Impact of Ascorbic Acid on Zero-Valent Iron Nanoparticle and UV-B Mediated Stress in the Cyanobacterium, Fremyella diplosiphon
Abstract
1. Introduction
2. Materials and Methods
2.1. Strain and Experimental Conditions
2.2. Effect of Ascorbic Acid on the Reversal of Zero-Valent Nanoparticle and UV-Mediated Stress in F. diplosiphon
2.3. Microscopic Observations of F. diplosiphon Treated with Zero-Valent Iron Nanoparticles and Ascorbic Acid
2.4. Total Lipid Extraction in F. diplosiphon
2.5. Transesterification of F. diplosiphon Extracted Lipids
2.6. Gas Chromatography-Mass Spectrometry Analysis of Transesterified Lipids
2.7. Statistical Analysis
3. Results
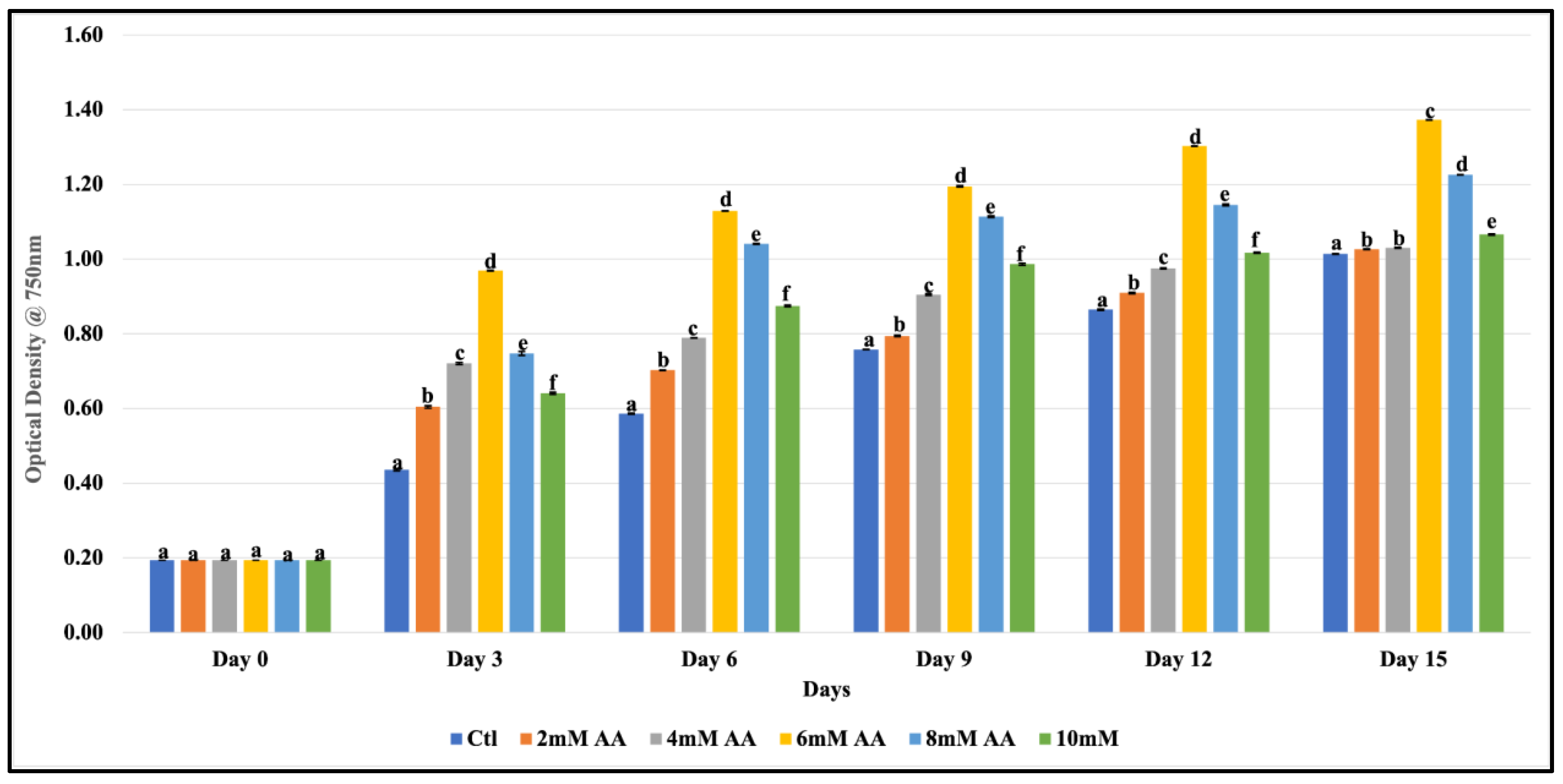
3.1. Impact of Ascorbic Acid on F. diplosiphon Growth
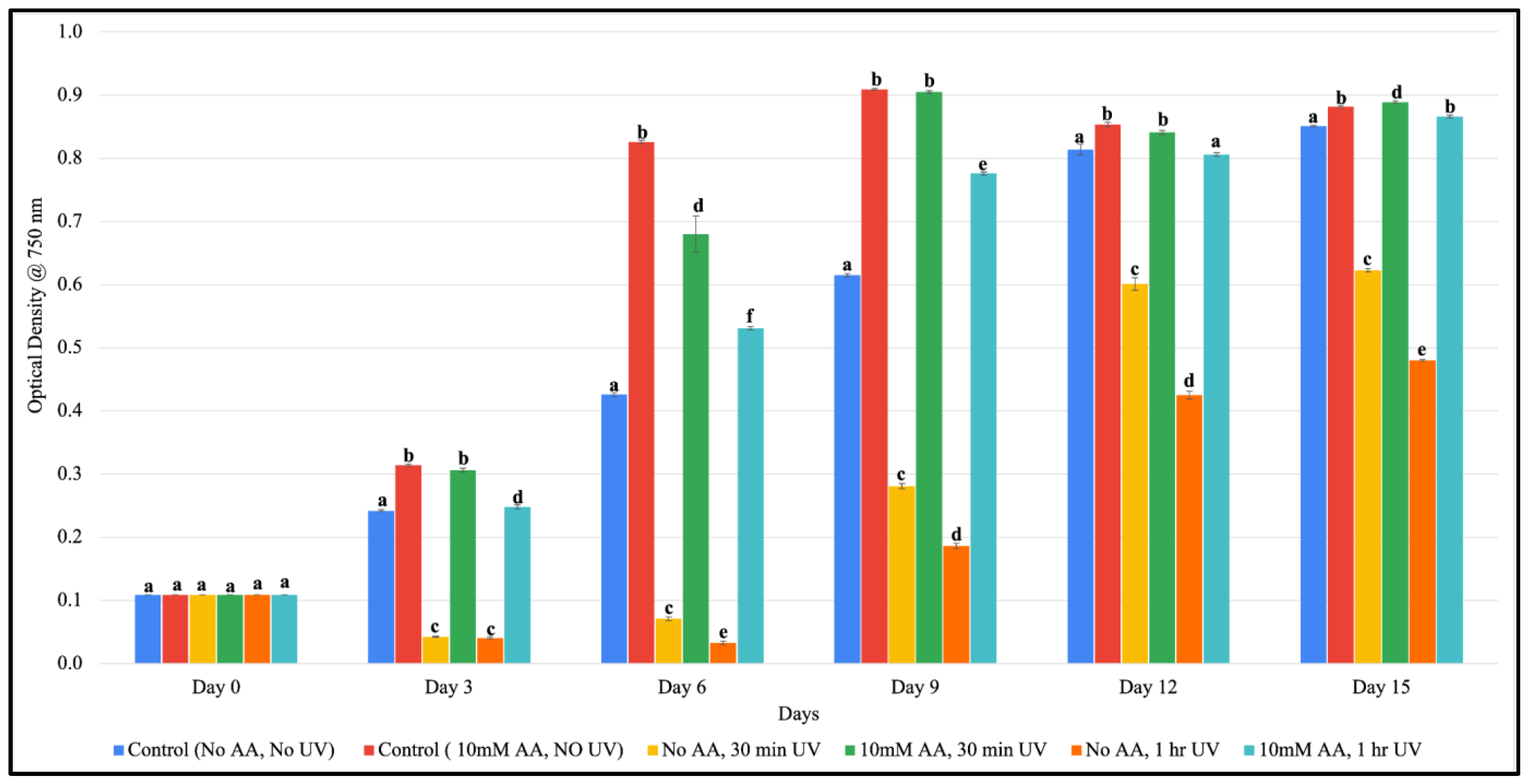
3.2. Ascorbic Acid-Mediated Growth in F. diplosiphon Exposed to Varying Zero-Valent Iron Nanoparticle Concentrations and UV-B Radiation
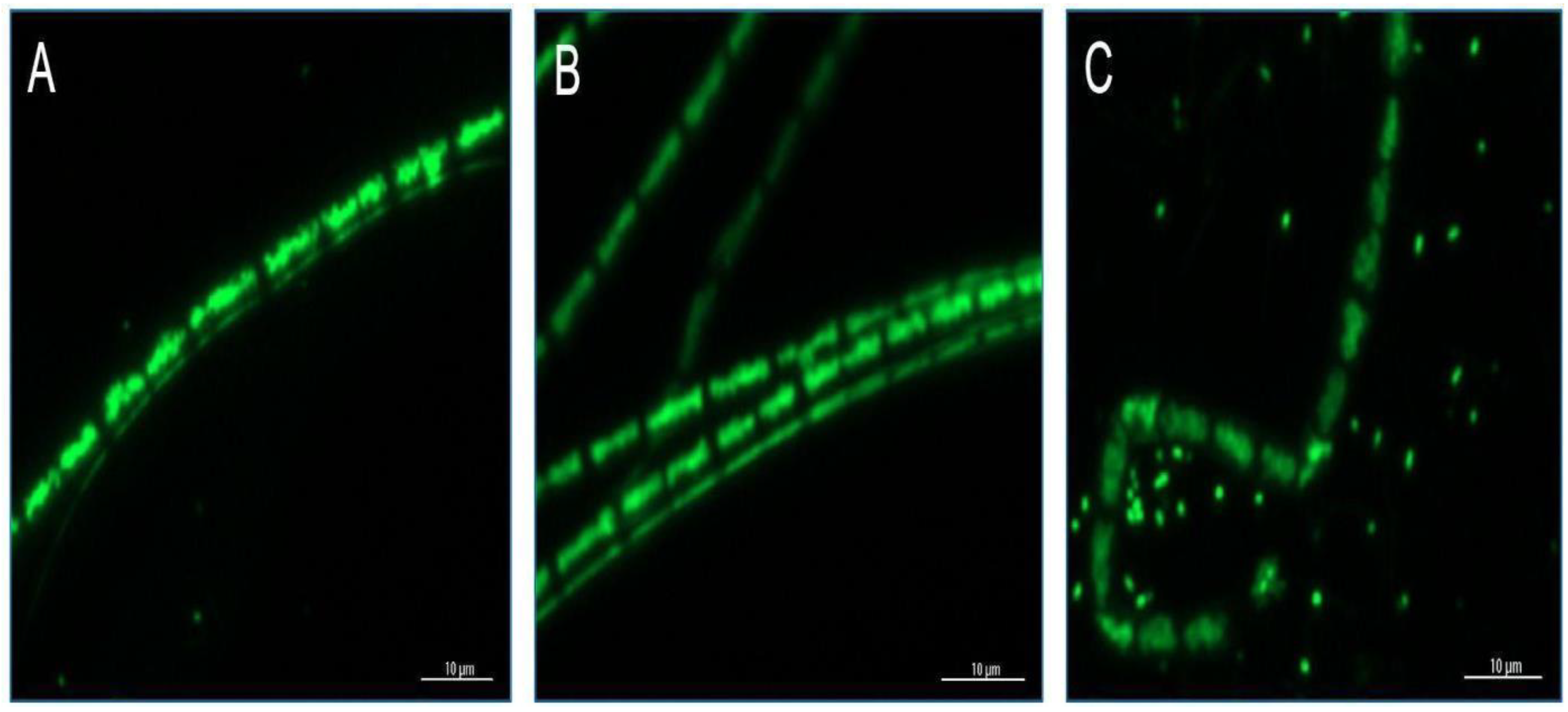
3.3. Microscopic Analysis of F. diplosiphon Treated with Ascorbic Acid and Varying Zero-Valent Iron Nanoparticle Concentrations
3.4. FAME Identification in F. diplosiphon Treated with Varying Zero-Valent Iron Nanoparticle Concentrations and Ascorbic Acid
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Calzadilla, P.I.; Muzzopappa, F.; Sétif, P.; Kirilovsky, D. Different roles for ApcD and ApcF in Synechococcus elongatus and Synechocystis Sp. PCC 6803 Phycobilisomes. Biochim. Biophys. Acta Bioenerg. 2019, 1860, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Whitton, B.A. Ecology of Cyanobacteria II: Their Diversity in Space and Time; Springer: Amsterdam, The Netherlands, 2012; ISBN 9789400738553. [Google Scholar]
- Gichuki, S.M.; Arumanayagam, A.S.; Tabatabai, B.; Yalcin, Y.S.; Wyatt, L.; Sitther, V. Augmentation of the photoreactivation gene in Fremyella diplosiphon confers UV-B tolerance. ACS Omega 2022, 7, 35092–35101. [Google Scholar] [CrossRef]
