Current Updates on the Role of Microbiome in Endometriosis: A Narrative Review
Abstract
1. Introduction
2. How Common Is Endometriosis?
3. Diagnosis and Management of Endometriosis
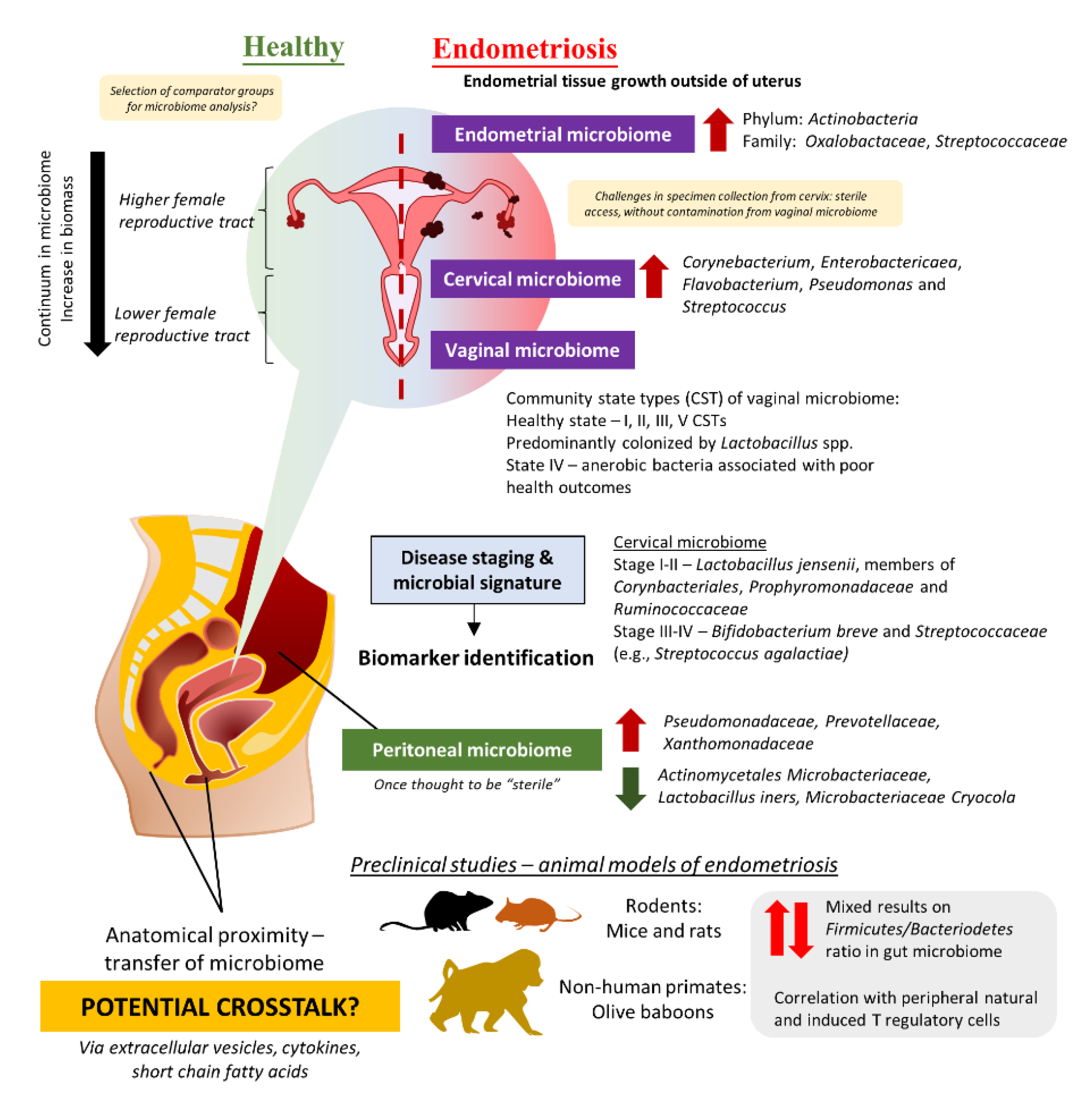
4. The Intricate Relationship between the Female Reproductive Tract Microbiome and Gut Microbiome in the Development and Progression of Endometriosis
4.1. Evidence from Clinical Studies: Are There Any Distinct Microbiome Changes in the Vaginal Microbiome?
4.2. Beyond the Female Reproductive Tract: Connections between Gut Microbiome, Peritoneal Microbiome, and Endometriosis
4.3. Establishment of Animal Model for Endometriosis
5. Potential Benefits of Probiotics in the Management of Endometriosis
6. Future Recommendations and Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Giudice, L.C. Clinical practice. Endometriosis. N. Engl. J. Med. 2010, 362, 2389–2398. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.H.; Monsanto, S.P.; Miller, C.; Singh, S.S.; Thomas, R.; Tayade, C. Pathophysiology and immune dysfunction in endometriosis. Biomed. Res. Int. 2015, 2015, 795976. [Google Scholar] [CrossRef] [PubMed]
- Dai, Y.; Zhou, Y.; Zhang, X.; Xue, M.; Sun, P.; Leng, J.; Chapron, C. Factors associated with deep infiltrating endometriosis versus ovarian endometrioma in China: A subgroup analysis from the FEELING study. BMC Womens Health 2018, 18, 205. [Google Scholar] [CrossRef] [PubMed]
- McKinnon, B.D.; Bertschi, D.; Bersinger, N.A.; Mueller, M.D. Inflammation and nerve fiber interaction in endometriotic pain. Trends Endocrinol. Metab. 2015, 26, 1–10. [Google Scholar] [CrossRef]
- de Almeida Asencio, F.; Ribeiro, H.A.; Ayrosa Ribeiro, P.; Malzoni, M.; Adamyan, L.; Ussia, A.; Gomel, V.; Martin, D.C.; Koninckx, P.R. Symptomatic endometriosis developing several years after menopause in the absence of increased circulating estrogen concentrations: A systematic review and seven case reports. Gynecol. Surg. 2019, 16, 1–11. [Google Scholar] [CrossRef]
- Ladanyi, C.; Boyd, S.; Sticco, P.; Mohling, S. Postmenopausal endometriosis, where are we now? Curr. Opin. Obs. Gynecol. 2019, 31, 267–278. [Google Scholar] [CrossRef]
- Secosan, C.; Balulescu, L.; Brasoveanu, S.; Balint, O.; Pirtea, P.; Dorin, G.; Pirtea, L. Endometriosis in Menopause-Renewed Attention on a Controversial Disease. Diagnostics 2020, 10, 134. [Google Scholar] [CrossRef]
- Giannella, L.; Marconi, C.; Di Giuseppe, J.; Delli Carpini, G.; Fichera, M.; Grelloni, C.; Giuliani, L.; Montanari, M.; Insinga, S.; Ciavattini, A. Malignant Transformation of Postmenopausal Endometriosis: A Systematic Review of the Literature. Cancers 2021, 13, 4026. [Google Scholar] [CrossRef]
- Dai, Y.; Zhang, X.; Xue, M.; Zhou, Y.; Sun, P.; Leng, J. Not having been breastfed may protect Chinese women from developing deep infiltrating endometriosis: Results from subgroup analyses of the FEELING study. Reprod. Sci. 2019, 26, 1158–1167. [Google Scholar] [CrossRef]
- Attar, E.; Bulun, S.E. Aromatase and other steroidogenic genes in endometriosis: Translational aspects. Hum. Reprod. Update 2006, 12, 49–56. [Google Scholar] [CrossRef]
- Burney, R.O.; Giudice, L.C. Pathogenesis and pathophysiology of endometriosis. Fertil. Steril. 2012, 98, 511–519. [Google Scholar] [CrossRef]
- Khan, K.N.; Kitajima, M.; Hiraki, K.; Fujishita, A.; Sekine, I.; Ishimaru, T.; Masuzaki, H. Immunopathogenesis of pelvic endometriosis: Role of hepatocyte growth factor, macrophages and ovarian steroids. Am. J. Reprod. Immunol. 2008, 60, 383–404. [Google Scholar] [CrossRef]
- Sampson, J.A. Metastatic or Embolic Endometriosis, due to the Menstrual Dissemination of Endometrial Tissue into the Venous Circulation. Am. J. Pathol. 1927, 3, 93–110, 143. [Google Scholar]
- Bellelis, P.; Frediani Barbeiro, D.; Gueuvoghlanian-Silva, B.Y.; Kalil, J.; Abrao, M.S.; Podgaec, S. Interleukin-15 and interleukin-7 are the major cytokines to maintain endometriosis. Gynecol. Obs. Investig. 2019, 84, 435–444. [Google Scholar] [CrossRef]
- Gueuvoghlanian-Silva, B.Y.; Bellelis, P.; Barbeiro, D.F.; Hernandes, C.; Podgaec, S. Treg and NK cells related cytokines are associated with deep rectosigmoid endometriosis and clinical symptoms related to the disease. J. Reprod. Immunol. 2018, 126, 32–38. [Google Scholar] [CrossRef]
- Khan, K.N.; Fujishita, A.; Hiraki, K.; Kitajima, M.; Nakashima, M.; Fushiki, S.; Kitawaki, J. Bacterial contamination hypothesis: A new concept in endometriosis. Reprod. Med. Biol. 2018, 17, 125–133. [Google Scholar] [CrossRef]
- Podgaec, S.; Dias, J.A., Jr.; Chapron, C.; Oliveira, R.M.; Baracat, E.C.; Abrao, M.S. Th1 and Th2 ummune responses related to pelvic endometriosis. Rev. Assoc. Med. Bras. 2010, 56, 92–98. [Google Scholar] [CrossRef]
- Podgaec, S.; Rizzo, L.V.; Fernandes, L.F.; Baracat, E.C.; Abrao, M.S. CD4(+) CD25(high) Foxp3(+) cells increased in the peritoneal fluid of patients with endometriosis. Am. J. Reprod. Immunol. 2012, 68, 301–308. [Google Scholar] [CrossRef]
- Sourial, S.; Tempest, N.; Hapangama, D.K. Theories on the pathogenesis of endometriosis. Int. J. Reprod. Med. 2014, 2014, 179515. [Google Scholar] [CrossRef]
- Zhang, T.; De Carolis, C.; Man, G.C.W.; Wang, C.C. The link between immunity, autoimmunity and endometriosis: A literature update. Autoimmun. Rev. 2018, 17, 945–955. [Google Scholar] [CrossRef]
- Akiyama, K.; Nishioka, K.; Khan, K.N.; Tanaka, Y.; Mori, T.; Nakaya, T.; Kitawaki, J. Molecular detection of microbial colonization in cervical mucus of women with and without endometriosis. Am. J. Reprod. Immunol. 2019, 82, e13147. [Google Scholar] [CrossRef] [PubMed]
- Jiang, I.; Yong, P.J.; Allaire, C.; Bedaiwy, M.A. Intricate Connections between the Microbiota and Endometriosis. Int. J. Mol. Sci. 2021, 22, 5644. [Google Scholar] [CrossRef] [PubMed]
- Nih Hmp Working Group. Peterson, J.; Garges, S.; Giovanni, M.; McInnes, P.; Wang, L.; Schloss, J.A.; Bonazzi, V.; McEwen, J.E.; Wetterstrand, K.A.; et al. The NIH Human Microbiome Project. Genome Res. 2009, 19, 2317–2323. [Google Scholar] [CrossRef]
