Year-Long Microbial Succession on Microplastics in Wastewater: Chaotic Dynamics Outweigh Preferential Growth
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Setup
2.2. Extraction and Sequencing
2.3. Data Analysis
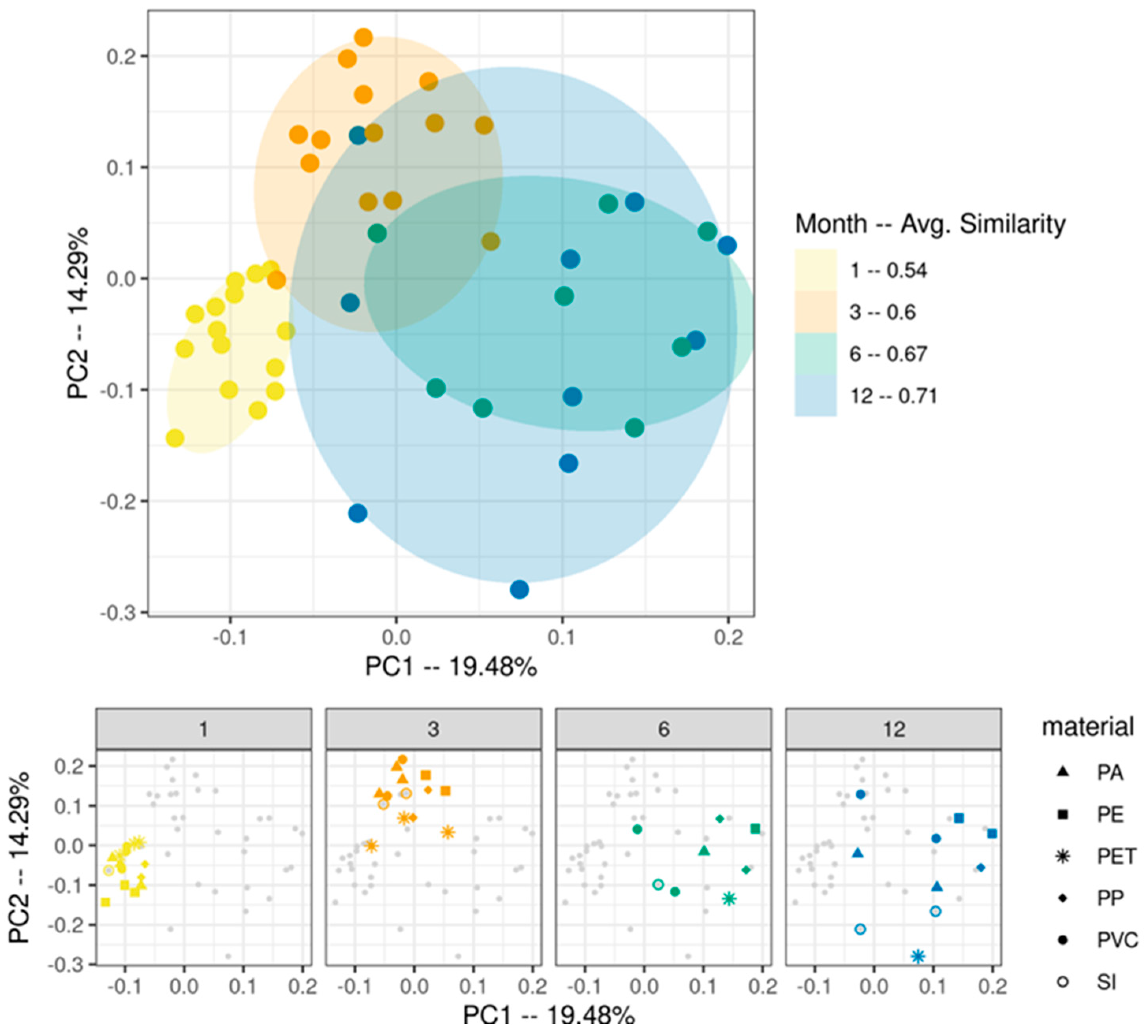
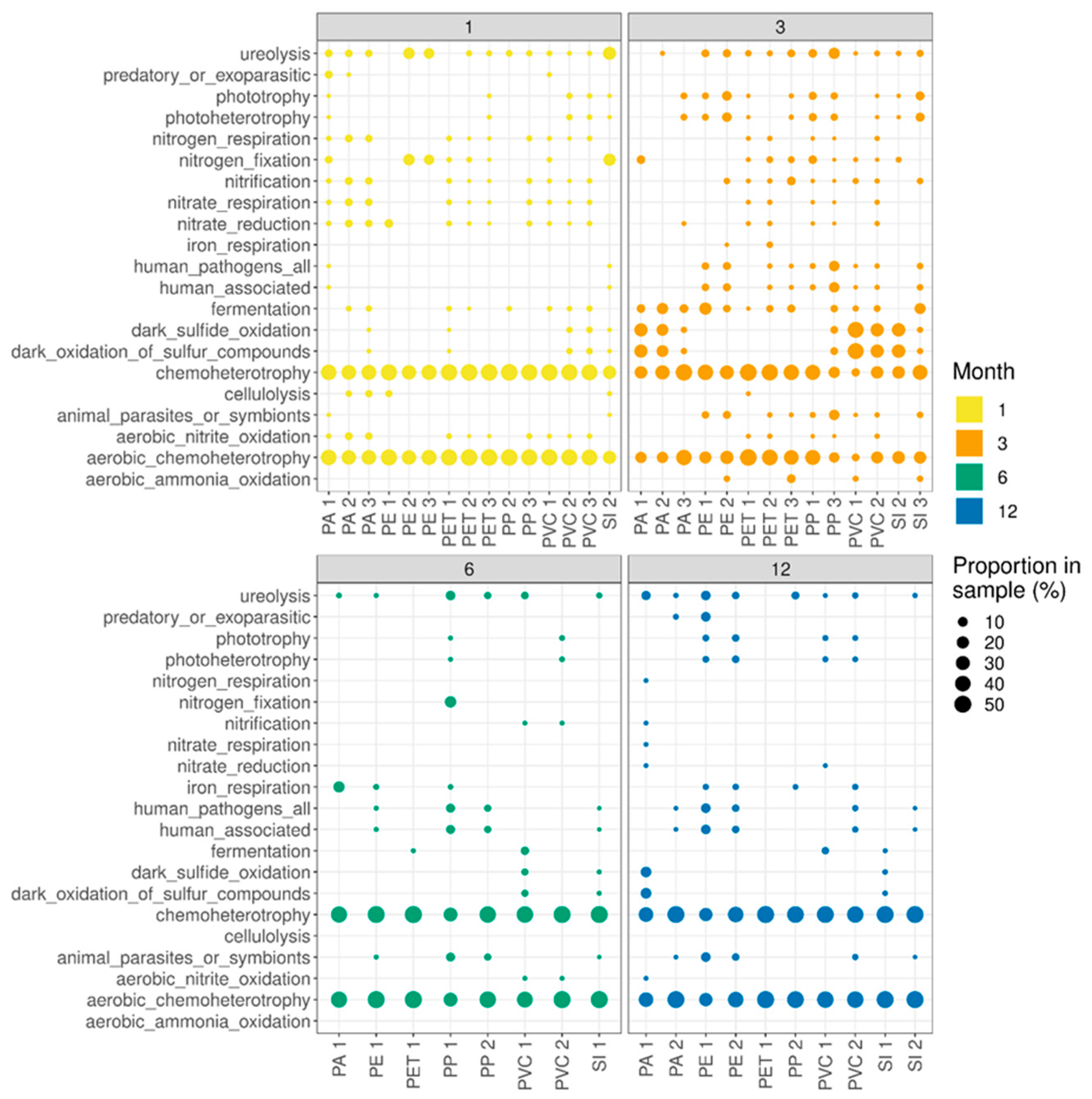