- Pádrová, K.; Lukavský, J.; Nedbalová, L.; Čejková, A.; Cajthaml, T.; Sigler, K.; Vítová, M.; Řezanka, T. Trace concentrations of Iron nanoparticles cause overproduction of biomass and lipids during cultivation of cyanobacteria and microalgae. J. Appl. Phycol. 2015, 27, 1443–1451. [Google Scholar] [CrossRef]
- Gharaie Fathabad, S.; Arumanayagam, A.C.S.; Tabatabai, B.; Chen, H.; Lu, J.; Sitther, V. Augmenting Fremyella diplosiphon cellular lipid content and unsaturated fatty acid methyl esters via sterol desaturase gene overexpression. Appl. Biochem. Biotechnol. 2019, 189, 1127–1140. [Google Scholar] [CrossRef] [PubMed]
- Gichuki, S.M.; Yalcin, Y.S.; Wyatt, L.; Ghann, W.; Uddin, J.; Kang, H.; Sitther, V. Zero-valent iron nanoparticles induce reactive oxygen species in the cyanobacterium, Fremyella diplosiphon. ACS Omega 2021, 6, 32730–32738. [Google Scholar] [CrossRef] [PubMed]
- Latifi, A.; Ruiz, M.; Zhang, C.C. Oxidative stress in cyanobacteria. FEMS Microbiol. Rev. 2009, 33, 258–278. [Google Scholar] [CrossRef]
- Kumar, J.; Babele, P.K.; Singh, D.; Kumar, A. UV-B Radiation Stress Causes Alterations in whole cell protein profile and expression of certain genes in the rice phyllospheric bacterium Enterobacter cloacae. Front. Microbiol. 2016, 7, 1440. [Google Scholar] [CrossRef]
- Patti, A.M.; Al-Rasadi, K.; Giglio, R.V.; Nikolic, D.; Mannina, C.; Castellino, G.; Chianetta, R.; Banach, M.; Cicero, A.F.G.; Lippi, G.; et al. Natural approaches in metabolic syndrome management. Arch. Med. Sci. 2018, 14, 422–441. [Google Scholar] [CrossRef]
- Arrigoni, O.; De Tullio, M. Ascorbic acid: Much more than just an antioxidant. Biochim. Biophys. Acta 2002, 1569, 1–9. [Google Scholar] [CrossRef]
- Gallie, D.R. L-Ascorbic Acid: A multifunctional molecule supporting plant growth and development. Scientifica 2013, 2013, 795–964. [Google Scholar] [CrossRef]
