Molecular Epidemiology, Seasonality and Phylogenetic Investigations of Anaplasma ovis in Small Ruminants from Diverse Agro-Climatic Regions of Punjab, Pakistan
Abstract
:1. Introduction
2. Materials and Methods
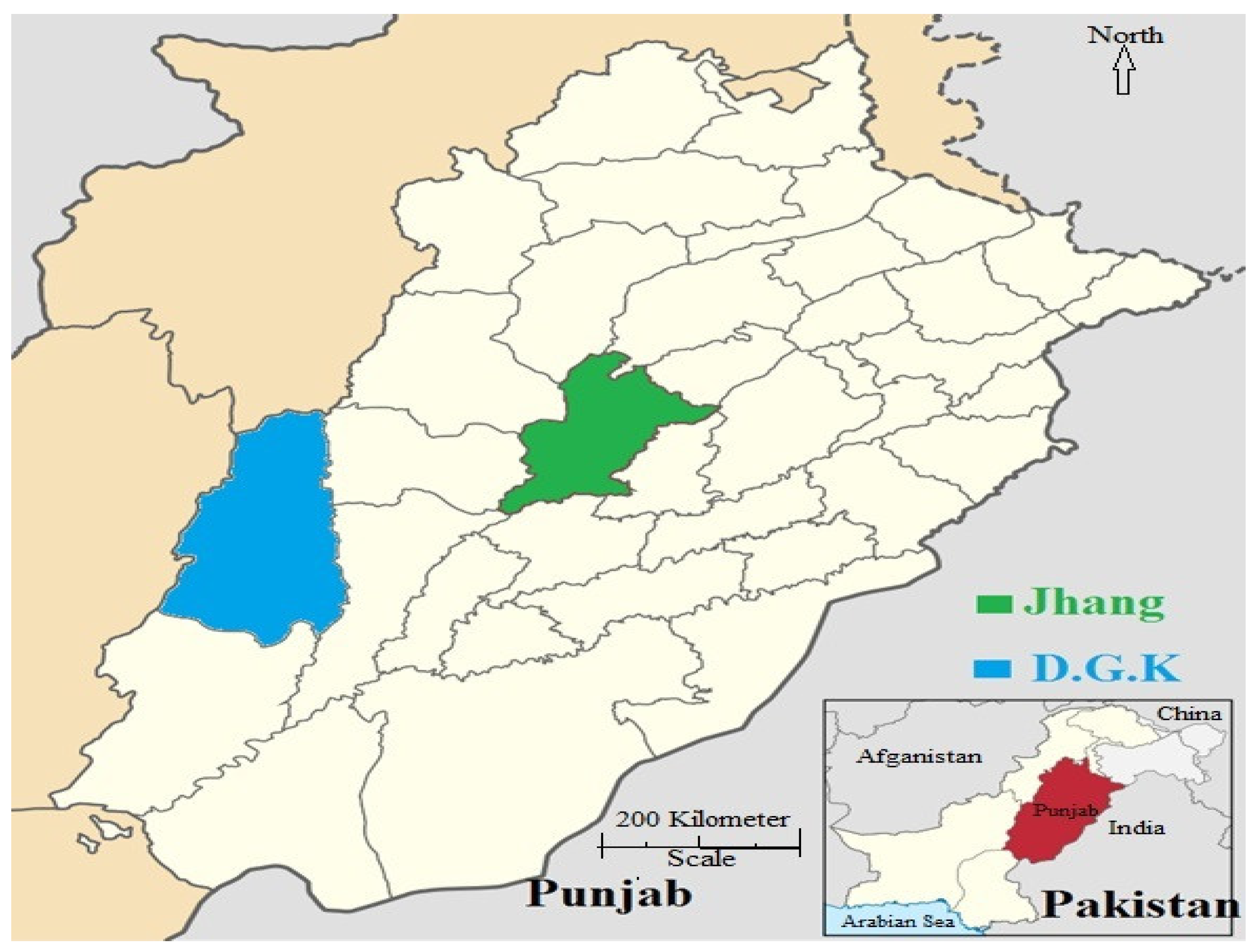
2.1. Study Area and Sample Collection
2.2. DNA Extraction
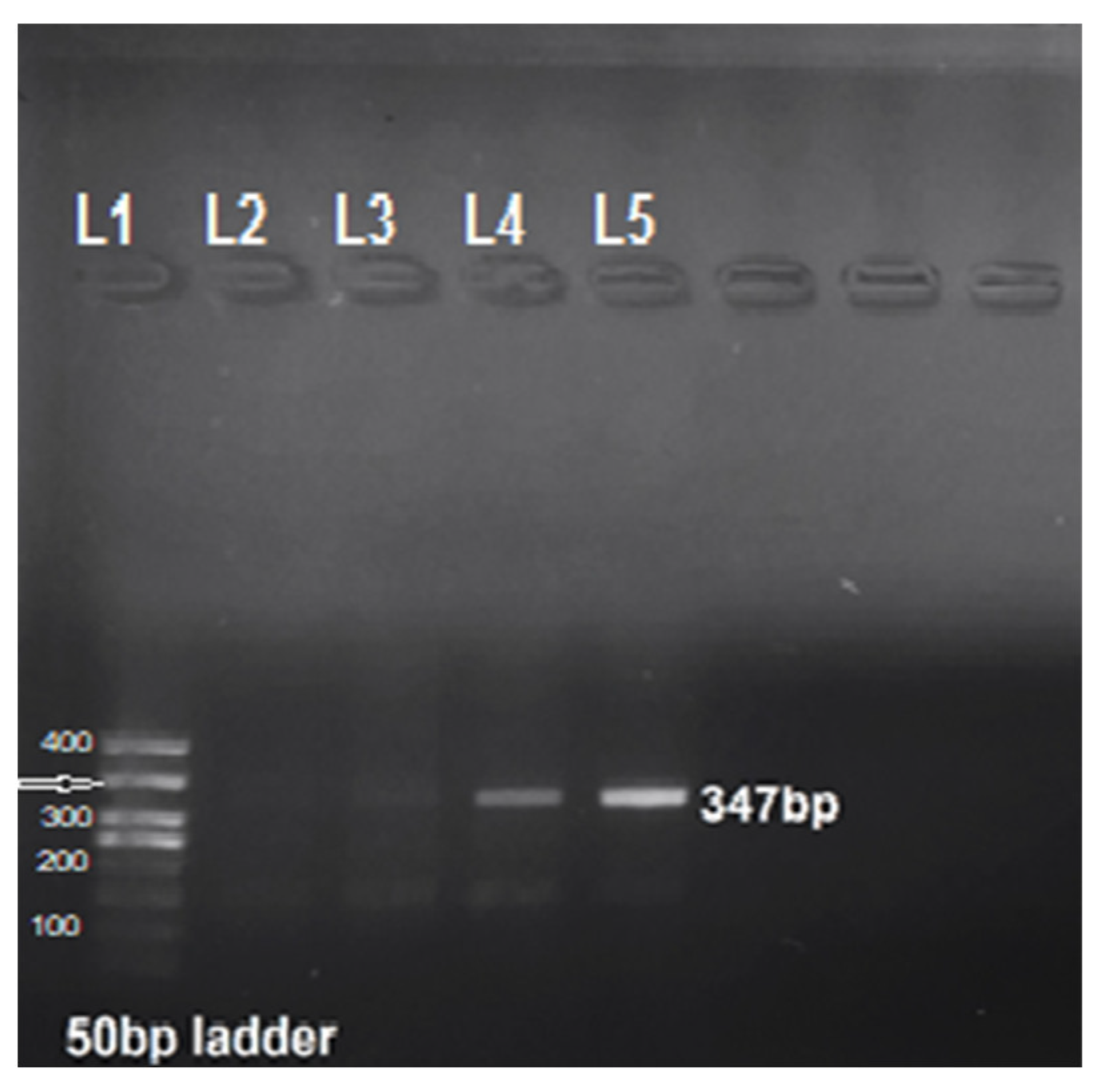
2.3. PCR
2.4. Estimation of Risk Factors
2.5. Sequencing and Phylogenetic Analysis
2.6. Statistical Analysis
2.7. Ethical Approval
3. Results
3.1. Molecular Prevalence
3.2. Risk Factor Study
3.3. Phylogenetic Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Economic Survey of Pakistan. Agriculture, Ministry of National Food Security and Research; Government of Pakistan: Islamabad, Pakistan, 2023; pp. 34–36.
