescens | No | 0.06 | - | 1.00 | 75 | 19 | 150 |
| LJP882 | P. libanensis | No | 0.06 | - | 0.50 | 300 | 600 | 600 |
| LJP841 | Pseudomonas sp. | Medium | 0.06 | 0.25 | 1.00 | 300 | 600 | 600 |
| LJP895 | P. veronii | Strong | 0.06 | - | 0.25 | 19 | 75 | 75 |
| LJP847 | S. liquefaciens | No | 0.06 | 0.13 | 0.50 | 300 | 600 | 300 |
| LJP900 | A. hydrophila | Medium | 0.13 | 0.13 | 1.00 | - | 19 | 75 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thomassen, G.M.B.; Reiche, T.; Hjørungnes, M.; Mehli, L. High Disinfectant Tolerance in Pseudomonas spp. Biofilm Aids the Survival of Listeria monocytogenes. Microorganisms 2023, 11, 1414. https://doi.org/10.3390/microorganisms11061414
Thomassen GMB, Reiche T, Hjørungnes M, Mehli L. High Disinfectant Tolerance in Pseudomonas spp. Biofilm Aids the Survival of Listeria monocytogenes. Microorganisms. 2023; 11(6):1414. https://doi.org/10.3390/microorganisms11061414
Chicago/Turabian StyleThomassen, Gunn Merethe Bjørge, Thorben Reiche, Martinus Hjørungnes, and Lisbeth Mehli. 2023. "High Disinfectant Tolerance in Pseudomonas spp. Biofilm Aids the Survival of Listeria monocytogenes" Microorganisms 11, no. 6: 1414. https://doi.org/10.3390/microorganisms11061414
APA StyleThomassen, G. M. B., Reiche, T., Hjørungnes, M., & Mehli, L. (2023). High Disinfectant Tolerance in Pseudomonas spp. Biofilm Aids the Survival of Listeria monocytogenes. Microorganisms, 11(6), 1414. https://doi.org/10.3390/microorganisms11061414