- Sharma, L.; Siedlewicz, G.; Pazdro, K. The toxic effects of antibiotics on freshwater and marine photosynthetic microorganisms: State of the Art. Plants 2021, 10, 591. [Google Scholar] [CrossRef] [PubMed]
- Foyer, C.H. Reactive oxygen species, oxidative signaling and the regulation of photosynthesis. Environ. Exp. Bot. 2018, 154, 134–142. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef] [PubMed]
- Wahlen, B.D.; Willis, R.M.; Seefeldt, L.C. Biodiesel production by simultaneous extraction and conversion of total lipids from microalgae, cyanobacteria, and wild mixed-cultures. Bioresour. Technol. 2011, 102, 2724–2730. [Google Scholar] [CrossRef] [PubMed]
- Tabatabai, B.; Chen, H.; Lu, J.; Giwa-Otusajo, J.; McKenna, A.M.; Shrivastava, A.K.; Sitther, V. Fremyella diplosiphon as a biodiesel agent: Identification of fatty acid methyl esters via microwave-assisted direct in situ transesterification. Bioenergy Res. 2018, 11, 528–537. [Google Scholar] [CrossRef] [PubMed]
- Malihan-Yap, L.; Grimm, H.C.; Kourist, R. Recent advances in cyanobacterial biotransformations. Chem. Ing. Tech. 2022, 94, 1628–1644. [Google Scholar] [CrossRef]
- Roco, M. Broader societal issues of nanotechnology. J. Nanoparticle Res. 2003, 5, 181–189. [Google Scholar] [CrossRef]
- Dong, D.; Kyung Choi, O.; Woo Lee, J. Influence of the continuous addition of zero valent iron (ZVI) and nano-scaled zero valent Iron (nZVI) on the anaerobic biomethanation of carbon dioxide. Chem. Eng. J. 2022, 430, 132233. [Google Scholar] [CrossRef]
- Kustka, A.; Carpenter, E.J.; Sañudo-Wilhelmy, S.A. Iron and marine nitrogen fixation: Progress and future directions. Res. Microbiol. 2002, 153, 255–262. [Google Scholar] [CrossRef]
- Sarma, M.K.; Kaushik, S.; Goswami, P. Cyanobacteria: A metabolic power house for harvesting solar energy to produce bio-electricity and biofuels. Biomass Bioenergy 2016, 90, 187–201. [Google Scholar] [CrossRef]