- Khan, M.; Khan, M.; Ahmad, S.; Mahmood, S. Genetic resources and diversity in Pakistani sheep. Int. J. Agric. Biol. 2007, 9, 941–944. [Google Scholar]
- Renneker, S.; Abdo, J.; Salih, D.E.; Karagenç, T.; Bilgiç, H.; Torina, A.; Oliva, A.G.; Campos, J.; Kullmann, B.; Ahmed, J.; et al. Can Anaplasma ovis in small ruminants be neglected any longer? Transbound. Emerg. Dis. 2013, 60 (Suppl. S2), 105–112. [Google Scholar] [CrossRef] [PubMed]
- Jabbar, A.; Abbas, T.; Sandhu, Z.-D.; Saddiqi, H.A.; Qamar, M.F.; Gasser, R.B. Tick-borne diseases of bovines in Pakistan: Major scope for future research and improved control. Parasites Vectors 2015, 8, 283. [Google Scholar] [CrossRef] [PubMed]
- Yasini, S.P.; Khaki, Z.; Rahbari, S.; Kazemi, B.; Amoli, J.S.; Gharabaghi, A.; Jalali, S.M. Hematologic and clinical aspects of experimental ovine anaplasmosis caused by Anaplasma ovis in Iran. Iran. J. Parasitol. 2012, 7, 91. [Google Scholar]
- Bevan, L.E.W. Anaplasmosis of sheep. Vet. J. 1912, 68, 400–401. [Google Scholar] [CrossRef]
- Li, H.; Zheng, Y.C.; Ma, L.; Jia, N.; Jiang, B.G.; Jiang, R.R.; Huo, Q.B.; Wang, Y.W.; Liu, H.B.; Chu, Y.L.; et al. Human infection with a novel tick-borne Anaplasma species in China: A surveillance study. Lancet Infect. Dis. 2015, 15, 663–670. [Google Scholar] [CrossRef]
- Atif, F.A. Alpha proteobacteria of genus Anaplasma (Rickettsiales: Anaplasmataceae): Epidemiology and characteristics of Anaplasma species related to veterinary and public health importance. Parasitology 2016, 143, 659–685. [Google Scholar] [CrossRef]
- Aouadi, A.; Leulmi, H.; Boucheikhchoukh, M.; Benakhla, A.; Raoult, D.; Parola, P. Molecular evidence of tick-borne hemoprotozoan-parasites (Theileria ovis and Babesia ovis) and bacteria in ticks and blood from small ruminants in Northern Algeria. Comp. Immunol. Microbiol. Infect. Dis. 2017, 50, 34–39. [Google Scholar] [CrossRef]
- Ruiz, H.; de Arcaute, M.R.; Benito, A.Á.; Villanueva-Saz, S.; Jiménez, J.C.; Lacasta, D. Long-lasting infection with Anaplasma ovis in sheep. Vet. Res. Commun. 2023, 1–5. [Google Scholar] [CrossRef]
- Niaz, S.; Ur Rahman, Z.; Ali, I.; Cossío-Bayúgar, R.; Amaro-Estrada, I.; Alanazi, A.D.; Khattak, I.; Zeb, J.; Nasreen, N.; Khan, A. Molecular prevalence, characterization and associated risk factors of Anaplasma spp. and Theileria spp. in small ruminants in Northern Pakistan. Parasite 2021, 28, 3. [Google Scholar] [CrossRef]
- Talat, R.; Khanum, T.; Hayat, A. Studies on mammalian haematozoan parasites of NWFP Pakistan. J. Biol. Sci. 2005, 8, 726–729. [Google Scholar]
- Ghaffar, A.; Ijaz, M.; Ali, A.; Farooqi, S.H.; Rehman, A.; Ali, M.M.; Zafar, M.Z.; Naeem, M.A. First report on molecular characterization of anaplasmosis in small ruminants in Pakistan. J. Parasitol. 2020, 106, 360–368. [Google Scholar] [CrossRef] [PubMed]
- Naeem, M.; Amaro-Estrada, I.; Taqadus, A.; Swelum, A.A.; Alqhtani, A.H.; Asif, M.; Sajid, M.; Khan, A.U.; Tariq, A.; Anjum, S.; et al. Molecular prevalence and associated risk factors of Anaplasma ovis in Pakistani sheep. Front. Vet. Sci. 2023, 10, 1096418. [Google Scholar] [CrossRef]