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef]
- Ramakrishna, B.S. Role of the gut microbiota in human nutrition and metabolism. J. Gastroenterol. Hepatol. 2013, 28, 9–17. [Google Scholar] [CrossRef]
- Schirbel, A.; Kessler, S.; Rieder, F.; West, G.; Rebert, N.; Asosingh, K.; McDonald, C.; Fiocchi, C. Pro-angiogenic activity of TLRs and NLRs: A novel link between gut microbiota and intestinal angiogenesis. Gastroenterology 2013, 144, 613–623.e619. [Google Scholar] [CrossRef]
- Shigesi, N.; Kvaskoff, M.; Kirtley, S.; Feng, Q.; Fang, H.; Knight, J.C.; Missmer, S.A.; Rahmioglu, N.; Zondervan, K.T.; Becker, C.M. The association between endometriosis and autoimmune diseases: A systematic review and meta-analysis. Hum. Reprod. Update 2019, 25, 486–503. [Google Scholar] [CrossRef]
- Hasain, Z.; Raja Ali, R.A.; Abdul Razak, S.; Azizan, K.A.; El-Omar, E.; Razalli, N.H.; Mokhtar, N.M. Gut microbiota signature among Asian post-gestational diabetes women linked to macronutrient intakes and metabolic phenotypes. Front. Microbiol. 2021, 12, 680622. [Google Scholar] [CrossRef]
- Khan, K.N.; Fujishita, A.; Kitajima, M.; Hiraki, K.; Nakashima, M.; Masuzaki, H. Intra-uterine microbial colonization and occurrence of endometritis in women with endometriosisdagger. Hum. Reprod. 2014, 29, 2446–2456. [Google Scholar] [CrossRef]
- Khan, K.N.; Fujishita, A.; Masumoto, H.; Muto, H.; Kitajima, M.; Masuzaki, H.; Kitawaki, J. Molecular detection of intrauterine microbial colonization in women with endometriosis. Eur. J. Obs. Gynecol. Reprod. Biol. 2016, 199, 69–75. [Google Scholar] [CrossRef]
- Khan, K.N.; Kitajima, M.; Hiraki, K.; Yamaguchi, N.; Katamine, S.; Matsuyama, T.; Nakashima, M.; Fujishita, A.; Ishimaru, T.; Masuzaki, H. Escherichia coli contamination of menstrual blood and effect of bacterial endotoxin on endometriosis. Fertil. Steril. 2010, 94, 2860–2863. [Google Scholar] [CrossRef]
- Grummer, R. Animal models in endometriosis research. Hum. Reprod. Update 2006, 12, 641–649. [Google Scholar] [CrossRef]
- Lagana, A.S.; Garzon, S.; Franchi, M.; Casarin, J.; Gullo, G.; Ghezzi, F. Translational animal models for endometriosis research: A long and windy road. Ann. Transl. Med. 2018, 6, 431. [Google Scholar] [CrossRef]
- Singh, S.; Soliman, A.M.; Rahal, Y.; Robert, C.; Defoy, I.; Nisbet, P.; Leyland, N. Prevalence, Symptomatic Burden, and Diagnosis of Endometriosis in Canada: Cross-Sectional Survey of 30,000 Women. J. Obs. Gynaecol. Can. 2020, 42, 829–838. [Google Scholar] [CrossRef]
- Fuldeore, M.J.; Soliman, A.M. Prevalence and symptomatic burden of diagnosed endometriosis in the United States: National estimates from a cross-sectional survey of 59,411 women. Gynecol. Obs. Investig. 2017, 82, 453–461. [Google Scholar] [CrossRef]
- Christ, J.P.; Yu, O.; Schulze-Rath, R.; Grafton, J.; Hansen, K.; Reed, S.D. Incidence, prevalence, and trends in endometriosis diagnosis: A United States population-based study from 2006 to 2015. Am. J. Obs. Gynecol. 2021, 225, 500.e1. [Google Scholar] [CrossRef]
- Medina-Perucha, L.; Pistillo, A.; Raventos, B.; Jacques-Avino, C.; Munros-Feliu, J.; Martinez-Bueno, C.; Valls-Llobet, C.; Carmona, F.; Lopez-Jimenez, T.; Pujolar-Diaz, G.; et al. Endometriosis prevalence and incidence trends in a large population-based study in Catalonia (Spain) from 2009 to 2018. Womens Health 2022, 18, 17455057221130566. [Google Scholar] [CrossRef]
- Yamamoto, A.; Johnstone, E.B.; Bloom, M.S.; Huddleston, H.G.; Fujimoto, V.Y. A higher prevalence of endometriosis among Asian women does not contribute to poorer IVF outcomes. J. Assist. Reprod. Genet. 2017, 34, 765–774. [Google Scholar] [CrossRef]
- Bougie, O.; Yap, M.I.; Sikora, L.; Flaxman, T.; Singh, S. Influence of race/ethnicity on prevalence and presentation of endometriosis: A systematic review and meta-analysis. BJOG Int. J. Obs. Gynaecol. 2019, 126, 1104–1115. [Google Scholar] [CrossRef]
- Ajayi, A.B.; Ajayi, V.D.; Biobaku, O.; Oyetunji, I.; Aikhuele, H.; Atiba, A.; Afolabi, B.M. A 10-year study of endometriosis in an indigenous Black African population. J. Endometr. Pelvic Pain Disord. 2016, 8, 157–166. [Google Scholar] [CrossRef]
- Gichuhi, J.; Ogengo, J.; Gichangi, P. Laparoscopic diagnosis of endometriosis at Kenyatta National Hospital, Kenya. East Afr. Med. J. 2021, 98, 4038–4046. [Google Scholar]
- Labinjo, T. A review of the prevalence of endometriosis in African women. J. Womens Health Issues Care 9 2020, 4, 2. [Google Scholar]
- Farland, L.V.; Horne, A.W. Disparity in endometriosis diagnoses between racial/ethnic groups. BJOG Int. J. Obs. Gynaecol. 2019, 126, 1115–1116. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, K. Incidence of endometriosis among Japanese women. Obs. Gynecol. 1976, 48, 407–409. [Google Scholar]
- Yasui, T.; Hayashi, K.; Nagai, K.; Mizunuma, H.; Kubota, T.; Lee, J.S.; Suzuki, S. Risk profiles for endometriosis in Japanese women: Results from a repeated survey of self-reports. J. Epidemiol. 2015, 25, 194–203. [Google Scholar] [CrossRef]
- Feng, J.; Zhang, S.; Chen, J.; Yang, J.; Zhu, J. Long-term trends in the incidence of endometriosis in China from 1990 to 2019: A joinpoint and age-period-cohort analysis. Gynecol. Endocrinol. 2021, 37, 1041–1045. [Google Scholar] [CrossRef]
- Bigambo, F.M.; Wang, D.; Zhang, Y.; Mzava, S.M.; Dai, R.; Wang, X. Current situation of menstruation and gynecological diseases prevalence among Chinese women: A cross-sectional study. BMC Womens Health 2022, 22, 270. [Google Scholar] [CrossRef]
- Rowlands, I.J.; Abbott, J.A.; Montgomery, G.W.; Hockey, R.; Rogers, P.; Mishra, G.D. Prevalence and incidence of endometriosis in Australian women: A data linkage cohort study. BJOG Int. J. Obs. Gynaecol. 2021, 128, 657–665. [Google Scholar] [CrossRef]
- Arumugam, K.; Templeton, A.A. Endometriosis and race. Aust. New Zealand J. Obs. Gynaecol. 1992, 32, 164–165. [Google Scholar] [CrossRef]