- Yalcin, Y.S.; Aydin, B.N.; Sayadujjhara, M.; Sitther, V. Antibiotic-induced changes in pigment accumulation, photosystem II, and membrane permeability in a model cyanobacterium. Front. Microbiol. 2022, 13, 930357. [Google Scholar] [CrossRef] [PubMed]
- Kaur Bhatia, K.; Puri, S.; Ahluwalia, A.S. Morphological and physiological performance of a cyanobacterium Spirulina platensis in presence of ascorbic acid: A growth facilitator. J. Algal Biomass Util. 2017, 8, 35–44. [Google Scholar]
- Kong, Y.; Ji, L.; Wang, Y.; Li, J.; Lu, H.; Mo, S.; Wang, X.; Zhu, L.; Xu, X.; Zheng, X. Combined effect of NZVI and H2O2 on the cyanobacterium Microcystis aeruginosa: Performance and mechanism. Nanomaterials 2022, 12, 3017. [Google Scholar] [CrossRef] [PubMed]
- Marslin, G.; Sheeba, C.J.; Franklin, G. Nanoparticles alter secondary metabolism in plants via ROS Burst. Front. Plant Sci. 2017, 8, 832. [Google Scholar] [CrossRef] [PubMed]
- Boden, J.S.; Konhauser, K.O.; Robbins, L.J.; Sánchez-Baracaldo, P. Timing the evolution of antioxidant enzymes in cyanobacteria. Nat. Commun. 2021, 12, 4742. [Google Scholar] [CrossRef]
- Checa, J.; Aran, J.M. Reactive oxygen species: Drivers of physiological and pathological processes. J. Inflamm. Res. 2020, 13, 1057–1073. [Google Scholar] [CrossRef]
- Kumar, M.; Sabu, S.; Sangela, V.; Meena, M.; Rajput, V.D.; Minkina, T.; Vinayak, V.; Harish. The mechanism of nanoparticle toxicity to cyanobacteria. Arch. Microbiol. 2023, 205, 30. [Google Scholar] [CrossRef]
- Dwivedi, S.; Ahmad, I.Z. Evaluation of the effect of UV-B radiation on growth, photosynthetic pigment, and antioxidant enzymes of some cyanobacteria. Environ. Res. 2023, 218, 114943. [Google Scholar] [CrossRef]
- He, Y.Y.; Klisch, M.; Häder, D.P. Adaptation of cyanobacteria to UV-B stress correlated with oxidative stress and oxidative damage. Photochem. Photobiol. 2002, 76, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Chen, X.; Sen, B.; Bai, M.; He, Y.; Wang, G. Exogenous antioxidants improve the accumulation of saturated and polyunsaturated fatty acids in Schizochytrium sp. PKU#Mn4. Mar. Drugs 2021, 19, 559. [Google Scholar] [CrossRef]