- Taqadus, A.; Chiou, C.C.; Amaro-Estrada, I.; Asif, M.; Nasreen, N.; Ahmad, G.; Iqbal, J.; Ali, M.; Khan, A.; Iqbal, F.; et al. Epidemiology and phylogeny of Anaplasma ovis with a note on hematological and biochemical changes in asymptomatic goats enrolled from four districts in Punjab, Pakistan. Vector-Borne Zoonotic Dis. 2023. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Nasreen; Mitchell, R.D.; Niaz, S.; Ayaz, S.; Khattak, I.; Naeem, H.; de León, A.A.P.; Zaman, M.A. Seroprevalence of Anaplasma spp. among sheep and goats in Charsadda District, Pakistan. Small Rumin. Res. 2019, 176, 5–10. [Google Scholar] [CrossRef]
- Splitter, E.J.; Twiehaus, M.J.; Castro, E.R. Anaplasmosis in sheep in the United States. J. Am. Vet. Med. Assoc. 1955, 127, 244–245. [Google Scholar]
- Lacasta, D.; Lorenzo, M.; González, J.M.; Ruiz de Arcaute, M.; Benito, A.Á.; Baselga, C.; Milian, M.E.; Lorenzo, N.; Jiménez, C.; Villanueva-Saz, S.; et al. Epidemiological study related to the first outbreak of ovine anaplasmosis in Spain. Animals 2021, 11, 2036. [Google Scholar] [CrossRef]
- Primo, M.E.; Bellezze, J.; Morel, N.; Panizza, M.M.; Valentini, B.S.; Torioni, S.M.; Thompson, C.S. Development and field evaluation of a nested polymerase chain reaction-restriction fragment length polymorphism (nPCR-RFLP) analysis to identify A. marginale-infected and A. centrale-vaccinated cattle. Ticks Tick-Borne Dis. 2022, 13, 101952. [Google Scholar] [CrossRef]
- Ahmadi-Hamedani, M.; Ahmadi-Hamedani, M.; Fathi, E.; Sani, R.N. Comparison of selected biochemical parameters between naturally infected and non-infected goats with Anaplasma ovis. Comp. Clin. Pathol. 2014, 23, 989–992. [Google Scholar] [CrossRef]
- Kocan, K.M.; de la Fuente, J.; Cabezas-Cruz, A. The genus Anaplasma: New challenges after reclassification. Rev. Sci. Tech. 2015, 34, 577–586. [Google Scholar] [CrossRef]
- Selim, A.; Attia, K.A.; Alsubki, R.A.; Albohairy, F.; Kimiko, I.; Said, M.B. The first study on the seroprevalence of Anaplasma spp. in small ruminants and assessment of associated risk factors in North Egypt. Vet. World 2022, 15, 1221–1227. [Google Scholar] [CrossRef]
- Friedhoff, K.T. Tick-borne diseases of sheep and goats caused by Babesia, Theileria or Anaplasma spp. Parasitologia 1997, 39, 99–109. [Google Scholar]
- Stiller, D.; Crosbie, P.R.; Boyce, W.M.; Goff, W.L. Dermacentor hunteri (Acari: Ixodidae): An experimental vector of Anaplasma marginale and A. ovis (Rickettsiales: Anaplasmataceae) to calves and sheep. J. Med. Entomol. 1997, 36, 321–324. [Google Scholar] [CrossRef] [PubMed]
- de la Fuente, J.; Atkinson, M.W.; Naranjo, V.; Fernández de Mera, I.G.; Mangold, A.J.; Keating, K.A.; Kocan, K.M. Sequence analysis of the msp4 gene of Anaplasma ovis strains. Vet. Microbiol. 2007, 119, 375–381. [Google Scholar] [CrossRef]
- Hornok, S.; Elek, V.; de la Fuente, J.; Naranjo, V.; Farkas, R.; Majoros, G.; Földvári, G. First serological and molecular evidence on the endemicity of Anaplasma ovis and A. marginale in Hungary. Vet. Microbiol. 2007, 122, 316–322. [Google Scholar] [CrossRef]