3. Results
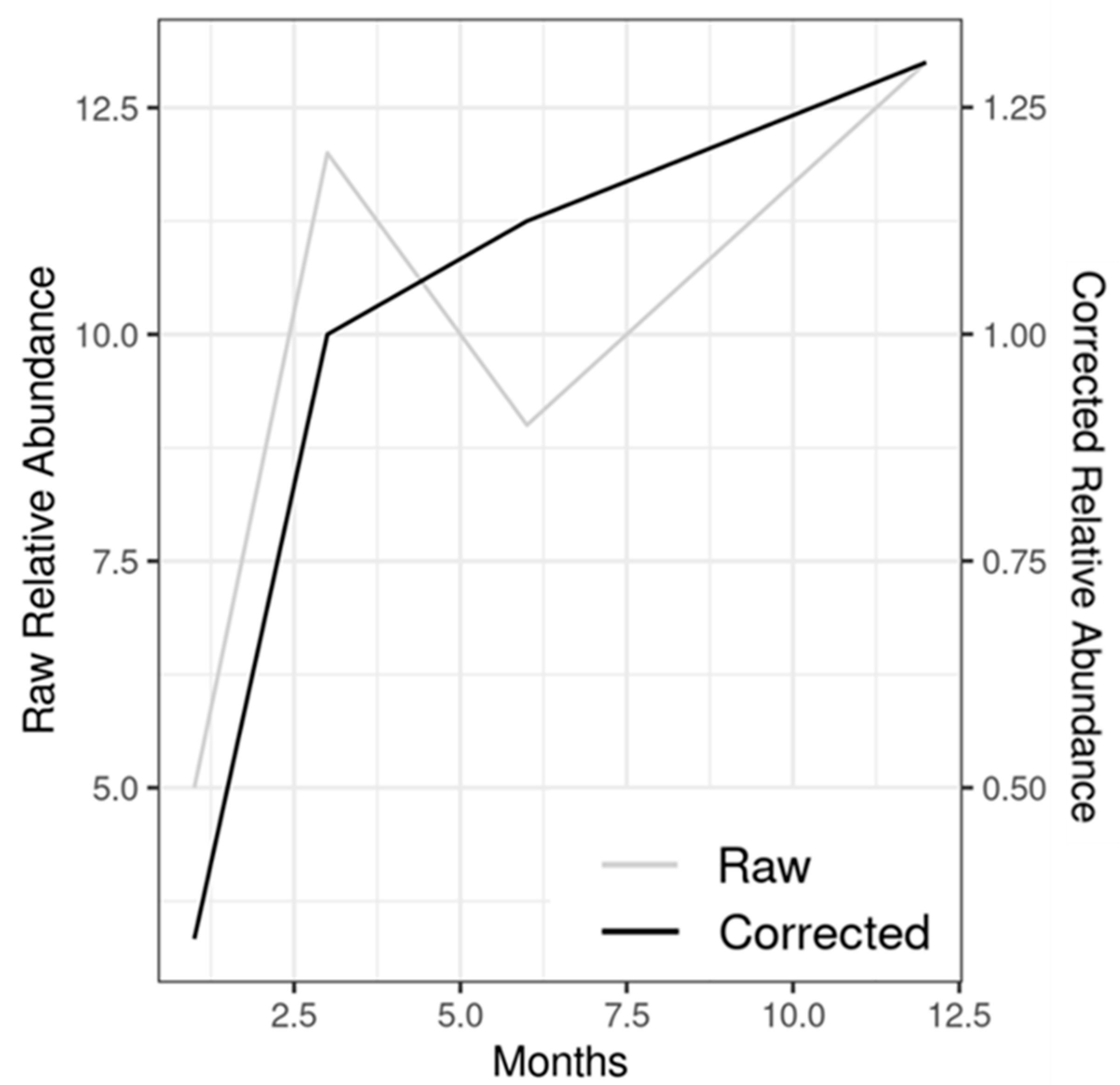
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Plastics Europe. Plastics—The Facts. 2021. Available online: https://plasticseurope.org/knowledge-hub/plastics-the-facts-2021/ (accessed on 2 November 2021).