- Tanmahasamut, P.; Noothong, S.; Sanga-Areekul, N.; Silprasit, K.; Dangrat, C. Prevalence of endometriosis in women undergoing surgery for benign gynecologic diseases. J. Med. Assoc. Thai. 2014, 97, 147–152. [Google Scholar]
- Yen, C.F.; Kim, M.R.; Lee, C.L. Epidemiologic Factors Associated with Endometriosis in East Asia. Gynecol. Minim. Invasive Ther. 2019, 8, 4–11. [Google Scholar] [CrossRef]
- Wisawasukmongchol, W.; Chalermchockcharoenkit, A.; Panyakhamlerd, K.; Ratchanon, S.; Luanratanakorn, S.; Sophonsritsuk, A.; Rungruxsirivorn, T.; Choksuchat, C.; Lertvikool, S.; Pantasri, T. Thai Interest Group for Endometriosis (TIGE) consensus statement on endometriosis-associated pain. J. Obs. Gynaecol. 2022, 42, 1607–1612. [Google Scholar] [CrossRef]
- Wilson, S.; Mogan, S.; Kaur, K. Understanding the role of Facebook to support women with endometriosis: A Malaysian perspective. Int. J. Nurs. Pract. 2020, 26, e12833. [Google Scholar] [CrossRef]
- Capezzuoli, T.; Clemenza, S.; Sorbi, F.; Campana, D.; Vannuccini, S.; Chapron, C.; Petraglia, F. Classification/staging systems for endometriosis: The state of the art. Gynecol. Reprod. Endocrinol. Metab. 2020, 1, 14–22. [Google Scholar]
- Vermeulen, N.; Abrao, M.S.; Einarsson, J.I.; Horne, A.W.; Johnson, N.P.; Lee, T.T.M.; Missmer, S.; Petrozza, J.; Tomassetti, C.; Zondervan, K.T.; et al. Endometriosis Classification, Staging and Reporting Systems: A Review on the Road to a Universally Accepted Endometriosis Classification. J. Minim. Invasive Gynecol. 2021, 28, 1822–1848. [Google Scholar] [CrossRef]
- Kuznetsov, L.; Dworzynski, K.; Davies, M.; Overton, C.; Guideline, C. Diagnosis and management of endometriosis: Summary of NICE guidance. BMJ 2017, 358, j3935. [Google Scholar] [CrossRef]
- Ballard, K.D.; Seaman, H.E.; de Vries, C.S.; Wright, J.T. Can symptomatology help in the diagnosis of endometriosis? Findings from a national case-control study—Part 1. BJOG Int. J. Obs. Gynaecol. 2008, 115, 1382–1391. [Google Scholar] [CrossRef]
- Becker, C.M.; Bokor, A.; Heikinheimo, O.; Horne, A.; Jansen, F.; Kiesel, L.; King, K.; Kvaskoff, M.; Nap, A.; Petersen, K.; et al. ESHRE guideline: Endometriosis. Hum. Reprod. Open 2022, 2022, hoac009. [Google Scholar] [CrossRef]
- Eskenazi, B.; Warner, M.; Bonsignore, L.; Olive, D.; Samuels, S.; Vercellini, P. Validation study of nonsurgical diagnosis of endometriosis. Fertil. Steril. 2001, 76, 929–935. [Google Scholar] [CrossRef]
- Nnoaham, K.E.; Hummelshoj, L.; Kennedy, S.H.; Jenkinson, C.; Zondervan, K.T.; World Endometriosis Research Foundation Women’s Health Symptom Survey. Developing symptom-based predictive models of endometriosis as a clinical screening tool: Results from a multicenter study. Fertil. Steril. 2012, 98, 692–701.e695. [Google Scholar] [CrossRef]
- Bazot, M.; Lafont, C.; Rouzier, R.; Roseau, G.; Thomassin-Naggara, I.; Darai, E. Diagnostic accuracy of physical examination, transvaginal sonography, rectal endoscopic sonography, and magnetic resonance imaging to diagnose deep infiltrating endometriosis. Fertil. Steril. 2009, 92, 1825–1833. [Google Scholar] [CrossRef] [PubMed]
- Parasar, P.; Ozcan, P.; Terry, K.L. Endometriosis: Epidemiology, Diagnosis and Clinical Management. Curr. Obs. Gynecol. Rep. 2017, 6, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Moura, A.P.C.; Ribeiro, H.; Bernardo, W.M.; Simoes, R.; Torres, U.S.; D’Ippolito, G.; Bazot, M.; Ribeiro, P. Accuracy of transvaginal sonography versus magnetic resonance imaging in the diagnosis of rectosigmoid endometriosis: Systematic review and meta-analysis. PLoS ONE 2019, 14, e0214842, Erratum in PLoS ONE 2019, 14, e0221499. [Google Scholar] [CrossRef]
- Nisenblat, V.; Bossuyt, P.M.; Farquhar, C.; Johnson, N.; Hull, M.L. Imaging modalities for the non-invasive diagnosis of endometriosis. Cochrane Database Syst. Rev. 2016, 2, CD009591. [Google Scholar] [CrossRef] [PubMed]
- Bazot, M.; Thomassin, I.; Hourani, R.; Cortez, A.; Darai, E. Diagnostic accuracy of transvaginal sonography for deep pelvic endometriosis. Ultrasound Obs. Gynecol. 2004, 24, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Dessole, S.; Farina, M.; Rubattu, G.; Cosmi, E.; Ambrosini, G.; Nardelli, G.B. Sonovaginography is a new technique for assessing rectovaginal endometriosis. Fertil. Steril. 2003, 79, 1023–1027. [Google Scholar] [CrossRef]
- Guerriero, S.; Martinez, L.; Gomez, I.; Pascual, M.A.; Ajossa, S.; Pagliuca, M.; Alcazar, J.L. Diagnostic accuracy of transvaginal sonography for detecting parametrial involvement in women with deep endometriosis: Systematic review and meta-analysis. Ultrasound Obs. Gynecol. 2021, 58, 669–676. [Google Scholar] [CrossRef]
- Kennedy, S.; Bergqvist, A.; Chapron, C.; D’Hooghe, T.; Dunselman, G.; Greb, R.; Hummelshoj, L.; Prentice, A.; Saridogan, E.; Endometriosis, E.S.I.G.f.; et al. ESHRE guideline for the diagnosis and treatment of endometriosis. Hum. Reprod. 2005, 20, 2698–2704. [Google Scholar] [CrossRef]
- Moore, J.; Copley, S.; Morris, J.; Lindsell, D.; Golding, S.; Kennedy, S. A systematic review of the accuracy of ultrasound in the diagnosis of endometriosis. Ultrasound Obs. Gynecol. 2002, 20, 630–634. [Google Scholar] [CrossRef]
- Guerriero, S.; Condous, G.; van den Bosch, T.; Valentin, L.; Leone, F.P.; Van Schoubroeck, D.; Exacoustos, C.; Installe, A.J.; Martins, W.P.; Abrao, M.S.; et al. Systematic approach to sonographic evaluation of the pelvis in women with suspected endometriosis, including terms, definitions and measurements: A consensus opinion from the International Deep Endometriosis Analysis (IDEA) group. Ultrasound Obs. Gynecol. 2016, 48, 318–332. [Google Scholar] [CrossRef]
- Leonardi, M.; Uzuner, C.; Mestdagh, W.; Lu, C.; Guerriero, S.; Zajicek, M.; Dueckelmann, A.; Filippi, F.; Buonomo, F.; Pascual, M.A.; et al. Diagnostic accuracy of transvaginal ultrasound for detection of endometriosis using International Deep Endometriosis Analysis (IDEA) approach: Prospective international pilot study. Ultrasound Obs. Gynecol. 2022, 60, 404–413. [Google Scholar] [CrossRef]