- Rai, S.; Sitther, V. Oxidative stress in cyanobacteria: Sources, mitigation and defense. In Expanding Horizon of Cyanobacterial Biology; Academic Press: Cambridge, MA, USA, 2020; pp. 163–178. [Google Scholar] [CrossRef]
- Sitther, V.; Tabatabai, B.; Fathabad, S.G.; Gichuki, S.; Chen, H.; Arumanayagam, A.C.S. Cyanobacteria as a biofuel source: Advances and applications. In Advances in Cyanobacterial Biology; Elsevier Academic Press: Cambridge, MA, USA, 2020; pp. 269–289. [Google Scholar]
- Han, Q.; Zhang, W.; Chen, W. Ascorbic acid alters the lipid metabolism and improves the antioxidant system in Microcystis aeruginosa. Environ. Pollut. 2019, 254, 112987. [Google Scholar]
- Gheda, S.F.; Ismail, G.A. Natural products from some soil cyanobacterial extracts with potent antimicrobial, antioxidant and cytotoxic activities. An. Acad. Bras. Cienc. 2020, 92, e20190934. [Google Scholar] [CrossRef] [PubMed]
- McCracken, M.D.; Middaugh, R.E.; Middaugh, R.S. A chemical characterization of an algal inhibitor obtained from Chlamydomonas. Hydrobiologia 1980, 70, 271–276. [Google Scholar] [CrossRef]
- Chien, L.T.; Hwang, D.F. Effects of thermal stress and vitamin c on lipid peroxidation and fatty acid composition in the liver of Thornfish Terapon jarbua. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2020, 128, 91–97. [Google Scholar] [CrossRef]

| Treatment | Percentage Area | Group Name | Identity |
|---|---|---|---|
| Control | 61.86 | Methyl esters of saturated straight chain fatty acids (including those labelled with stable isotopes) | Hexadecanoate (16:0) |
| 23.12 | Methyl esters of trienoic acids including (MTAD adducts) | Methyl 3c,9c,12c-octadecatrienoate (3,9,12 -18:3_ | |
| 6.95 | Methyl esters monoenoic fatty acids (including dimethyl disulfide adducts) | Methyl 9-tetradecenoate | |