- Setälä, O.; Fleming-Lehtinen, V.; Lehtiniemi, M. Ingestion and transfer of microplastics in the planktonic food web. Environ. Pollut. 2014, 185, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Farrell, P.; Nelson, K. Trophic level transfer of microplastic: Mytilus edulis (L.) to Carcinus maenas (L.). Environ. Pollut. 2013, 177, 1–3. [Google Scholar] [CrossRef]
- Amelia, T.S.M.; Khalik, W.M.A.W.M.; Ong, M.C.; Shao, Y.T.; Pan, H.-J.; Bhubalan, K. Marine microplastics as vectors of major ocean pollutants and its hazards to the marine ecosystem and humans. Prog. Earth Planet. Sci. 2021, 8, 1–26. [Google Scholar] [CrossRef]
- Koelmans, A.A.; Bakir, A.; Burton, G.A.; Janssen, C.R. Microplastic as a Vector for Chemicals in the Aquatic Environ-ment: Critical Review and Model-Supported Reinterpretation of Empirical Studies. Environ. Sci. Technol. 2016, 50, 3315–3326. [Google Scholar] [CrossRef]
- Bowley, J.; Baker-Austin, C.; Porter, A.; Hartnell, R.; Lewis, C. Oceanic Hitchhikers—Assessing Pathogen Risks from Ma-rine Microplastic. Trends Microbiol. 2021, 29, 107–116. [Google Scholar] [CrossRef]
- Barnes DK, A.; Galgani, F.; Thompson, R.C.; Barlaz, M. Accumulation and fragmentation of plastic debris in global environments. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1985–1998. [Google Scholar] [CrossRef] [PubMed]
- Desforges, J.-P.W.; Galbraith, M.; Dangerfield, N.; Ross, P.S. Widespread distribution of microplastics in subsurface sea-water in the NE Pacific Ocean. Mar. Pollut. Bull. 2014, 79, 94–99. [Google Scholar] [CrossRef]
- Cole, M.; Lindeque, P.; Halsband, C.; Galloway, T.S. Microplastics as contaminants in the marine environment: A review. Mar. Pollut. Bull. 2011, 62, 2588–2597. [Google Scholar] [CrossRef]
- Hogue, C. Microplastic Beads Pollute Great Lakes. Chem. Eng. News 2013, 91, 23–25. [Google Scholar] [CrossRef]
- Driedger, A.G.; Dürr, H.H.; Mitchell, K.; Van Cappellen, P. Plastic debris in the Laurentian Great Lakes: A review. J. Great Lakes Res. 2015, 41, 9–19. [Google Scholar] [CrossRef]
- Dodeva, S. Microorganisms in Lakes and Reservoirs; Springer: Dordrecht, The Netherlands, 2012; pp. 524–527. [Google Scholar] [CrossRef]
- Zbyszewski, M.; Corcoran, P.L. Distribution and Degradation of Fresh Water Plastic Particles Along the Beaches of Lake Huron, Canada. Water Air Soil Pollut. 2011, 220, 365–372. [Google Scholar] [CrossRef]
- Moore, C.; Lattin, G.; Zellers, A. Quantity and type of plastic debris flowing from two urban rivers to coastal waters and beaches of Southern California. Rev. De Gestão Costeira Integr. J. Integr. Coast. Zone Manag. 2011, 11, 65–73. [Google Scholar] [CrossRef]
- Klein, M.; Fischer, E.K. Microplastic abundance in atmospheric deposition within the Metropolitan area of Hamburg, Germany. Sci. Total Environ. 2019, 685, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Wright, S.; Ulke, J.; Font, A.; Chan, K.; Kelly, F. Atmospheric microplastic deposition in an urban environment and an evaluation of transport. Environ. Int. 2019, 136, 105411. [Google Scholar] [CrossRef]
- Allen, S.; Allen, D.; Phoenix, V.; Le Roux, G.; Jiménez, P.D.; Simonneau, A.; Binet, S.; Galop, D. Atmospheric transport and deposition of microplastics in a remote mountain catchment. Nat. Geosci. 2019, 12, 339–344. [Google Scholar] [CrossRef]
- Kanhai, L.D.K.; Gardfeldt, K.; Krumpen, T.; Thompson, R.C.; O’Connor, I. Microplastics in sea ice and seawater beneath ice floes from the Arctic Ocean. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef]
- Dyachenko, A.; Mitchell, J.; Arsem, N. Extraction and identification of microplastic particles from secondary wastewater treatment plant (WWTP) effluent. Anal. Methods 2016, 9, 1412–1418. [Google Scholar] [CrossRef]
- Tagg, A.S.; Sapp, M.; Harrison, J.P.; Sinclair, C.J.; Bradley, E.; Ju-Nam, Y.; Ojeda, J.J. Microplastic Monitoring at Different Stages in a Wastewater Treatment Plant Using Reflectance Micro-FTIR Imaging. Front. Environ. Sci. 2020, 8, 145. [Google Scholar] [CrossRef]
- Mintenig, S.; Int-Veen, I.; Löder, M.; Primpke, S.; Gerdts, G. Identification of microplastic in effluents of waste water treatment plants using focal plane array-based micro-Fourier-transform infrared imaging. Water Res. 2017, 108, 365–372. [Google Scholar] [CrossRef]
- Lusher, A.L.; Hurley, R.R.; Vogelsang, C.; Nizzetto, L.; Olsen, M. Mapping Microplastics in Sludge; M-907; Norsk Institutt for Vannforskning: Oslo, Norway, 2017. [Google Scholar] [CrossRef]
- Estahbanati, S.; Fahrenfeld, N. Influence of wastewater treatment plant discharges on microplastic concentrations in surface water. Chemosphere 2016, 162, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Oberbeckmann, S.; Labrenz, M. Marine Microbial Assemblages on Microplastics: Diversity, Adaptation, and Role in Degradation. Annu. Rev. Mar. Sci. 2020, 12, 209–232. [Google Scholar] [CrossRef]
- Oberbeckmann, S.; Löder, M.G.J.; Labrenz, M. Marine microplastic-associated biofilms—A review. Environ. Chem. 2015, 12, 551–562. [Google Scholar] [CrossRef]