- Taylor, H.S.; Adamson, G.D.; Diamond, M.P.; Goldstein, S.R.; Horne, A.W.; Missmer, S.A.; Snabes, M.C.; Surrey, E.; Taylor, R.N. An evidence-based approach to assessing surgical versus clinical diagnosis of symptomatic endometriosis. Int. J. Gynaecol. Obs. 2018, 142, 131–142. [Google Scholar] [CrossRef]
- Watkins, J.C.; DiVasta, A.D.; Vitonis, A.F.; Crum, C.P.; Laufer, M.R.; Terry, K.L.; Howitt, B.E.; Missmer, S.A. A Clinical and Pathologic Exploration of Suspected Peritoneal Endometriotic Lesions. Int. J. Gynecol. Pathol. 2021, 40, 602–610. [Google Scholar] [CrossRef]
- Lamvu, G.; Carrillo, J.; Ouyang, C.; Rapkin, A. Chronic Pelvic Pain in Women: A Review. JAMA 2021, 325, 2381–2391. [Google Scholar] [CrossRef]
- Zondervan, K.T.; Becker, C.M.; Missmer, S.A. Endometriosis. N. Engl. J. Med. 2020, 382, 1244–1256. [Google Scholar] [CrossRef]
- Abdul Karim, A.K.; Abd Aziz, N.H.; Md Zin, R.R.; Mohd Mokhtar, N.; Shafiee, M.N. The Effect of Surgical Intervention of Endometriosis to CA-125 and Pain. Malays. J. Med. Sci. 2020, 27, 7–14. [Google Scholar] [CrossRef]
- Kalaitzopoulos, D.R.; Samartzis, N.; Kolovos, G.N.; Mareti, E.; Samartzis, E.P.; Eberhard, M.; Dinas, K.; Daniilidis, A. Treatment of endometriosis: A review with comparison of 8 guidelines. BMC Womens Health 2021, 21, 397. [Google Scholar] [CrossRef]
- Subramaniam, R.S.; Damodaran, S.E.; Tham, S.W. Clinical Guidelines for the Management of Endometriosis 2016. OGSM: 2016. Available online: http://www.ogsm.org.my (accessed on 6 December 2022).
- Bafort, C.; Beebeejaun, Y.; Tomassetti, C.; Bosteels, J.; Duffy, J.M. Laparoscopic surgery for endometriosis. Cochrane Database Syst. Rev. 2020, 10, CD011031. [Google Scholar] [CrossRef]
- Huijs, E.; Nap, A. The effects of nutrients on symptoms in women with endometriosis: A systematic review. Reprod. Biomed. Online 2020, 41, 317–328. [Google Scholar] [CrossRef]
- Abokhrais, I.M.; Denison, F.C.; Whitaker, L.H.R.; Saunders, P.T.K.; Doust, A.; Williams, L.J.; Horne, A.W. Correction: A two-arm parallel double-blind randomised controlled pilot trial of the efficacy of Omega-3 polyunsaturated fatty acids for the treatment of women with endometriosis-associated pain (PurFECT1). PLoS ONE 2020, 15, e0230055. [Google Scholar] [CrossRef]
- Nodler, J.L.; DiVasta, A.D.; Vitonis, A.F.; Karevicius, S.; Malsch, M.; Sarda, V.; Fadayomi, A.; Harris, H.R.; Missmer, S.A. Supplementation with vitamin D or omega-3 fatty acids in adolescent girls and young women with endometriosis (SAGE): A double-blind, randomized, placebo-controlled trial. Am. J. Clin. Nutr. 2020, 112, 229–236. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.P.; Vase, L.; Hooten, W.M. Chronic pain: An update on burden, best practices, and new advances. Lancet 2021, 397, 2082–2097. [Google Scholar] [CrossRef]
- Williams, A.C.C.; Fisher, E.; Hearn, L.; Eccleston, C. Psychological therapies for the management of chronic pain (excluding headache) in adults. Cochrane Database Syst. Rev. 2020, 8, CD007407. [Google Scholar] [CrossRef] [PubMed]
- Loving, S.; Nordling, J.; Jaszczak, P.; Thomsen, T. Does evidence support physiotherapy management of adult female chronic pelvic pain? A systematic review. Scand. J. Pain. 2012, 3, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Zito, G.; Luppi, S.; Giolo, E.; Martinelli, M.; Venturin, I.; Di Lorenzo, G.; Ricci, G. Medical treatments for endometriosis-associated pelvic pain. Biomed. Res. Int. 2014, 2014, 191967. [Google Scholar] [CrossRef]
- Brown, J.; Pan, A.; Hart, R.J. Gonadotrophin-releasing hormone analogues for pain associated with endometriosis. Cochrane Database Syst. Rev. 2010, 2010, CD008475. [Google Scholar] [CrossRef]
- Donnez, J.; Taylor, H.S.; Taylor, R.N.; Akin, M.D.; Tatarchuk, T.F.; Wilk, K.; Gotteland, J.P.; Lecomte, V.; Bestel, E. Treatment of endometriosis-associated pain with linzagolix, an oral gonadotropin-releasing hormone-antagonist: A randomized clinical trial. Fertil. Steril. 2020, 114, 44–55. [Google Scholar] [CrossRef]
- Osuga, Y.; Seki, Y.; Tanimoto, M.; Kusumoto, T.; Kudou, K.; Terakawa, N. Relugolix, an oral gonadotropin-releasing hormone receptor antagonist, reduces endometriosis-associated pain in a dose-response manner: A randomized, double-blind, placebo-controlled study. Fertil. Steril. 2021, 115, 397–405. [Google Scholar] [CrossRef]
- Taylor, H.S.; Giudice, L.C.; Lessey, B.A.; Abrao, M.S.; Kotarski, J.; Archer, D.F.; Diamond, M.P.; Surrey, E.; Johnson, N.P.; Watts, N.B.; et al. Treatment of Endometriosis-Associated Pain with Elagolix, an Oral GnRH Antagonist. N. Engl. J. Med. 2017, 377, 28–40. [Google Scholar] [CrossRef]
- Coxon, L.; Horne, A.W.; Vincent, K. Pathophysiology of endometriosis-associated pain: A review of pelvic and central nervous system mechanisms. Best Pr. Res. Clin. Obs. Gynaecol. 2018, 51, 53–67. [Google Scholar] [CrossRef]
- Blum, H.E. The human microbiome. Adv. Med. Sci. 2017, 62, 414–420. [Google Scholar] [CrossRef]
- Martinez-Guryn, K.; Leone, V.; Chang, E.B. Regional Diversity of the Gastrointestinal Microbiome. Cell Host Microbe 2019, 26, 314–324. [Google Scholar] [CrossRef]
- Lopetuso, L.R.; Scaldaferri, F.; Franceschi, F.; Gasbarrini, A. The gastrointestinal microbiome—Functional interference between stomach and intestine. Best Pr. Res. Clin. Gastroenterol. 2014, 28, 995–1002. [Google Scholar] [CrossRef]
- Noto, J.M.; Peek, R.M., Jr. The gastric microbiome, its interaction with Helicobacter pylori, and its potential role in the progression to stomach cancer. PLoS Pathog. 2017, 13, e1006573. [Google Scholar] [CrossRef]
- Ruan, W.; Engevik, M.A.; Spinler, J.K.; Versalovic, J. Healthy Human Gastrointestinal Microbiome: Composition and Function After a Decade of Exploration. Dig. Dis. Sci. 2020, 65, 695–705. [Google Scholar] [CrossRef]