| 2.14 | Methyl esters of allenic acids | Methyl 4,5-trdecadienoate (4,5-13:2) | |
| 1.98 | Methyl esters of monoenoic fatty acids (including dimethyl disulfide adducts) | Methyl 9-tetradecenoate (9-14:1 | |
| 3.2 mg/L nZVIs | 70.85 | Methyl esters of saturated straight chain fatty acids (including those labelled with stable isotopes) | Hexadecanoate (16:0 |
| 20.23 | methyl esters of tetra-, penta-, and hexaenoic fatty acids | methyl 5,8,11,14-octadecatetraenoate (18:4(n-4)) | |
| 3.88 | methyl esters of acetylenic fatty acids | methyl octadeca-9-yn, trans-11-enoate | |
| 1.38 | Methyl esters of trienoic acids including (MTAD adducts) | methyl 8,11,14-heptadecatrienoate | |
| 0.92 | Methyl esters of natural cyclic fatty acids | Methyl 11-cyclopentylundecanoate | |
| 0.92 | Methyl esters of hydroxy fatty acids | Methyl 17-hydroxy-octadecanoate | |
| 0.57 | Methyl esters of trienoic acids including (MTAD adducts) | methyl 8,11, 14-heptadecatrienoate | |
| 0.55 | Methyl esters of trienoic acids including (MTAD adducts) | methyl 8,11, 14-heptadecatrienoate | |
| 12.8 mg/L nZVIs | 70.64 | Methyl esters of saturated straight chain fatty acids (including those labelled with stable isotopes) | Hexadecanoate (16:0) |
| 22.16 | Methyl esters if trienoic acids including (MTAD adducts) | Methyl 3c,9c,12c-octadecatrienoate (3,9,12 -18:3_ | |
| 2.06 | Methyl esters if trienoic acids including (MTAD adducts) | Methyl 3c,9c,12c-octadecatrienoate (3,9,12 -18:3_ | |
| 1.88 | Methyl esters of trienoic acids including (MTAD adducts) | Methyl 8,11,14-heptadecatrienoate | |
| 1.08 | Methyl esters if monoenoic fatty acids (including dimethyl disulfide adducts) | methyl trans-3-hexadecenoate (3t-16:1) | |
| 3.2 mg/L nZVIs + 6 mM AA | 62.9 | Methyl esters of saturated straight chain fatty acids (including those labelled with stable isotopes) | Hexadecanoate (16:0) |