- Sul, J.A.I.D.; Tagg, A.S.; Labrenz, M. Exploring the common denominator between microplastics and microbiology: A scientometric approach. Scientometrics 2018, 117, 2145–2157. [Google Scholar] [CrossRef]
- Harrison, J.P. The Spectroscopic Detection and Bacterial Colonisation of Synthetic Microplastics in Coastal Marine Sediments. Ph.D. Thesis, University of Sheffield, Sheffield, UK, 2012. [Google Scholar]
- Kesy, K.; Labrenz, M.; Scales, B.S.; Kreikemeyer, B.; Oberbeckmann, S. Vibrio Colonization Is Highly Dynamic in Early Microplastic-Associated Biofilms as Well as on Field-Collected Microplastics. Microorganisms 2020, 9, 76. [Google Scholar] [CrossRef] [PubMed]
- Zettler, E.R.; Mincer, T.J.; Amaral-Zettler, L.A. Life in the “Plastisphere”: Microbial Communities on Plastic Marine De-bris. Environ. Sci. Technol. 2013, 47, 7137–7146. [Google Scholar] [CrossRef]
- A Amaral-Zettler, L.; Zettler, E.; Slikas, B.; Boyd, G.D.; Melvin, D.W.; E Morrall, C.; Proskurowski, G.; Mincer, T.J. The biogeography of the Plastisphere: Implications for policy. Front. Ecol. Environ. 2015, 13, 541–546. [Google Scholar] [CrossRef]
- De Tender, C.; Schlundt, C.; Devriese, L.I.; Mincer, T.J.; Zettler, E.R.; Amaral-Zettler, L.A. A review of microscopy and comparative molecular-based methods to characterize ‘Plastisphere’ communities. Anal. Methods 2017, 9, 2132–2143. [Google Scholar] [CrossRef]
- Amaral-Zettler, L.A.; Zettler, E.R.; Mincer, T.J. Ecology of the plastisphere. Nat. Rev. Microbiol. 2020, 18, 139–151. [Google Scholar] [CrossRef]
- McCormick, A.; Hoellein, T.J.; Mason, S.A.; Schluep, J.; Kelly, J.J. Microplastic is an abundant and distinct microbial habitat in an urban river. Environ. Sci. Technol. 2014, 48, 11863–11871. [Google Scholar] [CrossRef]
- Weig, A.R.; Löder, M.G.J.; Ramsperger, A.F.R.M.; Laforsch, C. In situ Prokaryotic and Eukaryotic Communities on Microplastic Particles in a Small Headwater Stream in Germany. Front. Microbiol. 2021, 12, 660024. [Google Scholar] [CrossRef] [PubMed]
- Artham, T.; Sudhakar, M.; Venkatesan, R.; Nair, C.M.; Murty, K.V.G.K.; Doble, M. Biofouling and stability of synthetic polymers in sea water. Int. Biodeterior. Biodegrad. 2009, 63, 884–890. [Google Scholar] [CrossRef]
- Oberbeckmann, S.; Kreikemeyer, B.; Labrenz, M. Environmental factors support the formation of specific bacterial assemblages on microplastics. Front. Microbiol. 2018, 8, 2709. [Google Scholar] [CrossRef] [PubMed]
- Salter, S.J.; Cox, M.J.; Turek, E.M.; Calus, S.T.; Cookson, W.O.; Moffatt, M.F.; Turner, P.; Parkhill, J.; Loman, N.J.; Walker, A.W. Reagent and laboratory contamination can critically impact sequence-based microbiome analyses. BMC Biol. 2014, 12, 87. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Lozupone, C.A.; Turnbaugh, P.J.; Fierer, N.; Knight, R. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. 1), 4516–4522. [Google Scholar] [CrossRef] [PubMed]
- Toju, H.; Tanabe, A.S.; Yamamoto, S.; Sato, H. High-Coverage ITS Primers for the DNA-Based Identification of Ascomy-cetes and Basidiomycetes in Environmental Samples. PLoS ONE 2012, 7, e40863. [Google Scholar]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Gonzalez Peña, A.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Afgan, E.; Baker, D.; van den Beek, M.; Blankenberg, D.; Bouvier, D.; Čech, M.; Chilton, J.; Clements, D.; Coraor, N.; Eberhard, C.; et al. The Galaxy platform for accessible, reproducible and collaborative biomedical analyses: 2016 update. Nucleic Acids Res. 2016, 44, W3–W10. [Google Scholar] [CrossRef]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef]
- Debeljak, P.; Pinto, M.; Proietti, M.; Reisser, J.; Ferrari, F.F.; Abbas, B.; van Loosdrecht, M.C.M.; Slat, B.; Herndl, G.J. Extracting DNA from ocean microplastics: A method comparison study. Anal. Methods 2016, 9, 1521–1526. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2022. [Google Scholar]
- Goslee, S.C.; Urban, D. The ecodist package for dissimilarity-based analysis of ecological data. J. Stat. Softw. 2007, 22, 1–19. [Google Scholar] [CrossRef]
- Gu, Z.; Eils, R.; Schlesner, M. Complex heatmaps reveal patterns and correlations in multidimensional genomic data. Bioinformatics 2016, 32, 2847–2849. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Kassambara, A. ggpubr: ‘ggplot2′ Based Publication Ready Plots. 2020. Available online: https://rpkgs.datanovia.com/ggpubr (accessed on 5 May 2022).
- Pedersen, T. ggforce: Accelerating ‘ggplot2′. 2021. Available online: https://ggforce.data-imaginist.com (accessed on 5 May 2022).
- De Cáceres, M.; Legendre, P. Associations between species and groups of sites: Indices and statistical inference. Ecology 2009, 90, 3566–3574. [Google Scholar] [CrossRef] [PubMed]
- Louca, S.; Parfrey, L.W.; Doebeli, M. Decoupling function and taxonomy in the global ocean microbiome. Science 2016, 353, 1272–1277. [Google Scholar] [CrossRef] [PubMed]
- Oksanen, J.; Blanchet, F.G.; Kindt, R.; Legendre, P.; Minchin, P.R.; O’hara, R.B.; Simpson, G.L.; Solymos, P.; Stevens, M.H.H.; Wagner, H.H. Vegan: Community Ecology Package. R Package Version 2.0-10, 2013. Available online: https://cran.r-project.org/web/packages/vegan/index.html (accessed on 4 May 2017).