- Mason, K.L.; Huffnagle, G.B.; Noverr, M.C.; Kao, J.Y. Overview of gut immunology. Adv. Exp. Med. Biol. 2008, 635, 1–14. [Google Scholar] [CrossRef]
- Morbe, U.M.; Jorgensen, P.B.; Fenton, T.M.; von Burg, N.; Riis, L.B.; Spencer, J.; Agace, W.W. Human gut-associated lymphoid tissues (GALT); diversity, structure, and function. Mucosal Immunol. 2021, 14, 793–802. [Google Scholar] [CrossRef]
- Li, S.; Chen, S.; Nie, M.; Wen, L.; Zou, B.; Zhang, L.; Xie, J.; Ser, H.L.; Lee, L.H.; Wang, S.; et al. Salt-Sensitive Ileal Microbiota Plays a Role in Atrial Natriuretic Peptide Deficiency-Induced Cardiac Injury. Nutrients 2022, 14, 3129. [Google Scholar] [CrossRef]
- Ser, H.-L.; Wong, J.Y.-J.; Goh, B.-H.; Reginald, K. IDDF2022-ABS-0236 Healing the GUT with probiotics: Can probiotics help relieve allergic rhinitis? Gut 2022, 71, A63–A64. [Google Scholar]
- Joseph, R.J.; Ser, H.L.; Kuai, Y.H.; Tan, L.T.; Arasoo, V.J.T.; Letchumanan, V.; Wang, L.; Pusparajah, P.; Goh, B.H.; Ab Mutalib, N.S.; et al. Finding a Balance in the Vaginal Microbiome: How Do We Treat and Prevent the Occurrence of Bacterial Vaginosis? Antibiotics 2021, 10, 719. [Google Scholar] [CrossRef]
- Mestrovic, T.; Matijasic, M.; Peric, M.; Cipcic Paljetak, H.; Baresic, A.; Verbanac, D. The Role of Gut, Vaginal, and Urinary Microbiome in Urinary Tract Infections: From Bench to Bedside. Diagnostics 2020, 11, 7. [Google Scholar] [CrossRef] [PubMed]
- Ser, H.-L.; Wong, J.Y.J.; Letchumanan, V.; Law, J.W.-F.; Tan, L.T.-H.; Lee, L.-H. IDDF2021-ABS-0132 Moving beyond the gastrointestinal tract: The involvement of gut microbiome in endometriosis. Gut 2021, 70, A46–A47. [Google Scholar]
- Plesniarski, A.; Siddik, A.B.; Su, R.C. The Microbiome as a Key Regulator of Female Genital Tract Barrier Function. Front. Cell. Infect. Microbiol. 2021, 11, 790627. [Google Scholar] [CrossRef] [PubMed]
- Punzon-Jimenez, P.; Labarta, E. The impact of the female genital tract microbiome in women health and reproduction: A review. J. Assist. Reprod. Genet. 2021, 38, 2519–2541. [Google Scholar] [CrossRef]
- Gajer, P.; Brotman, R.M.; Bai, G.; Sakamoto, J.; Schutte, U.M.; Zhong, X.; Koenig, S.S.; Fu, L.; Ma, Z.S.; Zhou, X.; et al. Temporal dynamics of the human vaginal microbiota. Sci. Transl. Med. 2012, 4, 132ra152. [Google Scholar] [CrossRef]
- Ravel, J.; Gajer, P.; Abdo, Z.; Schneider, G.M.; Koenig, S.S.; McCulle, S.L.; Karlebach, S.; Gorle, R.; Russell, J.; Tacket, C.O.; et al. Vaginal microbiome of reproductive-age women. Proc. Natl. Acad. Sci. USA 2011, 108, 4680–4687. [Google Scholar] [CrossRef]
- Ata, B.; Yildiz, S.; Turkgeldi, E.; Brocal, V.P.; Dinleyici, E.C.; Moya, A.; Urman, B. The endobiota study: Comparison of vaginal, cervical and gut microbiota between women with stage 3/4 endometriosis and healthy controls. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Chen, S.; Gu, Z.; Zhang, W.; Jia, S.; Wu, Y.; Zheng, P.; Dai, Y.; Leng, J. Microbiome of the lower genital tract in Chinese women with endometriosis by 16s-rRNA sequencing technique: A pilot study. Ann. Transl. Med. 2020, 8, 1440. [Google Scholar] [CrossRef]
- Wei, W.; Zhang, X.; Tang, H.; Zeng, L.; Wu, R. Microbiota composition and distribution along the female reproductive tract of women with endometriosis. Ann. Clin. Microbiol. Antimicrob. 2020, 19, 15. [Google Scholar] [CrossRef]
- Lee, S.R.; Lee, J.C.; Kim, S.H.; Oh, Y.S.; Chae, H.D.; Seo, H.; Kang, C.S.; Shin, T.S. Altered Composition of Microbiota in Women with Ovarian Endometrioma: Microbiome Analyses of Extracellular Vesicles in the Peritoneal Fluid. Int. J. Mol. Sci. 2021, 22, 4608. [Google Scholar] [CrossRef]
- Huang, L.; Liu, B.; Liu, Z.; Feng, W.; Liu, M.; Wang, Y.; Peng, D.; Fu, X.; Zhu, H.; Cui, Z.; et al. Gut Microbiota Exceeds Cervical Microbiota for Early Diagnosis of Endometriosis. Front. Cell. Infect. Microbiol. 2021, 11, 788836. [Google Scholar] [CrossRef]
- Shan, J.; Ni, Z.; Cheng, W.; Zhou, L.; Zhai, D.; Sun, S.; Yu, C. Gut microbiota imbalance and its correlations with hormone and inflammatory factors in patients with stage 3/4 endometriosis. Arch. Gynecol. Obs. 2021, 304, 1363–1373. [Google Scholar] [CrossRef]
- Chao, X.; Liu, Y.; Fan, Q.; Shi, H.; Wang, S.; Lang, J. The role of the vaginal microbiome in distinguishing female chronic pelvic pain caused by endometriosis/adenomyosis. Ann. Transl. Med. 2021, 9, 771. [Google Scholar] [CrossRef]
- Chang, C.Y.; Chiang, A.J.; Lai, M.T.; Yan, M.J.; Tseng, C.C.; Lo, L.C.; Wan, L.; Li, C.J.; Tsui, K.H.; Chen, C.M.; et al. A more diverse cervical microbiome associates with better clinical outcomes in patients with endometriosis: A pilot study. Biomedicines 2022, 10, 174. [Google Scholar] [CrossRef]
- Oishi, S.; Mekaru, K.; Tanaka, S.E.; Arai, W.; Ashikawa, K.; Sakuraba, Y.; Nishioka, M.; Nakamura, R.; Miyagi, M.; Akamine, K. Microbiome analysis in women with endometriosis: Does a microbiome exist in peritoneal fluid and ovarian cystic fluid? Reprod. Med. Biol. 2022, 21, e12441. [Google Scholar] [CrossRef]
- Yuan, W.; Wu, Y.; Chai, X.; Wu, X. The colonized microbiota composition in the peritoneal fluid in women with endometriosis. Arch. Gynecol. Obs. 2022, 305, 1573–1580. [Google Scholar] [CrossRef]
- Lu, F.; Wei, J.; Zhong, Y.; Feng, Y.; Ma, B.; Xiong, Y.; Wei, K.; Tan, B.; Chen, T. Antibiotic Therapy and Vaginal Microbiota Transplantation Reduce Endometriosis Disease Progression in Female Mice via NF-kappaB Signaling Pathway. Front. Med. 2022, 9, 831115. [Google Scholar] [CrossRef]
- Hernandes, C.; Silveira, P.; Rodrigues Sereia, A.F.; Christoff, A.P.; Mendes, H.; Valter de Oliveira, L.F.; Podgaec, S. Microbiome Profile of Deep Endometriosis Patients: Comparison of Vaginal Fluid, Endometrium and Lesion. Diagnostics 2020, 10, 163. [Google Scholar] [CrossRef]