| 31.2 | Methyl esters of monoenoic fatty acids (including dimethyl disulfide adducts) | Methyl trans-2-octadecenoate (2-18:1) | |
| 2.63 | Methyl esters of hydroxy fatty acids | Methyl 13-hydroxy-hexadecanoate trimethylsilyl ether derivative | |
| 12.8 mg/L + nZVIs 6 mM AA | 72.48 | Methyl esters of saturated straight chain fatty acids (including those labelled with stable isotopes) | Hexadecanoate (16:0) |
| 25.77 | Methyl esters of saturated straight chain fatty acids (including those labelled with stable isotopes) | Methyl docosanoate (22:0) | |
| 0.79 | Methyl esters of trienoic acids including (MTAD adducts) | Methyl 8,11,14-heptadecatrienoate | |
| 0.52 | Methyl esters of hydroxy fatty acids | Dimethyl 9,10-dihydroxy-1,18-octadecanedioate TMS ether derivative | |
| 0.45 | Methyl esters of natural cyclic fatty acids | Methyl ferulate |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wyatt, L.; Gichuki, S.; Yalcin, Y.S.; Sitther, V. Impact of Ascorbic Acid on Zero-Valent Iron Nanoparticle and UV-B Mediated Stress in the Cyanobacterium, Fremyella diplosiphon. Microorganisms 2023, 11, 1245. https://doi.org/10.3390/microorganisms11051245
Wyatt L, Gichuki S, Yalcin YS, Sitther V. Impact of Ascorbic Acid on Zero-Valent Iron Nanoparticle and UV-B Mediated Stress in the Cyanobacterium, Fremyella diplosiphon. Microorganisms. 2023; 11(5):1245. https://doi.org/10.3390/microorganisms11051245
Chicago/Turabian StyleWyatt, LaDonna, Samson Gichuki, Yavuz S. Yalcin, and Viji Sitther. 2023. "Impact of Ascorbic Acid on Zero-Valent Iron Nanoparticle and UV-B Mediated Stress in the Cyanobacterium, Fremyella diplosiphon" Microorganisms 11, no. 5: 1245. https://doi.org/10.3390/microorganisms11051245
APA StyleWyatt, L., Gichuki, S., Yalcin, Y. S., & Sitther, V. (2023). Impact of Ascorbic Acid on Zero-Valent Iron Nanoparticle and UV-B Mediated Stress in the Cyanobacterium, Fremyella diplosiphon. Microorganisms, 11(5), 1245. https://doi.org/10.3390/microorganisms11051245