- Roeselers, G.; Zippel, B.; Staal, M.; Van Loosdrecht, M.; Muyzer, G. On the reproducibility of microcosm experiments—Different community composition in parallel phototrophic biofilm microcosms. FEMS Microbiol. Ecol. 2006, 58, 169–178. [Google Scholar] [CrossRef]
- Kangas, P.; Adey, W. Mesocosms and ecological engineering. Ecol. Eng. 1996, 6, 1–5. [Google Scholar] [CrossRef]
- May, R.M. Simple mathematical models with very complicated dynamics. Nature 1976, 261, 459–467. [Google Scholar] [CrossRef]
- Becks, L.; Hilker, F.; Malchow, H.; Jürgens, K.; Arndt, H. Experimental demonstration of chaos in a microbial food web. Nature 2005, 435, 1226–1229. [Google Scholar] [CrossRef] [PubMed]
- Rogers, T.L.; Johnson, B.J.; Munch, S.B. Chaos is not rare in natural ecosystems. Nat. Ecol. Evol. 2022, 68, 1105–1111. [Google Scholar] [CrossRef]
- McGowan, J.A.; Deyle, E.R.; Ye, H.; Carter, M.L.; Perretti, C.T.; Seger, K.D.; Verneil, A.; Sugihara, G. Predicting coastal algal blooms in southern California. Ecology 2017, 98, 1419–1433. [Google Scholar] [CrossRef] [PubMed]
- Kesy, K.; Oberbeckmann, S.; Kreikemeyer, B.; Labrenz, M. Spatial Environmental Heterogeneity Determines Young Biofilm Assemblages on Microplastics in Baltic Sea Mesocosms. Front. Microbiol. 2019, 10, 1665. [Google Scholar] [CrossRef] [PubMed]
- Oberbeckmann, S.; Loeder, M.G.; Gerdts, G.; Osborn, A.M. Spatial and seasonal variation in diversity and structure of microbial biofilms on marine plastics in Northern European waters. FEMS Microbiol. Ecol. 2014, 90, 478–492. [Google Scholar] [CrossRef] [PubMed]
- Lagarde, F.; Olivier, O.; Zanella, M.; Daniel, P.; Hiard, S.; Caruso, A. Microplastic interactions with freshwater microalgae: Hetero-aggregation and changes in plastic density appear strongly dependent on polymer type. Environ. Pollut. 2016, 215, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Parrish, K.; Fahrenfeld, N.L. Microplastic biofilm in fresh- and wastewater as a function of microparticle type and size class. Environ. Sci. Water Res. Technol. 2019, 5, 495–505. [Google Scholar] [CrossRef]
- Yoo, S.-H.; Weon, H.-Y.; Anandham, R.; Kim, B.-Y.; Hong, S.-B.; Jeon, Y.-A.; Koo, B.-S.; Kwon, S.-W. Dokdonella soli sp. nov., a gammaproteobacterium isolated from soil. Int. J. Syst. Evol. Microbiol. 2009, 59, 1965–1968. [Google Scholar] [CrossRef][Green Version]
- Liu, Y.; Jin, J.-H.; Liu, H.-C.; Liu, Z.-P. Dokdonella immobilis sp. nov., isolated from a batch reactor for the treatment of triphenylmethane dye effluent. Int. J. Syst. Evol. Microbiol. 2013, 63, 1557–1561. [Google Scholar] [CrossRef]
- Cunha, S.; Tiago, I.; Pires, A.L.; da Costa, M.S.; Veríssimo, A. Dokdonella fugitiva sp. nov., a Gammaproteobacterium isolated from potting soil. Syst. Appl. Microbiol. 2006, 29, 191–196. [Google Scholar] [CrossRef]
- Rawat, S.R.; Männistö, M.K.; Starovoytov, V.; Goodwin, L.; Nolan, M.; Hauser, L.; Land, M.; Davenport, K.W.; Woyke, T.; Häggblom, M.M. Complete genome sequence of Terriglobus saanensis type strain SP1PR4T, an Acidobacteria from tundra soil. Stand. Genom. Sci. 2012, 7, 59–69. [Google Scholar] [CrossRef]
- Eichorst, S.A.; Breznak, J.A.; Schmidt, T.M. Isolation and Characterization of Soil Bacteria That Define Terriglobus gen. nov., in the Phylum Acidobacteria. Appl. Environ. Microbiol. 2007, 73, 2708. [Google Scholar] [CrossRef]
- Azadi, D.; Shojaei, H.; Pourchangiz, M.; Dibaj, R.; Davarpanah, M.; Naser, A.D. Species diversity and molecular characterization of nontuberculous mycobacteria in hospital water system of a developing country, Iran. Microb. Pathog. 2016, 100, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.Z.; Li, J.; Zhu, W.Y.; Wei, D.Q.; Zhang, J.L.; Xu, L.H.; Li, W.J. Pseudonocardia xishanensis sp. nov., an endophytic actinomycete isolated from the roots of Artemisia annua L. Int. J. Syst. Evol. Microbiol. 2012, 62, 2395–2399. [Google Scholar] [CrossRef] [PubMed]
- Sato, I.; Ito, M.; Ishizaka, M.; Ikunaga, Y.; Sato, Y.; Yoshida, S.; Koitabashi, M.; Tsushima, S. Thirteen novel deoxynivalenol-degrading bacteria are classified within two genera with distinct degradation mechanisms. FEMS Microbiol. Lett. 2011, 327, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Zhu, Z.; Zhou, Y.; Ji, F.; Yao, Z.; Shi, J.; Xu, J. Complete genome sequence of deoxynivalenol-degrading bacterium Devosia sp. strain A16. J. Biotechnol. 2016, 218, 21–22. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Zhao, C.C.; Liu, Q.Y.; Zhang, Y.B. Isolation and Genetic Identification of Dibenzothiophene Degrading Bacteria from Contaminated Soil. Adv. Mater. Res. 2012, 610–613, 292–295. [Google Scholar] [CrossRef]
- Wallbank, J.A.; Lear, G.; Kingsbury, J.M.; Weaver, L.; Doake, F.; Smith, D.A.; Audrézet, F.; Maday, S.D.M.; Gambarini, V.; Donaldson, L.; et al. Into the Plastisphere, Where Only the Generalists Thrive: Early Insights in Plastisphere Microbial Community Succession. Front. Mar. Sci. 2022, 9, 626. [Google Scholar] [CrossRef]