- Perrotta, A.R.; Borrelli, G.M.; Martins, C.O.; Kallas, E.G.; Sanabani, S.S.; Griffith, L.G.; Alm, E.J.; Abrao, M.S. The vaginal microbiome as a tool to predict rASRM stage of disease in endometriosis: A pilot study. Reprod. Sci. 2020, 27, 1064–1073. [Google Scholar] [CrossRef]
- Le, N.; Cregger, M.; Fazleabas, A.; Braundmeier-Fleming, A. Effects of endometriosis on immunity and mucosal microbial community dynamics in female olive baboons. Sci. Rep. 2022, 12, 1590. [Google Scholar] [CrossRef]
- Svensson, A.; Brunkwall, L.; Roth, B.; Orho-Melander, M.; Ohlsson, B. Associations between endometriosis and gut microbiota. Reprod. Sci. 2021, 28, 2367–2377. [Google Scholar] [CrossRef] [PubMed]
- Wessels, J.M.; Domínguez, M.A.; Leyland, N.A.; Agarwal, S.K.; Foster, W.G. Endometrial microbiota is more diverse in people with endometriosis than symptomatic controls. Sci. Rep. 2021, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Le, N.; Cregger, M.; Brown, V.; Loret de Mola, J.; Bremer, P.; Nguyen, L.; Groesch, K.; Wilson, T.; Diaz-Sylvester, P.; Braundmeier-Fleming, A. Association of microbial dynamics with urinary estrogens and estrogen metabolites in patients with endometriosis. PLoS ONE 2021, 16, e0261362. [Google Scholar] [CrossRef] [PubMed]
- Simoes-Silva, L.; Araujo, R.; Pestana, M.; Soares-Silva, I.; Sampaio-Maia, B. Peritoneal Microbiome in End-Stage Renal Disease Patients and the Impact of Peritoneal Dialysis Therapy. Microorganisms 2020, 8, 173. [Google Scholar] [CrossRef]
- Gilbreath, J.J.; Semino-Mora, C.; Friedline, C.J.; Liu, H.; Bodi, K.L.; McAvoy, T.J.; Francis, J.; Nieroda, C.; Sardi, A.; Dubois, A.; et al. A core microbiome associated with the peritoneal tumors of pseudomyxoma peritonei. Orphanet J. Rare Dis. 2013, 8, 105. [Google Scholar] [CrossRef]
- Semino-Mora, C.; Liu, H.; McAvoy, T.; Nieroda, C.; Studeman, K.; Sardi, A.; Dubois, A. Pseudomyxoma peritonei: Is disease progression related to microbial agents? A study of bacteria, MUC2 AND MUC5AC expression in disseminated peritoneal adenomucinosis and peritoneal mucinous carcinomatosis. Ann. Surg. Oncol. 2008, 15, 1414–1423. [Google Scholar] [CrossRef]
- Ferrero, S.; Vellone, V.G.; Barra, F. Pathophysiology of pain in patients with peritoneal endometriosis. Ann. Transl. Med. 2019, 7, S8. [Google Scholar] [CrossRef]
- Taniguchi, F.; Wibisono, H.; Mon Khine, Y.; Harada, T. Animal models for research on endometriosis. Front. Biosci. 2021, 13, 37–53. [Google Scholar] [CrossRef]
- Vernon, M.W.; Wilson, E.A. Studies on the surgical induction of endometriosis in the rat. Fertil. Steril. 1985, 44, 684–694. [Google Scholar] [CrossRef]
- Yuan, M.; Li, D.; Zhang, Z.; Sun, H.; An, M.; Wang, G. Endometriosis induces gut microbiota alterations in mice. Hum. Reprod. 2018, 33, 607–616. [Google Scholar] [CrossRef]
- Chadchan, S.B.; Cheng, M.; Parnell, L.A.; Yin, Y.; Schriefer, A.; Mysorekar, I.U.; Kommagani, R. Antibiotic therapy with metronidazole reduces endometriosis disease progression in mice: A potential role for gut microbiota. Hum. Reprod. 2019, 34, 1106–1116. [Google Scholar] [CrossRef]
- Hantschel, J.; Weis, S.; Schafer, K.H.; Menger, M.D.; Kohl, M.; Egert, M.; Laschke, M.W. Effect of endometriosis on the fecal bacteriota composition of mice during the acute phase of lesion formation. PLoS ONE 2019, 14, e0226835. [Google Scholar] [CrossRef]
- Ni, Z.; Sun, S.; Bi, Y.; Ding, J.; Cheng, W.; Yu, J.; Zhou, L.; Li, M.; Yu, C. Correlation of fecal metabolomics and gut microbiota in mice with endometriosis. Am. J. Reprod. Immunol. 2020, 84, e13307. [Google Scholar] [CrossRef]
- Cao, Y.; Jiang, C.; Jia, Y.; Xu, D.; Yu, Y. Letrozole and the traditional chinese medicine, Shaofu Zhuyu decoction, reduce endometriotic disease progression in rats: A potential role for gut microbiota. Evid. Based Complement. Altern. Med. 2020, 2020, 3687498. [Google Scholar] [CrossRef]
- Ni, Z.; Ding, J.; Zhao, Q.; Cheng, W.; Yu, J.; Zhou, L.; Sun, S.; Yu, C. Alpha-linolenic acid regulates the gut microbiota and the inflammatory environment in a mouse model of endometriosis. Am. J. Reprod. Immunol. 2021, 86, e13471. [Google Scholar] [CrossRef]
- Higuchi, B.S.; Rodrigues, N.; Gonzaga, M.I.; Paiolo, J.C.C.; Stefanutto, N.; Omori, W.P.; Pinheiro, D.G.; Brisotti, J.L.; Matheucci, E., Jr.; Mariano, V.S.; et al. Intestinal Dysbiosis in Autoimmune Diabetes Is Correlated With Poor Glycemic Control and Increased Interleukin-6: A Pilot Study. Front. Immunol. 2018, 9, 1689. [Google Scholar] [CrossRef]
- Leite, A.Z.; Rodrigues, N.C.; Gonzaga, M.I.; Paiolo, J.C.C.; de Souza, C.A.; Stefanutto, N.A.V.; Omori, W.P.; Pinheiro, D.G.; Brisotti, J.L.; Matheucci, E., Jr.; et al. Detection of Increased Plasma Interleukin-6 Levels and Prevalence of Prevotella copri and Bacteroides vulgatus in the Feces of Type 2 Diabetes Patients. Front. Immunol. 2017, 8, 1107. [Google Scholar] [CrossRef]
- Henke, M.T.; Kenny, D.J.; Cassilly, C.D.; Vlamakis, H.; Xavier, R.J.; Clardy, J. Ruminococcus gnavus, a member of the human gut microbiome associated with Crohn’s disease, produces an inflammatory polysaccharide. Proc. Natl. Acad. Sci. USA 2019, 116, 12672–12677. [Google Scholar] [CrossRef]
- Jiang, W.; Wu, N.; Wang, X.; Chi, Y.; Zhang, Y.; Qiu, X.; Hu, Y.; Li, J.; Liu, Y. Dysbiosis gut microbiota associated with inflammation and impaired mucosal immune function in intestine of humans with non-alcoholic fatty liver disease. Sci. Rep. 2015, 5, 8096. [Google Scholar] [CrossRef]
- Hall, A.B.; Yassour, M.; Sauk, J.; Garner, A.; Jiang, X.; Arthur, T.; Lagoudas, G.K.; Vatanen, T.; Fornelos, N.; Wilson, R.; et al. A novel Ruminococcus gnavus clade enriched in inflammatory bowel disease patients. Genome Med. 2017, 9, 103. [Google Scholar] [CrossRef]
- Meng, J.; Banerjee, S.; Zhang, L.; Sindberg, G.; Moidunny, S.; Li, B.; Robbins, D.J.; Girotra, M.; Segura, B.; Ramakrishnan, S.; et al. Opioids Impair Intestinal Epithelial Repair in HIV-Infected Humanized Mice. Front. Immunol. 2019, 10, 2999. [Google Scholar] [CrossRef] [PubMed]