- Torina, A.; Alongi, A.; Naranjo, V.; Scimeca, S.; Nicosia, S.; Di Marco, V.; Caracappa, S.; Kocan, K.; De La Fuente, J. Characterization of anaplasma infections in Sicily, Italy. Ann. N. Y. Acad. Sci. 2008, 1149, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Aktas, M.; Altay, K.; Dumanli, N.; Kalkan, A. Molecular detection and identification of Ehrlichia and Anaplasma species in ixodid ticks. Parasitol. Res. 2009, 104, 1243–1248. [Google Scholar] [CrossRef]
- Hornok, S.; Micsutka, A.; de Mera, I.F.; Meli, M.L.; Gönczi, E.; Tánczos, B.; Mangold, A.J.; Farkas, R.; Lutz, H.; Hofmann-Lehmann, R.; et al. Fatal bovine anaplasmosis in a herd with new genotypes of Anaplasma marginale, Anaplasma ovis and concurrent haemoplasmosis. Res. Vet. Sci. 2012, 92, 30–35. [Google Scholar] [CrossRef]
- Belkahia, H.; Said, M.B.; Ghribi, R.; Selmi, R.; Asker, A.B.; Yahiaoui, M.; Bousrih, M.; Daaloul-Jedidi, M.; Messadi, L. Molecular detection, genotyping and phylogeny of Anaplasma spp. in Rhipicephalus ticks from Tunisia. Acta Trop. 2019, 191, 38–49. [Google Scholar] [CrossRef]
- Jabeen, F.; Mushtaq, M.; Qayyum, M.; Hasan, M.U.; Zafar, M.A.; Riaz, A.; Nasir, F. Tick taxonomy and nucleotide sequence analysis by internal transcribed spacer 2 (ITS 2) in large ruminants of Pothohar, Pakistan. Pak. Vet. J. 2022, 42, 554–560. [Google Scholar]
- Hosseini-Vasoukolaei, N.; Oshaghi, M.A.; Shayan, P.; Vatandoost, H.; Babamahmoudi, F.; Yaghoobi-Ershadi, M.R.; Telmadarraiy, Z.; Mohtarami, F. Anaplasma infection in ticks, livestock and human in Ghaemshahr, Mazandaran Province, Iran. J. Arthropod-Borne Dis. 2014, 8, 204–211. [Google Scholar] [PubMed]
- Yang, J.; Li, Y.; Liu, Z.; Liu, J.; Niu, Q.; Ren, Q.; Chen, Z.; Guan, G.; Luo, J.; Yin, H. Molecular detection and characterization of Anaplasma spp. in sheep and cattle from Xinjiang, northwest China. Parasites Vectors 2015, 8, 108. [Google Scholar] [CrossRef]
- Chochlakis, D.; Ioannou, I.; Tselentis, Y.; Psaroulaki, A. Human anaplasmosis and Anaplasma ovis variant. Emerg. Infect. Dis. 2010, 16, 1031. [Google Scholar] [CrossRef] [PubMed]
- Xue, J.; Ren, Q.; Yang, X.L.; Wang, J.; Xie, G.; Du, L.; Guo, W.P. Human pathogens in ticks removed from humans in Hebei, China. Heliyon 2023, 9, e13859. [Google Scholar] [CrossRef] [PubMed]
- Ben Said, M.B.; Attia, K.A.; Alsubki, R.A.; Mohamed, A.A.; Kimiko, I.; Selim, A. Molecular epidemiological survey, genetic characterization and phylogenetic analysis of Anaplasma ovis infecting sheep in Northern Egypt. Acta Trop. 2022, 229, 106370. [Google Scholar] [CrossRef]
- Ulucesme, M.C.; Ozubek, S.; Aktas, M. Molecular Prevalence and genetic diversity based on Msp1a Gene of Anaplasma ovis in goats from Türkiye. Life 2023, 13, 1101. [Google Scholar] [CrossRef]
- Weather Spark. 2023. Available online: https://weatherspark.com/y/106985/Average-Weather-in-Dera-Ghazi-Khan-Pakistan-Year-Round (accessed on 7 August 2023).
- Government of Punjab. Dera Ghazi Khan, Climate. 2023. Available online: https://dgkhandivision.punjab.gov.pk/division_climate#:~:text=The%20overall%20climate%20of%20the,F%20(4%C2%B0C) (accessed on 7 August 2023).