- Raghukumar, S. The Marine Environment and the Role of Fungi. In Fungi in Coastal and Oceanic Marine Ecosystems; Springer International Publishing: Cham, Switzerland, 2017; pp. 17–38. [Google Scholar]
- Tachibana, K.; Hashimoto, K.; Yoshikawa, M.; Okawa, H. Isolation and characterization of microorganisms degrading nylon 4 in the composted soil. Polym. Degrad. Stab. 2010, 95, 912–917. [Google Scholar] [CrossRef]
- Hamm, P.S.; Caimi, N.A.; Northup, D.E.; Valdez, E.W.; Buecher, D.C.; Dunlap, C.A.; Labeda, D.P.; Lueschow, S.; Porras-Alfaro, A. Western Bats as a Reservoir of Novel Streptomyces Species with Antifungal Activity. Appl. Environ. Microbiol. 2017, 83, e03057-16. [Google Scholar] [CrossRef]
- Thijs, S.; Weyens, N.; Sillen, W.; Gkorezis, P.; Carleer, R.; Vangronsveld, J. Potential for Plant Growth Promotion by a Consortium of Stress-Tolerant 2,4-Dinitrotoluene-Degrading Bacteria: Isolation and Characterization of a Military Soil. Microb. Biotechnol. 2014, 7, 294–306. [Google Scholar] [CrossRef]
- Colin, Y.; Nicolitch, O.; Turpault, M.-P.; Uroz, S. Mineral Types and Tree Species Determine the Functional and Taxonomic Structures of Forest Soil Bacterial Communities. Appl. Environ. Microbiol. 2017, 83, e02684-16. [Google Scholar] [CrossRef]
- Mikolasch, A.; Reinhard, A.; Alimbetova, A.; Omirbekova, A.; Pasler, L.; Schumann, P.; Kabisch, J.; Mukasheva, T.; Schauer, F. From Oil Spills to Barley Growth - Oil-Degrading Soil Bacteria and Their Promoting Effects. J. Basic Microbiol. 2016, 56, 1252–1273. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.L.; Pereira, M.A.; Janknecht, P.; Brito, A.G.; Nogueira, R. Biofilms Formed on Humic Substances: Response to Flow Conditions and Carbon Concentrations. Bioresour. Technol. 2010, 101, 6888–6894. [Google Scholar] [CrossRef]
- Suzuki, K.; Owen, R.; Mok, J.; Mochihara, H.; Hosokawa, T.; Kubota, H.; Sakamoto, H.; Matsuda, A.; Tashiro, Y.; Futamata, H. Comparison of Electrochemical and Microbiological Characterization of Microbial Fuel Cells Equipped with SPEEK and Nafion Membrane Electrode Assemblies. J. Biosci. Bioeng. 2016, 122, 322–328. [Google Scholar] [CrossRef]
- Zinger, L.; Shahnavaz, B.; Baptist, F.; Geremia, R.A.; Choler, P. Microbial Diversity in Alpine Tundra Soils Correlates with Snow Cover Dynamics. ISME J. 2009, 3, 850–859. [Google Scholar] [CrossRef]
- Zeglin, L.H.; Wang, B.; Waythomas, C.; Rainey, F.; Talbot, S.L. Organic Matter Quantity and Source Affects Microbial Community Structure and Function Following Volcanic Eruption on Kasatochi Island, Alaska. Environ. Microbiol. 2016, 18, 146–158. [Google Scholar] [CrossRef] [PubMed]
- Peipoch, M.; Jones, R.; Valett, H.M.; Sedell, J.; Cushing, C.; Hödl, I. Spatial Patterns in Biofilm Diversity across Hierarchical Levels of River-Floodplain Landscapes. PLoS ONE 2015, 10, e0144303. [Google Scholar] [CrossRef]
- Fonseca-García, C.; Coleman-Derr, D.; Garrido, E.; Visel, A.; Tringe, S.G.; Partida-Martínez, L.P. The Cacti Microbiome: Interplay between Habitat-Filtering and Host-Specificity. Front. Microbiol. 2016, 7, 150. [Google Scholar] [CrossRef]
- Sun, Y.; Wei, J.; Liang, P.; Huang, X. Microbial Community Analysis in Biocathode Microbial Fuel Cells Packed with Different Materials. AMB Express 2012, 2, 21. [Google Scholar] [CrossRef]
- Haskett, T.; Wang, P.; Ramsay, J.; O’Hara, G.; Reeve, W.; Howieson, J.; Terpolilli, J. Complete Genome Sequence of Mesorhizobium Ciceri Strain CC1192, an Efficient Nitrogen-Fixing Microsymbiont of Cicer Arietinum. Genome Announc. 2016, 4, 516–532. [Google Scholar] [CrossRef]
- De Meyer, S.E.; Willems, A.; Tan, H.; Andrews, M.; Heenan, P.B. Mesorhizobium waimense Sp. Nov. Isolated from Sophora longicarinata Root Nodules and Mesorhizobium cantuariense sp. Nov. Isolated from Sophora Microphylla Root Nodules. Int. J. Syst. Evol. Microbiol. 2015, 65, 3419–3426. [Google Scholar] [CrossRef]
- Yin, H.; Niu, J.; Ren, Y.; Cong, J.; Zhang, X.; Fan, F.; Xiao, Y.; Zhang, X.; Deng, J.; Xie, M.; et al. An Integrated Insight into the Response of Sedimentary Microbial Communities to Heavy Metal Contamination. Sci. Rep. 2015, 5, 14266. [Google Scholar] [CrossRef]
- Yergeau, E.; Newsham, K.K.; Pearce, D.A.; Kowalchuk, G.A. Patterns of Bacterial Diversity across a Range of Antarctic Terrestrial Habitats. Environ. Microbiol. 2007, 9, 2670–2682. [Google Scholar] [CrossRef]
- Dunbar, J.; Eichorst, S.A.; Gallegos-Graves, L.V.; Silva, S.; Xie, G.; Hengartner, N.W.; Evans, R.D.; Hungate, B.A.; Jackson, R.B.; Megonigal, J.P.; et al. Common Bacterial Responses in Six Ecosystems Exposed to 10 Years of Elevated Atmospheric Carbon Dioxide. Environ. Microbiol. 2012, 14, 1145–1158. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, G.; Stetler, L.D.; Peyton, B.M.; Sani, R.K. Molecular Analysis of Prokaryotic Diversity in the Deep Subsurface of the Former Homestake Gold Mine, South Dakota, USA. J. Microbiol. 2009, 47, 371–384. [Google Scholar] [CrossRef]