- Khodaverdi, S.; Mohammadbeigi, R.; Khaledi, M.; Mesdaghinia, L.; Sharifzadeh, F.; Nasiripour, S.; Gorginzadeh, M. Beneficial Effects of Oral Lactobacillus on Pain Severity in Women Suffering from Endometriosis: A Pilot Placebo-Controlled Randomized Clinical Trial. Int. J. Fertil. Steril. 2019, 13, 178–183. [Google Scholar] [CrossRef] [PubMed]
- Itoh, H.; Sashihara, T.; Hosono, A.; Kaminogawa, S.; Uchida, M. Lactobacillus gasseri OLL2809 inhibits development of ectopic endometrial cell in peritoneal cavity via activation of NK cells in a murine endometriosis model. Cytotechnology 2011, 63, 205–210. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.; Ali, R.A.R.; Manaf, M.R.A.; Ahmad, N.; Tajurruddin, F.W.; Qin, W.Z.; Desa, S.H.M.; Ibrahim, N.M. Multi-strain probiotics (Hexbio) containing MCP BCMC strains improved constipation and gut motility in Parkinson’s disease: A randomised controlled trial. PLoS ONE 2020, 15, e0244680. [Google Scholar] [CrossRef] [PubMed]
- Itoh, H.; Uchida, M.; Sashihara, T.; Ji, Z.S.; Li, J.; Tang, Q.; Ni, S.; Song, L.; Kaminogawa, S. Lactobacillus gasseri OLL2809 is effective especially on the menstrual pain and dysmenorrhea in endometriosis patients: Randomized, double-blind, placebo-controlled study. Cytotechnology 2011, 63, 153–161. [Google Scholar] [CrossRef]
- Uchida, M.; Kobayashi, O. Effects of Lactobacillus gasseri OLL2809 on the induced endometriosis in rats. Biosci. Biotechnol. Biochem. 2013, 77, 1879–1881. [Google Scholar] [CrossRef]
- Del Piano, M.; Carmagnola, S.; Ballare, M.; Sartori, M.; Orsello, M.; Balzarini, M.; Pagliarulo, M.; Tari, R.; Anderloni, A.; Strozzi, G.P.; et al. Is microencapsulation the future of probiotic preparations? The increased efficacy of gastro-protected probiotics. Gut Microbes 2011, 2, 120–123. [Google Scholar] [CrossRef]
- Sanders, M.E.; Klaenhammer, T.R.; Ouwehand, A.C.; Pot, B.; Johansen, E.; Heimbach, J.T.; Marco, M.L.; Tennila, J.; Ross, R.P.; Franz, C.; et al. Effects of genetic, processing, or product formulation changes on efficacy and safety of probiotics. Ann. New York Acad. Sci. 2014, 1309, 1–18. [Google Scholar] [CrossRef]
- Su, K.-Y.; Koh Kok, J.-Y.; Chua, Y.-W.; Ong, S.-D.; Ser, H.L.; Pusparajah, P.; San Saw, P.; Goh, B.H.; Lee, W.-L. Bacterial extracellular vesicles in biofluids as potential diagnostic biomarkers. Expert Rev. Mol. Diagn. 2023, 22, 1057–1062. [Google Scholar] [CrossRef]
- Anderson, G. Endometriosis Pathoetiology and Pathophysiology: Roles of Vitamin A, Estrogen, Immunity, Adipocytes, Gut Microbiome and Melatonergic Pathway on Mitochondria Regulation. Biomol. Concepts 2019, 10, 133–149. [Google Scholar] [CrossRef]
- Toson, B.; Simon, C.; Moreno, I. The Endometrial Microbiome and Its Impact on Human Conception. Int. J. Mol. Sci. 2022, 23, 485. [Google Scholar] [CrossRef]
- Baker, J.M.; Al-Nakkash, L.; Herbst-Kralovetz, M.M. Estrogen-gut microbiome axis: Physiological and clinical implications. Maturitas 2017, 103, 45–53. [Google Scholar] [CrossRef]
- Kaliannan, K.; Robertson, R.C.; Murphy, K.; Stanton, C.; Kang, C.; Wang, B.; Hao, L.; Bhan, A.K.; Kang, J.X. Estrogen-mediated gut microbiome alterations influence sexual dimorphism in metabolic syndrome in mice. Microbiome 2018, 6, 205. [Google Scholar] [CrossRef]
- Karim, A.K.A.; Shafiee, M.N.; Abd Aziz, N.H.; Omar, M.H.; Zin, R.R.M.; Mokhtar, N.M. Perturbations in progesterone receptor isoforms in endometriosis. J. Reprod. Med. 2019, 64, 275–281. [Google Scholar]
| Treatments for Pain | Endometriosis Guidelines | ||||||
|---|---|---|---|---|---|---|---|
| ESHRE a (2022) | TIGE (2022) | NICE (2017) | OGSM (2016) | ASRM (2014) | |||
| Surgical | Ablation | •• | ✓ | ✓ | ✓ | ✓ | |
| Excision | •• | ✓ | ✓ | ✓ | ✓ | ||
| Excision of Ovarian Endometrioma | •• | ✓ | ✓ | ✓ | ✓ | ||
| Ablation of Ovarian Endometrioma | • | ✓ | ✓ | ✓ | ✓ | ||
| Excision of Deep-Infiltrating Endometriosis | •• | ||||||
| Hysterectomy | •• | ✓ | ✓ | ✓ | ✓ | ||
| Adhesiolysis (Anti-adhesion Agents) | X | ||||||
| Presacral Neurectomy | X | (SE) | |||||
| Laparoscopic Uterosacral Nerve Ablation | |||||||
| Pharmacological | Combination Oral Contraceptives Pills | •• | ✓ | ✓ | ✓ | ✓ | |
| Danazol | X | ✓ (SE) | ✓ (SE) | ✓ (SE) | |||
| Dienogest | •• | ✓ | ✓ | ✓ | ✓ | ||
| Medroxyprogesterone Acetate | •• | ✓ | ✓ | ✓ | ✓ | ||
| Levonorgestrel-Releasing Intrauterine System | ••• | ✓ | ✓ | ✓ | ✓ | ||
| Anti-Progesterone (Gestrinone) | X | ✓ | |||||
| Analgesics (non-steroidal anti-inflammatory drugs) | • | GPP | GPP | ||||
| Aromatose Inhibitors | •• | ✓ | |||||
| GnRH-Agonist | •• | ✓ | ✓ | ✓ | ✓ | ||
| GnRH-Antagonist | ••• | ||||||
| Selective Progesterone Receptor Modulators | ••• | ||||||
| Selective Estrogen Receptor Modulators | ••• | ||||||
| First-Line of Treatment | Not Recommended d | •• | Low Evidence Level | X | Removed by GDG g | ||
| Second-Line of Treatment | No Recommendation c | ••• | Moderate Evidence Level | SE | Critical due to Side-Effect f | ||
| Third-Line of Treatment | Insufficient Evidence | GPP | Good Practice Point b | SC | Special Cases e | ||
| Additional/Other Treatment | • | Very Low Evidence Level | ✓ | No Information of Evidence | M | Mild Endometriosis | |
| Treatments for Infertility | Endometriosis Guidelines | ||||||
|---|---|---|---|---|---|---|---|
| ESHRE a (2022) | NICE (2017) | OGSM (2016) | ASRM (2014) | ||||
| Surgical | Ablation | •• (M) | ✓ | ✓ (M) | ✓ (M) | ||
| Excision | •• (M) | ✓ | ✓ (M) | ✓ (M) | |||
| Excision of Ovarian Endometrioma | ✓ | ✓ (M) | ✓ (M) | ||||
| Ablation of Ovarian Endometrioma | ✓ | ✓ (M) | ✓ (M) | ||||
| Excision of Deep-Infiltrating Endometriosis | |||||||
| Adhesiolysis (Anti-adhesion Agents) | ✓ | ||||||
| Pharmacological | Combination Oral Contraceptives Pills | •• | ✓ | ✓ | ✓ | ||
| Danazol | X | ✓ | ✓ | ✓ | |||
| Dienogest | ✓ | ✓ | ✓ | ||||
| Medroxyprogesterone Acetate | ✓ | ✓ | ✓ | ||||
| Anti-Progesterone (Gestrinone) | ✓ | ✓ | ✓ | ||||
| Aromatose Inhibitors | ✓ | ✓ | |||||