- Yousefi, A.; Rahbari, S.; Shayan, P.; Sadeghi-dehkordi, Z.; Bahonar, A. Molecular detection of Anaplasma marginale and Anaplasma ovis in sheep and goat in west highland pasture of Iran. Asian Pac. J. Trop. Biomed. 2017, 7, 455–459. [Google Scholar] [CrossRef]
- Tamura, K. Estimation of the number of nucleotide substitutions when there are strong transition-transversion and G+C-content biases. Mol. Biol. Evol. 1992, 9, 678–687. [Google Scholar]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis, Version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef]
- Lew, A.E.; Gale, K.R.; Minchin, C.M.; Shkap, V.; de Waal, D.T. Phylogenetic analysis of the erythrocytic Anaplasma species based on 16S rDNA and GroEL (HSP60) sequences of A. marginale, A. centrale, and A. ovis and the specific detection of A. centrale vaccine strain. Vet. Microbiol. 2003, 92, 145–160. [Google Scholar] [CrossRef]
- Cabezas-Cruz, A.; Gallois, M.; Fontugne, M.; Allain, E.; Denoual, M.; Moutailler, S.; Devillers, E.; Zientara, S.; Memmi, M.; Chauvin, A.; et al. Epidemiology and genetic diversity of Anaplasma ovis in goats in Corsica, France. Parasite Vectors 2019, 12, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ceylan, C.; Ekici, Ö.D. Molecular investigation of ovine and caprine anaplasmosis in south-eastern Anatolia region of Turkey. Pak. Vet. J. 2023, 43, 139–145. [Google Scholar]
- Djiba, M.L.; Mediannikov, O.; Mbengue, M.; Thiongane, Y.; Molez, J.-F.; Seck, M.T.; Fenollar, F.; Raoult, D.; Ndiaye, M. Survey of Anaplasmataceae bacteria in sheep from Senegal. Trop. Anim. Health Prod. 2013, 45, 1557–1561. [Google Scholar] [CrossRef]
- Derdáková, M.; Štefančíková, A.; Špitalská, E.; Tarageľová, V.; Košťálová, T.; Hrkľová, G.; Kybicová, K.; Schánilec, P.; Majláthová, V.; Várady, M.; et al. Emergence and genetic variability of Anaplasma species in small ruminants and ticks from Central Europe. Vet. Microbiol. 2011, 153, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Prajapati, A.; Prajapati, B.; Patel, A.; Chauhan, P.; Das, B.; Raval, S.; Suthar, A.; Sutaria, T.; Chaudhari, R.K.; Patel, P.; et al. Molecular identification and genetic characterization of Theileria and Anaplasma infection in sheep and goat of North Gujarat, India. Parasitol. Res. 2023, 122, 1427–1433. [Google Scholar] [CrossRef] [PubMed]
- Enkhtaivan, B.; Narantsatsral, S.; Davaasuren, B.; Otgonsuren, D.; Amgalanbaatar, T.; Uuganbayar, E.; Zoljargal, M.; Myagmarsuren, P.; Suganuma, K.; Molefe, N.I.; et al. Molecular detection of Anaplasma ovis in small ruminants and ixodid ticks from Mongolia. Parasitol. Int. 2019, 69, 47–53. [Google Scholar] [CrossRef]
- Cabezas-Cruz, A.; Estrada-Peña, A.; de la Fuente, J. The good, the bad and the tick. Front. Cell Dev. Biol. 2019, 7, 79. [Google Scholar] [CrossRef]
- Abid, K.; Bukhari, S.; Asif, M.; Sattar, A.; Arshad, M.; Aktas, M.; Ozubek, S.; Shaikh, R.S.; Iqbal, F. Molecular detection and prevalence of Theileria ovis and Anaplasma marginale in sheep blood samples collected from Layyah district in Punjab, Pakistan. Trop. Anim. Health Prod. 2021, 53, 439. [Google Scholar] [CrossRef]
- Noaman, V.; Sazmand, A. Anaplasma ovis infection in sheep from Iran: Molecular prevalence, associated risk factors, and spatial clustering. Trop. Animal Health Prod. 2021, 54, 6. [Google Scholar] [CrossRef]
- Eisawi, N.M.; El Hussein, A.R.M.; Hassan, D.A.; Musa, A.B.; Hussien, M.O.; Enan, K.A.; Bakheit, M.A. A molecular prevalence survey on Anaplasma infection among domestic ruminants in Khartoum State, Sudan. Trop. Anim. Health Prod. 2020, 52, 1845–1852. [Google Scholar] [CrossRef]