- Hodkinson, B.P.; Gottel, N.R.; Schadt, C.W.; Lutzoni, F. Photoautotrophic Symbiont and Geography Are Major Factors Affecting Highly Structured and Diverse Bacterial Communities in the Lichen Microbiome. Environ. Microbiol. 2012, 14, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Kelly, L.C.; Cockell, C.S.; Piceno, Y.M.; Andersen, G.L.; Thorsteinsson, T.; Marteinsson, V. Bacterial Diversity of Weathered Terrestrial Icelandic Volcanic Glasses. Microb. Ecol. 2010, 60, 740–752. [Google Scholar] [CrossRef]
- Ricaboni, D.; Mailhe, M.; Khelaifia, S.; Raoult, D.; Million, M. Romboutsia Timonensis, a New Species Isolated from Human Gut. New Microbes New Infect. 2016, 12, 6–7. [Google Scholar] [CrossRef] [PubMed]
- Falteisek, L.; Čepička, I. Microbiology of Diverse Acidic and Non-Acidic Microhabitats within a Sulfidic Ore Mine. Extremophiles 2012, 16, 911–922. [Google Scholar] [CrossRef]
- Chen, M.; Li, X.; Yang, Q.; Chi, X.; Pan, L.; Chen, N.; Yang, Z.; Wang, T.; Wang, M.; Yu, S. Dynamic Succession of Soil Bacterial Community during Continuous Cropping of Peanut (Arachis Hypogaea L.). PLoS ONE 2014, 9, e101355. [Google Scholar] [CrossRef]
- Hilyard, E.J.; Jones-Meehan, J.M.; Spargo, B.J.; Hill, R.T. Enrichment, Isolation, and Phylogenetic Identification of Polycyclic Aromatic Hydrocarbon-Degrading Bacteria from Elizabeth River Sediments. Appl. Environ. Microbiol. 2008, 74, 1176–1182. [Google Scholar] [CrossRef] [PubMed]
- Ganzert, L.; Bajerski, F.; Wagner, D. Bacterial Community Composition and Diversity of Five Different Permafrost-Affected Soils of Northeast Greenland. FEMS Microbiol. Ecol. 2014, 89, 426–441. [Google Scholar] [CrossRef] [PubMed]
- Al-Sadi, A.M.; Al-Zakwani, H.A.; Nasehi, A.; Al-Mazroui, S.S.; Al-Mahmooli, I.H. Analysis of Bacterial Communities Associated with Potting Media. Springerplus 2016, 5, 74. [Google Scholar] [CrossRef] [PubMed]
- Al-Kharousi, Z.S.; Guizani, N.; Al-Sadi, A.M.; Al-Bulushi, I.M.; Shaharoona, B. Hiding in Fresh Fruits and Vegetables: Opportunistic Pathogens May Cross Geographical Barriers. Int. J. Microbiol. 2016, 2016, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lesaulnier, C.; Papamichail, D.; McCorkle, S.; Ollivier, B.; Skiena, S.; Taghavi, S.; Zak, D.; van der Lelie, D. Elevated Atmospheric CO 2 Affects Soil Microbial Diversity Associated with Trembling Aspen. Environ. Microbiol. 2008, 10, 926–941. [Google Scholar] [CrossRef]
- Zhao, J.; Wang, B.; Jia, Z. Phylogenetically Distinct Phylotypes Modulate Nitrification in a Paddy Soil. Appl. Environ. Microbiol. 2015, 81, 3218–3227. [Google Scholar] [CrossRef]
- Sepúlveda, F.A.; Torres, J.F.; Infante, C.D.; González, M.T. Potential Role of Ectoparasites (Zeuxapta Seriolae and Caligus Lalandei) in the Transmission of Pathogenic Bacteria in Yellowtail Kingfish Seriola Lalandi, Inferred from Cultivable Microbiota and Molecular Analyses. J. Fish Dis. 2016, 40, 979–985. [Google Scholar] [CrossRef] [PubMed]
- Lueders, T.; Kindler, R.; Miltner, A.; Friedrich, M.W.; Kaestner, M. Identification of Bacterial Micropredators Distinctively Active in a Soil Microbial Food Web. Appl. Environ. Microbiol. 2006, 72, 5342–5348. [Google Scholar] [CrossRef]
- Sanchez-Andrea, I.; Rodriguez, N.; Amils, R.; Sanz, J.L. Microbial Diversity in Anaerobic Sediments at Rio Tinto, a Naturally Acidic Environment with a High Heavy Metal Content. Appl. Environ. Microbiol. 2011, 77, 6085–6093. [Google Scholar] [CrossRef]
- Auffret, M.D.; Yergeau, E.; Labbé, D.; Fayolle-Guichard, F.; Greer, C.W. Importance of Rhodococcus Strains in a Bacterial Consortium Degrading a Mixture of Hydrocarbons, Gasoline, and Diesel Oil Additives Revealed by Metatranscriptomic Analysis. Appl. Microbiol. Biotechnol. 2015, 99, 2419–2430. [Google Scholar] [CrossRef]
- Xie, J.; Strobel, G.; Xu, W.-F.; Chen, J.; Ren, H.-S.; An, D.-J.; Geary, B. Fungi as Architects of the Rimstone Dams in Huanglong, NSD, Sichuan, China. Microb. Ecol. 2017, 73, 29–38. [Google Scholar] [CrossRef]
- Poosakkannu, A.; Nissinen, R.; Kytöviita, M.-M. Culturable Endophytic Microbial Communities in the Circumpolar Grass, Deschampsia Flexuosa in a Sub-Arctic Inland Primary Succession Are Habitat and Growth Stage Specific. Environ. Microbiol. Rep. 2015, 7, 164–165. [Google Scholar] [CrossRef] [PubMed]
- Egert, M.; Schmidt, I.; Bussey, K.; Breves, R. A Glimpse under the Rim - the Composition of Microbial Biofilm Communities in Domestic Toilets. J. Appl. Microbiol. 2010, 108, 1167–1174. [Google Scholar] [CrossRef] [PubMed]
- Ceja-Navarro, J.A.; Rivera-Orduna, F.N.; Patino-Zuniga, L.; Vila-Sanjurjo, A.; Crossa, J.; Govaerts, B.; Dendooven, L. Phylogenetic and Multivariate Analyses To Determine the Effects of Different Tillage and Residue Management Practices on Soil Bacterial Communities. Appl. Environ. Microbiol. 2010, 76, 3685–3691. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, D.J.; Korenblum, E.; van Elsas, J.D. Novel Multispecies Microbial Consortia Involved in Lignocellulose and 5-Hydroxymethylfurfural Bioconversion. Appl. Microbiol. Biotechnol. 2014, 98, 2789–2803. [Google Scholar] [CrossRef]