| GnRH-Agonist | •• | ✓ | ✓ | ✓ | |||
| GnRH-Antagonist | ✓ | ||||||
| Selective Progesterone Receptor Modulators | ✓ | ||||||
| Selective Estrogen Receptor Modulators | ✓ | ||||||
| Assisted Reproduction Techniques (ART) | Intrauterine Insemination (IUI) | • | ✓ | ✓ (M) | |||
| In vitro fertilization/Intracytoplasmic sperm injection | •• | ✓ | ✓ | ||||
| First-Line of Treatment | Not Recommended d | •• | Low Evidence Level | M | Mild Endometriosis | ||
| Second-Line of Treatment | No Recommendation c | ••• | Moderate Evidence Level | ||||
| Third-Line of Treatment | Insufficient Evidence | GPP | Good Practice Point b | ||||
| Additional/Other Treatment | • | Very Low Evidence Level | ✓ | No Information of Evidence | |||
| References | Year | Study Location | Mean Age (Years Old) | Microbiome Analysis | Findings |
|---|---|---|---|---|---|
| Akiyama et al. [21] | 2019 | Japan | Endometriosis: 33.9 ± 5.7 Control: 32.5 ± 6.0 | Cervical Mucus |
|
| Ata et al. [108] | 2019 | Turkey | # Endometriosis: 28.5 (range: 26–31.3) Control: 27.5 (range: 25.8–30) | Fecal Sample, Vaginal and Endocervical Swabs |
|
| Chen et al. [109] | 2020 | China | 36.07 ± 5.57 (range: 18–45) | Cervical swabs and Vaginal swabs |
|
| Hernandes et al. [119] | 2020 | Brazil | 18–50 | Vaginal fluid, eutopic endometrium, and endometriotic lesion |
|
| Wei et al. [110] | 2020 | China | 31.47 (range: 23–44) | Vagina swab, posterior vaginal fornix swab, cervical mucus swab, endometrium, and peritoneal fluid |
|
| Perrotta et al. [120] | 2020 | Brazil | Endometriosis: 34.9 ± 6.8 Control: 35.25 ± 6.9 | Rectal and vaginal swab |
|
| Lee et al. [111] | 2021 | Korea | Endometriosis: 36.20 ± 1.30 Control: 39.40 ± 1.10 | Extracellular vesicles in peritoneal fluid |
|
| Le et al. [124] | 2021 | United States | Endometriosis: 32.5 ± 1.1 Control: 32.6 ± 2.0 | Urine, fecal and vaginal swab |
|
| Svensson et al. [122] | 2021 | Sweden | Endometriosis: 37.8 (range: 32.8–43.3) Control: 37.0 (range: 32.0–44.0) | Feces (self-collected) |
|
| Wessels et al. [123] | 2021 | Canada | Endometriosis: 33.8 ± 5.8 Control: 35.1 ± 3.3 | Endometrial biopsy tissue |
|
| Huang et al. [112] | 2021 | China | Endometriosis: 38.3 ± 7.88 Control: 34.0 ± 10.8 | Peritoneal fluid, cervical swab, and feces (self-collected) |
|
| Shan et al. [113] | 2021 | China | Endometriosis/Control: 32 ± 2, 32 ± 3 | Feces (self-collected) |
|
| Chao et al. [114] | 2021 | China | Endometriosis: 39.89 ± 6.24 Control: 38.23 ± 7.80 | Vaginal swab |
|
| Chang et al. [115] | 2022 | Taiwan | Endometriosis: 35.4 ± 6.7 Control: N.A. | Cervical swab |
|
| Oishi et al. [116] | 2022 | Japan | Endometriosis: 37.9 ± 6.4 Control: 35.2 ± 8.6 | Vaginal fluid, endometrial fluid, peritoneal fluid, ovarian cystic fluid |
|
| Yuan et al. [117] | 2022 | China | Endometriosis: 35.28 ± 7.24 Control: 33.32 ± 8.04 | Peritoneal fluid |
|
| Lu et al. [118] | 2022 | China | Endometriosis: 36.75 ± 7.11 Control: 35 ± 6.61 | Vaginal fluid |
|
| References | Year | Animal Used and Age | Endometriosis Model | Specimen and Methods Used to Evaluate Microbiome Changes | Important Findings on Microbiome Changes |
|---|---|---|---|---|---|
| Yuan et al. [131] | 2018 | Female C57BL6 mice | Intraperitoneal injection of endometrial segments | Fecal pellet (7, 14, 28, 42 days post-induction)—16S rRNA analysis (V4 region) on Illumina HiSeq platform |
|
| Chadchan et al. [132] | 2019 | Female C57BL6 mice | Intraperitoneal injection of endometrial segments (autologous) | Fecal pellet (21 days post-induction)—16S rRNA analysis on Illumina MiSeq platform |
|
| Hantschel et al. [133] | 2019 | 12–16 weeks old female C57BL6 mice (Wild-type and Transgenic TgN (ACTB-EGFP) | Intraperitoneal injection of uterine tissue fragments (biopsy punch) | Fecal pellet (7 and 21 days post-induction)—16S rRNA analysis (V4-V5 region) on Illumina Miseq platform |
|
| Ni et al. [134] | 2020 | 6 weeks old female C57BL6 mice | Intraperitoneal injection of endometrial segments | Feces from cecum segment (21 days post-induction)—16S rRNA analysis (V3-V4 region) on Illumina MiSeq platform |
|
| Cao et al. [135] | 2020 | 6–8 weeks old female SD Rats | Intraperitoneal injection of uterine tissue fragments (autologous) | Fecal pellet (28 days post-induction)—16S rRNA analysis (V3-V4 region) on Ion S5TMXL sequencer |
|
| Ni et al. [136] | 2021 | 6 weeks old female C57BL6 mice | Intraperitoneal injection of endometrial segments, fecal microbiota transplant | Fecal pellet (21 days post-induction)—16S rRNA analysis (V4-V5 region) on Illumina MiSeq platform |
|
| Le et al. [121] | 2022 | Non-human primates (Papio Anubis), In reproductive age | Intraperitoneal injection of autologous menstrual tissues for 2 consecutive months during menses | Fecal pellets, urine, vaginal swab, and peritoneal fluid (3, 6, 9, 15 months post-induction)—16S rRNA analysis (V4 region) on Illumina MiSeq platform |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ser, H.-L.; Au Yong, S.-J.; Shafiee, M.N.; Mokhtar, N.M.; Ali, R.A.R. Current Updates on the Role of Microbiome in Endometriosis: A Narrative Review. Microorganisms 2023, 11, 360. https://doi.org/10.3390/microorganisms11020360
Ser H-L, Au Yong S-J, Shafiee MN, Mokhtar NM, Ali RAR. Current Updates on the Role of Microbiome in Endometriosis: A Narrative Review. Microorganisms. 2023; 11(2):360. https://doi.org/10.3390/microorganisms11020360
Chicago/Turabian StyleSer, Hooi-Leng, Siu-Jung Au Yong, Mohamad Nasir Shafiee, Norfilza Mohd Mokhtar, and Raja Affendi Raja Ali. 2023. "Current Updates on the Role of Microbiome in Endometriosis: A Narrative Review" Microorganisms 11, no. 2: 360. https://doi.org/10.3390/microorganisms11020360
APA StyleSer, H.-L., Au Yong, S.-J., Shafiee, M. N., Mokhtar, N. M., & Ali, R. A. R. (2023). Current Updates on the Role of Microbiome in Endometriosis: A Narrative Review. Microorganisms, 11(2), 360. https://doi.org/10.3390/microorganisms11020360