- Atif, F.A.; Abbas, R.Z.; Mehnaz, S.; Qamar, M.F.; Hussain, K.; Nazir, M.U.; Zaman, M.A.; Khan, A.U.; Said, M.B. First report on molecular surveillance based on duplex detection of Anaplasma marginale and Theileria annulata in dairy cattle from Punjab, Pakistan. Trop. Anim. Health Prod. 2022, 54, 155. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, S.; Parveen, A.; Muhammad Awais, M.; Gillani, Q.; Aktas, M.; Ozubek, S.; Iqbal, F. A report on molecular detection and phylogenetic evaluation of Anaplasma marginale in ticks and blood samples collected from cattle in district Layyah in Punjab (Pakistan). Curr. Microbiol. 2021, 78, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Faruque, M.R.; Rahman, M.M.; Chowdhury, M.Y.E. Epidemiology and molecular detection of Anaplasma spp. in goats from Chattogram district, Bangladesh. Vet. Med. Sci. 2022, 8, 1240–1249. [Google Scholar] [CrossRef] [PubMed]

| Variable | β | p-Value | OR (95% CI) | Lower (CI) | Upper (CI) |
|---|---|---|---|---|---|
| Area | 0.149 | 0.536 | 1.161 | 0.724 | 1.860 |
| Species | 0.935 | 0.070 | 2.546 | 0.925 | 7.007 |
| Tick infestation load | −0.206 | 0.221 | 0.814 | 0.585 | 1.132 |
| Age | 0.504 | 0.001 | 1.655 | 1.214 | 2.258 |
| Grazing pattern | −0.486 | 0.000 | 0.615 | 0.469 | 0.808 |
| Acaricide use | 1.317 | 0.000 | 3.734 | 2.611 | 5.340 |
| Sex | 0.534 | 0.031 | 1.705 | 1.050 | 2.769 |
| Breed | 0.020 | 0.860 | 1.020 | 0.818 | 1.272 |
| Season | −0.098 | 0.443 | 0.906 | 0.705 | 1.165 |
| Variable | Category | β | Wald | p-Value | OR (95% CI) | Lower (CI) | Upper (CI) |
|---|---|---|---|---|---|---|---|
| Species | Sheep * | -- | -- | -- | -- | -- | -- |
| Goat | 1.167 | 21.806 | 0.000 | 3.212 | 1.968 | 5.242 | |
| Age | <6 months * | -- | 9.504 | 0.009 | -- | -- | -- |
| 6–12 months | 0.532 | 2.469 | 0.116 | 1.702 | 0.877 | 3.304 | |
| >12 months | 0.958 | 9.089 | 0.003 | 2.606 | 1.398 | 4.858 | |
| Acaricide use | Regular * | -- | 62.246 | 0.000 | -- | -- | -- |
| Irregular | 1.094 | 7.479 | 0.006 | 2.986 | 1.363 | 6.541 | |
| Never | 2.615 | 47.031 | 0.000 | 13.671 | 6.474 | 28.867 | |
| Sex | Male * | -- | -- | -- | -- | -- | -- |
| Female | 0.461 | 3.692 | 0.055 | 1.586 | 0.991 | 2.538 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atif, F.A.; Ullah, S.; Cossío-Bayúgar, R.; Kashif, M.; Khan, A.U.; Wu, W.-F. Molecular Epidemiology, Seasonality and Phylogenetic Investigations of Anaplasma ovis in Small Ruminants from Diverse Agro-Climatic Regions of Punjab, Pakistan. Microorganisms 2023, 11, 2430. https://doi.org/10.3390/microorganisms11102430
Atif FA, Ullah S, Cossío-Bayúgar R, Kashif M, Khan AU, Wu W-F. Molecular Epidemiology, Seasonality and Phylogenetic Investigations of Anaplasma ovis in Small Ruminants from Diverse Agro-Climatic Regions of Punjab, Pakistan. Microorganisms. 2023; 11(10):2430. https://doi.org/10.3390/microorganisms11102430
Chicago/Turabian StyleAtif, Farhan Ahmad, Sami Ullah, Raquel Cossío-Bayúgar, Muhammad Kashif, Aman Ullah Khan, and Wen-Feng Wu. 2023. "Molecular Epidemiology, Seasonality and Phylogenetic Investigations of Anaplasma ovis in Small Ruminants from Diverse Agro-Climatic Regions of Punjab, Pakistan" Microorganisms 11, no. 10: 2430. https://doi.org/10.3390/microorganisms11102430
APA StyleAtif, F. A., Ullah, S., Cossío-Bayúgar, R., Kashif, M., Khan, A. U., & Wu, W.-F. (2023). Molecular Epidemiology, Seasonality and Phylogenetic Investigations of Anaplasma ovis in Small Ruminants from Diverse Agro-Climatic Regions of Punjab, Pakistan. Microorganisms, 11(10), 2430. https://doi.org/10.3390/microorganisms11102430