- Anees, M.; Tronsmo, A.; Edel-Hermann, V.; Hjeljord, L.G.; Heraud, C.; Steinberg, C. Characterization of Field Isolates of Trichoderma Antagonistic against Rhizoctonia Solani. Fungal Biol. 2010, 114, 691–701. [Google Scholar] [CrossRef]
- Wen, Y.Z.; Lun, Z.R.; Zhu, X.Q.; Hide, G.; Lai, D.H. Further Evidence from SSCP and ITS DNA Sequencing Support Trypanosoma Evansi and Trypanosoma Equiperdum as Subspecies or Even Strains of Trypanosoma Brucei. Infect. Genet. Evol. 2016, in press. [Google Scholar] [CrossRef]
- Taylor, D.L.; Booth, M.G.; McFarland, J.W.; Herriott, I.C.; Lennon, N.J.; Nusbaum, C.; Marr, T.G. Increasing Ecological Inference from High Throughput Sequencing of Fungi in the Environment through a Tagging Approach. Mol. Ecol. Resour. 2008, 8, 742–752. [Google Scholar] [CrossRef]
- Lamit, L.J.; Lau, M.K.; Sthultz, C.M.; Wooley, S.C.; Whitham, T.G.; Gehring, C.A. Tree Genotype and Genetically Based Growth Traits Structure Twig Endophyte Communities. Am. J. Bot. 2014, 101, 467–478. [Google Scholar] [CrossRef]
- Bukovská, P.; Jelínková, M.; Hršelová, H.; Sýkorová, Z.; Gryndler, M. Terminal Restriction Fragment Length Measurement Errors Are Affected Mainly by Fragment Length, G+C Nucleotide Content and Secondary Structure Melting Point. J. Microbiol. Methods 2010, 82, 223–228. [Google Scholar] [CrossRef]
- Adams, R.I.; Miletto, M.; Taylor, J.W.; Bruns, T.D. The Diversity and Distribution of Fungi on Residential Surfaces. PLoS ONE 2013, 8, e78866. [Google Scholar] [CrossRef]
- De Oliveira Santos, A.R.; Perri, A.M.; Andrietta, M.D.; Rosa, C.A.; Lachance, M.A. The Expanding Large-Spored Metschnikowia Clade: Metschnikowia Matae Sp. Nov., a Yeast Species with Two Varieties from the Brazilian Atlantic Forest. Antonie Van Leeuwenhoek 2015, in press. [Google Scholar] [CrossRef] [PubMed]
- Cordier, T.; Robin, C.; Capdevielle, X.; Fabreguettes, O.; Desprez-Loustau, M.L.; Vacher, C. The Composition of Phyllosphere Fungal Assemblages of European Beech (Fagus Sylvatica) Varies Significantly along an Elevation Gradient. New Phytol. 2012, 196, 510–519. [Google Scholar] [CrossRef] [PubMed]
- Kartzinel, T.R.; Trapnell, D.W.; Shefferson, R.P. Highly Diverse and Spatially Heterogeneous Mycorrhizal Symbiosis in a Rare Epiphyte Is Unrelated to Broad Biogeographic or Environmental Features. Mol. Ecol. 2013, 22, 5949–5961. [Google Scholar] [CrossRef] [PubMed]
- Alaei, H.; De Backer, M.; Nuytinck, J.; Maes, M.; Höfte, M.; Heungens, K. Phylogenetic Relationships of Puccinia Horiana and Other Rust Pathogens of Chrysanthemum×morifolium Based on RDNA ITS Sequence Analysis. Mycol. Res. 2009, 113, 668–683. [Google Scholar] [CrossRef]
- Scoble, J.M.; Cavalier-Smith, T. Scale Evolution, Sequence Phylogeny, and Taxonomy of Thaumatomonad Cercozoa: 11 New Species and New Genera Scutellomonas, Cowlomonas, Thaumatospina and Ovaloplaca. Eur. J. Protistol. 2014, 50, 270–313. [Google Scholar] [CrossRef]
- Waldrop, M.P.; Zak, D.R.; Blackwood, C.B.; Curtis, C.D.; Tilman, D. Resource Availability Controls Fungal Diversity across a Plant Diversity Gradient. Ecol. Lett. 2006, 9, 1127–1135. [Google Scholar] [CrossRef]
- Carter, J.P.; Spink, J.; Cannon, P.F.; Daniels, M.J.; Osbourn, A.E. Isolation, Characterization, and Avenacin Sensitivity of a Diverse Collection of Cereal-Root-Colonizing Fungi. Appl. Environ. Microbiol. 1999, 65, 3364–3372. [Google Scholar] [CrossRef]
- Liu, Y.; de Bruijn, I.; Jack, A.L.; Drynan, K.; van den Berg, A.H.; Thoen, E.; Sandoval-Sierra, V.; Skaar, I.; van West, P.; Diéguez-Uribeondo, J.; et al. Deciphering Microbial Landscapes of Fish Eggs to Mitigate Emerging Diseases. ISME J. 2014, 8, 2002–2014. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tagg, A.S.; Sperlea, T.; Labrenz, M.; Harrison, J.P.; Ojeda, J.J.; Sapp, M. Year-Long Microbial Succession on Microplastics in Wastewater: Chaotic Dynamics Outweigh Preferential Growth. Microorganisms 2022, 10, 1775. https://doi.org/10.3390/microorganisms10091775
Tagg AS, Sperlea T, Labrenz M, Harrison JP, Ojeda JJ, Sapp M. Year-Long Microbial Succession on Microplastics in Wastewater: Chaotic Dynamics Outweigh Preferential Growth. Microorganisms. 2022; 10(9):1775. https://doi.org/10.3390/microorganisms10091775
Chicago/Turabian StyleTagg, Alexander S., Theodor Sperlea, Matthias Labrenz, Jesse P. Harrison, Jesús J. Ojeda, and Melanie Sapp. 2022. "Year-Long Microbial Succession on Microplastics in Wastewater: Chaotic Dynamics Outweigh Preferential Growth" Microorganisms 10, no. 9: 1775. https://doi.org/10.3390/microorganisms10091775
APA StyleTagg, A. S., Sperlea, T., Labrenz, M., Harrison, J. P., Ojeda, J. J., & Sapp, M. (2022). Year-Long Microbial Succession on Microplastics in Wastewater: Chaotic Dynamics Outweigh Preferential Growth. Microorganisms, 10(9), 1775. https://doi.org/10.3390/microorganisms10091775






